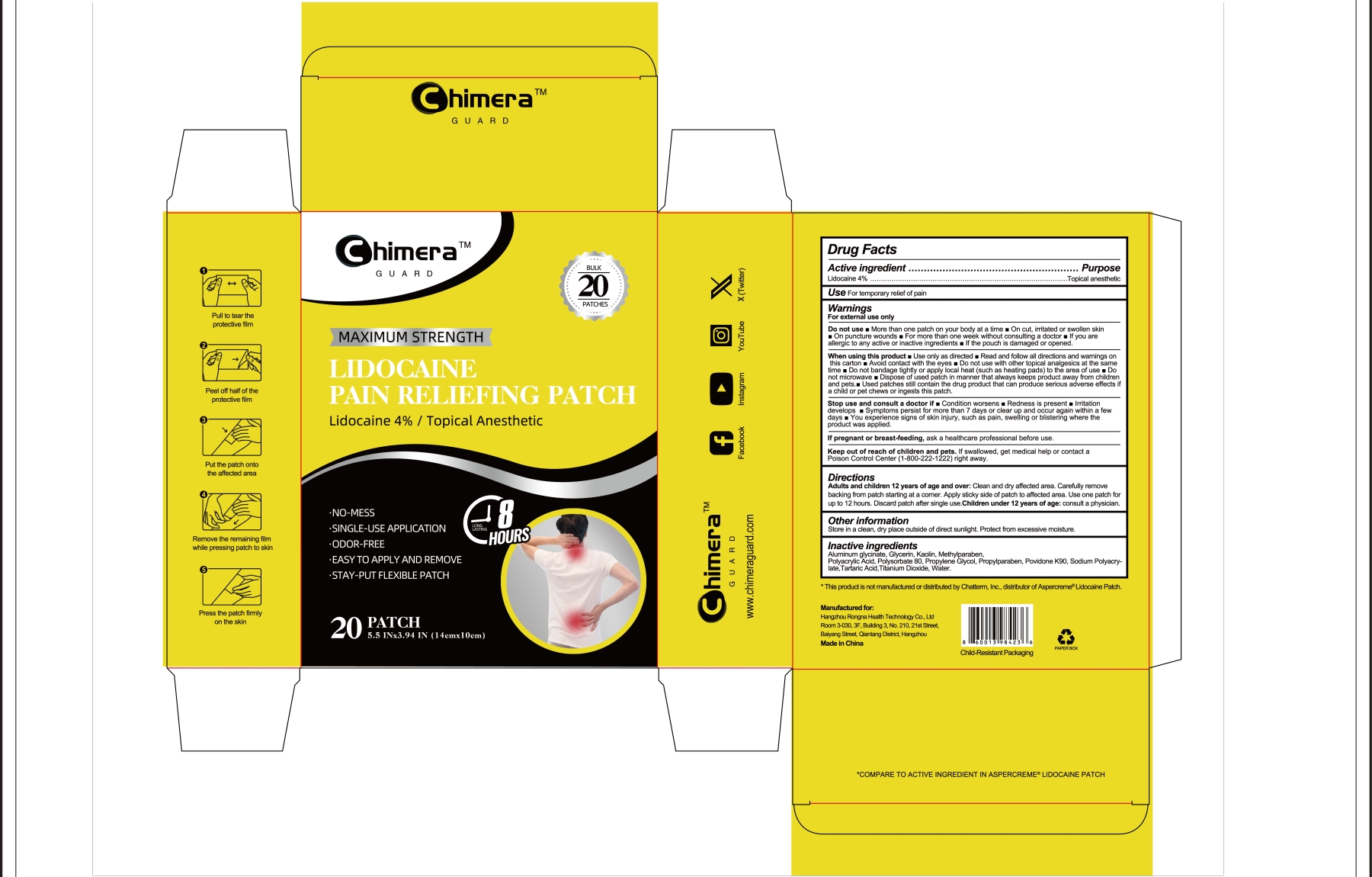
Hangzhou Rongna Health Technology Co., Ltd
3758d625-010f-7441-e063-6394a90a066d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4% ...... Purpose: Topical Anesthetic
Purpose
Topical Anesthetic
Medication Information
Purpose
Topical Anesthetic
Description
Lidocaine 4% ...... Purpose: Topical Anesthetic
Uses
For temporary relief of pain
Warnings
For external use only
Directions
Adult and Children 12 years of age and over :
- Clean and dry affected area
- Carefully remove backing from patch starting at a corner
- Apply sticky side of patch to affected area.
- Use one patch forup to 12 hours
- Discard patch after single use
Children under 12 years of age : Consult a physician
Do Not Use
- More than 1 patch on your body at a time
- On cut,irritated orswollen skin
- On puncture wounds
- For more than oneweek without consulting a doctor
- if you are allergic to any active or inactive ingredients
- If the pouch is damaged or opened.
85761 159 20
85761 159 25
Stop Use ... If
- Condition worsens
- Redness is present
- Irritationn develops
- Symptoms persist for more than 7 days or clear up and occur again within a few days
- You experience signs of skin injury,suchaspain,swelling or blistering where the
product was applied
Active Ingredient
Lidocaine 4% ...... Purpose: Topical Anesthetic
Other Information
Store ina clean,dry place outside of direct sunlight .Protect from excessive moisture.
... Ask A Doctor If
Condition worsens
Redness is present
Irritationn develops
Symptoms persist for more than 7 days or clear up and occur again within a few days
You experience signs of skin injury,suchaspain,swelling or blistering where the product was applied
Inactive Ingredients
Aluminum glycinate, Glycerin, Kaolin, Methylparaben, Polyacrylic Acid,Polysorbate 80, Propylene Glycol, , Propylparaben, Povidone K90, Sodium Polyacrylate, Tartaric Acid, Titanium Dioxide, Water
Questions Or Comments
www.chimeraguard.com
When Using This Product
- Use only as directed
- Read and follow all directions and warnings on this carton
- Avoid contact with the eyes
- Do not use with other topical analgesics at the same time
- Do not bandage tightly or apply local heat(such as heating pads) to the area of use
- Do not microwave
- Dispose of used patch in manner that always keeps product away from children and pets.
- Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
If Pregnant Or Breast Feeding
ask a healthcare professional before use
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center(1-800-222-1222) right away.
Structured Label Content
Uses
For temporary relief of pain
Purpose
Topical Anesthetic
Warnings
For external use only
Directions
Adult and Children 12 years of age and over :
- Clean and dry affected area
- Carefully remove backing from patch starting at a corner
- Apply sticky side of patch to affected area.
- Use one patch forup to 12 hours
- Discard patch after single use
Children under 12 years of age : Consult a physician
Do Not Use (Do not use)
- More than 1 patch on your body at a time
- On cut,irritated orswollen skin
- On puncture wounds
- For more than oneweek without consulting a doctor
- if you are allergic to any active or inactive ingredients
- If the pouch is damaged or opened.
85761 159 20 (85761-159-20)
85761 159 25 (85761-159-25)
Stop Use ... If (Stop use ... if)
- Condition worsens
- Redness is present
- Irritationn develops
- Symptoms persist for more than 7 days or clear up and occur again within a few days
- You experience signs of skin injury,suchaspain,swelling or blistering where the
product was applied
Active Ingredient
Lidocaine 4% ...... Purpose: Topical Anesthetic
Other Information (Other information)
Store ina clean,dry place outside of direct sunlight .Protect from excessive moisture.
... Ask A Doctor If (... ask a doctor if)
Condition worsens
Redness is present
Irritationn develops
Symptoms persist for more than 7 days or clear up and occur again within a few days
You experience signs of skin injury,suchaspain,swelling or blistering where the product was applied
Inactive Ingredients
Aluminum glycinate, Glycerin, Kaolin, Methylparaben, Polyacrylic Acid,Polysorbate 80, Propylene Glycol, , Propylparaben, Povidone K90, Sodium Polyacrylate, Tartaric Acid, Titanium Dioxide, Water
Questions Or Comments (Questions or Comments)
www.chimeraguard.com
When Using This Product (When using this product)
- Use only as directed
- Read and follow all directions and warnings on this carton
- Avoid contact with the eyes
- Do not use with other topical analgesics at the same time
- Do not bandage tightly or apply local heat(such as heating pads) to the area of use
- Do not microwave
- Dispose of used patch in manner that always keeps product away from children and pets.
- Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
ask a healthcare professional before use
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center(1-800-222-1222) right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:38.792492 · Updated: 2026-03-14T23:09:31.593516