These Highlights Do Not Include All The Information Needed To Use Cotempla Xr-odt Safely And Effectively. See Full Prescribing Information For Cotempla Xr-odt.
33f70f58-c871-42c8-8adb-345caeafefcd
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
COTEMPLA XR-ODT has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death (5.1, 9.2, 10). Before prescribing COTEMPLA XR-ODT, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout treatment, reassess each patient’s risk and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
Indications and Usage
COTEMPLA XR-ODT is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in pediatric patients 6 to 17 years of age [see Clinical Studies (14) ]. Limitations of Use The use of COTEMPLA XR-ODT is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.7). Use in Specific Populations (8.4)] .
Dosage and Administration
Recommended starting dose for pediatric patients 6 to 17 years of age is 17.3 mg given orally once daily in the morning. Dosage may be increased weekly in increments of 8.6 mg to 17.3 mg per day. Daily dosage above 51.8 mg is not recommended. ( 2.2 ) Patients are advised to take COTEMPLA XR-ODT consistently either with food or without food. ( 2.2 )
Warnings and Precautions
.
Contraindications
COTEMPLA XR-ODT is contraindicated in patients with: Known hypersensitivity to methylphenidate or other components of COTEMPLA XR-ODT. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.2) ] . Concomitant treatment with monoamine oxidase inhibitors (MAOIs), and also within a minimum of 14 days following discontinuation of treatment with a monoamine oxidase inhibitor because of the risk of hypertensive crisis [see Drug Interactions (7.1) ] .
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Known hypersensitivity to methylphenidate or other ingredients of Cotempla XR-ODT [see Contraindications (4) ] Hypertensive crisis when used concomitantly with monoamine oxidase inhibitors [see Contraindications (4) and Drug Interactions (7.1) ] Abuse, Misuse, and Addiction [see Boxed Warning , Warnings and Precautions (5.1) , and Drug Abuse and Dependence (9.2 , 9.3) ] Risks to patients with serious cardiac disease [see Warnings and Precautions (5.2) ] Increased blood pressure and heart rate [see Warnings and Precautions (5.3) ] Psychiatric adverse reactions [see Warnings and Precautions (5.4) ] Priapism [see Warnings and Precautions (5.5) ] Peripheral vasculopathy, including Raynaud's phenomenon [see Warnings and Precautions (5.6) ] Long-term suppression of growth in pediatric patients [see Warnings and Precautions (5.7) ] Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8)] Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9)] Motor and Verbal Tics, and Worsening of Tourette’s Syndrome [ see Warnings and Precautions (5.10)]
Drug Interactions
Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. ( 7 )
Storage and Handling
COTEMPLA XR-ODT Extended Release Orally Disintegrating Tablets are available in three strengths: 8.6 mg tablets, round, purple to light purple, mottled, and debossed "T1" on one side of the tablet; 17.3 mg tablets, round, purple to light purple, mottled, and debossed "T2" on one side of the tablet; 25.9 mg tablets, round, purple to light purple, mottled, and debossed "T3" on one side of the tablet. They are available as follows: NDC 70165-100-30 8.6 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-200-30 17.3 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-300-30 25.9 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case.
How Supplied
COTEMPLA XR-ODT Extended Release Orally Disintegrating Tablets are available in three strengths: 8.6 mg tablets, round, purple to light purple, mottled, and debossed "T1" on one side of the tablet; 17.3 mg tablets, round, purple to light purple, mottled, and debossed "T2" on one side of the tablet; 25.9 mg tablets, round, purple to light purple, mottled, and debossed "T3" on one side of the tablet. They are available as follows: NDC 70165-100-30 8.6 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-200-30 17.3 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-300-30 25.9 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case.
Medication Information
Warnings and Precautions
.
Indications and Usage
COTEMPLA XR-ODT is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in pediatric patients 6 to 17 years of age [see Clinical Studies (14) ]. Limitations of Use The use of COTEMPLA XR-ODT is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.7). Use in Specific Populations (8.4)] .
Dosage and Administration
Recommended starting dose for pediatric patients 6 to 17 years of age is 17.3 mg given orally once daily in the morning. Dosage may be increased weekly in increments of 8.6 mg to 17.3 mg per day. Daily dosage above 51.8 mg is not recommended. ( 2.2 ) Patients are advised to take COTEMPLA XR-ODT consistently either with food or without food. ( 2.2 )
Contraindications
COTEMPLA XR-ODT is contraindicated in patients with: Known hypersensitivity to methylphenidate or other components of COTEMPLA XR-ODT. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.2) ] . Concomitant treatment with monoamine oxidase inhibitors (MAOIs), and also within a minimum of 14 days following discontinuation of treatment with a monoamine oxidase inhibitor because of the risk of hypertensive crisis [see Drug Interactions (7.1) ] .
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Known hypersensitivity to methylphenidate or other ingredients of Cotempla XR-ODT [see Contraindications (4) ] Hypertensive crisis when used concomitantly with monoamine oxidase inhibitors [see Contraindications (4) and Drug Interactions (7.1) ] Abuse, Misuse, and Addiction [see Boxed Warning , Warnings and Precautions (5.1) , and Drug Abuse and Dependence (9.2 , 9.3) ] Risks to patients with serious cardiac disease [see Warnings and Precautions (5.2) ] Increased blood pressure and heart rate [see Warnings and Precautions (5.3) ] Psychiatric adverse reactions [see Warnings and Precautions (5.4) ] Priapism [see Warnings and Precautions (5.5) ] Peripheral vasculopathy, including Raynaud's phenomenon [see Warnings and Precautions (5.6) ] Long-term suppression of growth in pediatric patients [see Warnings and Precautions (5.7) ] Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8)] Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9)] Motor and Verbal Tics, and Worsening of Tourette’s Syndrome [ see Warnings and Precautions (5.10)]
Drug Interactions
Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. ( 7 )
Storage and Handling
COTEMPLA XR-ODT Extended Release Orally Disintegrating Tablets are available in three strengths: 8.6 mg tablets, round, purple to light purple, mottled, and debossed "T1" on one side of the tablet; 17.3 mg tablets, round, purple to light purple, mottled, and debossed "T2" on one side of the tablet; 25.9 mg tablets, round, purple to light purple, mottled, and debossed "T3" on one side of the tablet. They are available as follows: NDC 70165-100-30 8.6 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-200-30 17.3 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-300-30 25.9 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case.
How Supplied
COTEMPLA XR-ODT Extended Release Orally Disintegrating Tablets are available in three strengths: 8.6 mg tablets, round, purple to light purple, mottled, and debossed "T1" on one side of the tablet; 17.3 mg tablets, round, purple to light purple, mottled, and debossed "T2" on one side of the tablet; 25.9 mg tablets, round, purple to light purple, mottled, and debossed "T3" on one side of the tablet. They are available as follows: NDC 70165-100-30 8.6 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-200-30 17.3 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. NDC 70165-300-30 25.9 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case.
Description
COTEMPLA XR-ODT has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death (5.1, 9.2, 10). Before prescribing COTEMPLA XR-ODT, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout treatment, reassess each patient’s risk and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
Section 42229-5
Exacerbation of Pre-Existing Psychosis
CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a pre-existing psychotic disorder.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 09/2025 | ||
|
Medication Guide
COTEMPLA XR-ODT (koh-TEM-pluh - oh dee tee) (methylphenidate) extended-release orally disintegrating tablets, CII |
|||
|
What is the most important information I should know about COTEMPLA XR-ODT? |
|||
COTEMPLA XR-ODT may cause serious side effects, including:
Tell your healthcare provider if your child has ever abused or been dependent on alcohol, prescription medicines, or street drugs.
Your healthcare provider should check your child carefully for heart problems before starting COTEMPLA XR-ODT. Tell your healthcare provider if your child has any heart problems, heart disease, or heart defects. Call your healthcare provider or go to the nearest hospital emergency room right away if your child has any signs of heart problems such as chest pain, shortness of breath, or fainting during treatment with COTEMPLA XR-ODT.
Your healthcare provider should check your child's blood pressure and heart rate regulary during treatment with COTEMPLA XR-ODT.
Tell your healthcare provider about any mental problems your child has, or about a family history of suicide, bipolar illness, or depression.
|
|||
| What is COTEMPLA XR-ODT? | |||
| COTEMPLA XR-ODT is a central nervous system (CNS) stimulant prescription medicine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in children 6 to 17 years of age. COTEMPLA XR-ODT may help increase attention and decrease impulsiveness and hyperactivity in children 6 to 17 years of age with ADHD. | |||
| COTEMPLA XR-ODT is not recommended for use in children under 6 years of age with ADHD. | |||
| COTEMPLA XR-ODT is a federally controlled substance (CII) because it contains methylphenidate that can be a target for people who abuse prescription medicines or street drugs. Keep COTEMPLA XR-ODT in a safe place to protect it from theft. Never give your COTEMPLA XR-ODT to anyone else, because it may cause death or harm them. Selling or giving away COTEMPLA XR-ODT may harm others and is against the law. | |||
Do not give COTEMPLA XR-ODT to your child if they are:
|
|||
Before taking COTEMPLA XR-ODT tell your child's healthcare provider about all medical conditions, including if your child:
|
|||
| Tell your healthcare provider about all of the medicines that your child takes, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| COTEMPLA XR-ODT and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be adjusted during treatment with COTEMPLA XR-ODT. | |||
Your healthcare provider will decide whether COTEMPLA XR-ODT can be taken with other medicines.
Especially tell your healthcare provider if your child takes:
|
|||
| Know the medicines that your child takes. Keep a list of the medicines with you to show your healthcare provider and pharmacist. Do not start any new medicine during treatment with COTEMPLA XR-ODT without talking to your healthcare provider first. | |||
How should COTEMPLA XR-ODT be taken?
|
|||
| Take COTEMPLA XR-ODT as follows: | |||
|
|||
|
If your child takes too much COTEMPLA XR-ODT, call your healthcare provider or Poison Help Line at 1-800-222-1222 or go to the nearest hospital emergency room right away. |
|||
| What should I avoid during treatment with COTEMPLA XR-ODT? | |||
| You should avoid drinking alcohol during treatment with COTEMPLA XR-ODT. | |||
| What are possible side effects of COTEMPLA XR-ODT? | |||
COTEMPLA XR-ODT may cause serious side effects, including:
|
|||
| The most common side effects of methylphenidate products include: | |||
|
|
||
| These are not all the possible side effects of COTEMPLA XR-ODT. | |||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
How should I store COTEMPLA XR-ODT?
|
|||
| Keep COTEMPLA XR-ODT and all medicines out of the reach of children. | |||
| General information about the safe and effective use of COTEMPLA XR-ODT | |||
| Medicines are sometimes prescribed for purposes other than those listed in the Medication Guide. Do not use COTEMPLA XR-ODT for a condition for which it was not prescribed. Do not give COTEMPLA XR-ODT to other people, even if they have the same condition. It may harm them and it is against the law. You can ask your healthcare provider or pharmacist for information about COTEMPLA XR-ODT that is written for healthcare professionals. | |||
| What are the ingredients in COTEMPLA XR-ODT? | |||
| Active Ingredient: Methylphenidate | |||
| Inactive Ingredients: Mannitol, Fructose, Microcrystalline Cellulose, Crospovidone, Methacrylic Acid, Polystyrene Sulfonate, Citric Acid, Colloidal Silicon Dioxide, Grape Flavor, Natural Masking Type Powder, Triethyl Citrate, Magnesium Stearate, Ethylcellulose, Sucralose, Lake Blend Purple, and Polyethylene Glycol 3350 | |||
| Manufactured for Neos Therapeutics Brands, LLC, Denver, CO 80237 | |||
| For more information go to https://linkprotect.cudasvc.com/url?a=http%3a%2f%2fwww.COTEMPLAXRODT.com&c=E,1,uJnpRo76RJYGQDo8GMW8zCnFW9Wn0k16nND89VJMYg7anbFT-Thnek2P1Uts7soE5kxS-99rv0KJLMK4Fi0tUhQLe2IT7HZHVjJZmOQANFuP3n-FX1c,&typo=1 or call 1-888-319-1789 | |||
| COTEMPLA XR-ODT is registered in the US Patent and Trademark Office © 2014 Neos Therapeutics, Inc. |
Section 43683-2
Indications and Usage (1) 09/2025
Warnings and Precautions (5.7) 09/2025
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F); excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Store COTEMPLA XR-ODT blister packages in the reusable travel case after removal from the carton.
9.2 Abuse
COTEMPLA XR-ODT has a high potential for abuse and misuse which can lead to the development of a substance use disorder, including addiction [see Warnings and Precautions (5.1)]. COTEMPLA XR-ODT can be diverted for non-medical use into illicit channels or distribution.
Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of methylphenidate may cause increased heart rate, respiratory rate, or blood pressure; sweating; dilated pupils; hyperactivity; restlessness; insomnia; decreased appetite; loss of coordination; tremors; flushed skin; vomiting; and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed with CNS stimulants abuse and/or misuse. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
5.5 Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate use in both adult and pediatric male patients. Although priapism was not reported with methylphenidate initiation, it developed after some time on methylphenidate, often subsequent to an increase in dosage. Priapism also occurred during methylphenidate withdrawal (drug holidays or during discontinuation).
COTEMPLA XR-ODT-treated patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
11 Description
COTEMPLA XR-ODT contains methylphenidate, a central nervous system (CNS) stimulant. COTEMPLA XR-ODT is an extended-release orally disintegrating tablet intended for once daily administration. COTEMPLA XR-ODT contains approximately 25% immediate-release and 75% extended-release methylphenidate. Methylphenidate is ionically-bound to the sulfonate of polystyrene sulfonate particles.
COTEMPLA XR-ODT contains 8.6 mg, 17.3 mg or 25.9 mg of methylphenidate which is the same as the amount of methylphenidate (base equivalent) found, respectively, in 10 mg, 20 mg and 30 mg strength methylphenidate hydrochloride products.
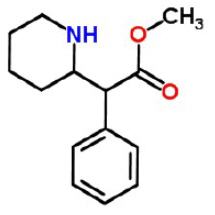
The chemical name of methylphenidate is methyl α-phenyl-2-piperidineacetate, and its structural formula is shown in Figure 1.
Figure 1: Methylphenidate Structure
C 14H 19NO 2 Mol. Wt. 233.31
COTEMPLA XR-ODT also contains the following inactive ingredients: Mannitol, Fructose, Microcrystalline Cellulose, Crospovidone, Methacrylic Acid, Polystyrene Sulfonate, Citric Acid, Colloidal Silicon Dioxide, Grape Flavor, Natural Masking Type Powder, Triethyl Citrate, Magnesium Stearate, Ethylcellulose, Sucralose, Lake Blend Purple, and Polyethylene Glycol 3350.
8.4 Pediatric Use
The safety and effectiveness of COTEMPLA XR-ODT have not been established in pediatric patients below the age of 6 years.
In studies evaluating extended-release methylphenidate products, patients 4 to <6 years of age had higher systemic methylphenidate exposures than those observed in older pediatric patients at the same dosage. Pediatric patients 4 to <6 years of age also had a higher incidence of adverse reactions, including weight loss.
The safety and effectiveness of COTEMPLA XR-ODT have been established in pediatric patients 6 to 17 years of age in one adequate and well-controlled study in pediatric patients 6 to 12 years, pharmacokinetic data in adolescents, and safety information from other methyphenidate-containing products [see Clinical Pharmacology (12) and Clinical Studies (14)] .
8.5 Geriatric Use
COTEMPLA XR-ODT has not been studied in patients over the age of 65 years.
14 Clinical Studies
The efficacy of COTEMPLA XR-ODT was evaluated in a laboratory classroom study conducted in 87 pediatric patients (Aged 6 to 12 years) with ADHD. Following washout of previous methylphenidate medication, there was an open-label dose-optimization period (4 weeks) with an initial dose of 17.3 mg of COTEMPLA XR-ODT once daily in the morning. The dose could be titrated on a weekly basis from 17.3 mg, to 25.9 mg, to 34.6 mg, and up to 51.8 mg until an optimal dose or the maximum dose of 51.8 mg/day was reached. At the end of this period, subjects remained on their optimized dose for an additional week. Subjects then entered a 1-week randomized, double-blind, parallel group treatment period with the individually optimized dose of COTEMPLA XR-ODT or placebo. At the end of this week, raters evaluated the attention and behavior of the subjects in a laboratory classroom setting, using the Swanson, Kotkin, Agler, M-Flynn, and Pelham (SKAMP) rating scale SKAMP is a validated 13-item teacher-rated scale that assesses manifestations of ADHD in a classroom setting.
The primary efficacy endpoint was the average of the SKAMP-Combined (Attention and Deportment) scores over the test day (not including the baseline score), with assessments conducted at baseline, and 1, 3, 5, 7, 10, 12, and 13 hours post-dosing. The key secondary efficacy endpoints were onset and duration of effect, defined as the first point at which active drug separated from placebo on SKAMP-Combined scores and the last time point at which active drug separated from placebo on SKAMP-Combined scores, respectively.
The SKAMP-Combined scores test day average was statistically significantly lower (improved) with COTEMPLA XR-ODT compared to placebo (difference of -11 (95% CI: -13.9, -8.2)) (Table 3).
| Study Number | Treatment Group | Baseline Score at Randomization a (SD) | Pre-dose Score on Classroom Day b (SD) | LS Mean c (SE) | Placebo-subtracted Difference d (95% CI) |
|---|---|---|---|---|---|
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: confidence interval.
a Visit 7 baseline score (Visit 7 occurred prior to the 1-week randomized, double-blind, parallel group treatment period). b Visit 8 baseline score (Visit 8 occurred at the end of the 1-week randomized, double-blind, parallel group treatment period). c Visit 8 LS mean over hours 1, 3, 5, 7, 10, 12, and 13. d Difference (drug minus placebo) in least-squares means. |
|||||
| Study 1 | Cotempla XR-ODT
(17.3-51.8 mg/day) |
21.1 (9.56) | 26.8 (11.52) | 14.3 (1.07) | -11.0 (-13.9, -8.2) |
| Placebo | 20.4 (9.09) | 19.1 (11.04) | 25.3 (1.16) | -- |
The SKAMP-Combined scores were also statistically significantly lower (improved) at time points (1, 3, 5, 7, 10, 12 hours) post-dosing with COTEMPLA XR-ODT compared to placebo (Figure 3).
Figure 3: LS Mean SKAMP Combined Score After Treatment with COTEMPLA XR-ODT or Placebo During Classroom Day in Patients with ADHD
*SE = Standard Error
The database was not large enough to assess whether there were differences in effects in age, gender, or race subgroups.
4 Contraindications
COTEMPLA XR-ODT is contraindicated in patients with:
- Known hypersensitivity to methylphenidate or other components of COTEMPLA XR-ODT. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.2)] .
- Concomitant treatment with monoamine oxidase inhibitors (MAOIs), and also within a minimum of 14 days following discontinuation of treatment with a monoamine oxidase inhibitor because of the risk of hypertensive crisis [see Drug Interactions (7.1)] .
6 Adverse Reactions
The following are discussed in more detail in other sections of the labeling:
- Known hypersensitivity to methylphenidate or other ingredients of Cotempla XR-ODT [see Contraindications (4)]
- Hypertensive crisis when used concomitantly with monoamine oxidase inhibitors [see Contraindications (4) and Drug Interactions (7.1)]
- Abuse, Misuse, and Addiction [see Boxed Warning, Warnings and Precautions (5.1), and Drug Abuse and Dependence (9.2, 9.3)]
- Risks to patients with serious cardiac disease [see Warnings and Precautions (5.2)]
- Increased blood pressure and heart rate [see Warnings and Precautions (5.3)]
- Psychiatric adverse reactions [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.5)]
- Peripheral vasculopathy, including Raynaud's phenomenon [see Warnings and Precautions (5.6)]
- Long-term suppression of growth in pediatric patients [see Warnings and Precautions (5.7)]
- Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8)]
- Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9)]
- Motor and Verbal Tics, and Worsening of Tourette’s Syndrome [ see Warnings and Precautions (5.10)]
7 Drug Interactions
- Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. ( 7)
12.2 Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d- and 1-isomers. The d-isomer is more pharmacologically active than the l-isomer. Methylphenidate is thought to block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
After oral administration of COTEMPLA XR-ODT, circulation levels of l- methylphenidate (MPH) were about 2% of total MPH.
1 Indications and Usage
COTEMPLA XR-ODT is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in pediatric patients 6 to 17 years of age [see Clinical Studies (14)].
Limitations of Use
The use of COTEMPLA XR-ODT is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.7). Use in Specific Populations (8.4)].
12.1 Mechanism of Action
Methylphenidate is a central nervous system (CNS) stimulant. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
COTEMPLA XR-ODT contains methylphenidate, a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to treating patients with COTEMPLA XR-ODT, assess:
- for the presence of cardiac disease (i.e. perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2)] .
- the family history and clinically evaluate patients for motor or verbal tics or Tourette's sydrome before initiating COTEMPLA XR-ODT [see Warnings and Precautions (5.10)].
5 Warnings and Precautions
.
2 Dosage and Administration
- Recommended starting dose for pediatric patients 6 to 17 years of age is 17.3 mg given orally once daily in the morning. Dosage may be increased weekly in increments of 8.6 mg to 17.3 mg per day. Daily dosage above 51.8 mg is not recommended. ( 2.2)
- Patients are advised to take COTEMPLA XR-ODT consistently either with food or without food. ( 2.2)
3 Dosage Forms and Strengths
- 8.6 mg extended-release orally disintegrating tablet: round, purple to light purple mottled (debossed "T1" on one side and plain on the other)
- 17.3 mg extended-release orally disintegrating tablet: round, purple to light purple mottled (debossed "T2" on one side and plain on the other)
- 25.9 mg extended-release orally disintegrating tablet: round, purple to light purple mottled (debossed "T3" on one side and plain on the other)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of methylphenidate products. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These adverse reactions are as follows:
Blood and Lymphatic System Disorders: Pancytopenia, Thrombocytopenia, Thrombocytopenic purpura
Cardiac Disorders: Angina pectoris, Bradycardia, Extrasystole, Supraventricular tachycardia, Ventricular extrasystole
Eye Disorders: Diplopia, Increased intraocolar pressure, Mydriasis, Visual impairment
General Disorders: Chest pain, Chest discomfort, Hyperpyrexia
Immune System Disorders: Hypersensitivity reactions such as Angioedema, Anaphylactic reactions, Auricular swelling, Bullous conditions, Exfoliative conditions, Urticarias, Pruritis NEC, Rashes, Eruptions, and Exanthemas NEC
Investigations: Alkaline phosphatase increased, Bilirubin increased, Hepatic enzyme increased, Platelet count decreased, White blood cell count abnormal
Musculoskeletal, Connective Tissue and Bone Disorders: Arthralgia, Myalgia, Muscle twitching, Rhabdomyolysis
Nervous System Disorders: Convulsion, Grand mal convulsion, Dyskinesia, Serotonin syndrome in combination with serotonergic drugs, Motor and Verbal Tics
Psychiatric Disorders: Disorientation, Hallucination, Hallucination auditory, Hallucination visual, Libido changes, Mania
Urogenital System: Priapism
Skin and Subcutaneous Tissue Disorders: Alopecia, Erythema
Vascular Disorders: Raynaud's phenomenon
8 Use in Specific Populations
.
2.2 General Dosing Information
COTEMPLA XR-ODT is given orally once daily in the morning.
Advise patients to take COTEMPLA XR-ODT consistently either with food or without food [see Clinical Pharmacology (12.3)] .
The recommended starting dose of COTEMPLA XR-ODT for patients 6 to 17 years of age is 17.3 mg once daily in the morning. The dose may be titrated weekly in increments of 8.6 mg to 17.3 mg. Daily doses above 51.8 mg have not been studied and are not recommended.
The dose should be individualized according to the needs and responses of the patient.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.1 Abuse, Misuse, and Addiction
COTEMPLA XR-ODT has a high potential for abuse and misuse. The use of COTEMPLA XR-ODT exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. COTEMPLA XR-ODT can be diverted for nonmedical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2)]. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing COTEMPLA XR-ODT, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store COTEMPLA XR-ODT in a safe place, preferably locked, and instruct patients to not give COTEMPLA XR-ODT to anyone else. Throughout COTEMPLA XR-ODT treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
5.8 Acute Angle Closure Glaucoma
There have been reports of angle closure glaucoma associated with methylphenidate treatment.
Although the mechanism is not clear, COTEMPLA XR-ODT-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.4 Cotempla Xr Odt Administration
Instruct the patient or caregiver on the following administration instructions:
- Do not remove the tablet from the blister pack until just prior to dosing. Take the tablet immediately after opening the blister pack. Do not store the tablet for future use.
- Use dry hands when opening the blister pack.
- Remove the tablet by peeling back the foil on the blister pack. Do not push the tablet through the foil.
- As soon as the blister is opened, remove the tablet and place on the patient's tongue.
- Place the whole tablet on the tongue and allow it to disintegrate without chewing or crushing.
- The tablet will disintegrate in saliva so that it can be swallowed. No liquid is needed to take the tablet.
16 How Supplied/storage and Handling
COTEMPLA XR-ODT Extended Release Orally Disintegrating Tablets are available in three strengths:
- 8.6 mg tablets, round, purple to light purple, mottled, and debossed "T1" on one side of the tablet;
- 17.3 mg tablets, round, purple to light purple, mottled, and debossed "T2" on one side of the tablet;
- 25.9 mg tablets, round, purple to light purple, mottled, and debossed "T3" on one side of the tablet.
They are available as follows:
| NDC 70165-100-30 | 8.6 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. |
| NDC 70165-200-30 | 17.3 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. |
| NDC 70165-300-30 | 25.9 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. |
Warning: Abuse, Misuse, and Addiction
COTEMPLA XR-ODT has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death (5.1, 9.2, 10). Before prescribing COTEMPLA XR-ODT, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout treatment, reassess each patient’s risk and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
2.3 Dosage Reduction and Discontinuation
If paradoxical aggravation of symptoms or other adverse reactions occur, reduce dosage, or, if necessary, discontinue COTEMPLA XR-ODT. If improvement is not observed after appropriate dosage adjustment over a one-month period, discontinue COTEMPLA XR-ODT.
5.3 Increased Blood Pressure and Heart Rate
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mm Hg) and heart rate (mean increase approximately 3 to 6 bpm). Some patients may have larger increases.
Monitor all COTEMPLA XR-ODT-treated patients for hypertension and tachycardia.
5.9 Increased Intraocular Pressure and Glaucoma
There have been reports of an elevation of intraocular pressure (IOP) associated with methylphenidate treatment [see Adverse Reactions (6.2)].
Prescribe COTEMPLA XR-ODT to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor COTEMPLA XR-ODT-treated patients with a history of abnormally increased IOP or open angle glaucoma.
5.2 Risks to Patients With Serious Cardiac Disease
Sudden death has occurred in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosages. Avoid COTEMPLA XR-ODT use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmia, coronary artery disease, or other serious cardiac disease.
5.7 Long Term Suppression of Growth in Pediatric Patients
COTEMPLA[A1] XR-ODT is not approved for use and is not recommended in pediatric patients below 6 years of age [see Use in Specific Populations (8.4)].
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or nonmedication-treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and nonmedication-treated pediatric patients over 36 months (to the ages of 10 to 13 years), suggests that pediatric patients who received methylphenidate for 7 days per week throughout the year had a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this development period.
Closely monitor growth (weight and height) in COTEMPLA XR-ODT-treated pediatric patients. Pediatric patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
7.1 Clinically Important Interactions With Cotempla Xr Odt
| Monoamine Oxidase Inhibitors (MAOI) | |
| Clinical Impact: | Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure [see Contraindications (4)] . |
| Intervention: | Do not administer COTEMPLA-XR ODT concomitantly with MAOIs or within 14 days after discontinuing MAOI treatment. |
| Gastric pH Modulators | |
| Clinical Impact: | May change the release profile and alter the pharmacodynamics of COTEMPLA-XR ODT. |
| Intervention: | Concomitant use of Cotempla XR-ODT with a gastric pH modulator (i.e., a H2-blocker or a proton pump inhibitor) is not recommended. |
| Antihypertensive Drugs | |
| Clinical Impact: | Cotempla XR-ODT may decrease the effectiveness of drug used to treat hypertension [see Warnings and Precautions (5.3)]. |
| Intervention: | Monitor blood pressure and adjust the dosage of the antihypertensive drug as needed. |
| Halogenated Anesthetics | |
| Clinical Impact: | Concomitant use of halogenated anesthetics and COTEMPLA XR-ODT may increase the risk of
sudden blood pressure and heart rate increase during surgery. |
| Intervention: | Avoid use of COTEMPLA XR-ODT in patients being treated with anesthetics on the day of
surgery. |
| Risperidone | |
| Clinical Impact: | Combined use of methylphenidate with risperidone when there is a change, whether an increase or decrease, in dosage of either or both medications, may increase the risk of extrapyramidal symptoms (EPS). |
| Intervention: | Monitor for signs of EPS. |
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon
CNS stimulants, including COTEMPLA XR-ODT, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud’s phenomenon, were observed in post-marketing reports and at the therapeutic dosages of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during COTEMPLA XR-ODT treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for COTEMPLA XRODT-treated patients who develop signs or symptoms of peripheral vasculopathy.
Principal Display Panel 8.6 Mg Tablet Blister Pack Carton
Contains:
NDC 70165-100-30
30 Tablets (5 x 6-count blister cards)
Travel Case
Rx Only
Cotempla XR-ODT™CII
Methylphenidate Extended-Release
Orally Disintegrating Tablets
Do not crush or chew tablets
Each tablet contains 8.6 mg of methylphenidate
(equivalent to that in a 10 mg strength
methylphenidate hydrochloride product)
8.6
mg
NEOS™
Therapeutics
PHARMACIST: Dispense the enclosed Medication Guide to each patient.
Principal Display Panel 17.3 Mg Tablet Blister Pack Carton
Contains:
NDC 70165-200-30
30 Tablets (5 x 6-count blister cards)
Travel Case
Rx Only
Cotempla XR-ODT™CII
Methylphenidate Extended-Release
Orally Disintegrating Tablets
Do not crush or chew tablets
Each tablet contains 17.3 mg of methylphenidate
(equivalent to that in a 20 mg strength
methylphenidate hydrochloride product)
17.3
mg
NEOS™
Therapeutics
PHARMACIST: Dispense the enclosed Medication Guide to each patient.
Principal Display Panel 25.9 Mg Tablet Blister Pack Carton
Contains:
NDC 70165-300-30
30 Tablets (5 x 6-count blister cards)
Travel Case
Rx Only
Cotempla XR-ODT™CII
Methylphenidate Extended-Release
Orally Disintegrating Tablets
Do not crush or chew tablets
Each tablet contains 25.9 mg of methylphenidate
(equivalent to that in a 30 mg strength
methylphenidate hydrochloride product)
25.9
mg
NEOS™
Therapeutics
PHARMACIST: Dispense the enclosed Medication Guide to each patient.
5.10 Motor and Verbal Tics, and Worsening of Tourette's Syndrome
CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette's syndrome has also been reported [see Adverse Reactions (6.2)].
Before initiating COTEMPLA XR-ODT, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regulary monitor COTEMPLA XR-ODT-treated patients for the emergence or worsening of tics or Tourette's syndrome, and discontine treatment if clinically appropriate.
Structured Label Content
Section 42229-5 (42229-5)
Exacerbation of Pre-Existing Psychosis
CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a pre-existing psychotic disorder.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 09/2025 | ||
|
Medication Guide
COTEMPLA XR-ODT (koh-TEM-pluh - oh dee tee) (methylphenidate) extended-release orally disintegrating tablets, CII |
|||
|
What is the most important information I should know about COTEMPLA XR-ODT? |
|||
COTEMPLA XR-ODT may cause serious side effects, including:
Tell your healthcare provider if your child has ever abused or been dependent on alcohol, prescription medicines, or street drugs.
Your healthcare provider should check your child carefully for heart problems before starting COTEMPLA XR-ODT. Tell your healthcare provider if your child has any heart problems, heart disease, or heart defects. Call your healthcare provider or go to the nearest hospital emergency room right away if your child has any signs of heart problems such as chest pain, shortness of breath, or fainting during treatment with COTEMPLA XR-ODT.
Your healthcare provider should check your child's blood pressure and heart rate regulary during treatment with COTEMPLA XR-ODT.
Tell your healthcare provider about any mental problems your child has, or about a family history of suicide, bipolar illness, or depression.
|
|||
| What is COTEMPLA XR-ODT? | |||
| COTEMPLA XR-ODT is a central nervous system (CNS) stimulant prescription medicine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in children 6 to 17 years of age. COTEMPLA XR-ODT may help increase attention and decrease impulsiveness and hyperactivity in children 6 to 17 years of age with ADHD. | |||
| COTEMPLA XR-ODT is not recommended for use in children under 6 years of age with ADHD. | |||
| COTEMPLA XR-ODT is a federally controlled substance (CII) because it contains methylphenidate that can be a target for people who abuse prescription medicines or street drugs. Keep COTEMPLA XR-ODT in a safe place to protect it from theft. Never give your COTEMPLA XR-ODT to anyone else, because it may cause death or harm them. Selling or giving away COTEMPLA XR-ODT may harm others and is against the law. | |||
Do not give COTEMPLA XR-ODT to your child if they are:
|
|||
Before taking COTEMPLA XR-ODT tell your child's healthcare provider about all medical conditions, including if your child:
|
|||
| Tell your healthcare provider about all of the medicines that your child takes, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| COTEMPLA XR-ODT and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be adjusted during treatment with COTEMPLA XR-ODT. | |||
Your healthcare provider will decide whether COTEMPLA XR-ODT can be taken with other medicines.
Especially tell your healthcare provider if your child takes:
|
|||
| Know the medicines that your child takes. Keep a list of the medicines with you to show your healthcare provider and pharmacist. Do not start any new medicine during treatment with COTEMPLA XR-ODT without talking to your healthcare provider first. | |||
How should COTEMPLA XR-ODT be taken?
|
|||
| Take COTEMPLA XR-ODT as follows: | |||
|
|||
|
If your child takes too much COTEMPLA XR-ODT, call your healthcare provider or Poison Help Line at 1-800-222-1222 or go to the nearest hospital emergency room right away. |
|||
| What should I avoid during treatment with COTEMPLA XR-ODT? | |||
| You should avoid drinking alcohol during treatment with COTEMPLA XR-ODT. | |||
| What are possible side effects of COTEMPLA XR-ODT? | |||
COTEMPLA XR-ODT may cause serious side effects, including:
|
|||
| The most common side effects of methylphenidate products include: | |||
|
|
||
| These are not all the possible side effects of COTEMPLA XR-ODT. | |||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
How should I store COTEMPLA XR-ODT?
|
|||
| Keep COTEMPLA XR-ODT and all medicines out of the reach of children. | |||
| General information about the safe and effective use of COTEMPLA XR-ODT | |||
| Medicines are sometimes prescribed for purposes other than those listed in the Medication Guide. Do not use COTEMPLA XR-ODT for a condition for which it was not prescribed. Do not give COTEMPLA XR-ODT to other people, even if they have the same condition. It may harm them and it is against the law. You can ask your healthcare provider or pharmacist for information about COTEMPLA XR-ODT that is written for healthcare professionals. | |||
| What are the ingredients in COTEMPLA XR-ODT? | |||
| Active Ingredient: Methylphenidate | |||
| Inactive Ingredients: Mannitol, Fructose, Microcrystalline Cellulose, Crospovidone, Methacrylic Acid, Polystyrene Sulfonate, Citric Acid, Colloidal Silicon Dioxide, Grape Flavor, Natural Masking Type Powder, Triethyl Citrate, Magnesium Stearate, Ethylcellulose, Sucralose, Lake Blend Purple, and Polyethylene Glycol 3350 | |||
| Manufactured for Neos Therapeutics Brands, LLC, Denver, CO 80237 | |||
| For more information go to https://linkprotect.cudasvc.com/url?a=http%3a%2f%2fwww.COTEMPLAXRODT.com&c=E,1,uJnpRo76RJYGQDo8GMW8zCnFW9Wn0k16nND89VJMYg7anbFT-Thnek2P1Uts7soE5kxS-99rv0KJLMK4Fi0tUhQLe2IT7HZHVjJZmOQANFuP3n-FX1c,&typo=1 or call 1-888-319-1789 | |||
| COTEMPLA XR-ODT is registered in the US Patent and Trademark Office © 2014 Neos Therapeutics, Inc. |
Section 43683-2 (43683-2)
Indications and Usage (1) 09/2025
Warnings and Precautions (5.7) 09/2025
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F); excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Store COTEMPLA XR-ODT blister packages in the reusable travel case after removal from the carton.
9.2 Abuse
COTEMPLA XR-ODT has a high potential for abuse and misuse which can lead to the development of a substance use disorder, including addiction [see Warnings and Precautions (5.1)]. COTEMPLA XR-ODT can be diverted for non-medical use into illicit channels or distribution.
Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of methylphenidate may cause increased heart rate, respiratory rate, or blood pressure; sweating; dilated pupils; hyperactivity; restlessness; insomnia; decreased appetite; loss of coordination; tremors; flushed skin; vomiting; and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed with CNS stimulants abuse and/or misuse. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
5.5 Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate use in both adult and pediatric male patients. Although priapism was not reported with methylphenidate initiation, it developed after some time on methylphenidate, often subsequent to an increase in dosage. Priapism also occurred during methylphenidate withdrawal (drug holidays or during discontinuation).
COTEMPLA XR-ODT-treated patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
11 Description (11 DESCRIPTION)
COTEMPLA XR-ODT contains methylphenidate, a central nervous system (CNS) stimulant. COTEMPLA XR-ODT is an extended-release orally disintegrating tablet intended for once daily administration. COTEMPLA XR-ODT contains approximately 25% immediate-release and 75% extended-release methylphenidate. Methylphenidate is ionically-bound to the sulfonate of polystyrene sulfonate particles.
COTEMPLA XR-ODT contains 8.6 mg, 17.3 mg or 25.9 mg of methylphenidate which is the same as the amount of methylphenidate (base equivalent) found, respectively, in 10 mg, 20 mg and 30 mg strength methylphenidate hydrochloride products.
The chemical name of methylphenidate is methyl α-phenyl-2-piperidineacetate, and its structural formula is shown in Figure 1.
Figure 1: Methylphenidate Structure
C 14H 19NO 2 Mol. Wt. 233.31
COTEMPLA XR-ODT also contains the following inactive ingredients: Mannitol, Fructose, Microcrystalline Cellulose, Crospovidone, Methacrylic Acid, Polystyrene Sulfonate, Citric Acid, Colloidal Silicon Dioxide, Grape Flavor, Natural Masking Type Powder, Triethyl Citrate, Magnesium Stearate, Ethylcellulose, Sucralose, Lake Blend Purple, and Polyethylene Glycol 3350.
8.4 Pediatric Use
The safety and effectiveness of COTEMPLA XR-ODT have not been established in pediatric patients below the age of 6 years.
In studies evaluating extended-release methylphenidate products, patients 4 to <6 years of age had higher systemic methylphenidate exposures than those observed in older pediatric patients at the same dosage. Pediatric patients 4 to <6 years of age also had a higher incidence of adverse reactions, including weight loss.
The safety and effectiveness of COTEMPLA XR-ODT have been established in pediatric patients 6 to 17 years of age in one adequate and well-controlled study in pediatric patients 6 to 12 years, pharmacokinetic data in adolescents, and safety information from other methyphenidate-containing products [see Clinical Pharmacology (12) and Clinical Studies (14)] .
8.5 Geriatric Use
COTEMPLA XR-ODT has not been studied in patients over the age of 65 years.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of COTEMPLA XR-ODT was evaluated in a laboratory classroom study conducted in 87 pediatric patients (Aged 6 to 12 years) with ADHD. Following washout of previous methylphenidate medication, there was an open-label dose-optimization period (4 weeks) with an initial dose of 17.3 mg of COTEMPLA XR-ODT once daily in the morning. The dose could be titrated on a weekly basis from 17.3 mg, to 25.9 mg, to 34.6 mg, and up to 51.8 mg until an optimal dose or the maximum dose of 51.8 mg/day was reached. At the end of this period, subjects remained on their optimized dose for an additional week. Subjects then entered a 1-week randomized, double-blind, parallel group treatment period with the individually optimized dose of COTEMPLA XR-ODT or placebo. At the end of this week, raters evaluated the attention and behavior of the subjects in a laboratory classroom setting, using the Swanson, Kotkin, Agler, M-Flynn, and Pelham (SKAMP) rating scale SKAMP is a validated 13-item teacher-rated scale that assesses manifestations of ADHD in a classroom setting.
The primary efficacy endpoint was the average of the SKAMP-Combined (Attention and Deportment) scores over the test day (not including the baseline score), with assessments conducted at baseline, and 1, 3, 5, 7, 10, 12, and 13 hours post-dosing. The key secondary efficacy endpoints were onset and duration of effect, defined as the first point at which active drug separated from placebo on SKAMP-Combined scores and the last time point at which active drug separated from placebo on SKAMP-Combined scores, respectively.
The SKAMP-Combined scores test day average was statistically significantly lower (improved) with COTEMPLA XR-ODT compared to placebo (difference of -11 (95% CI: -13.9, -8.2)) (Table 3).
| Study Number | Treatment Group | Baseline Score at Randomization a (SD) | Pre-dose Score on Classroom Day b (SD) | LS Mean c (SE) | Placebo-subtracted Difference d (95% CI) |
|---|---|---|---|---|---|
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: confidence interval.
a Visit 7 baseline score (Visit 7 occurred prior to the 1-week randomized, double-blind, parallel group treatment period). b Visit 8 baseline score (Visit 8 occurred at the end of the 1-week randomized, double-blind, parallel group treatment period). c Visit 8 LS mean over hours 1, 3, 5, 7, 10, 12, and 13. d Difference (drug minus placebo) in least-squares means. |
|||||
| Study 1 | Cotempla XR-ODT
(17.3-51.8 mg/day) |
21.1 (9.56) | 26.8 (11.52) | 14.3 (1.07) | -11.0 (-13.9, -8.2) |
| Placebo | 20.4 (9.09) | 19.1 (11.04) | 25.3 (1.16) | -- |
The SKAMP-Combined scores were also statistically significantly lower (improved) at time points (1, 3, 5, 7, 10, 12 hours) post-dosing with COTEMPLA XR-ODT compared to placebo (Figure 3).
Figure 3: LS Mean SKAMP Combined Score After Treatment with COTEMPLA XR-ODT or Placebo During Classroom Day in Patients with ADHD
*SE = Standard Error
The database was not large enough to assess whether there were differences in effects in age, gender, or race subgroups.
4 Contraindications (4 CONTRAINDICATIONS)
COTEMPLA XR-ODT is contraindicated in patients with:
- Known hypersensitivity to methylphenidate or other components of COTEMPLA XR-ODT. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.2)] .
- Concomitant treatment with monoamine oxidase inhibitors (MAOIs), and also within a minimum of 14 days following discontinuation of treatment with a monoamine oxidase inhibitor because of the risk of hypertensive crisis [see Drug Interactions (7.1)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are discussed in more detail in other sections of the labeling:
- Known hypersensitivity to methylphenidate or other ingredients of Cotempla XR-ODT [see Contraindications (4)]
- Hypertensive crisis when used concomitantly with monoamine oxidase inhibitors [see Contraindications (4) and Drug Interactions (7.1)]
- Abuse, Misuse, and Addiction [see Boxed Warning, Warnings and Precautions (5.1), and Drug Abuse and Dependence (9.2, 9.3)]
- Risks to patients with serious cardiac disease [see Warnings and Precautions (5.2)]
- Increased blood pressure and heart rate [see Warnings and Precautions (5.3)]
- Psychiatric adverse reactions [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.5)]
- Peripheral vasculopathy, including Raynaud's phenomenon [see Warnings and Precautions (5.6)]
- Long-term suppression of growth in pediatric patients [see Warnings and Precautions (5.7)]
- Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8)]
- Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9)]
- Motor and Verbal Tics, and Worsening of Tourette’s Syndrome [ see Warnings and Precautions (5.10)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. ( 7)
12.2 Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d- and 1-isomers. The d-isomer is more pharmacologically active than the l-isomer. Methylphenidate is thought to block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
After oral administration of COTEMPLA XR-ODT, circulation levels of l- methylphenidate (MPH) were about 2% of total MPH.
1 Indications and Usage (1 INDICATIONS AND USAGE)
COTEMPLA XR-ODT is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in pediatric patients 6 to 17 years of age [see Clinical Studies (14)].
Limitations of Use
The use of COTEMPLA XR-ODT is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions (e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.7). Use in Specific Populations (8.4)].
12.1 Mechanism of Action
Methylphenidate is a central nervous system (CNS) stimulant. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
COTEMPLA XR-ODT contains methylphenidate, a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to treating patients with COTEMPLA XR-ODT, assess:
- for the presence of cardiac disease (i.e. perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2)] .
- the family history and clinically evaluate patients for motor or verbal tics or Tourette's sydrome before initiating COTEMPLA XR-ODT [see Warnings and Precautions (5.10)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended starting dose for pediatric patients 6 to 17 years of age is 17.3 mg given orally once daily in the morning. Dosage may be increased weekly in increments of 8.6 mg to 17.3 mg per day. Daily dosage above 51.8 mg is not recommended. ( 2.2)
- Patients are advised to take COTEMPLA XR-ODT consistently either with food or without food. ( 2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 8.6 mg extended-release orally disintegrating tablet: round, purple to light purple mottled (debossed "T1" on one side and plain on the other)
- 17.3 mg extended-release orally disintegrating tablet: round, purple to light purple mottled (debossed "T2" on one side and plain on the other)
- 25.9 mg extended-release orally disintegrating tablet: round, purple to light purple mottled (debossed "T3" on one side and plain on the other)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of methylphenidate products. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These adverse reactions are as follows:
Blood and Lymphatic System Disorders: Pancytopenia, Thrombocytopenia, Thrombocytopenic purpura
Cardiac Disorders: Angina pectoris, Bradycardia, Extrasystole, Supraventricular tachycardia, Ventricular extrasystole
Eye Disorders: Diplopia, Increased intraocolar pressure, Mydriasis, Visual impairment
General Disorders: Chest pain, Chest discomfort, Hyperpyrexia
Immune System Disorders: Hypersensitivity reactions such as Angioedema, Anaphylactic reactions, Auricular swelling, Bullous conditions, Exfoliative conditions, Urticarias, Pruritis NEC, Rashes, Eruptions, and Exanthemas NEC
Investigations: Alkaline phosphatase increased, Bilirubin increased, Hepatic enzyme increased, Platelet count decreased, White blood cell count abnormal
Musculoskeletal, Connective Tissue and Bone Disorders: Arthralgia, Myalgia, Muscle twitching, Rhabdomyolysis
Nervous System Disorders: Convulsion, Grand mal convulsion, Dyskinesia, Serotonin syndrome in combination with serotonergic drugs, Motor and Verbal Tics
Psychiatric Disorders: Disorientation, Hallucination, Hallucination auditory, Hallucination visual, Libido changes, Mania
Urogenital System: Priapism
Skin and Subcutaneous Tissue Disorders: Alopecia, Erythema
Vascular Disorders: Raynaud's phenomenon
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
.
2.2 General Dosing Information
COTEMPLA XR-ODT is given orally once daily in the morning.
Advise patients to take COTEMPLA XR-ODT consistently either with food or without food [see Clinical Pharmacology (12.3)] .
The recommended starting dose of COTEMPLA XR-ODT for patients 6 to 17 years of age is 17.3 mg once daily in the morning. The dose may be titrated weekly in increments of 8.6 mg to 17.3 mg. Daily doses above 51.8 mg have not been studied and are not recommended.
The dose should be individualized according to the needs and responses of the patient.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.1 Abuse, Misuse, and Addiction
COTEMPLA XR-ODT has a high potential for abuse and misuse. The use of COTEMPLA XR-ODT exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. COTEMPLA XR-ODT can be diverted for nonmedical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2)]. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing COTEMPLA XR-ODT, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store COTEMPLA XR-ODT in a safe place, preferably locked, and instruct patients to not give COTEMPLA XR-ODT to anyone else. Throughout COTEMPLA XR-ODT treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
5.8 Acute Angle Closure Glaucoma
There have been reports of angle closure glaucoma associated with methylphenidate treatment.
Although the mechanism is not clear, COTEMPLA XR-ODT-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.4 Cotempla Xr Odt Administration (2.4 COTEMPLA XR-ODT Administration)
Instruct the patient or caregiver on the following administration instructions:
- Do not remove the tablet from the blister pack until just prior to dosing. Take the tablet immediately after opening the blister pack. Do not store the tablet for future use.
- Use dry hands when opening the blister pack.
- Remove the tablet by peeling back the foil on the blister pack. Do not push the tablet through the foil.
- As soon as the blister is opened, remove the tablet and place on the patient's tongue.
- Place the whole tablet on the tongue and allow it to disintegrate without chewing or crushing.
- The tablet will disintegrate in saliva so that it can be swallowed. No liquid is needed to take the tablet.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
COTEMPLA XR-ODT Extended Release Orally Disintegrating Tablets are available in three strengths:
- 8.6 mg tablets, round, purple to light purple, mottled, and debossed "T1" on one side of the tablet;
- 17.3 mg tablets, round, purple to light purple, mottled, and debossed "T2" on one side of the tablet;
- 25.9 mg tablets, round, purple to light purple, mottled, and debossed "T3" on one side of the tablet.
They are available as follows:
| NDC 70165-100-30 | 8.6 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. |
| NDC 70165-200-30 | 17.3 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. |
| NDC 70165-300-30 | 25.9 mg tablets: carton containing 5 blister cards of 6 tablets each, for a total of 30 tablets with a reusable travel case. |
Warning: Abuse, Misuse, and Addiction (WARNING: ABUSE, MISUSE, AND ADDICTION)
COTEMPLA XR-ODT has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including COTEMPLA XR-ODT, can result in overdose and death (5.1, 9.2, 10). Before prescribing COTEMPLA XR-ODT, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout treatment, reassess each patient’s risk and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
2.3 Dosage Reduction and Discontinuation
If paradoxical aggravation of symptoms or other adverse reactions occur, reduce dosage, or, if necessary, discontinue COTEMPLA XR-ODT. If improvement is not observed after appropriate dosage adjustment over a one-month period, discontinue COTEMPLA XR-ODT.
5.3 Increased Blood Pressure and Heart Rate
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mm Hg) and heart rate (mean increase approximately 3 to 6 bpm). Some patients may have larger increases.
Monitor all COTEMPLA XR-ODT-treated patients for hypertension and tachycardia.
5.9 Increased Intraocular Pressure and Glaucoma
There have been reports of an elevation of intraocular pressure (IOP) associated with methylphenidate treatment [see Adverse Reactions (6.2)].
Prescribe COTEMPLA XR-ODT to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor COTEMPLA XR-ODT-treated patients with a history of abnormally increased IOP or open angle glaucoma.
5.2 Risks to Patients With Serious Cardiac Disease (5.2 Risks to Patients with Serious Cardiac Disease)
Sudden death has occurred in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosages. Avoid COTEMPLA XR-ODT use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmia, coronary artery disease, or other serious cardiac disease.
5.7 Long Term Suppression of Growth in Pediatric Patients (5.7 Long-Term Suppression of Growth in Pediatric Patients)
COTEMPLA[A1] XR-ODT is not approved for use and is not recommended in pediatric patients below 6 years of age [see Use in Specific Populations (8.4)].
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or nonmedication-treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and nonmedication-treated pediatric patients over 36 months (to the ages of 10 to 13 years), suggests that pediatric patients who received methylphenidate for 7 days per week throughout the year had a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this development period.
Closely monitor growth (weight and height) in COTEMPLA XR-ODT-treated pediatric patients. Pediatric patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
7.1 Clinically Important Interactions With Cotempla Xr Odt (7.1 Clinically Important Interactions with COTEMPLA XR-ODT)
| Monoamine Oxidase Inhibitors (MAOI) | |
| Clinical Impact: | Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure [see Contraindications (4)] . |
| Intervention: | Do not administer COTEMPLA-XR ODT concomitantly with MAOIs or within 14 days after discontinuing MAOI treatment. |
| Gastric pH Modulators | |
| Clinical Impact: | May change the release profile and alter the pharmacodynamics of COTEMPLA-XR ODT. |
| Intervention: | Concomitant use of Cotempla XR-ODT with a gastric pH modulator (i.e., a H2-blocker or a proton pump inhibitor) is not recommended. |
| Antihypertensive Drugs | |
| Clinical Impact: | Cotempla XR-ODT may decrease the effectiveness of drug used to treat hypertension [see Warnings and Precautions (5.3)]. |
| Intervention: | Monitor blood pressure and adjust the dosage of the antihypertensive drug as needed. |
| Halogenated Anesthetics | |
| Clinical Impact: | Concomitant use of halogenated anesthetics and COTEMPLA XR-ODT may increase the risk of
sudden blood pressure and heart rate increase during surgery. |
| Intervention: | Avoid use of COTEMPLA XR-ODT in patients being treated with anesthetics on the day of
surgery. |
| Risperidone | |
| Clinical Impact: | Combined use of methylphenidate with risperidone when there is a change, whether an increase or decrease, in dosage of either or both medications, may increase the risk of extrapyramidal symptoms (EPS). |
| Intervention: | Monitor for signs of EPS. |
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon (5.6 Peripheral Vasculopathy, including Raynaud's Phenomenon)
CNS stimulants, including COTEMPLA XR-ODT, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud’s phenomenon, were observed in post-marketing reports and at the therapeutic dosages of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during COTEMPLA XR-ODT treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for COTEMPLA XRODT-treated patients who develop signs or symptoms of peripheral vasculopathy.
Principal Display Panel 8.6 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 8.6 mg Tablet Blister Pack Carton)
Contains:
NDC 70165-100-30
30 Tablets (5 x 6-count blister cards)
Travel Case
Rx Only
Cotempla XR-ODT™CII
Methylphenidate Extended-Release
Orally Disintegrating Tablets
Do not crush or chew tablets
Each tablet contains 8.6 mg of methylphenidate
(equivalent to that in a 10 mg strength
methylphenidate hydrochloride product)
8.6
mg
NEOS™
Therapeutics
PHARMACIST: Dispense the enclosed Medication Guide to each patient.
Principal Display Panel 17.3 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 17.3 mg Tablet Blister Pack Carton)
Contains:
NDC 70165-200-30
30 Tablets (5 x 6-count blister cards)
Travel Case
Rx Only
Cotempla XR-ODT™CII
Methylphenidate Extended-Release
Orally Disintegrating Tablets
Do not crush or chew tablets
Each tablet contains 17.3 mg of methylphenidate
(equivalent to that in a 20 mg strength
methylphenidate hydrochloride product)
17.3
mg
NEOS™
Therapeutics
PHARMACIST: Dispense the enclosed Medication Guide to each patient.
Principal Display Panel 25.9 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 25.9 mg Tablet Blister Pack Carton)
Contains:
NDC 70165-300-30
30 Tablets (5 x 6-count blister cards)
Travel Case
Rx Only
Cotempla XR-ODT™CII
Methylphenidate Extended-Release
Orally Disintegrating Tablets
Do not crush or chew tablets
Each tablet contains 25.9 mg of methylphenidate
(equivalent to that in a 30 mg strength
methylphenidate hydrochloride product)
25.9
mg
NEOS™
Therapeutics
PHARMACIST: Dispense the enclosed Medication Guide to each patient.
5.10 Motor and Verbal Tics, and Worsening of Tourette's Syndrome
CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette's syndrome has also been reported [see Adverse Reactions (6.2)].
Before initiating COTEMPLA XR-ODT, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regulary monitor COTEMPLA XR-ODT-treated patients for the emergence or worsening of tics or Tourette's syndrome, and discontine treatment if clinically appropriate.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:59.666947 · Updated: 2026-03-14T22:48:57.475644