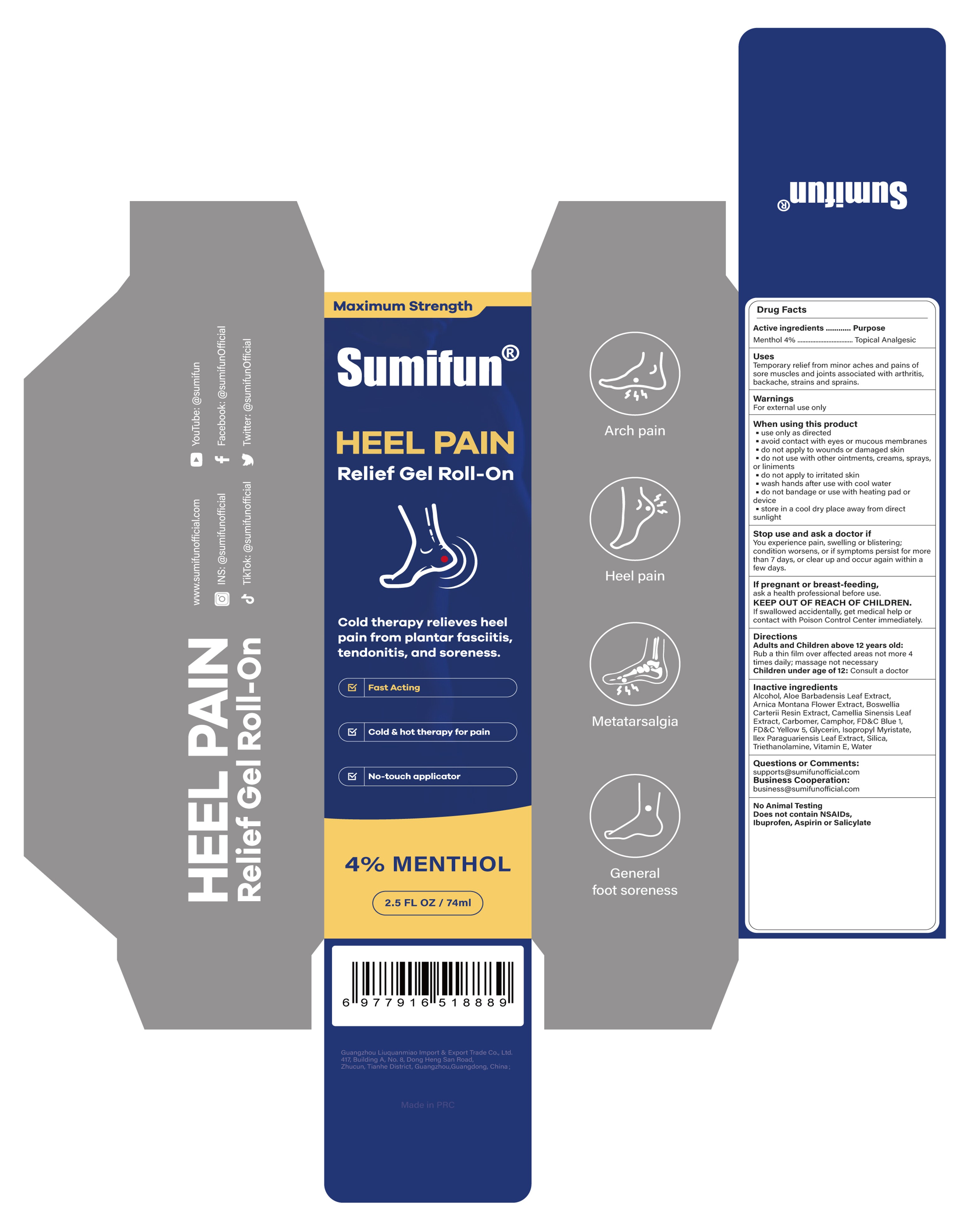
84165-011 Heel Pain Relief Gel Roll-on
36bacb5f-c1af-2e97-e063-6394a90afe5c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Menthol 4%
Medication Information
Warnings and Precautions
For external use only
Indications and Usage
Temporary relief from minor aches and pains ofsore muscles and joints associated with arthritis,backache, strains and sprains.
Dosage and Administration
Adults and Children above 12 years old:
Rub a thin film over affected areas not more 4 times daily; massage not necessary
Children under age of 12: Consult a doctor
Description
Menthol 4%
Section 50565-1
If swallowed accidentally, get medical help orcontact with Poison Control Center immediately.
Section 50566-9
You experience pain, swelling or blistering;condition worsens, or if symptoms persist for morethan 7 days, or clear up and occur again within afew days.
Section 50567-7
use only as directed
avoid contact with eyes or mucous membranes
store in a cool dry place away from directsunlight
Section 50570-1
do not apply to wounds or damaged skin
do not use with other ointments, creams, sprays
or liniments
do not apply to irritated skinwash hands after use with cool water
do not bandage or use with heating pad ordevice
Section 51727-6
Alcohol
Aloe barbadensis leaf extract
Arnica montana flower extract
Boswellia carterii resin extract
Camellia sinensis leaf extract
Carbomer
Camphor
FD&C Blue 1
FD&C Yellow 5
Glycerin
Ilex Paraguariensis Leaf Extract
Isopropyl Myristate
Silica
Triethanolamine
Vitamin E
Water
Section 51945-4
Section 55105-1
Topical Analgesic
Section 55106-9
Menthol 4%
Structured Label Content
Indications and Usage (34067-9)
Temporary relief from minor aches and pains ofsore muscles and joints associated with arthritis,backache, strains and sprains.
Dosage and Administration (34068-7)
Adults and Children above 12 years old:
Rub a thin film over affected areas not more 4 times daily; massage not necessary
Children under age of 12: Consult a doctor
Warnings and Precautions (34071-1)
For external use only
Section 50565-1 (50565-1)
If swallowed accidentally, get medical help orcontact with Poison Control Center immediately.
Section 50566-9 (50566-9)
You experience pain, swelling or blistering;condition worsens, or if symptoms persist for morethan 7 days, or clear up and occur again within afew days.
Section 50567-7 (50567-7)
use only as directed
avoid contact with eyes or mucous membranes
store in a cool dry place away from directsunlight
Section 50570-1 (50570-1)
do not apply to wounds or damaged skin
do not use with other ointments, creams, sprays
or liniments
do not apply to irritated skinwash hands after use with cool water
do not bandage or use with heating pad ordevice
Section 51727-6 (51727-6)
Alcohol
Aloe barbadensis leaf extract
Arnica montana flower extract
Boswellia carterii resin extract
Camellia sinensis leaf extract
Carbomer
Camphor
FD&C Blue 1
FD&C Yellow 5
Glycerin
Ilex Paraguariensis Leaf Extract
Isopropyl Myristate
Silica
Triethanolamine
Vitamin E
Water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Topical Analgesic
Section 55106-9 (55106-9)
Menthol 4%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:13.921148 · Updated: 2026-03-14T23:08:54.445441