These Highlights Do Not Include All The Information Needed To Use Lotronex Safely And Effectively. See Full Prescribing Information For Lotronex.
35cbb9d5-639b-207a-e054-00144ff88e88
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Infrequent but serious gastrointestinal adverse reactions have been reported with the use of LOTRONEX. These events, including ischemic colitis and serious complications of constipation, have resulted in hospitalization, and rarely, blood transfusion, surgery, and death. LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have not responded adequately to conventional therapy [see Indications and Usage ( 1 )] . LOTRONEX should be discontinued immediately in patients who develop constipation or symptoms of ischemic colitis. Patients should immediately report constipation or symptoms of ischemic colitis to their prescriber. LOTRONEX should not be resumed in patients who develop ischemic colitis. Patients who have constipation should immediately contact their prescriber if the constipation does not resolve after LOTRONEX is discontinued. Patients with resolved constipation should resume LOTRONEX only on the advice of their treating prescriber [see Dosage and Administration ( 2.1 ), Warnings and Precautions ( 5.1 ), ( 5.2 )].
Indications and Usage
LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have: chronic IBS symptoms (generally lasting 6 months or longer), had anatomic or biochemical abnormalities of the gastrointestinal tract excluded, and not responded adequately to conventional therapy. Diarrhea-predominant IBS is severe if it includes diarrhea and one or more of the following: frequent and severe abdominal pain/discomfort, frequent bowel urgency or fecal incontinence, disability or restriction of daily activities due to IBS. Because of infrequent but serious gastrointestinal adverse reactions associated with LOTRONEX, the indication is restricted to those patients for whom the benefit-to-risk balance is most favorable. Clinical studies have not been performed to adequately confirm the benefits of LOTRONEX in men.
Dosage and Administration
Starting dose is 0.5 mg twice a day ( 2.1 ) May increase dose to 1 mg twice a day after 4 weeks if starting dosage is well tolerated but does not adequately control IBS symptoms ( 2.1 ) Discontinue LOTRONEX in patients who have not had adequate control of IBS symptoms after 4 weeks of treatment with 1 mg twice a day. ( 2.1 )
Warnings and Precautions
Serious Complications of Constipation: May occur in some patients without warning. Includes obstruction, ileus, impaction, toxic megacolon, and secondary bowel ischemia and in rare cases perforation and death have been reported. Risk is increased in patients who are elderly, debilitated, or taking medications that decrease bowel motility. ( 5.1 ) Discontinue LOTRONEX immediately if constipation occurs. ( 5.1 ) Ischemic colitis: May occur in some patients without warning. Promptly evaluate patients with signs of ischemic colitis (e.g., rectal bleeding, bloody diarrhea, new or worsening abdominal pain). ( 5.2 ) Discontinue LOTRONEX immediately if signs of ischemic colitis occur, such as rectal bleeding, bloody diarrhea, or new or worsening abdominal pain. ( 5.2 )
Contraindications
Do not initiate in patients with constipation ( 4.1 ) History of chronic or severe constipation or sequelae from constipation; intestinal obstruction, stricture, toxic megacolon, gastrointestinal perforation, and/or adhesions; ischemic colitis; impaired intestinal circulation, thrombophlebitis, or hypercoagulable state; Crohn's disease or ulcerative colitis; diverticulitis; severe hepatic impairment ( 4.2 ) Concomitant use of fluvoxamine ( 4.3 )
Adverse Reactions
Infrequent but serious gastrointestinal adverse reactions have been reported with the use of LOTRONEX. These events, including ischemic colitis and serious complications of constipation, have resulted in hospitalization, and rarely, blood transfusion, surgery, and death. LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have not responded adequately to conventional therapy [see Indications and Usage ( 1 )] . LOTRONEX should be discontinued immediately in patients who develop constipation or symptoms of ischemic colitis. Patients should immediately report constipation or symptoms of ischemic colitis to their prescriber. LOTRONEX should not be resumed in patients who develop ischemic colitis. Patients who have constipation should immediately contact their prescriber if the constipation does not resolve after LOTRONEX is discontinued. Patients with resolved constipation should resume LOTRONEX only on the advice of their treating prescriber [see Dosage and Administration ( 2.1 ), Warnings and Precautions ( 5.1 ), ( 5.2 )].
Drug Interactions
In vivo data suggest that alosetron is primarily metabolized by cytochrome P450 (CYP) 1A2, with minor contributions from CYP3A4 and CYP2C9. Therefore, inducers or inhibitors of these enzymes may change the clearance of alosetron.
Storage and Handling
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron) are white, oval, film-coated tablets debossed with GX EX1 on one face. Bottles of 30 (NDC 65483-894-03) with child-resistant closures. LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face. Bottles of 30 (NDC 65483-895-03) with child-resistant closures. Store at 20-25°C (68-77°F) (USP Controlled Room Temperature). Protect from light and moisture.
How Supplied
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron) are white, oval, film-coated tablets debossed with GX EX1 on one face. Bottles of 30 (NDC 65483-894-03) with child-resistant closures. LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face. Bottles of 30 (NDC 65483-895-03) with child-resistant closures. Store at 20-25°C (68-77°F) (USP Controlled Room Temperature). Protect from light and moisture.
Medication Information
Warnings and Precautions
Serious Complications of Constipation: May occur in some patients without warning. Includes obstruction, ileus, impaction, toxic megacolon, and secondary bowel ischemia and in rare cases perforation and death have been reported. Risk is increased in patients who are elderly, debilitated, or taking medications that decrease bowel motility. ( 5.1 ) Discontinue LOTRONEX immediately if constipation occurs. ( 5.1 ) Ischemic colitis: May occur in some patients without warning. Promptly evaluate patients with signs of ischemic colitis (e.g., rectal bleeding, bloody diarrhea, new or worsening abdominal pain). ( 5.2 ) Discontinue LOTRONEX immediately if signs of ischemic colitis occur, such as rectal bleeding, bloody diarrhea, or new or worsening abdominal pain. ( 5.2 )
Indications and Usage
LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have: chronic IBS symptoms (generally lasting 6 months or longer), had anatomic or biochemical abnormalities of the gastrointestinal tract excluded, and not responded adequately to conventional therapy. Diarrhea-predominant IBS is severe if it includes diarrhea and one or more of the following: frequent and severe abdominal pain/discomfort, frequent bowel urgency or fecal incontinence, disability or restriction of daily activities due to IBS. Because of infrequent but serious gastrointestinal adverse reactions associated with LOTRONEX, the indication is restricted to those patients for whom the benefit-to-risk balance is most favorable. Clinical studies have not been performed to adequately confirm the benefits of LOTRONEX in men.
Dosage and Administration
Starting dose is 0.5 mg twice a day ( 2.1 ) May increase dose to 1 mg twice a day after 4 weeks if starting dosage is well tolerated but does not adequately control IBS symptoms ( 2.1 ) Discontinue LOTRONEX in patients who have not had adequate control of IBS symptoms after 4 weeks of treatment with 1 mg twice a day. ( 2.1 )
Contraindications
Do not initiate in patients with constipation ( 4.1 ) History of chronic or severe constipation or sequelae from constipation; intestinal obstruction, stricture, toxic megacolon, gastrointestinal perforation, and/or adhesions; ischemic colitis; impaired intestinal circulation, thrombophlebitis, or hypercoagulable state; Crohn's disease or ulcerative colitis; diverticulitis; severe hepatic impairment ( 4.2 ) Concomitant use of fluvoxamine ( 4.3 )
Adverse Reactions
Infrequent but serious gastrointestinal adverse reactions have been reported with the use of LOTRONEX. These events, including ischemic colitis and serious complications of constipation, have resulted in hospitalization, and rarely, blood transfusion, surgery, and death. LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have not responded adequately to conventional therapy [see Indications and Usage ( 1 )] . LOTRONEX should be discontinued immediately in patients who develop constipation or symptoms of ischemic colitis. Patients should immediately report constipation or symptoms of ischemic colitis to their prescriber. LOTRONEX should not be resumed in patients who develop ischemic colitis. Patients who have constipation should immediately contact their prescriber if the constipation does not resolve after LOTRONEX is discontinued. Patients with resolved constipation should resume LOTRONEX only on the advice of their treating prescriber [see Dosage and Administration ( 2.1 ), Warnings and Precautions ( 5.1 ), ( 5.2 )].
Drug Interactions
In vivo data suggest that alosetron is primarily metabolized by cytochrome P450 (CYP) 1A2, with minor contributions from CYP3A4 and CYP2C9. Therefore, inducers or inhibitors of these enzymes may change the clearance of alosetron.
Storage and Handling
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron) are white, oval, film-coated tablets debossed with GX EX1 on one face. Bottles of 30 (NDC 65483-894-03) with child-resistant closures. LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face. Bottles of 30 (NDC 65483-895-03) with child-resistant closures. Store at 20-25°C (68-77°F) (USP Controlled Room Temperature). Protect from light and moisture.
How Supplied
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron) are white, oval, film-coated tablets debossed with GX EX1 on one face. Bottles of 30 (NDC 65483-894-03) with child-resistant closures. LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face. Bottles of 30 (NDC 65483-895-03) with child-resistant closures. Store at 20-25°C (68-77°F) (USP Controlled Room Temperature). Protect from light and moisture.
Description
Infrequent but serious gastrointestinal adverse reactions have been reported with the use of LOTRONEX. These events, including ischemic colitis and serious complications of constipation, have resulted in hospitalization, and rarely, blood transfusion, surgery, and death. LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have not responded adequately to conventional therapy [see Indications and Usage ( 1 )] . LOTRONEX should be discontinued immediately in patients who develop constipation or symptoms of ischemic colitis. Patients should immediately report constipation or symptoms of ischemic colitis to their prescriber. LOTRONEX should not be resumed in patients who develop ischemic colitis. Patients who have constipation should immediately contact their prescriber if the constipation does not resolve after LOTRONEX is discontinued. Patients with resolved constipation should resume LOTRONEX only on the advice of their treating prescriber [see Dosage and Administration ( 2.1 ), Warnings and Precautions ( 5.1 ), ( 5.2 )].
Data
Animal Data
No adverse developmental effects were observed with oral administration of alosetron during the period of organogenesis to pregnant rats at doses up to 40 mg/kg/day (about 160 times the recommended human dose based on body surface area) or to pregnant rabbits at doses up to 30 mg/kg/day (about 240 times the recommended daily human dose based on body surface area).
Section 42229-5
Patients with Irritable Bowel Syndrome: Table 1 summarizes adverse reactions from 22 repeat-dose studies in patients with IBS who were treated with 1 mg of LOTRONEX twice daily for 8 to 24 weeks. The adverse reactions in Table 1 were reported in 1% or more of patients who received LOTRONEX and occurred more frequently on LOTRONEX than on placebo. A statistically significant difference was observed for constipation in patients treated with LOTRONEX compared to placebo (p<0.0001).
| LOTRONEX | ||
| Body System | Placebo | 1 mg twice daily |
| Adverse Reaction | (n = 2,363) | (n = 8,328) |
| Gastrointestinal | ||
| Constipation | 6% | 29% |
| Abdominal discomfort and pain | 4% | 7% |
| Nausea | 5% | 6% |
| Gastrointestinal discomfort and pain | 3% | 5% |
| Abdominal distention | 1% | 2% |
| Regurgitation and reflux | 2% | 2% |
| Hemorrhoids | 1% | 2% |
Section 42231-1
|
MEDICATION GUIDE LOTRONEX ® (LOW-trah-nex) (alosetron hydrochloride) Tablets |
| Read the Medication Guide you get with each refill for LOTRONEX. There may be new information. This Medication Guide does not take the place of talking with your doctor. |
|
What is the most important information I should know about LOTRONEX? LOTRONEX is a meeicine only for some women with severe chronic irritable bowel syndrome (IBS) whose:
Some people have developed serious bowel side effects while taking LOTRONEX. Serious bowel (intestine) side effects can happen suddenly, including the following: Serious complications of constipation: These complications may lead to a hospital stay and, in rare cases, blood transfusions, surgery, and death. People who are older, who are weak from illness, or who take other constipating medicines may be more likely to have serious complications of constipation with LOTRONEX. To lower your chances of getting serious complications of constipation, do the following:
Stop taking LOTRONEX and call your doctor or get medical help if you have symptoms of ischemic colitis such as new or worsening stomach-area (abdominal) pain, bloody diarrhea or blood in the stool. |
|
What is LOTRONEX? LOTRONEX is a prescription medicine used only for some women wih severe chronic IBS whose:
LOTRONEX does not cure IBS, and it may not help every person who takes it. For those who are helped, LOTRONEX reduces lower stomach area (abdominal) pain and discomfort, the sudden need to have a bowel movement (bowel urgency), and diarrhea from IBS. If you stop taking LOTRONEX, your IBS symptoms may return within 1 or 2 weeks to what they were before you started taking LOTRONEX. It is not known if LOTRONEX is safe and effective in men with IBS. It is not known if LOTRONEX is safe and effective in children. |
|
Who should not take LOTRONEX? Do not take LOTRONEX if you:
|
|
What should I talk about with my doctor before taking LOTRONEX? Talk with your doctor:
|
|
How should I take LOTRONEX?
effects from LOTRONEX, your doctor may increase your dose up to 1 mg two times a day.
your doctor.
|
|
What are the possible side effects of LOTRONEX? LOTRONEX may cause serious side effects, including: See "What is the most important information I should know about LOTRONEX?" The most common side effects of LOTRONEX include:
These are not all the possible side effects of LOTRONEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store LOTRONEX?
Keep LOTRONEX and all medicines out of the reach of children. |
|
General information about the safe and effective use of LOTRONEX Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use LOTRONEX for a condition for which it was not prescribed. Do not give LOTRONEX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LOTRONEX that is written for healthcare professionals. |
|
What are the ingredients of LOTRONEX? Active Ingredient: alosetron hydrochloride Inactive Ingredients: lactose (anhydrous), magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The white film-coat for the 0.5 mg tablet contains hypromellose, titanium dioxide, and triacetin. The blue film-coat for the 1 mg tablet contains hypromellose, titanium dioxide, triacetin, and indigo carmine. |
|
Manufactured for:
www.sebelapharma.com Toll Free 1-844-732-3521 Made in CANADA ©2019, Sebela Pharmaceuticals Inc. All rights reserved This product may be covered by one or more US pending or issued patents.
LOTRONEX is a registered trademark of Sebela International Bermuda Ltd.
This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: April 2019 |
Section 51945-4
Principal Display Panel - Lotronex 0.5 mg Bottle Label
NDC 54766-894-03
30 Tablets
LOTRONEX
(alosetron hydrochloride) Tablets 0.5mg
Each film-coated tablet contains alosetron
hydrochloride equivalent to 0.5 mg alosetron.
Rx only
See prescribing information
For Dosage and Administration
Store at 20-25˚C (68-77˚F) (USP Controlled Room Temperature)
Risk Summary
The available data with LOTRONEX use in pregnant women are insufficient to draw conclusions about any drug-associated risks for major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, no adverse developmental effects were observed with oral administration of alosetron in rats and rabbits during organogenesis at doses 160 to 240 times, respectively, the recommended human dosage (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and miscarriage is 15 to 20%, respectively.
10 Overdosage
There is no specific antidote for overdose of LOTRONEX. Patients should be managed with appropriate supportive therapy. Individual oral doses as large as 16 mg have been administered in clinical studies without significant adverse reactions. This dose is 8 times higher than the recommended total daily dose. Inhibition of the metabolic elimination and reduced first pass of other drugs might occur with overdoses of LOTRONEX [see Drug Interactions ( 7)].
15 References
1. Thompson WG, Creed F, Drossman DA, et al. Functional bowel disease and functional abdominal pain. Gastroenterol Int. 1992;5:75-91.
8.2 Lactation
Alosetron and/or metabolites of alosetron are excreted in the breast milk of lactating rats. It is not known whether alosetron is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when LOTRONEX is administered to a nursing woman.
11 Description
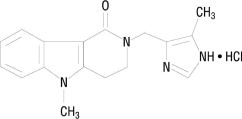
The active ingredient in LOTRONEX Tablets is alosetron hydrochloride (HCl), a potent and selective antagonist of the serotonin 5-HT 3 receptor type. Chemically, alosetron is designated as 2,3,4,5-tetrahydro-5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-1H-pyrido[4,3-b]indol-1-one, monohydrochloride. Alosetron is achiral and has the empirical formula C 17H 18N 4O•HCl, representing a molecular weight of 330.8. Alosetron is a white to beige solid that has a solubility of 61 mg/mL in water, 42 mg/mL in 0.1M hydrochloric acid, 0.3 mg/mL in pH 6 phosphate buffer, and <0.1 mg/mL in pH 8 phosphate buffer. The chemical structure of alosetron is:
LOTRONEX Tablets are supplied for oral administration as 0.5 mg (white) and 1 mg (blue) tablets. The 0.5 mg tablet contains 0.562 mg alosetron HCl equivalent to 0.5 mg alosetron, and the 1 mg tablet contains 1.124 mg alosetron HCl equivalent to 1 mg of alosetron. Each tablet also contains the inactive ingredients lactose (anhydrous), magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The white film coat for the 0.5 mg tablet contains hypromellose, titanium dioxide, and triacetin. The blue film coat for the 1 mg tablet contains hypromellose, titanium dioxide, triacetin, and indigo carmine.
4.1 Constipation
LOTRONEX should not be initiated in patients with constipation [see Warnings and Precautions ( 5.1)] .
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. Use of LOTRONEX is not recommended in the pediatric population, based upon the risk of serious complications of constipation and ischemic colitis in adults.
8.5 Geriatric Use
In some studies in healthy men or women, plasma concentrations were elevated by approximately 40% in individuals 65 years and older compared to young adults [see Warnings and Precautions ( 5.1)] . However, this effect was not consistently observed in men.
Postmarketing experience suggests that elderly patients may be at greater risk for complications of constipation therefore, appropriate caution and follow-up should be exercised if LOTRONEX is prescribed for these patients [see Warnings and Precautions ( 5.1)] .
14.3 Long Term Use
In a 48-week multinational, double-blind, placebo-controlled study, LOTRONEX 1 mg twice daily was evaluated in 714 women with non-constipated IBS. A retrospective analysis of the subset of women with severe diarrhea-predominant IBS (urgency on at least 10 days during the 2-week baseline period) was performed. Of the 417 patients with severe diarrhea-predominant IBS, 62% completed the trial.
LOTRONEX (n = 198) provided a greater average rate of adequate relief of IBS pain and discomfort (52% vs. 41%) and a greater average rate of satisfactory control of bowel urgency (60% vs. 48%) compared with placebo (n = 219). Significant improvement of these symptoms occurred for most of the 48-week treatment period with no evidence of tachyphylaxis.
2.1 Adult Patients
To lower the risk of constipation, LOTRONEX should be started at a dosage of 0.5 mg twice a day. Patients who become constipated at this dosage should stop taking LOTRONEX until the constipation resolves. They may be restarted at 0.5 mg once a day. If constipation recurs at the lower dose, LOTRONEX should be discontinued immediately.
Patients well controlled on 0.5 mg once or twice a day may be maintained on this regimen. If after 4 weeks the dosage is well tolerated but does not adequately control IBS symptoms, then the dosage can be increased to up to 1 mg twice a day . LOTRONEX should be discontinued in patients who have not had adequate control of IBS symptoms after 4 weeks of treatment with 1 mg twice a day.
LOTRONEX can be taken with or without food [see Clinical Pharmacology ( 12.3)] .
LOTRONEX should be discontinued immediately in patients who develop constipation or signs of ischemic colitis. LOTRONEX should not be restarted in patients who develop ischemic colitis.
Clinical trial and postmarketing experience suggest that debilitated patients or patients taking additional medications that decrease gastrointestinal motility may be at greater risk of serious complications of constipation. Therefore, appropriate caution and follow-up should be exercised if LOTRONEX is prescribed for these patients.
Postmarketing experience suggests that elderly patients may be at greater risk for complications of constipation; therefore, appropriate caution and follow-up should be exercised if LOTRONEX is prescribed for these patients [see Warnings and Precautions ( 5.1)] .
4 Contraindications
- Do not initiate in patients with constipation ( 4.1)
- History of chronic or severe constipation or sequelae from constipation; intestinal obstruction, stricture, toxic megacolon, gastrointestinal perforation, and/or adhesions; ischemic colitis; impaired intestinal circulation, thrombophlebitis, or hypercoagulable state; Crohn's disease or ulcerative colitis; diverticulitis; severe hepatic impairment ( 4.2)
- Concomitant use of fluvoxamine ( 4.3)
6 Adverse Reactions
The following adverse reactions are described in more detail in other sections of the label:
- Complications of constipation [see Boxed Warning, Warnings and Precautions ( 5.1)]
- Ischemic colitis [see Boxed Warning, Warnings and Precautions ( 5.2)]
7 Drug Interactions
In vivo data suggest that alosetron is primarily metabolized by cytochrome P450 (CYP) 1A2, with minor contributions from CYP3A4 and CYP2C9. Therefore, inducers or inhibitors of these enzymes may change the clearance of alosetron.
5.2 Ischemic Colitis
Some patients have experienced ischemic colitis without warning.
Ischemic colitis has been reported in patients receiving LOTRONEX in clinical trials as well as during marketed use of the drug. In IBS clinical trials, the cumulative incidence of ischemic colitis in women receiving LOTRONEX was 0.2% (2 per 1,000 patients, 95% confidence interval 1 to 3) through 3 months and was 0.3% (3 per 1,000 patients, 95% confidence interval 1 to 4) through 6 months. Ischemic colitis has been reported with use of 1 mg twice daily and with lower doses. A dose-response relationship has not been established. Ischemic colitis was reported in one patient receiving placebo. The patient experience in controlled clinical trials is insufficient to estimate the incidence of ischemic colitis in patients taking LOTRONEX for longer than 6 months.
LOTRONEX should be discontinued immediately in patients with signs of ischemic colitis such as rectal bleeding, bloody diarrhea, or new or worsening abdominal pain. Because ischemic colitis can be life-threatening, patients with signs or symptoms of ischemic colitis should be evaluated promptly and have appropriate diagnostic testing performed. Treatment with LOTRONEX should not be resumed in patients who develop ischemic colitis.
8.7 Renal Impairment
Renal impairment (creatinine clearance 4 to 56 mL/min) has no effect on the renal elimination of alosetron due to the minor contribution of this pathway to elimination. The effect of renal impairment on metabolite pharmacokinetics and the effect of end-stage renal disease have not been assessed.
12.2 Pharmacodynamics
In healthy volunteers and patients with IBS, alosetron (2 mg orally, twice daily for 8 days) increased colonic transit time without affecting orocecal transit time. In healthy volunteers, alosetron also increased basal jejunal water and sodium absorption after a single 4 mg dose. In patients with IBS, multiple oral dosages of alosetron (4 mg twice daily for 6.5 days) significantly increased colonic compliance.
Single oral doses of alosetron administered to healthy men produced a dose-dependent reduction in the flare response seen after intradermal injection of serotonin. Urinary 6-β-hydroxycortisol excretion decreased by 52% in elderly subjects after 27.5 days of alosetron 2 mg administered orally twice daily. This decrease was not statistically significant. In another study utilizing alosetron 1 mg administered orally twice daily for 4 days, there was a significant decrease in urinary 6-β-hydroxycortisol excretion. However, there was no change in the ratio of 6-β-hydroxycortisol to cortisol, indicating a possible decrease in cortisol production. The clinical significance of these findings is unknown.
12.3 Pharmacokinetics
The pharmacokinetics of alosetron have been studied after single oral doses ranging from 0.05 to 16 mg in healthy men. The pharmacokinetics of alosetron have also been evaluated in healthy women and men and in patients with IBS after repeated oral dosages ranging from 1 mg twice daily to 8 mg twice daily.
Absorption: Alosetron was rapidly absorbed after oral administration with a mean absolute bioavailability of approximately 50% to 60% (approximate range, 30% to >90%). After administration of radiolabeled alosetron, only 1% of the dose was recovered in the feces as unchanged drug. Following oral administration of a 1 mg alosetron dose to young men, a peak plasma concentration of approximately 5 ng/mL occurred at 1 hour. In young women, the mean peak plasma concentration was approximately 9 ng/mL, with a similar time to peak.
Plasma concentrations were 30% to 50% lower and less variable in men compared to women given the same oral dose. Population pharmacokinetic analysis in IBS patients confirmed that alosetron concentrations were influenced by gender (27% lower in men).
14.2 Efficacy Studies
LOTRONEX has been studied in women with IBS in five 12-week US multicenter, randomized, double-blind, placebo-controlled clinical studies.
| Study | Patient Population |
Placebo (n) |
Lotronex Dose (n) |
| 1 and 2 | Non-constipated women with IBS | (640) |
1 mg twice daily (633) |
| 3 and 4 |
Women with severe diarrhea-predominant IBS (defined as bowel urgency ≥50% of days) |
(515) |
1 mg twice daily (778) |
| 5 |
Women with severe diarrhea-predominant IBS (defined as average pain ≥moderate, urgency ≥50% of days, and/or restriction of daily activities ≥25% of days) |
(176) | 0.5 mg once daily
(177) |
| 1 mg once daily
(175) |
|||
| 1 mg once daily
(177) |
7.1 Cyp1a2 Inhibitors
Fluvoxamine is a known strong inhibitor of CYP1A2 and also inhibits CYP3A4, CYP2C9, and CYP2C19. In a pharmacokinetic study, 40 healthy female subjects received fluvoxamine in escalating doses from 50 to 200 mg/ day for 16 days, with coadministration of alosetron 1 mg on the last day. Fluvoxamine increased mean alosetron plasma concentrations (AUC) approximately 6-fold and prolonged the half-life by approximately 3-fold. Concomitant administration of alosetron and fluvoxamine is contraindicated [see Contraindications ( 4.3)] .
Concomitant administration of alosetron and moderate CYP1A2 inhibitors, including quinolone antibiotics and cimetidine, has not been evaluated, but should be avoided unless clinically necessary because of similar potential drug interactions.
7.2 Cyp3a4 Inhibitors
Ketoconazole is a known strong inhibitor of CYP3A4. In a pharmacokinetic study, 38 healthy female subjects received ketoconazole 200 mg twice daily for 7 days, with coadministration of alosetron 1 mg on the last day. Ketoconazole increased mean alosetron plasma concentrations (AUC) by 29%. Caution should be used when alosetron and ketoconazole are administered concomitantly. Coadministration of alosetron and strong CYP3A4 inhibitors such as clarithromycin, telithromycin, protease inhibitors, voriconazole, and itraconazole has not been evaluated but should be undertaken with caution because of similar potential drug interactions. The effect of induction or inhibition of other pathways on exposure to alosetron and its metabolites is not known.
7.3 Other Cyp Enzymes
In vitro human liver microsome studies and an in vivo metabolic probe study demonstrated that alosetron did not inhibit CYP enzymes 3A4, 2C9, or 2C19. In vitro at total drug concentrations 27-fold higher than peak plasma concentrations observed with the 1 mg dose, alosetron inhibited CYP enzymes 1A2 (60%) and 2E1 (50%). In an in vivo metabolic probe study, alosetron did not inhibit CYP2E1 but did produce 30% inhibition of both CYP1A2 and N-acetyltransferase. Although not studied with alosetron, inhibition of N-acetyltransferase may have clinically relevant consequences for drugs such as isoniazid, procainamide, and hydralazine. The effect on CYP1A2 was explored further in a clinical interaction study with theophylline and no effect on metabolism was observed. Another study showed that alosetron had no clinically significant effect on plasma concentrations of the oral contraceptive agents ethinyl estradiol and levonorgestrel (CYP3A4 substrates). A clinical interaction study was also conducted with alosetron and the CYP3A4 substrate cisapride. No significant effects on cisapride metabolism or QT interval were noted. The effects of alosetron on monoamine oxidases and on intestinal first pass secondary to high intraluminal concentrations have not been examined. Based on the above data from in vitro and in vivo studies, it is unlikely that alosetron will inhibit the hepatic metabolic clearance of drugs metabolized by the CYP enzymes 2C9, 2C19, or 2E1.
Alosetron does not appear to induce the major cytochrome P450 drug-metabolizing enzyme 3A. Alosetron also does not appear to induce CYP enzymes 2E1 or 2C19. It is not known whether alosetron might induce other enzymes.
8.6 Hepatic Impairment
Due to the extensive hepatic metabolism of alosetron, increased exposure to alosetron and/or its metabolites is likely to occur in patients with hepatic impairment. Alosetron should not be used in patients with severe hepatic impairment and should be used with caution in patients with mild or moderate hepatic impairment.
A single 1 mg oral dose of alosetron was administered to 1 female and 5 male patients with moderate hepatic impairment (Child-Pugh score of 7 to 9) and to 1 female and 2 male patients with severe hepatic impairment (Child-Pugh score of >9). In comparison with historical data from healthy subjects, patients with severe hepatic impairment displayed higher systemic exposure to alosetron. The female with severe hepatic impairment displayed approximately 14-fold higher exposure, while the female with moderate hepatic impairment displayed approximately 1.6-fold higher exposure, than healthy females. Due to the small number of subjects and high intersubject variability in the pharmacokinetic findings, no definitive quantitative conclusions can be made. However, due to the greater exposure to alosetron in the female with severe hepatic impairment, alosetron should not be used in females with severe hepatic impairment [see Dosage and Administration ( 2.2), Contraindications ( 4)] .
1 Indications and Usage
LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have:
- chronic IBS symptoms (generally lasting 6 months or longer),
- had anatomic or biochemical abnormalities of the gastrointestinal tract excluded, and
- not responded adequately to conventional therapy.
Diarrhea-predominant IBS is severe if it includes diarrhea and one or more of the following:
- frequent and severe abdominal pain/discomfort,
- frequent bowel urgency or fecal incontinence,
- disability or restriction of daily activities due to IBS.
Because of infrequent but serious gastrointestinal adverse reactions associated with LOTRONEX, the indication is restricted to those patients for whom the benefit-to-risk balance is most favorable.
Clinical studies have not been performed to adequately confirm the benefits of LOTRONEX in men.
14.1 Dose Ranging Study
Data from a dose-ranging study of women (n = 85) who received LOTRONEX 0.5 mg twice daily indicated that the incidence of constipation (14%) was lower than that experienced by women receiving 1 mg twice daily (29%). Therefore, to lower the risk of constipation, LOTRONEX should be started at a dosage of 0.5 mg twice a day. The efficacy of the 0.5 mg twice-daily dosage in treating severe diarrhea-predominant IBS has not been adequately evaluated in clinical trials. [See Dosage and Administration ( 2.1)]
Clinical Considerations
Monitor infants exposed to LOTRONEX through breast milk for severe constipation and blood in stools.
12.1 Mechanism of Action
Alosetron is a potent and selective 5-HT 3 receptor antagonist. 5-HT 3 receptors are ligand-gated cation channels that are extensively distributed on enteric neurons in the human gastrointestinal tract, as well as other peripheral and central locations. Activation of these channels and the resulting neuronal depolarization affect the regulation of visceral pain, colonic transit, and gastrointestinal secretions, processes that relate to the pathophysiology of IBS. 5-HT 3 receptor antagonists such as alosetron inhibit activation of non-selective cation channels, which results in the modulation of the enteric nervous system.
The cause of IBS is unknown. IBS is characterized by visceral hypersensitivity and hyperactivity of the gastrointestinal tract, which lead to abnormal sensations of pain and motor activity. Following distention of the rectum, patients with IBS exhibit pain and discomfort at lower volumes than healthy volunteers. Following such distention, alosetron reduced pain and exaggerated motor responses, possibly due to blockade of 5-HT 3 receptors.
5 Warnings and Precautions
- Serious Complications of Constipation: May occur in some patients without warning. Includes obstruction, ileus, impaction, toxic megacolon, and secondary bowel ischemia and in rare cases perforation and death have been reported. Risk is increased in patients who are elderly, debilitated, or taking medications that decrease bowel motility. ( 5.1)
- Discontinue LOTRONEX immediately if constipation occurs. ( 5.1)
- Ischemic colitis: May occur in some patients without warning. Promptly evaluate patients with signs of ischemic colitis (e.g., rectal bleeding, bloody diarrhea, new or worsening abdominal pain). ( 5.2)
- Discontinue LOTRONEX immediately if signs of ischemic colitis occur, such as rectal bleeding, bloody diarrhea, or new or worsening abdominal pain. ( 5.2)
2 Dosage and Administration
- Starting dose is 0.5 mg twice a day ( 2.1)
- May increase dose to 1 mg twice a day after 4 weeks if starting dosage is well tolerated but does not adequately control IBS symptoms ( 2.1)
- Discontinue LOTRONEX in patients who have not had adequate control of IBS symptoms after 4 weeks of treatment with 1 mg twice a day. ( 2.1)
3 Dosage Forms and Strengths
0.5 mg and 1 mg tablets
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron), are white, oval, film-coated tablets debossed with GX EX1 on one face.
LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face.
6.2 Postmarketing Experience
In addition to events reported in clinical trials, the following events have been identified during use of LOTRONEX in clinical practice. Because they were reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to LOTRONEX.
- Gastrointestinal: Impaction, perforation, ulceration, small bowel mesenteric ischemia.
- Neurological: Headache.
- Skin: Rash.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved labeling (Medication Guide).
4.3 Concomitant Use of Fluvoxamine
Concomitant administration of LOTRONEX with fluvoxamine is contraindicated. Fluvoxamine, a known strong inhibitor of CYP1A2, has been shown to increase mean alosetron plasma concentrations (AUC) approximately 6-fold and prolong the half-life by approximately 3-fold [see Drug Interactions ( 7.1)] .
16 How Supplied/storage and Handling
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron) are white, oval, film-coated tablets debossed with GX EX1 on one face.
Bottles of 30 (NDC 65483-894-03) with child-resistant closures.
LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face.
Bottles of 30 (NDC 65483-895-03) with child-resistant closures.
Store at 20-25°C (68-77°F) (USP Controlled Room Temperature). Protect from light and moisture.
2.2 Patients With Hepatic Impairment
LOTRONEX is extensively metabolized by the liver, and increased exposure to LOTRONEX is likely to occur in patients with hepatic impairment. Increased drug exposure may increase the risk of serious adverse reactions. LOTRONEX should be used with caution in patients with mild or moderate hepatic impairment and is contraindicated in patients with severe hepatic impairment [see Contraindications ( 4), Use in Specific Populations ( 8.6)].
5.1 Serious Complications of Constipation
Some patients have experienced serious complications of constipation without warning.
Serious complications of constipation, including obstruction, ileus, impaction, toxic megacolon, and secondary bowel ischemia, have been reported with use of LOTRONEX during clinical trials. Complications of constipation have been reported with use of 1 mg twice daily and with lower doses. A dose response relationship has not been established for serious complications of constipation. The incidence of serious complications of constipation was approximately 0.1% (1 per 1,000 patients) in women receiving either LOTRONEX or placebo. In addition, rare cases of perforation and death have been reported from postmarketing clinical practice. In some cases, complications of constipation required intestinal surgery, including colectomy. Patients who are elderly, debilitated, or taking additional medications that decrease gastrointestinal motility may be at greater risk for complications of constipation.
LOTRONEX should be discontinued immediately in patients who develop constipation [see Boxed Warning].
4.2 History of Severe Bowel Or Hepatic Disorders
LOTRONEX is contraindicated in patients with a history of the following:
- chronic or severe constipation or sequelae from constipation
- intestinal obstruction, stricture, toxic megacolon, gastrointestinal perforation, and/or adhesions
- ischemic colitis, impaired intestinal circulation, thrombophlebitis, or hypercoagulable state
- Crohn's disease or ulcerative colitis
- diverticulitis
- severe hepatic impairment
Warning: Serious Gastrointestinal Adverse Reactions
Infrequent but serious gastrointestinal adverse reactions have been reported with the use of LOTRONEX. These events, including ischemic colitis and serious complications of constipation, have resulted in hospitalization, and rarely, blood transfusion, surgery, and death.
- LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have not responded adequately to conventional therapy [see Indications and Usage ( 1)] .
- LOTRONEX should be discontinued immediately in patients who develop constipation or symptoms of ischemic colitis. Patients should immediately report constipation or symptoms of ischemic colitis to their prescriber. LOTRONEX should not be resumed in patients who develop ischemic colitis. Patients who have constipation should immediately contact their prescriber if the constipation does not resolve after LOTRONEX is discontinued. Patients with resolved constipation should resume LOTRONEX only on the advice of their treating prescriber [see Dosage and Administration ( 2.1), Warnings and Precautions ( 5.1), ( 5.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In 2-year oral studies, alosetron was not carcinogenic in mice at doses up to 30 mg/kg/day or in rats at doses up to 40 mg/kg/day. These doses are about 60 to 160 times, respectively, the recommended human dose of alosetron of 2 mg/day (1 mg twice daily) based on body surface area. Alosetron was not genotoxic in the Ames tests, the mouse lymphoma cell (L5178Y/TK ±) forward gene mutation test, the human lymphocyte chromosome aberration test, the ex vivo rat hepatocyte unscheduled DNA synthesis (UDS) test, or the in vivo rat micronucleus test for mutagenicity. Alosetron at oral doses up to 40 mg/kg/day (about 160 times the recommended daily human dose based on body surface area) was found to have no effect on fertility and reproductive performance of male or female rats.
Structured Label Content
Data
Animal Data
No adverse developmental effects were observed with oral administration of alosetron during the period of organogenesis to pregnant rats at doses up to 40 mg/kg/day (about 160 times the recommended human dose based on body surface area) or to pregnant rabbits at doses up to 30 mg/kg/day (about 240 times the recommended daily human dose based on body surface area).
Section 42229-5 (42229-5)
Patients with Irritable Bowel Syndrome: Table 1 summarizes adverse reactions from 22 repeat-dose studies in patients with IBS who were treated with 1 mg of LOTRONEX twice daily for 8 to 24 weeks. The adverse reactions in Table 1 were reported in 1% or more of patients who received LOTRONEX and occurred more frequently on LOTRONEX than on placebo. A statistically significant difference was observed for constipation in patients treated with LOTRONEX compared to placebo (p<0.0001).
| LOTRONEX | ||
| Body System | Placebo | 1 mg twice daily |
| Adverse Reaction | (n = 2,363) | (n = 8,328) |
| Gastrointestinal | ||
| Constipation | 6% | 29% |
| Abdominal discomfort and pain | 4% | 7% |
| Nausea | 5% | 6% |
| Gastrointestinal discomfort and pain | 3% | 5% |
| Abdominal distention | 1% | 2% |
| Regurgitation and reflux | 2% | 2% |
| Hemorrhoids | 1% | 2% |
Section 42231-1 (42231-1)
|
MEDICATION GUIDE LOTRONEX ® (LOW-trah-nex) (alosetron hydrochloride) Tablets |
| Read the Medication Guide you get with each refill for LOTRONEX. There may be new information. This Medication Guide does not take the place of talking with your doctor. |
|
What is the most important information I should know about LOTRONEX? LOTRONEX is a meeicine only for some women with severe chronic irritable bowel syndrome (IBS) whose:
Some people have developed serious bowel side effects while taking LOTRONEX. Serious bowel (intestine) side effects can happen suddenly, including the following: Serious complications of constipation: These complications may lead to a hospital stay and, in rare cases, blood transfusions, surgery, and death. People who are older, who are weak from illness, or who take other constipating medicines may be more likely to have serious complications of constipation with LOTRONEX. To lower your chances of getting serious complications of constipation, do the following:
Stop taking LOTRONEX and call your doctor or get medical help if you have symptoms of ischemic colitis such as new or worsening stomach-area (abdominal) pain, bloody diarrhea or blood in the stool. |
|
What is LOTRONEX? LOTRONEX is a prescription medicine used only for some women wih severe chronic IBS whose:
LOTRONEX does not cure IBS, and it may not help every person who takes it. For those who are helped, LOTRONEX reduces lower stomach area (abdominal) pain and discomfort, the sudden need to have a bowel movement (bowel urgency), and diarrhea from IBS. If you stop taking LOTRONEX, your IBS symptoms may return within 1 or 2 weeks to what they were before you started taking LOTRONEX. It is not known if LOTRONEX is safe and effective in men with IBS. It is not known if LOTRONEX is safe and effective in children. |
|
Who should not take LOTRONEX? Do not take LOTRONEX if you:
|
|
What should I talk about with my doctor before taking LOTRONEX? Talk with your doctor:
|
|
How should I take LOTRONEX?
effects from LOTRONEX, your doctor may increase your dose up to 1 mg two times a day.
your doctor.
|
|
What are the possible side effects of LOTRONEX? LOTRONEX may cause serious side effects, including: See "What is the most important information I should know about LOTRONEX?" The most common side effects of LOTRONEX include:
These are not all the possible side effects of LOTRONEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store LOTRONEX?
Keep LOTRONEX and all medicines out of the reach of children. |
|
General information about the safe and effective use of LOTRONEX Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use LOTRONEX for a condition for which it was not prescribed. Do not give LOTRONEX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LOTRONEX that is written for healthcare professionals. |
|
What are the ingredients of LOTRONEX? Active Ingredient: alosetron hydrochloride Inactive Ingredients: lactose (anhydrous), magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The white film-coat for the 0.5 mg tablet contains hypromellose, titanium dioxide, and triacetin. The blue film-coat for the 1 mg tablet contains hypromellose, titanium dioxide, triacetin, and indigo carmine. |
|
Manufactured for:
www.sebelapharma.com Toll Free 1-844-732-3521 Made in CANADA ©2019, Sebela Pharmaceuticals Inc. All rights reserved This product may be covered by one or more US pending or issued patents.
LOTRONEX is a registered trademark of Sebela International Bermuda Ltd.
This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: April 2019 |
Section 51945-4 (51945-4)
Principal Display Panel - Lotronex 0.5 mg Bottle Label
NDC 54766-894-03
30 Tablets
LOTRONEX
(alosetron hydrochloride) Tablets 0.5mg
Each film-coated tablet contains alosetron
hydrochloride equivalent to 0.5 mg alosetron.
Rx only
See prescribing information
For Dosage and Administration
Store at 20-25˚C (68-77˚F) (USP Controlled Room Temperature)
Risk Summary
The available data with LOTRONEX use in pregnant women are insufficient to draw conclusions about any drug-associated risks for major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, no adverse developmental effects were observed with oral administration of alosetron in rats and rabbits during organogenesis at doses 160 to 240 times, respectively, the recommended human dosage (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and miscarriage is 15 to 20%, respectively.
10 Overdosage (10 OVERDOSAGE)
There is no specific antidote for overdose of LOTRONEX. Patients should be managed with appropriate supportive therapy. Individual oral doses as large as 16 mg have been administered in clinical studies without significant adverse reactions. This dose is 8 times higher than the recommended total daily dose. Inhibition of the metabolic elimination and reduced first pass of other drugs might occur with overdoses of LOTRONEX [see Drug Interactions ( 7)].
15 References (15 REFERENCES)
1. Thompson WG, Creed F, Drossman DA, et al. Functional bowel disease and functional abdominal pain. Gastroenterol Int. 1992;5:75-91.
8.2 Lactation
Alosetron and/or metabolites of alosetron are excreted in the breast milk of lactating rats. It is not known whether alosetron is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when LOTRONEX is administered to a nursing woman.
11 Description (11 DESCRIPTION)
The active ingredient in LOTRONEX Tablets is alosetron hydrochloride (HCl), a potent and selective antagonist of the serotonin 5-HT 3 receptor type. Chemically, alosetron is designated as 2,3,4,5-tetrahydro-5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-1H-pyrido[4,3-b]indol-1-one, monohydrochloride. Alosetron is achiral and has the empirical formula C 17H 18N 4O•HCl, representing a molecular weight of 330.8. Alosetron is a white to beige solid that has a solubility of 61 mg/mL in water, 42 mg/mL in 0.1M hydrochloric acid, 0.3 mg/mL in pH 6 phosphate buffer, and <0.1 mg/mL in pH 8 phosphate buffer. The chemical structure of alosetron is:
LOTRONEX Tablets are supplied for oral administration as 0.5 mg (white) and 1 mg (blue) tablets. The 0.5 mg tablet contains 0.562 mg alosetron HCl equivalent to 0.5 mg alosetron, and the 1 mg tablet contains 1.124 mg alosetron HCl equivalent to 1 mg of alosetron. Each tablet also contains the inactive ingredients lactose (anhydrous), magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The white film coat for the 0.5 mg tablet contains hypromellose, titanium dioxide, and triacetin. The blue film coat for the 1 mg tablet contains hypromellose, titanium dioxide, triacetin, and indigo carmine.
4.1 Constipation
LOTRONEX should not be initiated in patients with constipation [see Warnings and Precautions ( 5.1)] .
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. Use of LOTRONEX is not recommended in the pediatric population, based upon the risk of serious complications of constipation and ischemic colitis in adults.
8.5 Geriatric Use
In some studies in healthy men or women, plasma concentrations were elevated by approximately 40% in individuals 65 years and older compared to young adults [see Warnings and Precautions ( 5.1)] . However, this effect was not consistently observed in men.
Postmarketing experience suggests that elderly patients may be at greater risk for complications of constipation therefore, appropriate caution and follow-up should be exercised if LOTRONEX is prescribed for these patients [see Warnings and Precautions ( 5.1)] .
14.3 Long Term Use (14.3 Long-Term Use)
In a 48-week multinational, double-blind, placebo-controlled study, LOTRONEX 1 mg twice daily was evaluated in 714 women with non-constipated IBS. A retrospective analysis of the subset of women with severe diarrhea-predominant IBS (urgency on at least 10 days during the 2-week baseline period) was performed. Of the 417 patients with severe diarrhea-predominant IBS, 62% completed the trial.
LOTRONEX (n = 198) provided a greater average rate of adequate relief of IBS pain and discomfort (52% vs. 41%) and a greater average rate of satisfactory control of bowel urgency (60% vs. 48%) compared with placebo (n = 219). Significant improvement of these symptoms occurred for most of the 48-week treatment period with no evidence of tachyphylaxis.
2.1 Adult Patients
To lower the risk of constipation, LOTRONEX should be started at a dosage of 0.5 mg twice a day. Patients who become constipated at this dosage should stop taking LOTRONEX until the constipation resolves. They may be restarted at 0.5 mg once a day. If constipation recurs at the lower dose, LOTRONEX should be discontinued immediately.
Patients well controlled on 0.5 mg once or twice a day may be maintained on this regimen. If after 4 weeks the dosage is well tolerated but does not adequately control IBS symptoms, then the dosage can be increased to up to 1 mg twice a day . LOTRONEX should be discontinued in patients who have not had adequate control of IBS symptoms after 4 weeks of treatment with 1 mg twice a day.
LOTRONEX can be taken with or without food [see Clinical Pharmacology ( 12.3)] .
LOTRONEX should be discontinued immediately in patients who develop constipation or signs of ischemic colitis. LOTRONEX should not be restarted in patients who develop ischemic colitis.
Clinical trial and postmarketing experience suggest that debilitated patients or patients taking additional medications that decrease gastrointestinal motility may be at greater risk of serious complications of constipation. Therefore, appropriate caution and follow-up should be exercised if LOTRONEX is prescribed for these patients.
Postmarketing experience suggests that elderly patients may be at greater risk for complications of constipation; therefore, appropriate caution and follow-up should be exercised if LOTRONEX is prescribed for these patients [see Warnings and Precautions ( 5.1)] .
4 Contraindications (4 CONTRAINDICATIONS)
- Do not initiate in patients with constipation ( 4.1)
- History of chronic or severe constipation or sequelae from constipation; intestinal obstruction, stricture, toxic megacolon, gastrointestinal perforation, and/or adhesions; ischemic colitis; impaired intestinal circulation, thrombophlebitis, or hypercoagulable state; Crohn's disease or ulcerative colitis; diverticulitis; severe hepatic impairment ( 4.2)
- Concomitant use of fluvoxamine ( 4.3)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in more detail in other sections of the label:
- Complications of constipation [see Boxed Warning, Warnings and Precautions ( 5.1)]
- Ischemic colitis [see Boxed Warning, Warnings and Precautions ( 5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
In vivo data suggest that alosetron is primarily metabolized by cytochrome P450 (CYP) 1A2, with minor contributions from CYP3A4 and CYP2C9. Therefore, inducers or inhibitors of these enzymes may change the clearance of alosetron.
5.2 Ischemic Colitis
Some patients have experienced ischemic colitis without warning.
Ischemic colitis has been reported in patients receiving LOTRONEX in clinical trials as well as during marketed use of the drug. In IBS clinical trials, the cumulative incidence of ischemic colitis in women receiving LOTRONEX was 0.2% (2 per 1,000 patients, 95% confidence interval 1 to 3) through 3 months and was 0.3% (3 per 1,000 patients, 95% confidence interval 1 to 4) through 6 months. Ischemic colitis has been reported with use of 1 mg twice daily and with lower doses. A dose-response relationship has not been established. Ischemic colitis was reported in one patient receiving placebo. The patient experience in controlled clinical trials is insufficient to estimate the incidence of ischemic colitis in patients taking LOTRONEX for longer than 6 months.
LOTRONEX should be discontinued immediately in patients with signs of ischemic colitis such as rectal bleeding, bloody diarrhea, or new or worsening abdominal pain. Because ischemic colitis can be life-threatening, patients with signs or symptoms of ischemic colitis should be evaluated promptly and have appropriate diagnostic testing performed. Treatment with LOTRONEX should not be resumed in patients who develop ischemic colitis.
8.7 Renal Impairment
Renal impairment (creatinine clearance 4 to 56 mL/min) has no effect on the renal elimination of alosetron due to the minor contribution of this pathway to elimination. The effect of renal impairment on metabolite pharmacokinetics and the effect of end-stage renal disease have not been assessed.
12.2 Pharmacodynamics
In healthy volunteers and patients with IBS, alosetron (2 mg orally, twice daily for 8 days) increased colonic transit time without affecting orocecal transit time. In healthy volunteers, alosetron also increased basal jejunal water and sodium absorption after a single 4 mg dose. In patients with IBS, multiple oral dosages of alosetron (4 mg twice daily for 6.5 days) significantly increased colonic compliance.
Single oral doses of alosetron administered to healthy men produced a dose-dependent reduction in the flare response seen after intradermal injection of serotonin. Urinary 6-β-hydroxycortisol excretion decreased by 52% in elderly subjects after 27.5 days of alosetron 2 mg administered orally twice daily. This decrease was not statistically significant. In another study utilizing alosetron 1 mg administered orally twice daily for 4 days, there was a significant decrease in urinary 6-β-hydroxycortisol excretion. However, there was no change in the ratio of 6-β-hydroxycortisol to cortisol, indicating a possible decrease in cortisol production. The clinical significance of these findings is unknown.
12.3 Pharmacokinetics
The pharmacokinetics of alosetron have been studied after single oral doses ranging from 0.05 to 16 mg in healthy men. The pharmacokinetics of alosetron have also been evaluated in healthy women and men and in patients with IBS after repeated oral dosages ranging from 1 mg twice daily to 8 mg twice daily.
Absorption: Alosetron was rapidly absorbed after oral administration with a mean absolute bioavailability of approximately 50% to 60% (approximate range, 30% to >90%). After administration of radiolabeled alosetron, only 1% of the dose was recovered in the feces as unchanged drug. Following oral administration of a 1 mg alosetron dose to young men, a peak plasma concentration of approximately 5 ng/mL occurred at 1 hour. In young women, the mean peak plasma concentration was approximately 9 ng/mL, with a similar time to peak.
Plasma concentrations were 30% to 50% lower and less variable in men compared to women given the same oral dose. Population pharmacokinetic analysis in IBS patients confirmed that alosetron concentrations were influenced by gender (27% lower in men).
14.2 Efficacy Studies
LOTRONEX has been studied in women with IBS in five 12-week US multicenter, randomized, double-blind, placebo-controlled clinical studies.
| Study | Patient Population |
Placebo (n) |
Lotronex Dose (n) |
| 1 and 2 | Non-constipated women with IBS | (640) |
1 mg twice daily (633) |
| 3 and 4 |
Women with severe diarrhea-predominant IBS (defined as bowel urgency ≥50% of days) |
(515) |
1 mg twice daily (778) |
| 5 |
Women with severe diarrhea-predominant IBS (defined as average pain ≥moderate, urgency ≥50% of days, and/or restriction of daily activities ≥25% of days) |
(176) | 0.5 mg once daily
(177) |
| 1 mg once daily
(175) |
|||
| 1 mg once daily
(177) |
7.1 Cyp1a2 Inhibitors (7.1 CYP1A2 Inhibitors)
Fluvoxamine is a known strong inhibitor of CYP1A2 and also inhibits CYP3A4, CYP2C9, and CYP2C19. In a pharmacokinetic study, 40 healthy female subjects received fluvoxamine in escalating doses from 50 to 200 mg/ day for 16 days, with coadministration of alosetron 1 mg on the last day. Fluvoxamine increased mean alosetron plasma concentrations (AUC) approximately 6-fold and prolonged the half-life by approximately 3-fold. Concomitant administration of alosetron and fluvoxamine is contraindicated [see Contraindications ( 4.3)] .
Concomitant administration of alosetron and moderate CYP1A2 inhibitors, including quinolone antibiotics and cimetidine, has not been evaluated, but should be avoided unless clinically necessary because of similar potential drug interactions.
7.2 Cyp3a4 Inhibitors (7.2 CYP3A4 Inhibitors)
Ketoconazole is a known strong inhibitor of CYP3A4. In a pharmacokinetic study, 38 healthy female subjects received ketoconazole 200 mg twice daily for 7 days, with coadministration of alosetron 1 mg on the last day. Ketoconazole increased mean alosetron plasma concentrations (AUC) by 29%. Caution should be used when alosetron and ketoconazole are administered concomitantly. Coadministration of alosetron and strong CYP3A4 inhibitors such as clarithromycin, telithromycin, protease inhibitors, voriconazole, and itraconazole has not been evaluated but should be undertaken with caution because of similar potential drug interactions. The effect of induction or inhibition of other pathways on exposure to alosetron and its metabolites is not known.
7.3 Other Cyp Enzymes (7.3 Other CYP Enzymes)
In vitro human liver microsome studies and an in vivo metabolic probe study demonstrated that alosetron did not inhibit CYP enzymes 3A4, 2C9, or 2C19. In vitro at total drug concentrations 27-fold higher than peak plasma concentrations observed with the 1 mg dose, alosetron inhibited CYP enzymes 1A2 (60%) and 2E1 (50%). In an in vivo metabolic probe study, alosetron did not inhibit CYP2E1 but did produce 30% inhibition of both CYP1A2 and N-acetyltransferase. Although not studied with alosetron, inhibition of N-acetyltransferase may have clinically relevant consequences for drugs such as isoniazid, procainamide, and hydralazine. The effect on CYP1A2 was explored further in a clinical interaction study with theophylline and no effect on metabolism was observed. Another study showed that alosetron had no clinically significant effect on plasma concentrations of the oral contraceptive agents ethinyl estradiol and levonorgestrel (CYP3A4 substrates). A clinical interaction study was also conducted with alosetron and the CYP3A4 substrate cisapride. No significant effects on cisapride metabolism or QT interval were noted. The effects of alosetron on monoamine oxidases and on intestinal first pass secondary to high intraluminal concentrations have not been examined. Based on the above data from in vitro and in vivo studies, it is unlikely that alosetron will inhibit the hepatic metabolic clearance of drugs metabolized by the CYP enzymes 2C9, 2C19, or 2E1.
Alosetron does not appear to induce the major cytochrome P450 drug-metabolizing enzyme 3A. Alosetron also does not appear to induce CYP enzymes 2E1 or 2C19. It is not known whether alosetron might induce other enzymes.
8.6 Hepatic Impairment
Due to the extensive hepatic metabolism of alosetron, increased exposure to alosetron and/or its metabolites is likely to occur in patients with hepatic impairment. Alosetron should not be used in patients with severe hepatic impairment and should be used with caution in patients with mild or moderate hepatic impairment.
A single 1 mg oral dose of alosetron was administered to 1 female and 5 male patients with moderate hepatic impairment (Child-Pugh score of 7 to 9) and to 1 female and 2 male patients with severe hepatic impairment (Child-Pugh score of >9). In comparison with historical data from healthy subjects, patients with severe hepatic impairment displayed higher systemic exposure to alosetron. The female with severe hepatic impairment displayed approximately 14-fold higher exposure, while the female with moderate hepatic impairment displayed approximately 1.6-fold higher exposure, than healthy females. Due to the small number of subjects and high intersubject variability in the pharmacokinetic findings, no definitive quantitative conclusions can be made. However, due to the greater exposure to alosetron in the female with severe hepatic impairment, alosetron should not be used in females with severe hepatic impairment [see Dosage and Administration ( 2.2), Contraindications ( 4)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have:
- chronic IBS symptoms (generally lasting 6 months or longer),
- had anatomic or biochemical abnormalities of the gastrointestinal tract excluded, and
- not responded adequately to conventional therapy.
Diarrhea-predominant IBS is severe if it includes diarrhea and one or more of the following:
- frequent and severe abdominal pain/discomfort,
- frequent bowel urgency or fecal incontinence,
- disability or restriction of daily activities due to IBS.
Because of infrequent but serious gastrointestinal adverse reactions associated with LOTRONEX, the indication is restricted to those patients for whom the benefit-to-risk balance is most favorable.
Clinical studies have not been performed to adequately confirm the benefits of LOTRONEX in men.
14.1 Dose Ranging Study (14.1 Dose-Ranging Study)
Data from a dose-ranging study of women (n = 85) who received LOTRONEX 0.5 mg twice daily indicated that the incidence of constipation (14%) was lower than that experienced by women receiving 1 mg twice daily (29%). Therefore, to lower the risk of constipation, LOTRONEX should be started at a dosage of 0.5 mg twice a day. The efficacy of the 0.5 mg twice-daily dosage in treating severe diarrhea-predominant IBS has not been adequately evaluated in clinical trials. [See Dosage and Administration ( 2.1)]
Clinical Considerations
Monitor infants exposed to LOTRONEX through breast milk for severe constipation and blood in stools.
12.1 Mechanism of Action
Alosetron is a potent and selective 5-HT 3 receptor antagonist. 5-HT 3 receptors are ligand-gated cation channels that are extensively distributed on enteric neurons in the human gastrointestinal tract, as well as other peripheral and central locations. Activation of these channels and the resulting neuronal depolarization affect the regulation of visceral pain, colonic transit, and gastrointestinal secretions, processes that relate to the pathophysiology of IBS. 5-HT 3 receptor antagonists such as alosetron inhibit activation of non-selective cation channels, which results in the modulation of the enteric nervous system.
The cause of IBS is unknown. IBS is characterized by visceral hypersensitivity and hyperactivity of the gastrointestinal tract, which lead to abnormal sensations of pain and motor activity. Following distention of the rectum, patients with IBS exhibit pain and discomfort at lower volumes than healthy volunteers. Following such distention, alosetron reduced pain and exaggerated motor responses, possibly due to blockade of 5-HT 3 receptors.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serious Complications of Constipation: May occur in some patients without warning. Includes obstruction, ileus, impaction, toxic megacolon, and secondary bowel ischemia and in rare cases perforation and death have been reported. Risk is increased in patients who are elderly, debilitated, or taking medications that decrease bowel motility. ( 5.1)
- Discontinue LOTRONEX immediately if constipation occurs. ( 5.1)
- Ischemic colitis: May occur in some patients without warning. Promptly evaluate patients with signs of ischemic colitis (e.g., rectal bleeding, bloody diarrhea, new or worsening abdominal pain). ( 5.2)
- Discontinue LOTRONEX immediately if signs of ischemic colitis occur, such as rectal bleeding, bloody diarrhea, or new or worsening abdominal pain. ( 5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Starting dose is 0.5 mg twice a day ( 2.1)
- May increase dose to 1 mg twice a day after 4 weeks if starting dosage is well tolerated but does not adequately control IBS symptoms ( 2.1)
- Discontinue LOTRONEX in patients who have not had adequate control of IBS symptoms after 4 weeks of treatment with 1 mg twice a day. ( 2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
0.5 mg and 1 mg tablets
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron), are white, oval, film-coated tablets debossed with GX EX1 on one face.
LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face.
6.2 Postmarketing Experience
In addition to events reported in clinical trials, the following events have been identified during use of LOTRONEX in clinical practice. Because they were reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to LOTRONEX.
- Gastrointestinal: Impaction, perforation, ulceration, small bowel mesenteric ischemia.
- Neurological: Headache.
- Skin: Rash.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved labeling (Medication Guide).
4.3 Concomitant Use of Fluvoxamine
Concomitant administration of LOTRONEX with fluvoxamine is contraindicated. Fluvoxamine, a known strong inhibitor of CYP1A2, has been shown to increase mean alosetron plasma concentrations (AUC) approximately 6-fold and prolong the half-life by approximately 3-fold [see Drug Interactions ( 7.1)] .
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
LOTRONEX Tablets, 0.5 mg (0.562 mg alosetron HCl equivalent to 0.5 mg alosetron) are white, oval, film-coated tablets debossed with GX EX1 on one face.
Bottles of 30 (NDC 65483-894-03) with child-resistant closures.
LOTRONEX Tablets, 1 mg (1.124 mg alosetron HCl equivalent to 1 mg alosetron), are blue, oval, film-coated tablets debossed with GX CT1 on one face.
Bottles of 30 (NDC 65483-895-03) with child-resistant closures.
Store at 20-25°C (68-77°F) (USP Controlled Room Temperature). Protect from light and moisture.
2.2 Patients With Hepatic Impairment (2.2 Patients with Hepatic Impairment)
LOTRONEX is extensively metabolized by the liver, and increased exposure to LOTRONEX is likely to occur in patients with hepatic impairment. Increased drug exposure may increase the risk of serious adverse reactions. LOTRONEX should be used with caution in patients with mild or moderate hepatic impairment and is contraindicated in patients with severe hepatic impairment [see Contraindications ( 4), Use in Specific Populations ( 8.6)].
5.1 Serious Complications of Constipation
Some patients have experienced serious complications of constipation without warning.
Serious complications of constipation, including obstruction, ileus, impaction, toxic megacolon, and secondary bowel ischemia, have been reported with use of LOTRONEX during clinical trials. Complications of constipation have been reported with use of 1 mg twice daily and with lower doses. A dose response relationship has not been established for serious complications of constipation. The incidence of serious complications of constipation was approximately 0.1% (1 per 1,000 patients) in women receiving either LOTRONEX or placebo. In addition, rare cases of perforation and death have been reported from postmarketing clinical practice. In some cases, complications of constipation required intestinal surgery, including colectomy. Patients who are elderly, debilitated, or taking additional medications that decrease gastrointestinal motility may be at greater risk for complications of constipation.
LOTRONEX should be discontinued immediately in patients who develop constipation [see Boxed Warning].
4.2 History of Severe Bowel Or Hepatic Disorders (4.2 History of Severe Bowel or Hepatic Disorders)
LOTRONEX is contraindicated in patients with a history of the following:
- chronic or severe constipation or sequelae from constipation
- intestinal obstruction, stricture, toxic megacolon, gastrointestinal perforation, and/or adhesions
- ischemic colitis, impaired intestinal circulation, thrombophlebitis, or hypercoagulable state
- Crohn's disease or ulcerative colitis
- diverticulitis
- severe hepatic impairment
Warning: Serious Gastrointestinal Adverse Reactions (WARNING: SERIOUS GASTROINTESTINAL ADVERSE REACTIONS)
Infrequent but serious gastrointestinal adverse reactions have been reported with the use of LOTRONEX. These events, including ischemic colitis and serious complications of constipation, have resulted in hospitalization, and rarely, blood transfusion, surgery, and death.
- LOTRONEX is indicated only for women with severe diarrhea-predominant irritable bowel syndrome (IBS) who have not responded adequately to conventional therapy [see Indications and Usage ( 1)] .
- LOTRONEX should be discontinued immediately in patients who develop constipation or symptoms of ischemic colitis. Patients should immediately report constipation or symptoms of ischemic colitis to their prescriber. LOTRONEX should not be resumed in patients who develop ischemic colitis. Patients who have constipation should immediately contact their prescriber if the constipation does not resolve after LOTRONEX is discontinued. Patients with resolved constipation should resume LOTRONEX only on the advice of their treating prescriber [see Dosage and Administration ( 2.1), Warnings and Precautions ( 5.1), ( 5.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In 2-year oral studies, alosetron was not carcinogenic in mice at doses up to 30 mg/kg/day or in rats at doses up to 40 mg/kg/day. These doses are about 60 to 160 times, respectively, the recommended human dose of alosetron of 2 mg/day (1 mg twice daily) based on body surface area. Alosetron was not genotoxic in the Ames tests, the mouse lymphoma cell (L5178Y/TK ±) forward gene mutation test, the human lymphocyte chromosome aberration test, the ex vivo rat hepatocyte unscheduled DNA synthesis (UDS) test, or the in vivo rat micronucleus test for mutagenicity. Alosetron at oral doses up to 40 mg/kg/day (about 160 times the recommended daily human dose based on body surface area) was found to have no effect on fertility and reproductive performance of male or female rats.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:40.697378 · Updated: 2026-03-14T22:49:18.583726