These Highlights Do Not Include All The Information Needed To Use Wynzora Cream Safely And Effectively. See Full Prescribing Information For Wynzora Cream.
36c5c818-0d40-445e-a3c0-0f46723a25e2
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WYNZORA Cream is indicated for the topical treatment of plaque psoriasis in patients 18 years of age and older.
Indications and Usage
WYNZORA Cream is indicated for the topical treatment of plaque psoriasis in patients 18 years of age and older.
Dosage and Administration
Apply WYNZORA Cream to affected areas once daily for up to 8 weeks. Rub in gently to ensure that the plaques are saturated with the cream. Do not use more than 100 g per week. Discontinue therapy when control is achieved. Do not use: with occlusive dressings unless directed by a healthcare provider on the face, groin, or axillae, or if skin atrophy is present at the treatment site WYNZORA Cream is not for oral, ophthalmic, or intravaginal use.
Warnings and Precautions
Hypercalcemia and Hypercalciuria : Hypercalcemia and hypercalciuria have been observed with use of topical calcipotriene. If either occurs, discontinue until parameters of calcium metabolism normalize. ( 5.1 ) Effects on Endocrine System: Can cause reversible hypothalamicpituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area, or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and young age. Modify use should HPA axis suppression develop. ( 5.2 ) Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. ( 5.5 )
Contraindications
None.
Adverse Reactions
The most common adverse reactions reported by more than 1% of subjects treated with WYNZORA Cream were upper respiratory infection, headache, and application site irritation. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact MC2 Therapeutics Ltd at 1-800-644-8240 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
WYNZORA ® (calcipotriene and betamethasone dipropionate) Cream, 0.005%/0.064%, is a white cream. Each gram of cream contains 50 mcg of anhydrous calcipotriene and 0.644 mg of betamethasone dipropionate. It is available as 60 g tubes individually packaged (NDC 73499-001-01).
How Supplied
WYNZORA ® (calcipotriene and betamethasone dipropionate) Cream, 0.005%/0.064%, is a white cream. Each gram of cream contains 50 mcg of anhydrous calcipotriene and 0.644 mg of betamethasone dipropionate. It is available as 60 g tubes individually packaged (NDC 73499-001-01).
Medication Information
Warnings and Precautions
Hypercalcemia and Hypercalciuria : Hypercalcemia and hypercalciuria have been observed with use of topical calcipotriene. If either occurs, discontinue until parameters of calcium metabolism normalize. ( 5.1 ) Effects on Endocrine System: Can cause reversible hypothalamicpituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area, or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and young age. Modify use should HPA axis suppression develop. ( 5.2 ) Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. ( 5.5 )
Indications and Usage
WYNZORA Cream is indicated for the topical treatment of plaque psoriasis in patients 18 years of age and older.
Dosage and Administration
Apply WYNZORA Cream to affected areas once daily for up to 8 weeks. Rub in gently to ensure that the plaques are saturated with the cream. Do not use more than 100 g per week. Discontinue therapy when control is achieved. Do not use: with occlusive dressings unless directed by a healthcare provider on the face, groin, or axillae, or if skin atrophy is present at the treatment site WYNZORA Cream is not for oral, ophthalmic, or intravaginal use.
Contraindications
None.
Adverse Reactions
The most common adverse reactions reported by more than 1% of subjects treated with WYNZORA Cream were upper respiratory infection, headache, and application site irritation. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact MC2 Therapeutics Ltd at 1-800-644-8240 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
WYNZORA ® (calcipotriene and betamethasone dipropionate) Cream, 0.005%/0.064%, is a white cream. Each gram of cream contains 50 mcg of anhydrous calcipotriene and 0.644 mg of betamethasone dipropionate. It is available as 60 g tubes individually packaged (NDC 73499-001-01).
How Supplied
WYNZORA ® (calcipotriene and betamethasone dipropionate) Cream, 0.005%/0.064%, is a white cream. Each gram of cream contains 50 mcg of anhydrous calcipotriene and 0.644 mg of betamethasone dipropionate. It is available as 60 g tubes individually packaged (NDC 73499-001-01).
Description
WYNZORA Cream is indicated for the topical treatment of plaque psoriasis in patients 18 years of age and older.
Section 42229-5
WYNZORA Cream can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of treatment. Factors that predispose a patient to HPA axis suppression include the use of high-potency steroids, large treatment surface areas, prolonged use, use of occlusive dressings, altered skin barrier, liver failure, and young age.
Evaluation for HPA axis suppression may be done by using the adrenocorticotropic hormone (ACTH) stimulation test. If HPA axis suppression is documented, gradually withdraw WYNZORA Cream, reduce the frequency of application, or substitute with a less potent corticosteroid.
The following trial evaluated the effects of WYNZORA Cream on HPA axis suppression:
HPA axis suppression was evaluated in adult subjects (N=27) with extensive psoriasis (including scalp). Adrenal suppression was seen in 6 out of 26 subjects (23%) after 4 weeks of treatment, and in 3 out of 25 subjects (12%) after 8 weeks of treatment [see Clinical Pharmacology (12.2)].
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 11/2023 | |
|
PATIENT INFORMATION WYNZORA® (win-ZOR-uh) (calcipotriene and betamethasone dipropionate) Cream |
||
| Important information: WYNZORA® Cream is for use on the skin only (topical use only). Do not get WYNZORA® Cream near or in your mouth, eyes, or vagina. | ||
| There are other medicines that contain the same medicine that is in WYNZORA® Cream and are used to treat plaque psoriasis. Do not use other products containing calcipotriene or a corticosteroid medicine with WYNZORA® Cream without talking to your healthcare provider first. | ||
|
What is WYNZORA® Cream?
WYNZORA® Cream is a prescription medicine used on the skin (topical) to treat plaque psoriasis in people 18 years of age and older. It is not known if WYNZORA® Cream is safe and effective in children under 18 years of age. |
||
Before using WYNZORA® Cream, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I use WYNZORA® Cream?
|
||
|
What are the possible side effects of WYNZORA® Cream? WYNZORA® Cream may cause serious side effects, including:
|
||
|
|
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1- 800-FDA-1088. |
||
How should I store WYNZORA® Cream?
|
||
|
General information about WYNZORA® Cream
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use WYNZORA® Cream for a condition for which it was not prescribed. Do not give WYNZORA® Cream to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about WYNZORA® Cream that is written for health professionals. |
||
|
What are the ingredients in WYNZORA® Cream?
Active ingredients: calcipotriene and betamethasone dipropionate Inactive ingredients: isopropyl myristate, mineral oil, medium-chain triglycerides, isopropyl alcohol, polyoxyl lauryl ether, poloxamer (407), polyoxyl 40 hydrogenated castor oil, carbomer interpolymer (type A), butylated hydroxyanisole, trolamine, dibasic sodium phosphate, heptahydrate, monobasic sodium phosphate, monohydrate, alpha-tocopherol and purified water. Manufactured for: MC2 Therapeutics, Ltd. 1A Guildford Business Park, Guildford, GU2 8XG, United Kingdom. For more information, go to www.wynzora.com or call 1-800-644-8240 |
Section 44425-7
Store between 20°C-25°C (68°F-77°F); excursions permitted between 15°C-30°C (59°F-86°F). [See USP controlled room temperature.]
Do not freeze. Protect from light and excessive heat.
Unused product should be discarded six months after the tube has been opened.
11 Description
WYNZORA (calcipotriene and betamethasone dipropionate) Cream contains anhydrous calcipotriene and betamethasone dipropionate intended for topical use.
Calcipotriene is a synthetic vitamin D3 analog.
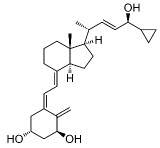
Chemically, calcipotriene is (5Z,7E,22E,24S)-24-Cyclopropyl-9,10-secochola-5,7,10(19),22 tetraene-1α,3β,24-triol, with the empirical formula C27H40O3, a molecular weight of 412.6, and the following structural formula:
Calcipotriene is a white or almost white powder. It is insoluble in water, freely soluble in ethanol and slightly soluble in methylene chloride.
Betamethasone dipropionate is a synthetic corticosteroid.
Betamethasone dipropionate has the chemical name Pregna-1,4-diene-3,20-dione,9-fluoro-11-hydroxy-16-methyl-17,21-bis(1-oxypropoxy)-,(11β,16β), with the empirical formula C28H37FO7, a molecular weight of 504.6, and the following structural formula:
Betamethasone dipropionate is a white to almost white crystalline powder. It is practically insoluble in water, freely soluble in acetone and in methylene chloride, sparingly soluble in alcohol.
Each gram of WYNZORA Cream contains 50 mcg of calcipotriene and 0.644 mg of betamethasone dipropionate (equivalent to 0.5 mg of betamethasone). WYNZORA Cream also contains the following inactive ingredients: isopropyl myristate, mineral oil, medium-chain triglycerides, isopropyl alcohol, polyoxyl lauryl ether, poloxamer (407), polyoxyl 40 hydrogenated castor oil, carbomer interpolymer (type A), butylated hydroxyanisole, trolamine, dibasic sodium phosphate, heptahydrate, monobasic sodium phosphate, monohydrate, alpha-tocopherol and purified water.
8.4 Pediatric Use
Safety and effectiveness of the use of WYNZORA Cream in adolescents and pediatric patients under the age of 18 years have not been established.
Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of systemic toxicity when treated with topical corticosteroids. Pediatric patients are, therefore, also at greater risk of HPA axis suppression and adrenal insufficiency with the use of topical corticosteroids including WYNZORA Cream [see Clinical Pharmacology (12.2)].
Systemic toxicities such as Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients, especially those with prolonged exposure to large doses of high potency topical corticosteroids. Local adverse reactions including striae have also been reported with use of topical corticosteroids in pediatric patients.
8.5 Geriatric Use
The trial included 66 subjects ≥ 65 years of age treated with WYNZORA Cream.
No overall differences in safety or effectiveness of WYNZORA Cream were observed between these subjects and younger subjects. All other reported clinical experience has not identified any differences in response between elderly and younger patients. However, greater sensitivity of some older individuals cannot be ruled out.
14 Clinical Studies
The safety and efficacy of WYNZORA Cream were evaluated in a randomized, multicenter, vehicle and active-comparator controlled, clinical trial (NCT03308799) in adult subjects with mild to moderate plaque psoriasis. In the trial, 794 subjects were randomized to 1 of 3 treatment groups: WYNZORA Cream, vehicle cream, or calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064%. The majority of subjects in the trial (81.7%) had disease of moderate severity at baseline, while 18.3% of subjects had disease of mild severity. Disease severity was determined by a 5-grade Physician's Global Assessment (PGA) scale.
The primary efficacy endpoint was the proportion of subjects with treatment success at Week 8. Treatment success was defined as at least a 2-grade improvement from baseline in the PGA score and an PGA score equating to "clear" or "almost clear". Other evaluated outcomes included reduction in itch as defined by at least a 4-point improvement in the 11-point peak pruritus numeric rating scale (NRS) from baseline to Week 4. Table 2 presents the primary efficacy results.
| WYNZORA Cream (N=342) |
Vehicle Cream (N=115) |
|
|---|---|---|
| PGA of Clear or Almost Clear And ≥2-grade Improvement | 37.4% | 3.7% |
| Difference from Vehicle (95% CI) |
33.7% (27.4%, 40.0%) |
WYNZORA Cream was non-inferior to calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064% for the primary endpoint of treatment success at Week 8 [Difference (95% CI): 14.6% (7.6%, 21.6%)].
Among subjects who had at least a peak pruritus NRS score of 4 at baseline, there was a higher percentage of subjects that achieved at least a 4-point improvement from baseline on the peak pruritus NRS score at Week 4 in the WYNZORA Cream group compared to the vehicle cream group (60.3% vs. 21.4%).
4 Contraindications
None.
6 Adverse Reactions
The most common adverse reactions reported by more than 1% of subjects treated with WYNZORA Cream were upper respiratory infection, headache, and application site irritation. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact MC2 Therapeutics Ltd at 1-800-644-8240 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
1 Indications and Usage
WYNZORA Cream is indicated for the topical treatment of plaque psoriasis in patients 18 years of age and older.
12.1 Mechanism of Action
WYNZORA Cream combines the pharmacological effects of calcipotriene as a synthetic vitamin D3 analog and betamethasone dipropionate as a synthetic corticosteroid. However, while their pharmacologic and clinical effects are known, the exact mechanisms of their actions in plaque psoriasis are unknown.
5 Warnings and Precautions
- Hypercalcemia and Hypercalciuria: Hypercalcemia and hypercalciuria have been observed with use of topical calcipotriene. If either occurs, discontinue until parameters of calcium metabolism normalize. (5.1)
- Effects on Endocrine System: Can cause reversible hypothalamicpituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area, or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and young age. Modify use should HPA axis suppression develop. (5.2)
- Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. (5.5)
2 Dosage and Administration
Apply WYNZORA Cream to affected areas once daily for up to 8 weeks. Rub in gently to ensure that the plaques are saturated with the cream.
Do not use more than 100 g per week.
Discontinue therapy when control is achieved.
Do not use:
- with occlusive dressings unless directed by a healthcare provider
- on the face, groin, or axillae, or if skin atrophy is present at the treatment site
WYNZORA Cream is not for oral, ophthalmic, or intravaginal use.
3 Dosage Forms and Strengths
Cream: 0.005%/0.064%.
Each gram of WYNZORA Cream contains 50 mcg of calcipotriene and 0.644 mg of betamethasone dipropionate in a white cream.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of topical corticosteroids. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Postmarketing reports for local adverse reactions to topical corticosteroids included: atrophy, striae, telangiectasias, itching, dryness, hypopigmentation, perioral dermatitis, secondary infection, and miliaria.
Ophthalmic adverse reactions of cataracts, glaucoma, and increased intraocular pressure, have been reported during use of topical corticosteroids, including topical betamethasone products.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The rates of adverse reactions given below were reported in a randomized, multicenter, prospective, vehicle and active controlled clinical trial in adult subjects with plaque psoriasis. Subjects applied WYNZORA Cream, calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064% or vehicle once daily for 8 weeks. The mean weekly dose of WYNZORA Cream was 33.8 g. A total of 342 subjects were treated with WYNZORA Cream, 337 with calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064% and 115 with vehicle. The majority of subjects were White (87%) and male (62%). Approximately 72% were non-Hispanic/Latino. The mean age was 52 years and ages ranged from 18 to 89 years.
The most common adverse reactions reported by ≥1% of subjects treated with WYNZORA Cream and more frequently than vehicle are presented in Table 1 below.
| Preferred Term | WYNZORA Cream (N=342) |
Vehicle Cream (N=115) |
|---|---|---|
| Upper Respiratory Infection (URI) Includes nasopharyngitis, upper respiratory tract infection (URTI), and viral URTI
|
7% | 5% |
| Headache | 2% | 0% |
| Application site irritation | 1% | 0% |
5.5 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including WYNZORA Cream, may increase the risks of glaucoma and posterior subcapsular cataract. Cataracts and glaucoma have been reported with the postmarketing use of topical corticosteroid products [see Adverse Reactions (6.2)].
Avoid contact of WYNZORA Cream with eyes. WYNZORA Cream may cause eye irritation. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
WYNZORA® (calcipotriene and betamethasone dipropionate) Cream, 0.005%/0.064%, is a white cream. Each gram of cream contains 50 mcg of anhydrous calcipotriene and 0.644 mg of betamethasone dipropionate. It is available as 60 g tubes individually packaged (NDC 73499-001-01).
5.1 Hypercalcemia and Hypercalciuria
Hypercalcemia and hypercalciuria have been observed with use of topical calcipotriene. If hypercalcemia or hypercalciuria develop, discontinue treatment until parameters of calcium metabolism have normalized.
Principal Display Panel 5 G Tube Carton
NDC 73499-001-03
Wynzora®
(calcipotriene and betamethasone dipropionate)
Cream, 0.005%/0.064%
Net Wt. 10 x 5 g
Samples – Not for sale
For Topical Use Only
Not for ophthalmic, oral or intravaginal use
Principal Display Panel 60 G Tube Carton
Wynzora®
(calcipotriene and betamethasone dipropionate)
Cream, 0.005%/0.064%
NDC 73499-001-01
Rx only
Net Wt. 60 g
For Topical Use Only
Not for ophthalmic, oral or intravaginal use
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
When calcipotriene was applied topically to mice for up to 24 months at dosages of 3, 10, and 30 mcg/kg/day (corresponding to 9, 30, and 90 mcg/m2/day), no significant changes in tumor incidence were observed when compared to control.
A 104-week oral carcinogenicity study was conducted with calcipotriene in male and female rats at doses of 1, 5 and 15 mcg/kg/day (corresponding to dosages of approximately 6, 30, and 90 mcg/m2/day). Beginning week 71, the dosage for high-dose animals of both genders was reduced to 10 mcg/kg/day (corresponding to a dosage of approximately 60 mcg/m2/day). A treatment-related increase in benign C-cell adenomas was observed in the thyroid of females that received 15 mcg/kg/day. A treatment-related increase in benign pheochromocytomas was observed in the adrenal glands of males that received 15 mcg/kg/day. No other statistically significant differences in tumor incidence were observed when compared to control. The relevance of these findings to patients is unknown.
When betamethasone dipropionate was applied topically to CD-1 mice for up to 24 months at dosages approximating 1.3, 4.2, and 8.5 mcg/kg/day in females, and 1.3, 4.2, and 12.9 mcg/kg/day in males (corresponding to dosages of up to approximately 26 mcg/m2/day and 39 mcg/m2/day, in females and males, respectively), no significant changes in tumor incidence were observed when compared to control.
When betamethasone dipropionate was administered via oral gavage to male and female Sprague Dawley rats for up to 24 months at dosages of 20, 60, and 200 mcg/kg/day (corresponding to dosages of approximately 120, 360, and 1200 mcg/m2/day), no significant changes in tumor incidence were observed when compared to control.
Calcipotriene did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, the human lymphocyte chromosome aberration test, or the mouse micronucleus test. Betamethasone dipropionate did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, or in the rat micronucleus test.
Studies in rats with oral doses of up to 54 mcg/kg/day (324 mcg/m2/day) of calcipotriene indicated no impairment of fertility or general reproductive performance. Studies in male rats at oral doses of up to 200 mcg/kg/day (1200 mcg/m2/day), and in female rats at oral doses of up to 1000 mcg/kg/day (6000 mcg/m2/day), of betamethasone dipropionate indicated no impairment of fertility.
5.4 Allergic Contact Dermatitis With Topical Calcipotriene
Allergic contact dermatitis has been observed with use of topical calcipotriene. Such an observation should be corroborated with appropriate diagnostic patch testing.
5.3 Allergic Contact Dermatitis With Topical Corticosteroids
Allergic contact dermatitis to a topical corticosteroid is usually diagnosed by observing a failure to heal rather than a clinical exacerbation. Such an observation should be corroborated with appropriate diagnostic patch testing.
Structured Label Content
Section 42229-5 (42229-5)
WYNZORA Cream can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of treatment. Factors that predispose a patient to HPA axis suppression include the use of high-potency steroids, large treatment surface areas, prolonged use, use of occlusive dressings, altered skin barrier, liver failure, and young age.
Evaluation for HPA axis suppression may be done by using the adrenocorticotropic hormone (ACTH) stimulation test. If HPA axis suppression is documented, gradually withdraw WYNZORA Cream, reduce the frequency of application, or substitute with a less potent corticosteroid.
The following trial evaluated the effects of WYNZORA Cream on HPA axis suppression:
HPA axis suppression was evaluated in adult subjects (N=27) with extensive psoriasis (including scalp). Adrenal suppression was seen in 6 out of 26 subjects (23%) after 4 weeks of treatment, and in 3 out of 25 subjects (12%) after 8 weeks of treatment [see Clinical Pharmacology (12.2)].
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 11/2023 | |
|
PATIENT INFORMATION WYNZORA® (win-ZOR-uh) (calcipotriene and betamethasone dipropionate) Cream |
||
| Important information: WYNZORA® Cream is for use on the skin only (topical use only). Do not get WYNZORA® Cream near or in your mouth, eyes, or vagina. | ||
| There are other medicines that contain the same medicine that is in WYNZORA® Cream and are used to treat plaque psoriasis. Do not use other products containing calcipotriene or a corticosteroid medicine with WYNZORA® Cream without talking to your healthcare provider first. | ||
|
What is WYNZORA® Cream?
WYNZORA® Cream is a prescription medicine used on the skin (topical) to treat plaque psoriasis in people 18 years of age and older. It is not known if WYNZORA® Cream is safe and effective in children under 18 years of age. |
||
Before using WYNZORA® Cream, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I use WYNZORA® Cream?
|
||
|
What are the possible side effects of WYNZORA® Cream? WYNZORA® Cream may cause serious side effects, including:
|
||
|
|
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1- 800-FDA-1088. |
||
How should I store WYNZORA® Cream?
|
||
|
General information about WYNZORA® Cream
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use WYNZORA® Cream for a condition for which it was not prescribed. Do not give WYNZORA® Cream to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about WYNZORA® Cream that is written for health professionals. |
||
|
What are the ingredients in WYNZORA® Cream?
Active ingredients: calcipotriene and betamethasone dipropionate Inactive ingredients: isopropyl myristate, mineral oil, medium-chain triglycerides, isopropyl alcohol, polyoxyl lauryl ether, poloxamer (407), polyoxyl 40 hydrogenated castor oil, carbomer interpolymer (type A), butylated hydroxyanisole, trolamine, dibasic sodium phosphate, heptahydrate, monobasic sodium phosphate, monohydrate, alpha-tocopherol and purified water. Manufactured for: MC2 Therapeutics, Ltd. 1A Guildford Business Park, Guildford, GU2 8XG, United Kingdom. For more information, go to www.wynzora.com or call 1-800-644-8240 |
Section 44425-7 (44425-7)
Store between 20°C-25°C (68°F-77°F); excursions permitted between 15°C-30°C (59°F-86°F). [See USP controlled room temperature.]
Do not freeze. Protect from light and excessive heat.
Unused product should be discarded six months after the tube has been opened.
11 Description (11 DESCRIPTION)
WYNZORA (calcipotriene and betamethasone dipropionate) Cream contains anhydrous calcipotriene and betamethasone dipropionate intended for topical use.
Calcipotriene is a synthetic vitamin D3 analog.
Chemically, calcipotriene is (5Z,7E,22E,24S)-24-Cyclopropyl-9,10-secochola-5,7,10(19),22 tetraene-1α,3β,24-triol, with the empirical formula C27H40O3, a molecular weight of 412.6, and the following structural formula:
Calcipotriene is a white or almost white powder. It is insoluble in water, freely soluble in ethanol and slightly soluble in methylene chloride.
Betamethasone dipropionate is a synthetic corticosteroid.
Betamethasone dipropionate has the chemical name Pregna-1,4-diene-3,20-dione,9-fluoro-11-hydroxy-16-methyl-17,21-bis(1-oxypropoxy)-,(11β,16β), with the empirical formula C28H37FO7, a molecular weight of 504.6, and the following structural formula:
Betamethasone dipropionate is a white to almost white crystalline powder. It is practically insoluble in water, freely soluble in acetone and in methylene chloride, sparingly soluble in alcohol.
Each gram of WYNZORA Cream contains 50 mcg of calcipotriene and 0.644 mg of betamethasone dipropionate (equivalent to 0.5 mg of betamethasone). WYNZORA Cream also contains the following inactive ingredients: isopropyl myristate, mineral oil, medium-chain triglycerides, isopropyl alcohol, polyoxyl lauryl ether, poloxamer (407), polyoxyl 40 hydrogenated castor oil, carbomer interpolymer (type A), butylated hydroxyanisole, trolamine, dibasic sodium phosphate, heptahydrate, monobasic sodium phosphate, monohydrate, alpha-tocopherol and purified water.
8.4 Pediatric Use
Safety and effectiveness of the use of WYNZORA Cream in adolescents and pediatric patients under the age of 18 years have not been established.
Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of systemic toxicity when treated with topical corticosteroids. Pediatric patients are, therefore, also at greater risk of HPA axis suppression and adrenal insufficiency with the use of topical corticosteroids including WYNZORA Cream [see Clinical Pharmacology (12.2)].
Systemic toxicities such as Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients, especially those with prolonged exposure to large doses of high potency topical corticosteroids. Local adverse reactions including striae have also been reported with use of topical corticosteroids in pediatric patients.
8.5 Geriatric Use
The trial included 66 subjects ≥ 65 years of age treated with WYNZORA Cream.
No overall differences in safety or effectiveness of WYNZORA Cream were observed between these subjects and younger subjects. All other reported clinical experience has not identified any differences in response between elderly and younger patients. However, greater sensitivity of some older individuals cannot be ruled out.
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of WYNZORA Cream were evaluated in a randomized, multicenter, vehicle and active-comparator controlled, clinical trial (NCT03308799) in adult subjects with mild to moderate plaque psoriasis. In the trial, 794 subjects were randomized to 1 of 3 treatment groups: WYNZORA Cream, vehicle cream, or calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064%. The majority of subjects in the trial (81.7%) had disease of moderate severity at baseline, while 18.3% of subjects had disease of mild severity. Disease severity was determined by a 5-grade Physician's Global Assessment (PGA) scale.
The primary efficacy endpoint was the proportion of subjects with treatment success at Week 8. Treatment success was defined as at least a 2-grade improvement from baseline in the PGA score and an PGA score equating to "clear" or "almost clear". Other evaluated outcomes included reduction in itch as defined by at least a 4-point improvement in the 11-point peak pruritus numeric rating scale (NRS) from baseline to Week 4. Table 2 presents the primary efficacy results.
| WYNZORA Cream (N=342) |
Vehicle Cream (N=115) |
|
|---|---|---|
| PGA of Clear or Almost Clear And ≥2-grade Improvement | 37.4% | 3.7% |
| Difference from Vehicle (95% CI) |
33.7% (27.4%, 40.0%) |
WYNZORA Cream was non-inferior to calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064% for the primary endpoint of treatment success at Week 8 [Difference (95% CI): 14.6% (7.6%, 21.6%)].
Among subjects who had at least a peak pruritus NRS score of 4 at baseline, there was a higher percentage of subjects that achieved at least a 4-point improvement from baseline on the peak pruritus NRS score at Week 4 in the WYNZORA Cream group compared to the vehicle cream group (60.3% vs. 21.4%).
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions reported by more than 1% of subjects treated with WYNZORA Cream were upper respiratory infection, headache, and application site irritation. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact MC2 Therapeutics Ltd at 1-800-644-8240 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
1 Indications and Usage (1 INDICATIONS AND USAGE)
WYNZORA Cream is indicated for the topical treatment of plaque psoriasis in patients 18 years of age and older.
12.1 Mechanism of Action
WYNZORA Cream combines the pharmacological effects of calcipotriene as a synthetic vitamin D3 analog and betamethasone dipropionate as a synthetic corticosteroid. However, while their pharmacologic and clinical effects are known, the exact mechanisms of their actions in plaque psoriasis are unknown.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypercalcemia and Hypercalciuria: Hypercalcemia and hypercalciuria have been observed with use of topical calcipotriene. If either occurs, discontinue until parameters of calcium metabolism normalize. (5.1)
- Effects on Endocrine System: Can cause reversible hypothalamicpituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area, or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and young age. Modify use should HPA axis suppression develop. (5.2)
- Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Apply WYNZORA Cream to affected areas once daily for up to 8 weeks. Rub in gently to ensure that the plaques are saturated with the cream.
Do not use more than 100 g per week.
Discontinue therapy when control is achieved.
Do not use:
- with occlusive dressings unless directed by a healthcare provider
- on the face, groin, or axillae, or if skin atrophy is present at the treatment site
WYNZORA Cream is not for oral, ophthalmic, or intravaginal use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Cream: 0.005%/0.064%.
Each gram of WYNZORA Cream contains 50 mcg of calcipotriene and 0.644 mg of betamethasone dipropionate in a white cream.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of topical corticosteroids. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Postmarketing reports for local adverse reactions to topical corticosteroids included: atrophy, striae, telangiectasias, itching, dryness, hypopigmentation, perioral dermatitis, secondary infection, and miliaria.
Ophthalmic adverse reactions of cataracts, glaucoma, and increased intraocular pressure, have been reported during use of topical corticosteroids, including topical betamethasone products.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The rates of adverse reactions given below were reported in a randomized, multicenter, prospective, vehicle and active controlled clinical trial in adult subjects with plaque psoriasis. Subjects applied WYNZORA Cream, calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064% or vehicle once daily for 8 weeks. The mean weekly dose of WYNZORA Cream was 33.8 g. A total of 342 subjects were treated with WYNZORA Cream, 337 with calcipotriene/betamethasone dipropionate topical suspension, 0.005%/0.064% and 115 with vehicle. The majority of subjects were White (87%) and male (62%). Approximately 72% were non-Hispanic/Latino. The mean age was 52 years and ages ranged from 18 to 89 years.
The most common adverse reactions reported by ≥1% of subjects treated with WYNZORA Cream and more frequently than vehicle are presented in Table 1 below.
| Preferred Term | WYNZORA Cream (N=342) |
Vehicle Cream (N=115) |
|---|---|---|
| Upper Respiratory Infection (URI) Includes nasopharyngitis, upper respiratory tract infection (URTI), and viral URTI
|
7% | 5% |
| Headache | 2% | 0% |
| Application site irritation | 1% | 0% |
5.5 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including WYNZORA Cream, may increase the risks of glaucoma and posterior subcapsular cataract. Cataracts and glaucoma have been reported with the postmarketing use of topical corticosteroid products [see Adverse Reactions (6.2)].
Avoid contact of WYNZORA Cream with eyes. WYNZORA Cream may cause eye irritation. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
WYNZORA® (calcipotriene and betamethasone dipropionate) Cream, 0.005%/0.064%, is a white cream. Each gram of cream contains 50 mcg of anhydrous calcipotriene and 0.644 mg of betamethasone dipropionate. It is available as 60 g tubes individually packaged (NDC 73499-001-01).
5.1 Hypercalcemia and Hypercalciuria
Hypercalcemia and hypercalciuria have been observed with use of topical calcipotriene. If hypercalcemia or hypercalciuria develop, discontinue treatment until parameters of calcium metabolism have normalized.
Principal Display Panel 5 G Tube Carton (PRINCIPAL DISPLAY PANEL - 5 g Tube Carton)
NDC 73499-001-03
Wynzora®
(calcipotriene and betamethasone dipropionate)
Cream, 0.005%/0.064%
Net Wt. 10 x 5 g
Samples – Not for sale
For Topical Use Only
Not for ophthalmic, oral or intravaginal use
Principal Display Panel 60 G Tube Carton (PRINCIPAL DISPLAY PANEL - 60 g Tube Carton)
Wynzora®
(calcipotriene and betamethasone dipropionate)
Cream, 0.005%/0.064%
NDC 73499-001-01
Rx only
Net Wt. 60 g
For Topical Use Only
Not for ophthalmic, oral or intravaginal use
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
When calcipotriene was applied topically to mice for up to 24 months at dosages of 3, 10, and 30 mcg/kg/day (corresponding to 9, 30, and 90 mcg/m2/day), no significant changes in tumor incidence were observed when compared to control.
A 104-week oral carcinogenicity study was conducted with calcipotriene in male and female rats at doses of 1, 5 and 15 mcg/kg/day (corresponding to dosages of approximately 6, 30, and 90 mcg/m2/day). Beginning week 71, the dosage for high-dose animals of both genders was reduced to 10 mcg/kg/day (corresponding to a dosage of approximately 60 mcg/m2/day). A treatment-related increase in benign C-cell adenomas was observed in the thyroid of females that received 15 mcg/kg/day. A treatment-related increase in benign pheochromocytomas was observed in the adrenal glands of males that received 15 mcg/kg/day. No other statistically significant differences in tumor incidence were observed when compared to control. The relevance of these findings to patients is unknown.
When betamethasone dipropionate was applied topically to CD-1 mice for up to 24 months at dosages approximating 1.3, 4.2, and 8.5 mcg/kg/day in females, and 1.3, 4.2, and 12.9 mcg/kg/day in males (corresponding to dosages of up to approximately 26 mcg/m2/day and 39 mcg/m2/day, in females and males, respectively), no significant changes in tumor incidence were observed when compared to control.
When betamethasone dipropionate was administered via oral gavage to male and female Sprague Dawley rats for up to 24 months at dosages of 20, 60, and 200 mcg/kg/day (corresponding to dosages of approximately 120, 360, and 1200 mcg/m2/day), no significant changes in tumor incidence were observed when compared to control.
Calcipotriene did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, the human lymphocyte chromosome aberration test, or the mouse micronucleus test. Betamethasone dipropionate did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, or in the rat micronucleus test.
Studies in rats with oral doses of up to 54 mcg/kg/day (324 mcg/m2/day) of calcipotriene indicated no impairment of fertility or general reproductive performance. Studies in male rats at oral doses of up to 200 mcg/kg/day (1200 mcg/m2/day), and in female rats at oral doses of up to 1000 mcg/kg/day (6000 mcg/m2/day), of betamethasone dipropionate indicated no impairment of fertility.
5.4 Allergic Contact Dermatitis With Topical Calcipotriene (5.4 Allergic Contact Dermatitis with Topical Calcipotriene)
Allergic contact dermatitis has been observed with use of topical calcipotriene. Such an observation should be corroborated with appropriate diagnostic patch testing.
5.3 Allergic Contact Dermatitis With Topical Corticosteroids (5.3 Allergic Contact Dermatitis with Topical Corticosteroids)
Allergic contact dermatitis to a topical corticosteroid is usually diagnosed by observing a failure to heal rather than a clinical exacerbation. Such an observation should be corroborated with appropriate diagnostic patch testing.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:30.087965 · Updated: 2026-03-14T21:58:01.008833