34156655-8706-f0a4-e063-6294a90acc90
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
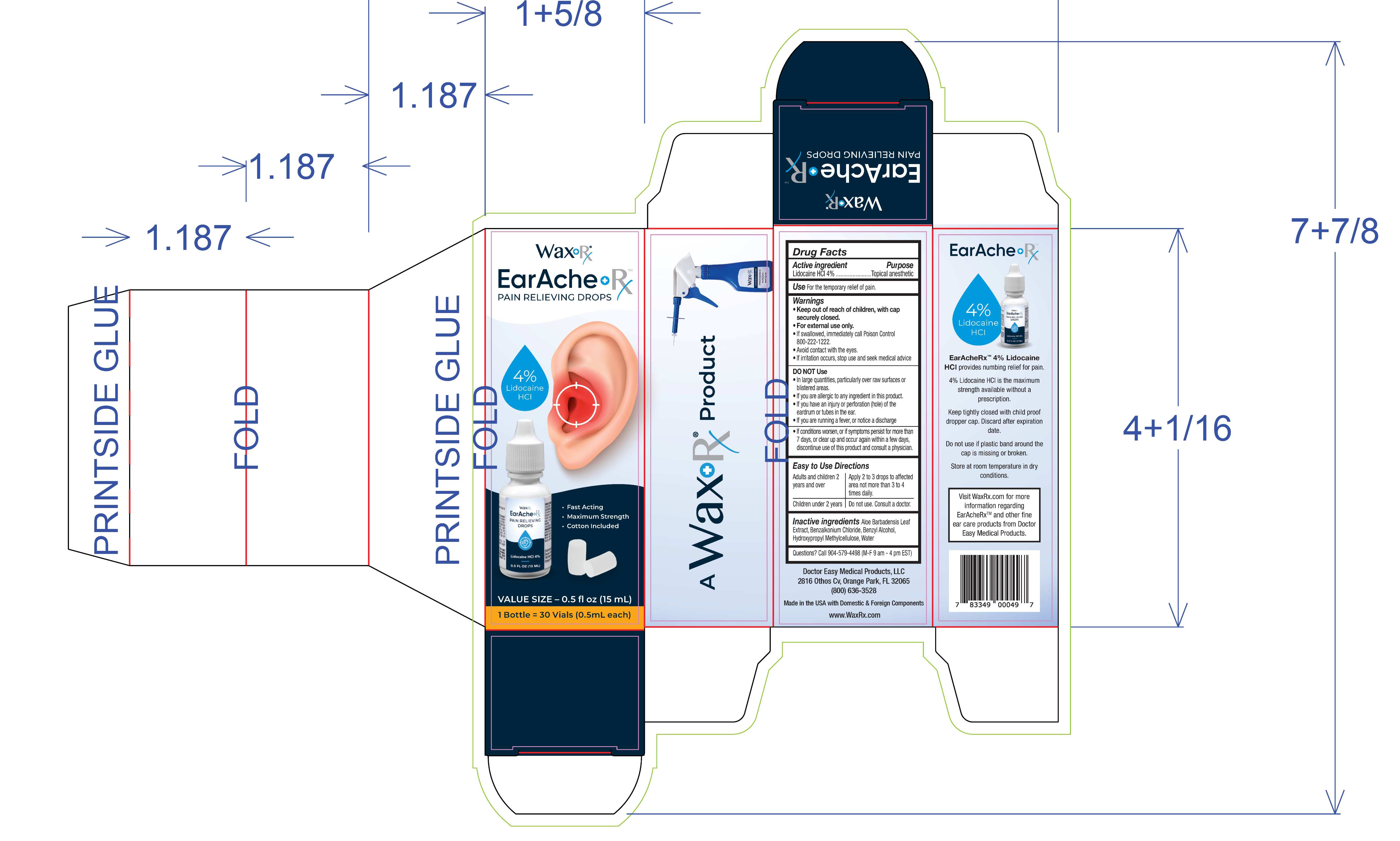
Identifiers & Packaging
Description
Lidocaine HCL 4%
Medication Information
Warnings and Precautions
- Keep out of reach of children, with cap securely closed.
- For external use only.
- If swallowed, immediately call Poison Control 800-222-1222
- Avoid contact with the eyes.
- If irritation occurs, stop and seek medical advice
Indications and Usage
For the temporary relief of pain.
Description
Lidocaine HCL 4%
Section 50565-1
Keep out of reach of children, with cap securely closed.
If swallowed, immediately call Poison Control 800-222-1222
Section 50566-9
DO NOT Use
- In large quantities, particularly over raw surfaces or blistered areas.
- If you are allergic to any ingredient in this product.
- If you have an injury or perforation (hole) of the eardrum or tubes in the ear.
- If you are running a fever, or notice a discharge.
- If conditions worsen, or if symptoms persist for more than 7 days, or clear up and occur again within a few days, discontinue use of this product and consult a physician.
Section 51727-6
Aloe Barbadensis Leaf Extract, Benzalkonium Chloride, Benzyl Alcohol, Hydroxypropyl Methycellulose, Water
Section 51945-4
WaxRx
EarAcheRx
Pain Relieving Drops
4% Lidocaine HC
0.5 FL OZ (15 ML)
Section 55105-1
Lidocaine HCL 4%...................Topical Anesthetic
Section 55106-9
Lidocaine HCL 4%
Easy to Use Directions
Adults and children 2 years and over
Apply 2 to 3 drops to affected area not more than 3 to 4 times daily.
Children under 2 years Consult a doctor.
Structured Label Content
Indications and Usage (34067-9)
For the temporary relief of pain.
Warnings and Precautions (34071-1)
- Keep out of reach of children, with cap securely closed.
- For external use only.
- If swallowed, immediately call Poison Control 800-222-1222
- Avoid contact with the eyes.
- If irritation occurs, stop and seek medical advice
Section 50565-1 (50565-1)
Keep out of reach of children, with cap securely closed.
If swallowed, immediately call Poison Control 800-222-1222
Section 50566-9 (50566-9)
DO NOT Use
- In large quantities, particularly over raw surfaces or blistered areas.
- If you are allergic to any ingredient in this product.
- If you have an injury or perforation (hole) of the eardrum or tubes in the ear.
- If you are running a fever, or notice a discharge.
- If conditions worsen, or if symptoms persist for more than 7 days, or clear up and occur again within a few days, discontinue use of this product and consult a physician.
Section 51727-6 (51727-6)
Aloe Barbadensis Leaf Extract, Benzalkonium Chloride, Benzyl Alcohol, Hydroxypropyl Methycellulose, Water
Section 51945-4 (51945-4)
WaxRx
EarAcheRx
Pain Relieving Drops
4% Lidocaine HC
0.5 FL OZ (15 ML)
Section 55105-1 (55105-1)
Lidocaine HCL 4%...................Topical Anesthetic
Section 55106-9 (55106-9)
Lidocaine HCL 4%
Easy to Use Directions
Adults and children 2 years and over
Apply 2 to 3 drops to affected area not more than 3 to 4 times daily.
Children under 2 years Consult a doctor.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:02.405568 · Updated: 2026-03-14T23:08:30.723878