Dermoplast Post Partum Spray
374dced3-6527-b458-e063-6394a90a911e
34390-5
HUMAN OTC DRUG LABEL
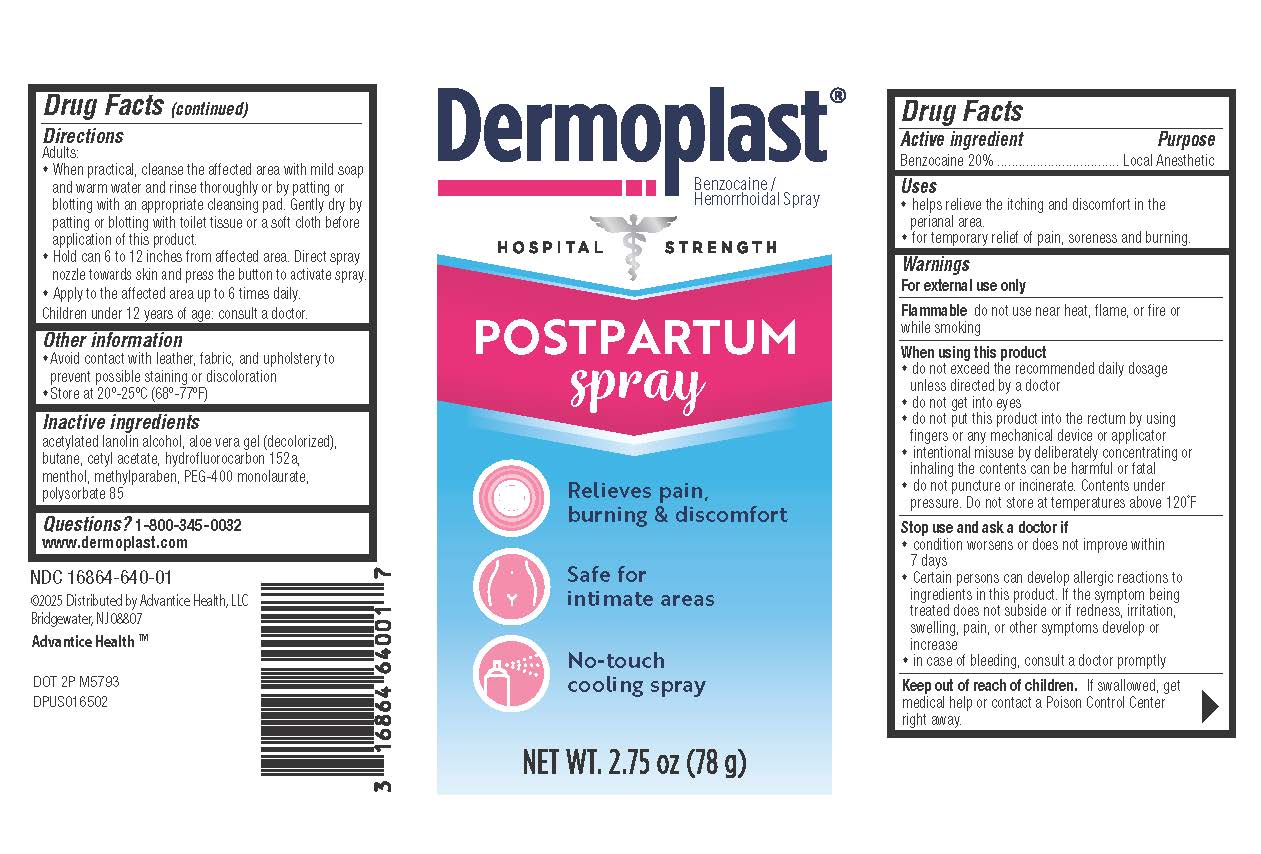
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ingredient: Benzocaine 20% Purpose: Local Anesthetic
Medication Information
Warnings and Precautions
For external use only
Flammable: do not use near heat, flame, or fire or while smoking
When using this product:
• Do not exceed the recommended daily dosage unless directed by a doctor.
• Do not get into eyes.
• Do not put this product into the rectum by using fingers or any mechanical device or applicator.
• Intentional misuse by deliberately concentrating or inhaling the contents can be harmful or fatal.
• Do not puncture or incinerate. Contents under pressure.
• Do not store at temperatures above 120°F.
Stop use and ask a doctor if:
• Condition worsens or does not improve within 7 days.
• Certain persons can develop allergic reactions to ingredients in this product. If the symptom being treated does not subside or if redness, irritation, swelling, pain, or other symptoms develop or increase.
• In case of bleeding, consult a doctor promptly
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Indications and Usage
• helps relieve the itching and discomfort in the perianal area. • for temporary relief of pain, soreness and burning.
Dosage and Administration
• When practical, cleanse the affected area with mild soap and warm water and rinse thoroughly or by patting and blotting with an appropriate cleansing pad.
• Gently dry by patting or blotting with toilet tissue or a soft cloth before application of this product.
• Hold can 6 to 12 inches away from affected area. Direct the spray nozzle towards the skin and press the button to activate spray.
• Apply to the affected area up to 6 times daily.
• Children under 12 years of age: consult a doctor.
Description
Ingredient: Benzocaine 20% Purpose: Local Anesthetic
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
acetylated lanolin alcohol, aloe vera gel (decolorized), butane, cetyl acetate, hydrofluorocarbon 152a, methylparaben USP, PEG-400 monolaurate, menthol, polysorbate 85
Section 51945-4
Section 55105-1
Local Anesthetic
Section 55106-9
Ingredient: Benzocaine 20% Purpose: Local Anesthetic
Structured Label Content
Indications and Usage (34067-9)
• helps relieve the itching and discomfort in the perianal area. • for temporary relief of pain, soreness and burning.
Dosage and Administration (34068-7)
• When practical, cleanse the affected area with mild soap and warm water and rinse thoroughly or by patting and blotting with an appropriate cleansing pad.
• Gently dry by patting or blotting with toilet tissue or a soft cloth before application of this product.
• Hold can 6 to 12 inches away from affected area. Direct the spray nozzle towards the skin and press the button to activate spray.
• Apply to the affected area up to 6 times daily.
• Children under 12 years of age: consult a doctor.
Warnings and Precautions (34071-1)
For external use only
Flammable: do not use near heat, flame, or fire or while smoking
When using this product:
• Do not exceed the recommended daily dosage unless directed by a doctor.
• Do not get into eyes.
• Do not put this product into the rectum by using fingers or any mechanical device or applicator.
• Intentional misuse by deliberately concentrating or inhaling the contents can be harmful or fatal.
• Do not puncture or incinerate. Contents under pressure.
• Do not store at temperatures above 120°F.
Stop use and ask a doctor if:
• Condition worsens or does not improve within 7 days.
• Certain persons can develop allergic reactions to ingredients in this product. If the symptom being treated does not subside or if redness, irritation, swelling, pain, or other symptoms develop or increase.
• In case of bleeding, consult a doctor promptly
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
acetylated lanolin alcohol, aloe vera gel (decolorized), butane, cetyl acetate, hydrofluorocarbon 152a, methylparaben USP, PEG-400 monolaurate, menthol, polysorbate 85
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Local Anesthetic
Section 55106-9 (55106-9)
Ingredient: Benzocaine 20% Purpose: Local Anesthetic
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:54.811020 · Updated: 2026-03-14T23:13:23.763125