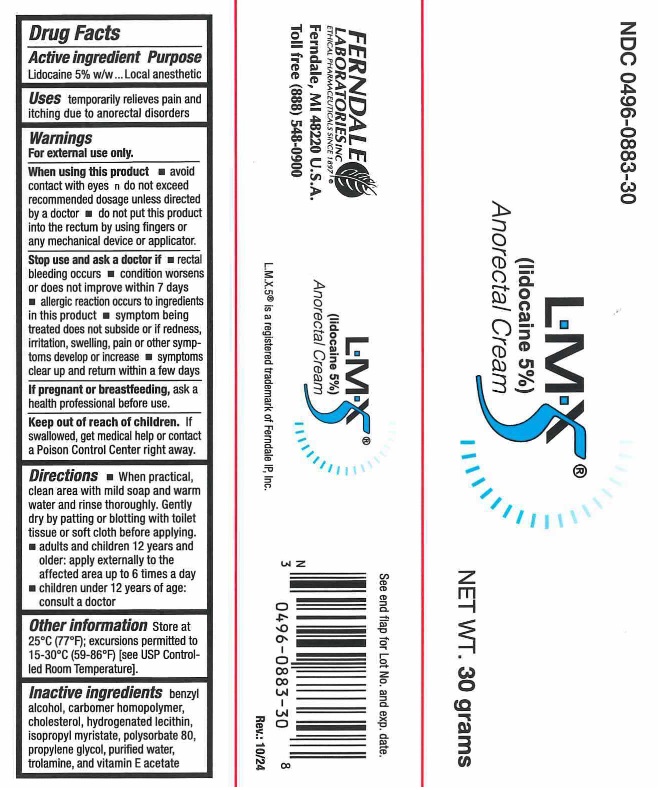
Lmx5 (lidocaine 5%) Anorectal Cream
34f8ed6b-4a52-4a99-9c1b-254196dc29b0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 5% w/w
Purpose
Local anesthetic
Medication Information
Purpose
Local anesthetic
Description
Lidocaine 5% w/w
Uses
temporarily relieves pain and itching due to anorectal disorders
Warnings
Directions
- When practical, clean area with mild soap and warm water and rinse thoroughly. Gently dry by patting or blotting with toilet tissue or soft cloth before applying.
- Adults and Children 12 years and older: Apply to the affected area up to 6 times a day.
- Children under 12 years of age: Consult a doctor.
Package Label
Manufactured for Ferndale Healthcare Inc.
Ferndale, MI 48220 U.S.A.
Toll free (888) 548-0900
www.ferndalehealthcare.com
L.M.X.5® is a registered trademark of Ferndale, IP Inc.
30 gram NDC 0496-0883-30
Active Ingredient
Lidocaine 5% w/w
Other Information
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Inactive Ingredients
benzyl alcohol, carbomer homopolymer, chloesterol, hydrogenated lecithin, isopropyl myristate, polysorbate 80, propylene glycol, purified water, trolamine, and vitamin E acetate
When Using This Product
- avoid contact with eyes
- do not exceed recommended dosage unless directed by a doctor
Stop Use and Ask A Doctor If
- rectal bleeding occurs
- condition worsens or does not improve within 7 days
- allergic reaction occurs
- redness, irritation, swelling, pain or other symptoms begin or increase
- symptoms clear up and return within a few days
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
temporarily relieves pain and itching due to anorectal disorders
Purpose
Local anesthetic
Warnings
Directions
- When practical, clean area with mild soap and warm water and rinse thoroughly. Gently dry by patting or blotting with toilet tissue or soft cloth before applying.
- Adults and Children 12 years and older: Apply to the affected area up to 6 times a day.
- Children under 12 years of age: Consult a doctor.
Package Label
Manufactured for Ferndale Healthcare Inc.
Ferndale, MI 48220 U.S.A.
Toll free (888) 548-0900
www.ferndalehealthcare.com
L.M.X.5® is a registered trademark of Ferndale, IP Inc.
30 gram NDC 0496-0883-30
Active Ingredient (Active ingredient)
Lidocaine 5% w/w
Other Information (Other information)
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Inactive Ingredients
benzyl alcohol, carbomer homopolymer, chloesterol, hydrogenated lecithin, isopropyl myristate, polysorbate 80, propylene glycol, purified water, trolamine, and vitamin E acetate
When Using This Product (When using this product)
- avoid contact with eyes
- do not exceed recommended dosage unless directed by a doctor
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- rectal bleeding occurs
- condition worsens or does not improve within 7 days
- allergic reaction occurs
- redness, irritation, swelling, pain or other symptoms begin or increase
- symptoms clear up and return within a few days
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:45.145635 · Updated: 2026-03-14T23:11:30.674942