3557859e-f8b3-1049-e063-6394a90ad737
34390-5
HUMAN OTC DRUG LABEL
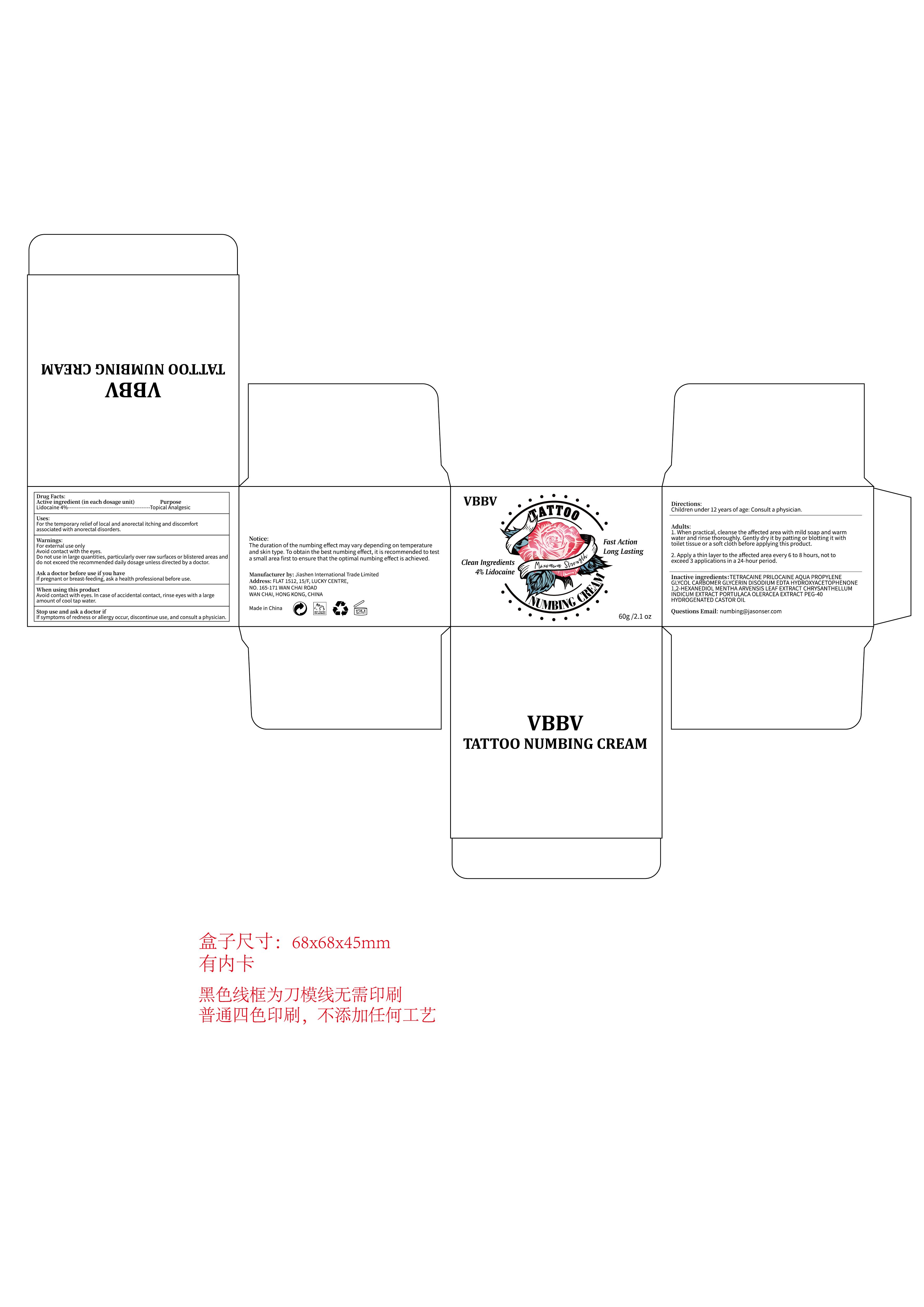
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%
Medication Information
Warnings and Precautions
Warnings:For external use onlyAvoid contact with the eyes.Do not use in large quantities, particularly over raw surfaces or blistered areas anddo not exceed the recommended daily dosage unless directed by a doctor.
Indications and Usage
Directions:
Children under 12 years of age: Consult a physician.
Dosage and Administration
Adults:1. When practical, cleanse the affected area with mild soap and warmwater and rinse thoroughly. Gently dry it by patting or blotting it withtoilet tissue or a soft cloth before applying this product.
2. Apply a thin layer to the affected area every 6 to 8 hours, not toexceed 3 applications in a 24-hour period.
Description
Lidocaine 4%
Section 50565-1
Children under 12 years of age: Consult a physician. keep out of reach of children
Section 50566-9
Stop use and ask a doctor if
lf symptoms of redness or allergy occur, discontinue use, and consult a physician.
Section 50567-7
When using this productAvoid contact with eyes. In case of accidental contact, rinse eyes with a largeamount of cool tap water.
Section 50569-3
Ask a doctor before use if you havelf pregnant or breast-feeding, ask a health professional before use.
Section 51727-6
Inactive ingredientS:TETRACAINE PRILOCAINE AQUA PROPYLENEGLYCOL CARBOMER GLYCERIN DISODIUM EDTA HYDROXYACETOPHENONE1,2-HEXANEDIOL MENTHA ARVENSIS LEAF EXTRACT CHRYSANTHELLUMINDICUM EXTRACT PORTULACA OLERACEA EXTRACT PEG-40HYDROGENATED CASTOR OIL
Section 51945-4
Section 55105-1
Uses:For the temporary relief of local and anorectal itching and discomfortassociated with anorectal disorders.
Section 55106-9
Lidocaine 4%
Structured Label Content
Indications and Usage (34067-9)
Directions:
Children under 12 years of age: Consult a physician.
Dosage and Administration (34068-7)
Adults:1. When practical, cleanse the affected area with mild soap and warmwater and rinse thoroughly. Gently dry it by patting or blotting it withtoilet tissue or a soft cloth before applying this product.
2. Apply a thin layer to the affected area every 6 to 8 hours, not toexceed 3 applications in a 24-hour period.
Warnings and Precautions (34071-1)
Warnings:For external use onlyAvoid contact with the eyes.Do not use in large quantities, particularly over raw surfaces or blistered areas anddo not exceed the recommended daily dosage unless directed by a doctor.
Section 50565-1 (50565-1)
Children under 12 years of age: Consult a physician. keep out of reach of children
Section 50566-9 (50566-9)
Stop use and ask a doctor if
lf symptoms of redness or allergy occur, discontinue use, and consult a physician.
Section 50567-7 (50567-7)
When using this productAvoid contact with eyes. In case of accidental contact, rinse eyes with a largeamount of cool tap water.
Section 50569-3 (50569-3)
Ask a doctor before use if you havelf pregnant or breast-feeding, ask a health professional before use.
Section 51727-6 (51727-6)
Inactive ingredientS:TETRACAINE PRILOCAINE AQUA PROPYLENEGLYCOL CARBOMER GLYCERIN DISODIUM EDTA HYDROXYACETOPHENONE1,2-HEXANEDIOL MENTHA ARVENSIS LEAF EXTRACT CHRYSANTHELLUMINDICUM EXTRACT PORTULACA OLERACEA EXTRACT PEG-40HYDROGENATED CASTOR OIL
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Uses:For the temporary relief of local and anorectal itching and discomfortassociated with anorectal disorders.
Section 55106-9 (55106-9)
Lidocaine 4%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:08.616397 · Updated: 2026-03-14T23:08:46.104923