Cvs 44-347
382108c6-f814-4c84-acde-2fd1e3e38b74
34390-5
HUMAN OTC DRUG LABEL
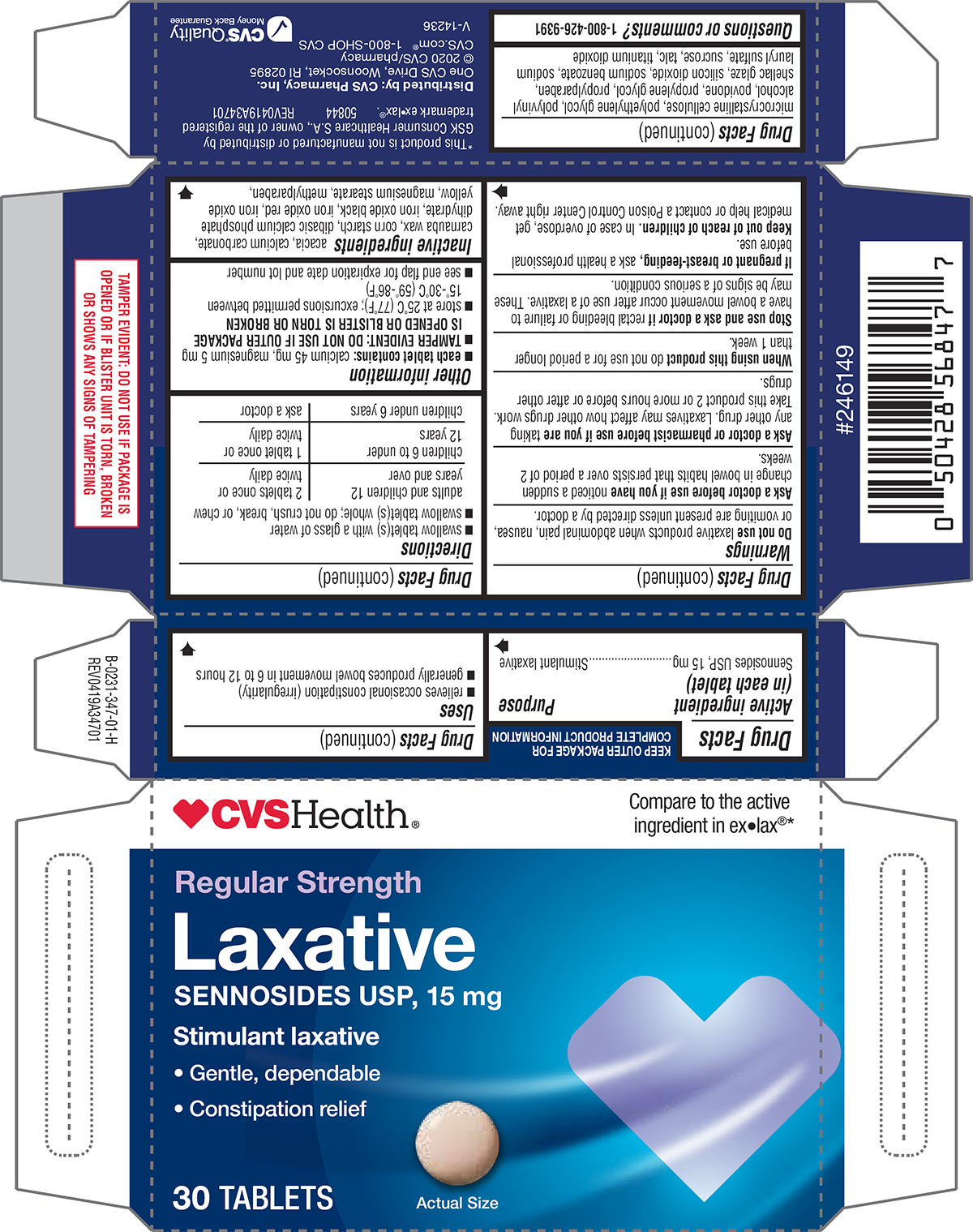
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sennosides USP, 15 mg
Purpose
Stimulant laxative
Medication Information
Purpose
Stimulant laxative
Description
Sennosides USP, 15 mg
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 6 to 12 hours
Directions
- swallow tablet(s) with a glass of water
- swallow tablet(s) whole; do not crush, break, or chew
| adults and children 12 years and over | 2 tablets once or twice daily |
| children 6 to under 12 years | 1 tablet once or twice daily |
| children under 6 years | ask a doctor |
Do Not Use
laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor.
Other Information
- each tablet contains: calcium 45 mg, magnesium 5 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive Ingredients
acacia, calcium carbonate, carnauba wax, corn starch, dibasic calcium phosphate dihydrate, iron oxide black, iron oxide red, iron oxide yellow, magnesium stearate, methylparaben, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, propylene glycol, propylparaben, shellac glaze, silicon dioxide, sodium benzoate, sodium lauryl sulfate, sucrose, talc, titanium dioxide
Questions Or Comments?
1-800-426-9391
Principal Display Panel
CVSHealth®
Compare to the active
ingredient in ex•lax®*
Regular Strength
Laxative
SENNOSIDES USP, 15 mg
Stimulant laxative
• Gentle, dependable
• Constipation relief
30 TABLETS
Actual Size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by
GSK Consumer Healthcare S.A., owner of the registered
trademark ex•lax®. 50844 REV0419A34701
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2020 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
V-14236
CVS®Quality
Money Back Guarantee
When Using This Product
do not use for a period longer than 1 week.
Stop Use and Ask A Doctor If
rectal bleeding or failure to have a bowel movement occur after use of a laxative. These may be signs of a serious condition.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet)
Sennosides USP, 15 mg
Ask A Doctor Before Use If You Have
noticed a sudden change in bowel habits that persists over a period of 2 weeks.
Ask A Doctor Or Pharmacist Before Use If You Are
taking any other drug. Laxatives may affect how other drugs work. Take this product 2 or more hours before or after other drugs.
Structured Label Content
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 6 to 12 hours
Purpose
Stimulant laxative
Directions
- swallow tablet(s) with a glass of water
- swallow tablet(s) whole; do not crush, break, or chew
| adults and children 12 years and over | 2 tablets once or twice daily |
| children 6 to under 12 years | 1 tablet once or twice daily |
| children under 6 years | ask a doctor |
Do Not Use (Do not use)
laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor.
Other Information (Other information)
- each tablet contains: calcium 45 mg, magnesium 5 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive Ingredients (Inactive ingredients)
acacia, calcium carbonate, carnauba wax, corn starch, dibasic calcium phosphate dihydrate, iron oxide black, iron oxide red, iron oxide yellow, magnesium stearate, methylparaben, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, propylene glycol, propylparaben, shellac glaze, silicon dioxide, sodium benzoate, sodium lauryl sulfate, sucrose, talc, titanium dioxide
Questions Or Comments? (Questions or comments?)
1-800-426-9391
Principal Display Panel (Principal display panel)
CVSHealth®
Compare to the active
ingredient in ex•lax®*
Regular Strength
Laxative
SENNOSIDES USP, 15 mg
Stimulant laxative
• Gentle, dependable
• Constipation relief
30 TABLETS
Actual Size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by
GSK Consumer Healthcare S.A., owner of the registered
trademark ex•lax®. 50844 REV0419A34701
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2020 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
V-14236
CVS®Quality
Money Back Guarantee
When Using This Product (When using this product)
do not use for a period longer than 1 week.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
rectal bleeding or failure to have a bowel movement occur after use of a laxative. These may be signs of a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Sennosides USP, 15 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
noticed a sudden change in bowel habits that persists over a period of 2 weeks.
Ask A Doctor Or Pharmacist Before Use If You Are (Ask a doctor or pharmacist before use if you are)
taking any other drug. Laxatives may affect how other drugs work. Take this product 2 or more hours before or after other drugs.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:15.506298 · Updated: 2026-03-14T23:08:56.104871