Drug Facts
376eb27a-63bf-87d6-e063-6294a90a9314
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
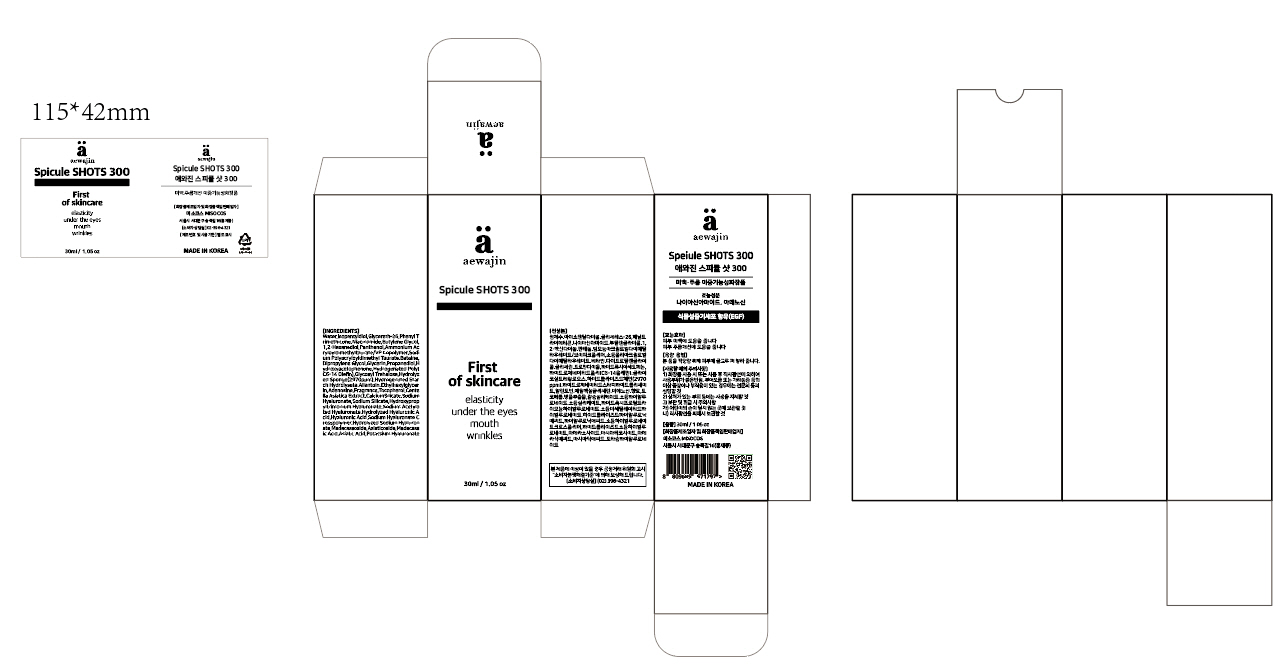
Composition & Product
Identifiers & Packaging
Description
Niacinamide Adenosine
Medication Information
Warnings and Precautions
■ if following abnormal symptoms occurs after use , stop use and consult with a skin specialist
red specks, swelling, itching
■ don’t use on the part where there is injury, eczema, or dermatitis
Keep out of reach of children
■ if swallowed, get medical help or contact a person control center immediately
Indications and Usage
take a suitable amount on the skin
Dosage and Administration
for external use only
Description
Niacinamide Adenosine
Section 50565-1
keep out of reach of the children
Section 51727-6
Water
Isopentyldiol
Glycereth-26
Phenyl Trimethicone
Butylene Glycol
1,2-Hexanediol
Panthenol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Sodium Polyacryloyldimethyl Taurate
Betaine
Dipropylene Glycol
Glycerin
Propanediol
Hydroxyacetophenone
Hydrogenated Poly(C6-14 Olefin)
Glycosyl Trehalose
Hydrolyzed Sponge
Hydrogenated Starch Hydrolysate
Allantoin
Ethylhexylglycerin
Fragrance
Tocopherol
Centella Asiatica Extract
Calcium Silicate
Sodium Hyaluronate
Sodium Silicate
Hydroxypropyltrimonium Hyaluronate
Sodium Acetylated Hyaluronate
Hydrolyzed Hyaluronic Acid
Hyaluronic Acid
Sodium Hyaluronate Crosspolymer
Hydrolyzed Sodium Hyaluronate
Madecassoside
Asiaticoside
Madecassic Acid
Asiatic Acid
Potassium Hyaluronate
Section 51945-4
Section 55105-1
skin protectant
Section 55106-9
Niacinamide
Adenosine
Structured Label Content
Indications and Usage (34067-9)
take a suitable amount on the skin
Dosage and Administration (34068-7)
for external use only
Warnings and Precautions (34071-1)
■ if following abnormal symptoms occurs after use , stop use and consult with a skin specialist
red specks, swelling, itching
■ don’t use on the part where there is injury, eczema, or dermatitis
Keep out of reach of children
■ if swallowed, get medical help or contact a person control center immediately
Section 50565-1 (50565-1)
keep out of reach of the children
Section 51727-6 (51727-6)
Water
Isopentyldiol
Glycereth-26
Phenyl Trimethicone
Butylene Glycol
1,2-Hexanediol
Panthenol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Sodium Polyacryloyldimethyl Taurate
Betaine
Dipropylene Glycol
Glycerin
Propanediol
Hydroxyacetophenone
Hydrogenated Poly(C6-14 Olefin)
Glycosyl Trehalose
Hydrolyzed Sponge
Hydrogenated Starch Hydrolysate
Allantoin
Ethylhexylglycerin
Fragrance
Tocopherol
Centella Asiatica Extract
Calcium Silicate
Sodium Hyaluronate
Sodium Silicate
Hydroxypropyltrimonium Hyaluronate
Sodium Acetylated Hyaluronate
Hydrolyzed Hyaluronic Acid
Hyaluronic Acid
Sodium Hyaluronate Crosspolymer
Hydrolyzed Sodium Hyaluronate
Madecassoside
Asiaticoside
Madecassic Acid
Asiatic Acid
Potassium Hyaluronate
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
skin protectant
Section 55106-9 (55106-9)
Niacinamide
Adenosine
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:18.924764 · Updated: 2026-03-14T23:09:01.566522