Dentalux
37011052-16ef-4ae4-aade-a86f9fa9cdac
34390-5
HUMAN OTC DRUG LABEL
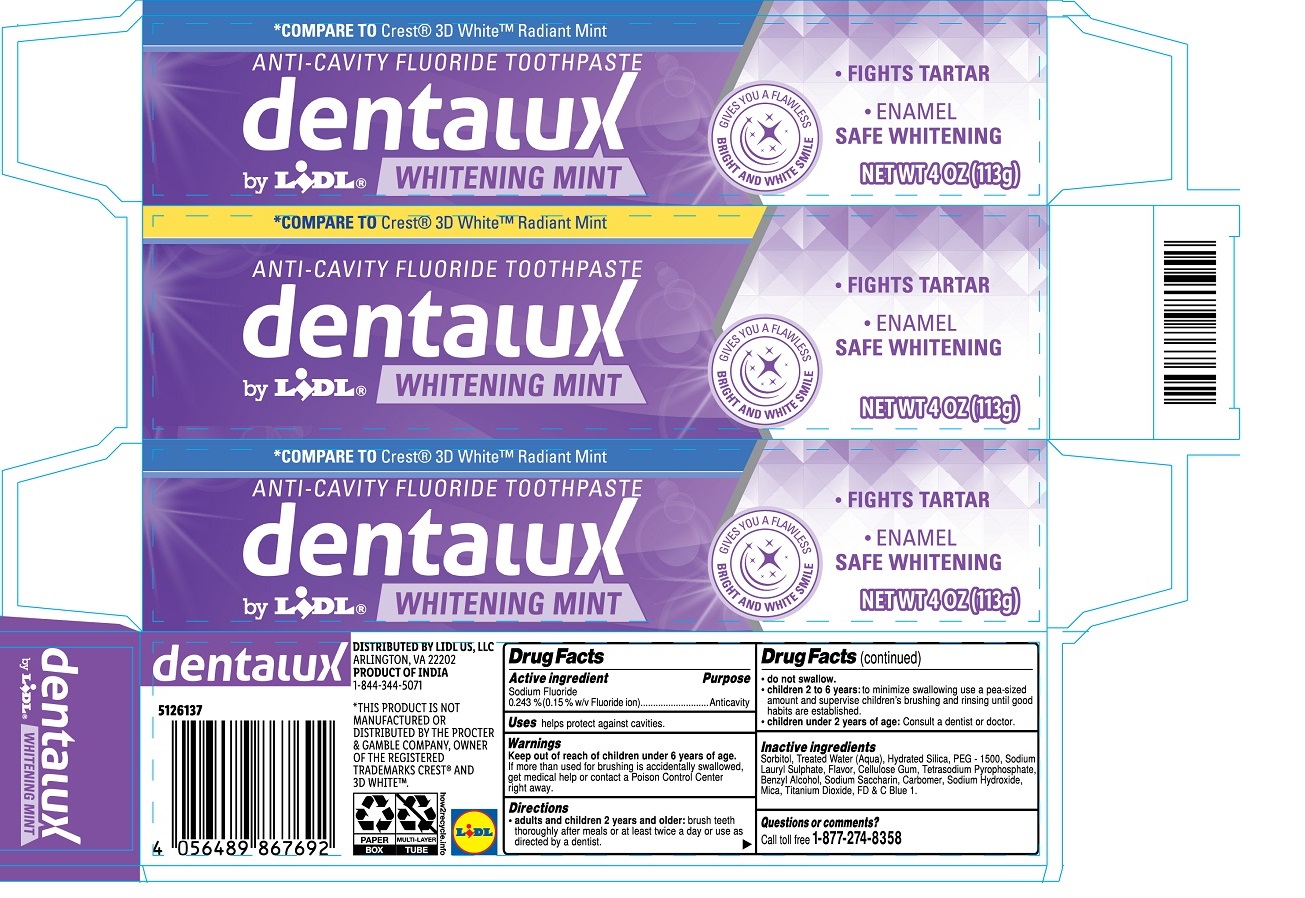
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sodium Fluoride 0.243 % (0.15 % w/v Fluoride ion)
Purpose
Anticavity
Medication Information
Purpose
Anticavity
Description
Sodium Fluoride 0.243 % (0.15 % w/v Fluoride ion)
Uses
helps protect against cavities.
Section 42229-5
*COMPARE TO Crest® 3D White™ Radiant Mint
• FIGHTS TARTAR
• ENAMEL SAFE WHITENING
GIVES YOU A FLAWLESS BRIGHT AND WHITE SMILE
DISTRIBUTED BY LIDL US, LLC
ARLINGTON, VA 22202
PRODUCT OF INDIA
1-844-344-5071
*THIS PRODUCT IS NOT MANUFACTURED OR DISTRIBUTED BY THE PROCTER & GAMBLE COMPANY, OWNER OF THE REGISTERED TRADEMARKS CREST® AND 3D WHITE™.
Section 50565-1
Keep out of reach of children under 6 years of age. If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Warnings
Packaging
Directions
• adults and children 2 years and older: brush teeth thoroughly after meals or at least twice a day or use as directed by a dentist.
• do not swallow.
• children 2 to 6 years: to minimize swallowing use a pea-sized amount and supervise children's brushing and rinsing until good habits are established.
• children under 2 years of age: Consult a dentist or doctor.
Drug Facts
Active Ingredient
Sodium Fluoride
0.243 % (0.15 % w/v Fluoride ion)
Inactive Ingredients
Sorbitol, Treated Water (Aqua), Hydrated Silica, PEG-1500, Sodium Lauryl Sulphate, Flavor, Cellulose Gum, Tetrasodium Pyrophosphate, Benzyl Alcohol, Sodium Saccharin, Carbomer, Sodium Hydroxide, Mica, Titanium Dioxide, FD&C Blue 1.
Questions Or Comments?
Call toll free 1-877-274-8358
Structured Label Content
Uses
helps protect against cavities.
Section 42229-5 (42229-5)
*COMPARE TO Crest® 3D White™ Radiant Mint
• FIGHTS TARTAR
• ENAMEL SAFE WHITENING
GIVES YOU A FLAWLESS BRIGHT AND WHITE SMILE
DISTRIBUTED BY LIDL US, LLC
ARLINGTON, VA 22202
PRODUCT OF INDIA
1-844-344-5071
*THIS PRODUCT IS NOT MANUFACTURED OR DISTRIBUTED BY THE PROCTER & GAMBLE COMPANY, OWNER OF THE REGISTERED TRADEMARKS CREST® AND 3D WHITE™.
Section 50565-1 (50565-1)
Keep out of reach of children under 6 years of age. If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Anticavity
Warnings
Packaging
Directions
• adults and children 2 years and older: brush teeth thoroughly after meals or at least twice a day or use as directed by a dentist.
• do not swallow.
• children 2 to 6 years: to minimize swallowing use a pea-sized amount and supervise children's brushing and rinsing until good habits are established.
• children under 2 years of age: Consult a dentist or doctor.
Drug Facts
Active Ingredient (Active ingredient)
Sodium Fluoride
0.243 % (0.15 % w/v Fluoride ion)
Inactive Ingredients (Inactive ingredients)
Sorbitol, Treated Water (Aqua), Hydrated Silica, PEG-1500, Sodium Lauryl Sulphate, Flavor, Cellulose Gum, Tetrasodium Pyrophosphate, Benzyl Alcohol, Sodium Saccharin, Carbomer, Sodium Hydroxide, Mica, Titanium Dioxide, FD&C Blue 1.
Questions Or Comments? (Questions or comments?)
Call toll free 1-877-274-8358
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:28.331924 · Updated: 2026-03-14T23:05:14.204136