These Highlights Do Not Include All The Information Needed To Use Anastrozole Tablets Safely And Effectively. See Full Prescribing Information For Anastrozole Tablets.
33f14d51-de4e-cbe4-e063-6294a90a964b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Anastrozole tablets are an aromatase inhibitor indicated for: Adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer ( 1.1 ) First-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer ( 1.2 ) Treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets ( 1.3 )
Indications and Usage
Anastrozole tablets are an aromatase inhibitor indicated for: Adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer ( 1.1 ) First-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer ( 1.2 ) Treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets ( 1.3 )
Dosage and Administration
One 1 mg tablet taken once daily ( 2.1 )
Warnings and Precautions
In women with pre-existing ischemic heart disease, an increased incidence of ischemic cardiovascular events occurred with anastrozole tablet use compared to tamoxifen use. Consider risks and benefits. ( 5.1 , 6.1 ) Decreases in bone mineral density may occur. Consider bone mineral density monitoring. ( 5.2 , 6.1 ) Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.3 , 6.1 ) Embryo-Fetal Toxicity: Anastrozole tablets may cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.4 , 8.1 )
Contraindications
Patients with demonstrated hypersensitivity to anastrozole tablets or any excipient ( 4 )
Adverse Reactions
Serious adverse reactions with anastrozole tablets occurring in less than 1 in 10,000 patients, are: 1) skin reactions such as lesions, ulcers, or blisters; 2) allergic reactions with swelling of the face, lips, tongue, and/or throat. This may cause difficulty in swallowing and/or breathing; and 3) changes in blood tests of the liver function, including inflammation of the liver with symptoms that may include a general feeling of not being well, with or without jaundice, liver pain or liver swelling [see Adverse Reactions (6.2) ]. Common adverse reactions (occurring with an incidence of ≥10%) in women taking anastrozole tablets included: hot flashes, asthenia, arthritis, pain, arthralgia, hypertension, depression, nausea and vomiting, rash, osteoporosis, fractures, back pain, insomnia, headache, bone pain, peripheral edema, increased cough, dyspnea, pharyngitis and lymphedema. In the ATAC trial, the most common reported adverse reaction (>0.1%) leading to discontinuation of therapy for both treatment groups was hot flashes, although there were fewer patients who discontinued therapy as a result of hot flashes in the anastrozole tablets group. Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
Tamoxifen: Do not use in combination with anastrozole tablets. No additional benefit seen over tamoxifen monotherapy ( 7.1 , 14.1 ) Estrogen-containing products: Combination use may diminish activity of anastrozole tablets ( 7.2 )
Storage and Handling
These tablets are supplied in bottles 90 tablets (NDC 680701-3838-9)
How Supplied
These tablets are supplied in bottles 90 tablets (NDC 680701-3838-9)
Patient Information
What is the most important information I should know about anastrozole tablets? Anastrozole tablets may cause serious side effects including: heart disease. Women with early breast cancer, who have a history of blockage in their heart arteries (ischemic heart disease) and who take anastrozole tablets, may have an increase in symptoms of decreased blood flow to their heart compared to similar women who take tamoxifen. Get medical help right away if you have new or worsening chest pain or shortness of breath during treatment with anastrozole tablets. What are anastrozole tablets? Anastrozole tablets are a prescription medicine used in women after menopause ("the change of life") for: treatment of early breast cancer after surgery in women whose breast cancer is hormone receptor-positive the first treatment of breast cancer that has spread to nearby tissue or lymph nodes (locally advanced) or has spread to other parts of the body (metastatic), in women whose breast cancer is hormone receptor-positive or the hormone receptors are not known. treatment of advanced breast cancer, if the cancer has grown, or the disease has spread after tamoxifen therapy Anastrozole tablets does not work in women with breast cancer who have not gone through menopause (premenopausal women). Who should not take anastrozole tablets? Do not take anastrozole tablet if you: have had a severe allergic reaction to anastrozole or any of the ingredients in anastrozole tablets. See the end of this leaflet for a complete list of ingredients in anastrozole tablets. Symptoms of a severe allergic reaction to anastrozole tablets include: swelling of the face, lips, tongue or throat, trouble breathing or swallowing, hives and itching. What should I tell my healthcare provider before taking anastrozole tablets? Before you take anastrozole tablets, tell your healthcare provider if you: are still having menstrual periods (are not past menopause). Talk to your healthcare provider if you are not sure. have or had a heart problem have been told you have bone thinning or weakness (osteoporosis) have high cholesterol have any other medical conditions are pregnant or plan to become pregnant. Taking anastrozole tablets during pregnancy or within 3 weeks of becoming pregnant may harm your unborn baby. Females who are able to become pregnant should have a pregnancy test before starting treatment with anastrozole tablets. Females who are able to become pregnant should use effective birth control (contraceptive) during treatment with anastrozole tablets and for 3 weeks after your last dose of anastrozole tablets. Tell your healthcare provider right away if you become pregnant or think you may be pregnant. are breastfeeding or plan to breastfeed. It is not known if anastrozole tablets passes into breast milk. Do not breastfeed during treatment with anastrozole tablets and for 2 weeks after your last dose of anastrozole tablets. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take: tamoxifen. You should not take anastrozole tablets if you take tamoxifen. Taking anastrozole tablets with tamoxifen may lower the amount of anastrozole tablets in your blood and may cause anastrozole tablets not to work as well. Medicines that contain estrogen. Anastrozole tablets may not work if taken with any of these medicines: hormone replacement therapy birth control pills estrogen creams vaginal rings vaginal suppositories Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. How should I take anastrozole tablets? Take anastrozole tablets exactly as your healthcare provider tells you to take it. Continue taking anastrozole tablets until your healthcare provider tells you to stop. Anastrozole tablets can be taken with or without food. If you miss a dose, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Take your next regularly scheduled dose. Do not take two doses at the same time. If you take too much anastrozole tablets, call your healthcare provider or go to the nearest hospital emergency room right away. What are the possible side effects of anastrozole tablets? Anastrozole tablets may cause serious side effects including: See " What is the most important information I should know about anastrozole tablets? " bone thinning or weakness (osteoporosis). Anastrozole tablets lowers estrogen in your body, which may cause your bones to become thinner and weaker. This may increase your risk of fractures, especially of your spine, hip and wrist. Your healthcare provider may order a bone mineral density test before you start and during treatment with anastrozole tablets to check you for bone changes. increased blood cholesterol (fat in the blood). Your healthcare provider may do blood tests to check your cholesterol while you are taking anastrozole tablets. skin reactions. Stop taking anastrozole tablets and call your healthcare provider right away if you get any skin lesions, ulcers, or blisters. severe allergic reactions. Get medical help right away if you get: swelling of your face, lips, tongue, or throat trouble swallowing or breathing liver problems. Anastrozole tablets can cause inflammation of your liver and changes in liver function blood tests. Your healthcare provider may check you for this. Stop taking anastrozole tablets and call your healthcare provider right away if you have any of these signs or symptoms of a liver problem: a general feeling of not being well yellowing of your skin or whites of your eyes pain on the right side of your stomach-area (abdomen) Common side effects in women taking anastrozole tablets include: hot flashes weakness joint aches joint pain, stiffness or swelling (arthritis) pain sore throat high blood pressure depression nausea and vomiting rash back pain sleep problems bone pain headache swelling of your legs, ankles, or feet increased cough shortness of breath build up of lymph fluid in the tissues of your affected arm (lymphedema) Anastrozole tablets may also cause you to have tickling, tingling or numbness of your skin. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of anastrozole tablets. For more information, ask your healthcare provider or pharmacist. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store anastrozole tablets? Store anastrozole tablets at room temperature between 68°F to 77°F (20°C to 25°C). Keep anastrozole tablets and all medicines out of the reach of children. General information about the safe and effective use of anastrozole tablets. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not take anastrozole tablets for a condition for which it was not prescribed. Do not give anastrozole tablets to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about anastrozole tablets that is written for health professionals. For more information, call 1-800-367-3395 or go to www.breckenridgepharma.com. What are the ingredients in anastrozole tablets? Active ingredient: anastrozole, USP Inactive ingredients: hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone, sodium starch glycolate, and titanium dioxide. This Patient Information has been approved by the U.S. Food and Drug Administration. Manufactured by: Natco Pharma Limited. Visakhapatnam - 531019 AP, India. Distributed by: Breckenridge Pharmaceutical, Inc. Berkeley Heights, NJ 07922 XXXXXXXX Revised: 10/2024
Medication Information
Warnings and Precautions
In women with pre-existing ischemic heart disease, an increased incidence of ischemic cardiovascular events occurred with anastrozole tablet use compared to tamoxifen use. Consider risks and benefits. ( 5.1 , 6.1 ) Decreases in bone mineral density may occur. Consider bone mineral density monitoring. ( 5.2 , 6.1 ) Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.3 , 6.1 ) Embryo-Fetal Toxicity: Anastrozole tablets may cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.4 , 8.1 )
Indications and Usage
Anastrozole tablets are an aromatase inhibitor indicated for: Adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer ( 1.1 ) First-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer ( 1.2 ) Treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets ( 1.3 )
Dosage and Administration
One 1 mg tablet taken once daily ( 2.1 )
Contraindications
Patients with demonstrated hypersensitivity to anastrozole tablets or any excipient ( 4 )
Adverse Reactions
Serious adverse reactions with anastrozole tablets occurring in less than 1 in 10,000 patients, are: 1) skin reactions such as lesions, ulcers, or blisters; 2) allergic reactions with swelling of the face, lips, tongue, and/or throat. This may cause difficulty in swallowing and/or breathing; and 3) changes in blood tests of the liver function, including inflammation of the liver with symptoms that may include a general feeling of not being well, with or without jaundice, liver pain or liver swelling [see Adverse Reactions (6.2) ]. Common adverse reactions (occurring with an incidence of ≥10%) in women taking anastrozole tablets included: hot flashes, asthenia, arthritis, pain, arthralgia, hypertension, depression, nausea and vomiting, rash, osteoporosis, fractures, back pain, insomnia, headache, bone pain, peripheral edema, increased cough, dyspnea, pharyngitis and lymphedema. In the ATAC trial, the most common reported adverse reaction (>0.1%) leading to discontinuation of therapy for both treatment groups was hot flashes, although there were fewer patients who discontinued therapy as a result of hot flashes in the anastrozole tablets group. Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
Tamoxifen: Do not use in combination with anastrozole tablets. No additional benefit seen over tamoxifen monotherapy ( 7.1 , 14.1 ) Estrogen-containing products: Combination use may diminish activity of anastrozole tablets ( 7.2 )
Storage and Handling
These tablets are supplied in bottles 90 tablets (NDC 680701-3838-9)
How Supplied
These tablets are supplied in bottles 90 tablets (NDC 680701-3838-9)
Patient Information
What is the most important information I should know about anastrozole tablets?
Anastrozole tablets may cause serious side effects including:
-
heart disease.Women with early breast cancer, who have a history of blockage in their heart arteries (ischemic heart disease) and who take anastrozole tablets, may have an increase in symptoms of decreased blood flow to their heart compared to similar women who take tamoxifen.
Get medical help right away if you have new or worsening chest pain or shortness of breath during treatment with anastrozole tablets.
What are anastrozole tablets?
Anastrozole tablets are a prescription medicine used in women after menopause ("the change of life") for:
- treatment of early breast cancer
- after surgery
- in women whose breast cancer is hormone receptor-positive
- the first treatment of breast cancer that has spread to nearby tissue or lymph nodes (locally advanced) or has spread to other parts of the body (metastatic), in women whose breast cancer is hormone receptor-positive or the hormone receptors are not known.
- treatment of advanced breast cancer, if the cancer has grown, or the disease has spread after tamoxifen therapy
Anastrozole tablets does not work in women with breast cancer who have not gone through menopause (premenopausal women).
Who should not take anastrozole tablets?
Do not take anastrozole tablet if you:
- have had a severe allergic reaction to anastrozole or any of the ingredients in anastrozole tablets. See the end of this leaflet for a complete list of ingredients in anastrozole tablets. Symptoms of a severe allergic reaction to anastrozole tablets include: swelling of the face, lips, tongue or throat, trouble breathing or swallowing, hives and itching.
What should I tell my healthcare provider before taking anastrozole tablets?
Before you take anastrozole tablets, tell your healthcare provider if you:
- are still having menstrual periods (are not past menopause). Talk to your healthcare provider if you are not sure.
- have or had a heart problem
- have been told you have bone thinning or weakness (osteoporosis)
- have high cholesterol
- have any other medical conditions
- are pregnant or plan to become pregnant. Taking anastrozole tablets during pregnancy or within 3 weeks of becoming pregnant may harm your unborn baby.
- Females who are able to become pregnant should have a pregnancy test before starting treatment with anastrozole tablets.
- Females who are able to become pregnant should use effective birth control (contraceptive) during treatment with anastrozole tablets and for 3 weeks after your last dose of anastrozole tablets. Tell your healthcare provider right away if you become pregnant or think you may be pregnant.
- are breastfeeding or plan to breastfeed. It is not known if anastrozole tablets passes into breast milk. Do not breastfeed during treatment with anastrozole tablets and for 2 weeks after your last dose of anastrozole tablets.
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take:
- tamoxifen.You should not take anastrozole tabletsif you take tamoxifen. Taking anastrozole tablets with tamoxifen may lower the amount of anastrozole tablets in your blood and may cause anastrozole tablets not to work as well.
-
Medicines that contain estrogen.Anastrozole tablets may not work if taken with any of these medicines:
- hormone replacement therapy
- birth control pills
- estrogen creams
- vaginal rings
- vaginal suppositories
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take anastrozole tablets?
- Take anastrozole tablets exactly as your healthcare provider tells you to take it.
- Continue taking anastrozole tablets until your healthcare provider tells you to stop.
- Anastrozole tablets can be taken with or without food.
- If you miss a dose, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Take your next regularly scheduled dose. Do not take two doses at the same time.
If you take too much anastrozole tablets, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of anastrozole tablets?
Anastrozole tablets may cause serious side effects including:
- See " What is the most important information I should know about anastrozole tablets?"
- bone thinning or weakness (osteoporosis).Anastrozole tablets lowers estrogen in your body, which may cause your bones to become thinner and weaker. This may increase your risk of fractures, especially of your spine, hip and wrist. Your healthcare provider may order a bone mineral density test before you start and during treatment with anastrozole tablets to check you for bone changes.
- increased blood cholesterol (fat in the blood).Your healthcare provider may do blood tests to check your cholesterol while you are taking anastrozole tablets.
- skin reactions.Stop taking anastrozole tablets and call your healthcare provider right away if you get any skin lesions, ulcers, or blisters.
-
severe allergic reactions.Get medical help right away if you get:
- swelling of your face, lips, tongue, or throat
- trouble swallowing or breathing
- liver problems.Anastrozole tablets can cause inflammation of your liver and changes in liver function blood tests. Your healthcare provider may check you for this.
Stop taking anastrozole tablets and call your healthcare provider right awayif you have any of these signs or symptoms of a liver problem:
- a general feeling of not being well
- yellowing of your skin or whites of your eyes
- pain on the right side of your stomach-area (abdomen)
Common side effects in women taking anastrozole tablets include:
- hot flashes
- weakness
- joint aches
- joint pain, stiffness or swelling (arthritis)
- pain
- sore throat
- high blood pressure
- depression
- nausea and vomiting
- rash
- back pain
- sleep problems
- bone pain
- headache
- swelling of your legs, ankles, or feet
- increased cough
- shortness of breath
- build up of lymph fluid in the tissues of your affected arm (lymphedema)
Anastrozole tablets may also cause you to have tickling, tingling or numbness of your skin.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of anastrozole tablets. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store anastrozole tablets?
- Store anastrozole tablets at room temperature between 68°F to 77°F (20°C to 25°C).
Keep anastrozole tablets and all medicines out of the reach of children.
General information about the safe and effective use of anastrozole tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not take anastrozole tablets for a condition for which it was not prescribed. Do not give anastrozole tablets to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about anastrozole tablets that is written for health professionals. For more information, call 1-800-367-3395 or go to www.breckenridgepharma.com.
What are the ingredients in anastrozole tablets?
Active ingredient:anastrozole, USP
Inactive ingredients:hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone, sodium starch glycolate, and titanium dioxide.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Natco Pharma Limited.
Visakhapatnam - 531019
AP, India.
Distributed by:
Breckenridge Pharmaceutical, Inc.
Berkeley Heights, NJ 07922
XXXXXXXX
Revised: 10/2024
Description
Anastrozole tablets are an aromatase inhibitor indicated for: Adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer ( 1.1 ) First-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer ( 1.2 ) Treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets ( 1.3 )
Section 42229-5
Hypersensitivity
Anastrozole tablets are contraindicated in any patient who has shown a hypersensitivity reaction to the drug or to any of the excipients. Observed reactions include anaphylaxis, angioedema, and urticaria [see Adverse Reactions (6.2)].
Section 44425-7
Storage
Store at controlled room temperature, 20-25°C (68-77°F) [see USP].
7.2 Estrogen
Estrogen-containing therapies should not be used with anastrozole tablets as they may diminish its pharmacological action.
7.3 Warfarin
In a study conducted in 16 male volunteers, anastrozole did not alter the exposure (as measured by C maxand AUC) and anticoagulant activity (as measured by prothrombin time, activated partial thromboplastin time, and thrombin time) of both R- and S-warfarin.
10 Overdosage
Clinical trials have been conducted with anastrozole tablets, up to 60 mg in a single dose given to healthy male volunteers and up to 10 mg daily given to postmenopausal women with advanced breast cancer; these dosages were tolerated. A single dose of anastrozole tablets that results in life-threatening symptoms has not been established. There is no specific antidote to overdosage and treatment must be symptomatic. In the management of an overdose, consider that multiple agents may have been taken. Vomiting may be induced if the patient is alert. Dialysis may be helpful because anastrozole tablets are not highly protein bound. General supportive care, including frequent monitoring of vital signs and close observation of the patient, is indicated.
7.1 Tamoxifen
Co-administration of anastrozole and tamoxifen in breast cancer patients reduced anastrozole plasma concentration by 27%. However, the co-administration of anastrozole and tamoxifen did not affect the pharmacokinetics of tamoxifen or N-desmethyltamoxifen. At a median follow-up of 33 months, the combination of anastrozole tablets and tamoxifen did not demonstrate any efficacy benefit when compared with tamoxifen in all patients as well as in the hormone receptor-positive subpopulation. This treatment arm was discontinued from the trial [see Clinical Studies (14.1)]. Based on clinical and pharmacokinetic results from the ATAC trial, tamoxifen should not be administered with anastrozole.
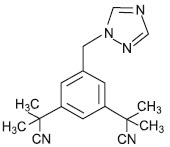
11 Description
Anastrozole Tablets, USP for oral administration contain 1 mg of anastrozole, a non-steroidal aromatase inhibitor. It is chemically described as 1,3-Benzenediacetonitrile, a, a, a', a'-tetramethyl-5-(1H-1,2,4-triazol-1-ylmethyl). Its molecular formula is C 17H 19N 5and its structural formula is:
Anastrozole is an off-white powder with a molecular weight of 293.4. Anastrozole has moderate aqueous solubility (0.5 mg/mL at 25°C); solubility is independent of pH in the physiological range. Anastrozole is freely soluble in methanol, acetone, ethanol, and tetrahydrofuran, and very soluble in acetonitrile.
Each tablet contains as inactive ingredients: hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone, sodium starch glycolate and titanium dioxide.
5.3 Cholesterol
During the ATAC trial, more patients receiving anastrozole tablets were reported to have elevated serum cholesterol compared to patients receiving tamoxifen (9% versus 3.5%, respectively) [see Adverse Reactions (6.1)].
5.2 Bone Effects
Results from the ATAC trial bone substudy at 12 and 24 months demonstrated that patients receiving anastrozole tablets had a mean decrease in both lumbar spine and total hip bone mineral density (BMD) compared to baseline. Patients receiving tamoxifen had a mean increase in both lumbar spine and total hip BMD compared to baseline. Consider bone mineral density monitoring in patients treated with anastrozole tablets [see Adverse Reactions (6.1)].
8.4 Pediatric Use
Clinical studies in pediatric patients included a placebo-controlled trial in pubertal boys of adolescent age with gynecomastia and a single-arm trial in girls with McCune-Albright Syndrome and progressive precocious puberty. The efficacy of anastrozole tablets in the treatment of pubertal gynecomastia in adolescent boys and in the treatment of precocious puberty in girls with McCune-Albright Syndrome has not been demonstrated.
8.5 Geriatric Use
In studies 0030 and 0027, about 50% of patients were 65 or older. Patients ≥ 65 years of age had moderately better tumor response and time to tumor progression than patients < 65 years of age regardless of randomized treatment. In studies 0004 and 0005, 50% of patients were 65 or older. Response rates and time to progression were similar for the over 65 and younger patients.
In the ATAC study, 45% of patients were 65 years of age or older. The efficacy of anastrozole tablets compared to tamoxifen in patients who were 65 years or older (N=1413 for anastrozole tablets and N=1410 for tamoxifen, the hazard ratio for disease-free survival was 0.93 [95% CI: 0.80, 1.08]) was less than efficacy observed in patients who were less than 65 years of age (N=1712 for anastrozole tablets and N=1706 for tamoxifen, the hazard ratio for disease-free survival was 0.79 [95% CI: 0.67, 0.94]).
The pharmacokinetics of anastrozole are not affected by age.
4 Contraindications
- Patients with demonstrated hypersensitivity to anastrozole tablets or any excipient ( 4)
6 Adverse Reactions
Serious adverse reactions with anastrozole tablets occurring in less than 1 in 10,000 patients, are: 1) skin reactions such as lesions, ulcers, or blisters; 2) allergic reactions with swelling of the face, lips, tongue, and/or throat. This may cause difficulty in swallowing and/or breathing; and 3) changes in blood tests of the liver function, including inflammation of the liver with symptoms that may include a general feeling of not being well, with or without jaundice, liver pain or liver swelling [see Adverse Reactions (6.2)].
Common adverse reactions (occurring with an incidence of ≥10%) in women taking anastrozole tablets included: hot flashes, asthenia, arthritis, pain, arthralgia, hypertension, depression, nausea and vomiting, rash, osteoporosis, fractures, back pain, insomnia, headache, bone pain, peripheral edema, increased cough, dyspnea, pharyngitis and lymphedema.
In the ATAC trial, the most common reported adverse reaction (>0.1%) leading to discontinuation of therapy for both treatment groups was hot flashes, although there were fewer patients who discontinued therapy as a result of hot flashes in the anastrozole tablets group.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions
7.4 Cytochrome P450
Based on in vitroand in vivoresults, it is unlikely that co-administration of anastrozole tablets 1 mg will affect other drugs as a result of inhibition of cytochrome P450 [see Clinical Pharmacology (12.3)].
2.1 Recommended Dose
The dose of anastrozole tablets are one 1 mg tablet taken once a day. For patients with advanced breast cancer, anastrozole tablets should be continued until tumor progression. Anastrozole tablets can be taken with or without food.
For adjuvant treatment of early breast cancer in postmenopausal women, the optimal duration of therapy is unknown. In the ATAC trial, anastrozole tablets were administered for five years [see Clinical Studies (14.1)] .
No dosage adjustment is necessary for patients with renal impairment or for elderly patients [see Use in Specific Populations (8.6)].
8.6 Renal Impairment
Since only about 10% of anastrozole is excreted unchanged in the urine, the renal impairment does not influence the total body clearance. Dosage adjustment in patients with renal impairment is not necessary [see Dosage and Administration (2.1)and Clinical Pharmacology (12.3)].
1.1 Adjuvant Treatment
Anastrozole tablets are indicated for adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer.
8.7 Hepatic Impairment
The plasma anastrozole concentrations in the subjects with hepatic cirrhosis were within the range of concentrations seen in normal subjects across all clinical trials. Therefore, dosage adjustment is also not necessary in patients with stable hepatic cirrhosis. Anastrozole tablets have not been studied in patients with severe hepatic impairment [see Dosage and Administration (2.2)and Clinical Pharmacology (12.3)].
1 Indications and Usage
Anastrozole tablets are an aromatase inhibitor indicated for:
- Adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer ( 1.1)
- First-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer ( 1.2)
- Treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets ( 1.3)
1.2 First Line Treatment
Anastrozole tablets are indicated for the first-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer.
12.1 Mechanism of Action
The growth of many cancers of the breast is stimulated or maintained by estrogens.
In postmenopausal women, estrogens are mainly derived from the action of the aromatase enzyme, which converts adrenal androgens (primarily androstenedione and testosterone) to estrone and estradiol. The suppression of estrogen biosynthesis in peripheral tissues and in the cancer tissue itself can therefore be achieved by specifically inhibiting the aromatase enzyme.
Anastrozole is a selective non-steroidal aromatase inhibitor. It significantly lowers serum estradiol concentrations and has no detectable effect on formation of adrenal corticosteroids or aldosterone.
1.3 Second Line Treatment
Anastrozole tablets are indicated for the treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets.
5.4 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, anastrozole tablets can cause fetal harm when administered to a pregnant woman. Anastrozole caused embryo-fetal toxicities in rats at maternal exposure that were 9 times the human clinical exposure, based on area under the curve (AUC). In rabbits, anastrozole caused pregnancy failure at doses equal to or greater than 16 times the recommended human dose on a mg/m 2basis. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during therapy with anastrozole tablets and for at least 3 weeks after the last dose [see Use in Specific Populations (8.1, 8.3)and Clinical Pharmacology (12.1)] .
5 Warnings and Precautions
- In women with pre-existing ischemic heart disease, an increased incidence of ischemic cardiovascular events occurred with anastrozole tablet use compared to tamoxifen use. Consider risks and benefits. ( 5.1, 6.1)
- Decreases in bone mineral density may occur. Consider bone mineral density monitoring. ( 5.2, 6.1)
- Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.3, 6.1)
- Embryo-Fetal Toxicity: Anastrozole tablets may cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.4, 8.1)
2 Dosage and Administration
One 1 mg tablet taken once daily ( 2.1)
3 Dosage Forms and Strengths
The tablets are white to off-white, round, film-coated containing 1 mg of anastrozole. The tablets are debossed with "AN" and "1" on one side and plain surface on the other side.
6.2 Post Marketing Experience
These adverse reactions are reported voluntarily from a population of uncertain size. Therefore, it is not always possible to estimate reliably their frequency or establish a causal relationship to drug exposure. The following have been reported in post-approval use of anastrozole tablets:
- Hepatobiliary events including increases in alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, gamma-GT, and bilirubin; hepatitis
- Rash including cases of mucocutaneous disorders such as erythema multiforme and Stevens-Johnson syndrome
- Cases of allergic reactions including angioedema, urticaria and anaphylaxis [see Contraindications (4)]
- Myalgia, trigger finger and hypercalcemia (with or without an increase in parathyroid hormone)
8 Use in Specific Populations
- Lactation: Do not breastfeed. ( 8.2)
- Females and Males of Reproductive Potential: Verify pregnancy status prior to initiation of anastrozole tablets. ( 8.3)
- Pediatric patients: Efficacy has not been demonstrated for pubertal boys of adolescent age with gynecomastia or girls with McCune-Albright Syndrome and progressive precocious puberty. ( 8.4)
17 Patient Counseling Information
See FDA approved patient labeling ( Patient Information).
5.1 Ischemic Cardiovascular Events
In women with pre-existing ischemic heart disease, an increased incidence of ischemic cardiovascular events was observed with anastrozole tablets in the ATAC trial (17% of patients on anastrozole tablets and 10% of patients on tamoxifen). Consider risk and benefits of anastrozole tablets therapy in patients with pre-existing ischemic heart disease [see Adverse Reactions (6.1)].
16 How Supplied/storage and Handling
These tablets are supplied in bottles 90 tablets (NDC 680701-3838-9)
2.2 Patients With Hepatic Impairment
No changes in dose are recommended for patients with mild-to-moderate hepatic impairment. Anastrozole tablets have not been studied in patients with severe hepatic impairment [see Use in Specific Populations (8.7)].
Principal Display Panel 1 Mg Tablet Bottle Label
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A conventional carcinogenesis study in rats at doses of 1.0 to 25 mg/kg/day (about 10 to 243 times the daily maximum recommended human dose on a mg/m 2basis) administered by oral gavage for up to 2 years revealed an increase in the incidence of hepatocellular adenoma and carcinoma and uterine stromal polyps in females and thyroid adenoma in males at the high dose. A dose-related increase was observed in the incidence of ovarian and uterine hyperplasia in females. At 25 mg/kg/day, plasma AUC 0-24 hrlevels in rats were 110 to 125 times higher than the level exhibited in postmenopausal volunteers at the recommended dose. A separate carcinogenicity study in mice at oral doses of 5 to 50 mg/kg/day (about 24 to 243 times the daily maximum recommended human dose on a mg/m 2basis) for up to 2 years produced an increase in the incidence of benign ovarian stromal, epithelial and granulosa cell tumors at all dose levels. A dose-related increase in the incidence of ovarian hyperplasia was also observed in female mice. These ovarian changes are considered to be rodent-specific effects of aromatase inhibition and are of questionable significance to humans. The incidence of lymphosarcoma was increased in males and females at the high dose. At 50 mg/kg/day, plasma AUC levels in mice were 35 to 40 times higher than the level exhibited in postmenopausal volunteers at the recommended dose.
Anastrozole tablets have not been shown to be mutagenic in in vitrotests (Ames and E. coli bacterial tests, CHO-K1 gene mutation assay) or clastogenic either in vitro(chromosome aberrations in human lymphocytes) or in vivo(micronucleus test in rats).
Oral administration of anastrozole to female rats (from 2 weeks before mating to pregnancy day 7) produced significant incidence of infertility and reduced numbers of viable pregnancies at 1 mg/kg/day (about 10 times the recommended human dose on a mg/m 2basis and 9 times higher than the AUC 0-24 hrfound in postmenopausal volunteers at the recommended dose). Pre-implantation loss of ova or fetus was increased at doses equal to or greater than 0.02 mg/kg/day (about one-fifth the recommended human dose on a mg/m 2basis). Recovery of fertility was observed following a 5-week non-dosing period which followed 3 weeks of dosing. It is not known whether these effects observed in female rats are indicative of impaired fertility in humans.
Multiple-dose studies in rats administered anastrozole for 6 months at doses equal to or greater than 1 mg/kg/day (which produced plasma anastrozole C ssmaxand AUC 0-24 hrthat were 19 and 9 times higher than the respective values found in postmenopausal volunteers at the recommended dose) resulted in hypertrophy of the ovaries and the presence of follicular cysts. In addition, hyperplastic uteri were observed in 6-month studies in female dogs administered doses equal to or greater than 1 mg/kg/day (which produced plasma anastrozole C ssmaxand AUC 0-24 hrthat were 22 times and 16 times higher than the respective values found in postmenopausal women at the recommended dose). It is not known whether these effects on the reproductive organs of animals are associated with impaired fertility in premenopausal women.
14.1 Adjuvant Treatment of Breast Cancer in Postmenopausal Women
A multicenter, double-blind trial (ATAC) randomized 9,366 postmenopausal women with operable breast cancer to adjuvant treatment with anastrozole tablets 1 mg daily, tamoxifen 20 mg daily, or a combination of the two treatments for five years or until recurrence of the disease.
The primary endpoint of the trial was disease-free survival (i.e., time to occurrence of a distant or local recurrence, or contralateral breast cancer or death from any cause). Secondary endpoints of the trial included distant disease-free survival, the incidence of contralateral breast cancer and overall survival. At a median follow-up of 33 months, the combination of anastrozole tablets and tamoxifen did not demonstrate any efficacy benefit when compared with tamoxifen in all patients as well as in the hormone receptor positive subpopulation. This treatment arm was discontinued from the trial. Based on clinical and pharmacokinetic results from the ATAC trial, tamoxifen should not be administered with anastrozole [see Drug Interactions (7.1)].
Demographic and other baseline characteristics were similar among the three treatment groups (see Table 7).
| Demographic Characteristic | Anastrozole tablets
1 mg (N N=Number of patients randomized to the treatment =3125)
|
Tamoxifen
20 mg (N =3116) |
Anastrozole tablets 1 mg plus Tamoxifen
The combination arm was discontinued due to lack of efficacy benefit at 33 months of follow-up 20 mg
(N =3125) |
|---|---|---|---|
| Mean age (yrs.) | 64.1 | 64.1 | 64.3 |
| Age Range (yrs.) | 38.1 - 92.8 | 32.8 - 94.9 | 37.0 – 92.2 |
| Age Distribution (%) | |||
| <45 yrs. | 0.7 | 0.4 | 0.5 |
| 45-60 yrs. | 34.6 | 35.0 | 34.5 |
| >60 <70 yrs. | 38.0 | 37.1 | 37.7 |
| >70 yrs. | 26.7 | 27.4 | 27.3 |
| Mean Weight (kg) | 70.8 | 71.1 | 71.3 |
| Receptor Status (%) | |||
| Positive
Includes patients who were estrogen receptor (ER) positive or progesterone receptor (PgR) positive, or both positive
|
83.5 | 83.1 | 84.0 |
| Negative
Includes patients with both ER negative and PgR negative receptor status
|
7.4 | 8.0 | 7.0 |
| Other
Includes all other combinations of ER and PgR receptor status unknown
|
8.8 | 8.6 | 9.0 |
| Other Treatment (%) prior to Randomization | |||
| Mastectomy | 47.8 | 47.3 | 48.1 |
| Breast conservation
Among the patients who had breast conservation, radiotherapy was administered to 95.0% of patients in the anastrozole tablets arm, 94.1% in the tamoxifen arm and 94.5% in the anastrozole tablets plus tamoxifen arm.
|
52.3 | 52.8 | 51.9 |
| Axillary surgery | 95.5 | 95.7 | 95.2 |
| Radiotherapy | 63.3 | 62.5 | 61.9 |
| Chemotherapy | 22.3 | 20.8 | 20.8 |
| Neoadjuvant Tamoxifen | 1.6 | 1.6 | 1.7 |
| Primary Tumor Size (%) | |||
| T1 (≤2 cm) | 63.9 | 62.9 | 64.1 |
| T2 (>2 cm and ≤5 cm) | 32.6 | 34.2 | 32.9 |
| T3 (>5 cm) | 2.7 | 2.2 | 2.3 |
| Nodal Status (%) | |||
| Node positive | 34.9 | 33.6 | 33.5 |
| 1-3 (# of nodes) | 24.4 | 24.4 | 24.3 |
| 4-9 | 7.5 | 6.4 | 6.8 |
| >9 | 2.9 | 2.7 | 2.3 |
| Tumor Grade (%) | |||
| Well-differentiated | 20.8 | 20.5 | 21.2 |
| Moderately differentiated | 46.8 | 47.8 | 46.5 |
| Poorly/undifferentiated | 23.7 | 23.3 | 23.7 |
| Not assessed/recorded | 8.7 | 8.4 | 8.5 |
Patients in the two monotherapy arms of the ATAC trial were treated for a median of 60 months (5 years) and followed for a median of 68 months. Disease-free survival in the intent-to-treat population was statistically significantly improved [Hazard Ratio (HR) = 0.87, 95% CI: 0.78, 0.97, p=0.0127] in the anastrozole tablets arm compared to the tamoxifen arm. In the hormone receptor-positive subpopulation representing about 84% of the trial patients, disease-free survival was also statistically significantly improved (HR =0.83, 95% CI: 0.73, 0.94, p=0.0049) in the anastrozole tablets arm compared to the tamoxifen arm.
Figure 1 – Disease-Free Survival Kaplan Meier Survival Curve for all Patients Randomized to Anastrozole Tablets or Tamoxifen Monotherapy in the ATAC Trial (Intent-to-Treat)
Figure 2 – Disease-Free Survival for Hormone Receptor-Positive Subpopulation of Patients Randomized to Anastrozole Tablets or Tamoxifen Monotherapy in the ATAC Trial
The survival data with 68 months follow-up is presented in Table 9.
In the group of patients who had previous adjuvant chemotherapy (N=698 for anastrozole tablets and N=647 for tamoxifen), the hazard ratio for disease-free survival was 0.91 (95% CI: 0.73 to 1.13) in the anastrozole tablets arm compared to the tamoxifen arm.
The frequency of individual events in the intent-to-treat population and the hormone receptor-positive subpopulation are described in Table 8.
| Intent-To-Treat Population
Patients may fall into more than one category.
|
Hormone Receptor-Positive Subpopulation | |||
|---|---|---|---|---|
| Anastrozole tablets
1 mg (N N=Number of patients randomized =3125)
|
Tamoxifen
20 mg (N =3116) |
Anastrozole tablets
1 mg (N =2618) |
Tamoxifen
20 mg (N =2598) |
|
| Median Duration of Therapy (mo) | 60 | 60 | 60 | 60 |
| Median Efficacy Follow-up (mo) | 68 | 68 | 68 | 68 |
| Loco-regional recurrence | 119 (3.8) | 149 (4.8) | 76 (2.9) | 101 (3.9) |
| Contralateral breast cancer | 35 (1.1) | 59 (1.9) | 26 (1.0) | 54 (2.1) |
| Invasive | 27 (0.9) | 52 (1.7) | 21 (0.8) | 48 (1.8) |
| Ductal carcinoma in situ | 8 (0.3) | 6 (0.2) | 5 (0.2) | 5 (0.2) |
| Unknown | 0 | 1 (<0.1) | 0 | 1 (<0.1) |
| Distant recurrence | 324 (10.4) | 375 (12.0) | 226 (8.6) | 265 (10.2) |
| Death from Any Cause | 411 (13.2) | 420 (13.5) | 296 (11.3) | 301 (11.6) |
| Death breast cancer | 218 (7.0) | 248 (8.0) | 138 (5.3) | 160 (6.2) |
| Death other reason (including unknown) | 193 (6.2) | 172 (5.5) | 158 (6.0) | 141 (5.4) |
A summary of the study efficacy results is provided in Table 9.
| Intent-To-Treat Population | Hormone Receptor-Positive Subpopulation | |||
|---|---|---|---|---|
| Anastrozole tablets
1 mg (N=3125) |
Tamoxifen
20 mg (N=3116) |
Anastrozole tablets
1 mg (N=2618) |
Tamoxifen
20 mg (N=2598) |
|
| Number of Events | Number of Events | |||
| Disease-free Survival | 575 | 651 | 424 | 497 |
| Hazard ratio | 0.87 | 0.83 | ||
| 2-sided 95% CI | 0.78 to 0.97 | 0.73 to 0.94 | ||
| p-value | 0.0127 | 0.0049 | ||
| Distant Disease-free Survival | 500 | 530 | 370 | 394 |
| Hazard ratio | 0.94 | 0.93 | ||
| 2-sided 95% CI | 0.83 to 1.06 | 0.80 to 1.07 | ||
| Overall Survival | 411 | 420 | 296 | 301 |
| Hazard ratio | 0.97 | 0.97 | ||
| 2-sided 95% CI | 0.85 to 1.12 | 0.83 to 1.14 |
14.2 First Line Therapy in Postmenopausal Women With Advanced Breast Cancer
Two double-blind, controlled clinical studies of similar design (0030, a North American study and 0027, a predominately European study) were conducted to assess the efficacy of anastrozole tablets compared with tamoxifen as first-line therapy for hormone receptor positive or hormone receptor unknown locally advanced or metastatic breast cancer in postmenopausal women. A total of 1021 patients between the ages of 30 and 92 years old were randomized to receive trial treatment. Patients were randomized to receive 1 mg of anastrozole tablets once daily or 20 mg of tamoxifen once daily. The primary end points for both trials were time to tumor progression, objective tumor response rate, and safety.
Demographics and other baseline characteristics, including patients who had measurable and no measurable disease, patients who were given previous adjuvant therapy, the site of metastatic disease and ethnic origin were similar for the two treatment groups for both trials. The following table summarizes the hormone receptor status at entry for all randomized patients in trials 0030 and 0027.
| Number (%) of subjects | ||||
|---|---|---|---|---|
| Trial 0030 | Trial 0027 | |||
| Receptor status | Anastrozole tablets
1 mg (N=171) |
Tamoxifen
20 mg (N=182) |
Anastrozole tablets
1 mg (N=340) |
Tamoxifen
20 mg (N=328) |
| ER
ER=Estrogen receptor and/or PgR
PgR=Progesterone receptor
|
151 (88.3) | 162 (89.0) | 154 (45.3) | 144 (43.9) |
| ER unknown, PgR Unknown | 19 (11.1) | 20 (11.0) | 185 (54.4) | 183 (55.8) |
For the primary endpoints, trial 0030 showed that anastrozole tablets had a statistically significant advantage over tamoxifen (p=0.006) for time to tumor progression; objective tumor response rates were similar for anastrozole tablets and tamoxifen. Trial 0027 showed that anastrozole tablets and tamoxifen had similar objective tumor response rates and time to tumor progression (see Table 12and Figures 5and 6).
Table 12 below summarizes the results of trial 0030 and trial 0027 for the primary efficacy endpoints.
| Endpoint | Trial 0030 | Trial 0027 | ||
|---|---|---|---|---|
| Anastrozole tablets
1 mg (N=171) |
Tamoxifen
20 mg (N=182) |
Anastrozole tablets
1 mg (N=340) |
Tamoxifen
20 mg (N=328) |
|
| Time to progression (TTP) | ||||
| Median TTP (months) | 11.1 | 5.6 | 8.2 | 8.3 |
| Number (%) of subjects who progressed | 114 (67%) | 138 (76%) | 249 (73%) | 247 (75%) |
| Hazard ratio (LCL
LCL=Lower Confidence Limit )
Tamoxifen:Anastrozole tablets
|
1.42 (1.15) | 1.01 (0.87) | ||
| 2-sided 95% CI
CI=Confidence Interval
|
(1.11, 1.82) | (0.85, 1.20) | ||
| p-value
Two-sided Log Rank
|
0.006 | 0.920 | ||
| Best objective response rate | ||||
| Number (%) of subjects with CR
CR=Complete Response + PR
PR=Partial Response
|
36 (21.1%) | 31 (17.0%) | 112 (32.9%) | 107 (32.6%) |
| Odds Ratio (LCL
)
Anastrozole tablets:Tamoxifen
|
1.30 (0.83) | 1.01 (0.77) |
Figure 5 — Kaplan-Meier probability of time to disease progression for all randomized patients (intent-to-treat) in Trial 0030
Figure 6 — Kaplan-Meier probability of time to progression for all randomized patients (intent-to-treat) in Trial 0027
Results from the secondary endpoints were supportive of the results of the primary efficacy endpoints. There were too few deaths occurring across treatment groups of both trials to draw conclusions on overall survival differences.
14.3 Second Line Therapy in Postmenopausal Women With Advanced Breast Cancer Who Had Disease Progression Following Tamoxifen Therapy
Anastrozole was studied in two controlled clinical trials (0004, a North American study; 0005, a predominately European study) in postmenopausal women with advanced breast cancer who had disease progression following tamoxifen therapy for either advanced or early breast cancer. Some of the patients had also received previous cytotoxic treatment. Most patients were ER-positive; a smaller fraction were ER-unknown or ER-negative; the ER-negative patients were eligible only if they had a positive response to tamoxifen. Eligible patients with measurable and non-measurable disease were randomized to receive either a single daily dose of 1 mg or 10 mg of anastrozole tablets or megestrol acetate 40 mg four times a day. The studies were double-blinded with respect to anastrozole tablets. Time to progression and objective response (only patients with measurable disease could be considered partial responders) rates were the primary efficacy variables. Objective response rates were calculated based on the Union Internationale Contre le Cancer (UICC) criteria. The rate of prolonged (more than 24 weeks) stable disease, the rate of progression, and survival were also calculated.
Both trials included over 375 patients; demographics and other baseline characteristics were similar for the three treatment groups in each trial. Patients in the 0005 trial had responded better to prior tamoxifen treatment. Of the patients entered who had prior tamoxifen therapy for advanced disease (58% in Trial 0004; 57% in Trial 0005), 18% of these patients in Trial 0004 and 42% in Trial 0005 were reported by the primary investigator to have responded. In Trial 0004, 81% of patients were ER-positive, 13% were ER-unknown, and 6% were ER-negative. In Trial 0005, 58% of patients were ER-positive, 37% were ER-unknown, and 5% were ER-negative. In Trial 0004, 62% of patients had measurable disease compared to 79% in Trial 0005. The sites of metastatic disease were similar among treatment groups for each trial. On average, 40% of the patients had soft tissue metastases; 60% had bone metastases; and 40% had visceral (15% liver) metastases.
Efficacy results from the two studies were similar as presented in Table 13. In both studies there were no significant differences between treatment arms with respect to any of the efficacy parameters listed in the table below.
| Anastrozole tablets
1 mg |
Anastrozole tablets
10 mg |
Megestrol Acetate
160 mg |
|
|---|---|---|---|
| Trial 0004 | |||
| ( N. America) | (N=128) | (N=130) | (N=128) |
| Median Follow-up (months)
Surviving Patients
|
31.3 | 30.9 | 32.9 |
| Median Time to Death (months) | 29.6 | 25.7 | 26.7 |
| 2 Year Survival Probability (%) | 62.0 | 58.0 | 53.1 |
| Median Time to Progression (months) | 5.7 | 5.3 | 5.1 |
| Objective Response (all patients ) (%) | 12.5 | 10.0 | 10.2 |
| Stable Disease for >24 weeks (%) | 35.2 | 29.2 | 32.8 |
| Progression (%) | 86.7 | 85.4 | 90.6 |
| Trial 0005 | |||
| ( Europe, Australia, S. Africa) | (N=135) | (N=118) | (N=125) |
| Median Follow-up (months) | 31.0 | 30.9 | 31.5 |
| Median Time to Death (months) | 24.3 | 24.8 | 19.8 |
| 2 Year Survival Probability (%) | 50.5 | 50.9 | 39.1 |
| Median Time to Progression (months) | 4.4 | 5.3 | 3.9 |
| Objective Response (all patients) (%) | 12.6 | 15.3 | 14.4 |
| Stable Disease for >24 weeks (%) | 24.4 | 25.4 | 23.2 |
| Progression (%) | 91.9 | 89.8 | 92.0 |
When data from the two controlled trials are pooled, the objective response rates and median times to progression and death were similar for patients randomized to anastrozole tablets 1 mg and megestrol acetate. There is, in this data, no indication that anastrozole tablets 10 mg are superior to anastrozole tablets 1 mg.
| Trials 0004 & 0005
(Pooled Data) |
Anastrozole tablets
1 mg N=263 |
Anastrozole tablets
10 mg N=248 |
Megestrol Acetate
160 mg N=253 |
|---|---|---|---|
| Median Time to Death (months) | 26.7 | 25.5 | 22.5 |
| 2 Year Survival Probability (%) | 56.1 | 54.6 | 46.3 |
| Median Time to Progression | 4.8 | 5.3 | 4.6 |
| Objective Response (all patients) (%) | 12.5 | 12.5 | 12.3 |
Structured Label Content
Section 42229-5 (42229-5)
Hypersensitivity
Anastrozole tablets are contraindicated in any patient who has shown a hypersensitivity reaction to the drug or to any of the excipients. Observed reactions include anaphylaxis, angioedema, and urticaria [see Adverse Reactions (6.2)].
Section 44425-7 (44425-7)
Storage
Store at controlled room temperature, 20-25°C (68-77°F) [see USP].
7.2 Estrogen
Estrogen-containing therapies should not be used with anastrozole tablets as they may diminish its pharmacological action.
7.3 Warfarin
In a study conducted in 16 male volunteers, anastrozole did not alter the exposure (as measured by C maxand AUC) and anticoagulant activity (as measured by prothrombin time, activated partial thromboplastin time, and thrombin time) of both R- and S-warfarin.
10 Overdosage (10 OVERDOSAGE)
Clinical trials have been conducted with anastrozole tablets, up to 60 mg in a single dose given to healthy male volunteers and up to 10 mg daily given to postmenopausal women with advanced breast cancer; these dosages were tolerated. A single dose of anastrozole tablets that results in life-threatening symptoms has not been established. There is no specific antidote to overdosage and treatment must be symptomatic. In the management of an overdose, consider that multiple agents may have been taken. Vomiting may be induced if the patient is alert. Dialysis may be helpful because anastrozole tablets are not highly protein bound. General supportive care, including frequent monitoring of vital signs and close observation of the patient, is indicated.
7.1 Tamoxifen
Co-administration of anastrozole and tamoxifen in breast cancer patients reduced anastrozole plasma concentration by 27%. However, the co-administration of anastrozole and tamoxifen did not affect the pharmacokinetics of tamoxifen or N-desmethyltamoxifen. At a median follow-up of 33 months, the combination of anastrozole tablets and tamoxifen did not demonstrate any efficacy benefit when compared with tamoxifen in all patients as well as in the hormone receptor-positive subpopulation. This treatment arm was discontinued from the trial [see Clinical Studies (14.1)]. Based on clinical and pharmacokinetic results from the ATAC trial, tamoxifen should not be administered with anastrozole.
11 Description (11 DESCRIPTION)
Anastrozole Tablets, USP for oral administration contain 1 mg of anastrozole, a non-steroidal aromatase inhibitor. It is chemically described as 1,3-Benzenediacetonitrile, a, a, a', a'-tetramethyl-5-(1H-1,2,4-triazol-1-ylmethyl). Its molecular formula is C 17H 19N 5and its structural formula is:
Anastrozole is an off-white powder with a molecular weight of 293.4. Anastrozole has moderate aqueous solubility (0.5 mg/mL at 25°C); solubility is independent of pH in the physiological range. Anastrozole is freely soluble in methanol, acetone, ethanol, and tetrahydrofuran, and very soluble in acetonitrile.
Each tablet contains as inactive ingredients: hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone, sodium starch glycolate and titanium dioxide.
5.3 Cholesterol
During the ATAC trial, more patients receiving anastrozole tablets were reported to have elevated serum cholesterol compared to patients receiving tamoxifen (9% versus 3.5%, respectively) [see Adverse Reactions (6.1)].
5.2 Bone Effects
Results from the ATAC trial bone substudy at 12 and 24 months demonstrated that patients receiving anastrozole tablets had a mean decrease in both lumbar spine and total hip bone mineral density (BMD) compared to baseline. Patients receiving tamoxifen had a mean increase in both lumbar spine and total hip BMD compared to baseline. Consider bone mineral density monitoring in patients treated with anastrozole tablets [see Adverse Reactions (6.1)].
8.4 Pediatric Use
Clinical studies in pediatric patients included a placebo-controlled trial in pubertal boys of adolescent age with gynecomastia and a single-arm trial in girls with McCune-Albright Syndrome and progressive precocious puberty. The efficacy of anastrozole tablets in the treatment of pubertal gynecomastia in adolescent boys and in the treatment of precocious puberty in girls with McCune-Albright Syndrome has not been demonstrated.
8.5 Geriatric Use
In studies 0030 and 0027, about 50% of patients were 65 or older. Patients ≥ 65 years of age had moderately better tumor response and time to tumor progression than patients < 65 years of age regardless of randomized treatment. In studies 0004 and 0005, 50% of patients were 65 or older. Response rates and time to progression were similar for the over 65 and younger patients.
In the ATAC study, 45% of patients were 65 years of age or older. The efficacy of anastrozole tablets compared to tamoxifen in patients who were 65 years or older (N=1413 for anastrozole tablets and N=1410 for tamoxifen, the hazard ratio for disease-free survival was 0.93 [95% CI: 0.80, 1.08]) was less than efficacy observed in patients who were less than 65 years of age (N=1712 for anastrozole tablets and N=1706 for tamoxifen, the hazard ratio for disease-free survival was 0.79 [95% CI: 0.67, 0.94]).
The pharmacokinetics of anastrozole are not affected by age.
4 Contraindications (4 CONTRAINDICATIONS)
- Patients with demonstrated hypersensitivity to anastrozole tablets or any excipient ( 4)
6 Adverse Reactions (6 ADVERSE REACTIONS)
Serious adverse reactions with anastrozole tablets occurring in less than 1 in 10,000 patients, are: 1) skin reactions such as lesions, ulcers, or blisters; 2) allergic reactions with swelling of the face, lips, tongue, and/or throat. This may cause difficulty in swallowing and/or breathing; and 3) changes in blood tests of the liver function, including inflammation of the liver with symptoms that may include a general feeling of not being well, with or without jaundice, liver pain or liver swelling [see Adverse Reactions (6.2)].
Common adverse reactions (occurring with an incidence of ≥10%) in women taking anastrozole tablets included: hot flashes, asthenia, arthritis, pain, arthralgia, hypertension, depression, nausea and vomiting, rash, osteoporosis, fractures, back pain, insomnia, headache, bone pain, peripheral edema, increased cough, dyspnea, pharyngitis and lymphedema.
In the ATAC trial, the most common reported adverse reaction (>0.1%) leading to discontinuation of therapy for both treatment groups was hot flashes, although there were fewer patients who discontinued therapy as a result of hot flashes in the anastrozole tablets group.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions (7 DRUG INTERACTIONS)
7.4 Cytochrome P450
Based on in vitroand in vivoresults, it is unlikely that co-administration of anastrozole tablets 1 mg will affect other drugs as a result of inhibition of cytochrome P450 [see Clinical Pharmacology (12.3)].
Patient Information
What is the most important information I should know about anastrozole tablets?
Anastrozole tablets may cause serious side effects including:
-
heart disease.Women with early breast cancer, who have a history of blockage in their heart arteries (ischemic heart disease) and who take anastrozole tablets, may have an increase in symptoms of decreased blood flow to their heart compared to similar women who take tamoxifen.
Get medical help right away if you have new or worsening chest pain or shortness of breath during treatment with anastrozole tablets.
What are anastrozole tablets?
Anastrozole tablets are a prescription medicine used in women after menopause ("the change of life") for:
- treatment of early breast cancer
- after surgery
- in women whose breast cancer is hormone receptor-positive
- the first treatment of breast cancer that has spread to nearby tissue or lymph nodes (locally advanced) or has spread to other parts of the body (metastatic), in women whose breast cancer is hormone receptor-positive or the hormone receptors are not known.
- treatment of advanced breast cancer, if the cancer has grown, or the disease has spread after tamoxifen therapy
Anastrozole tablets does not work in women with breast cancer who have not gone through menopause (premenopausal women).
Who should not take anastrozole tablets?
Do not take anastrozole tablet if you:
- have had a severe allergic reaction to anastrozole or any of the ingredients in anastrozole tablets. See the end of this leaflet for a complete list of ingredients in anastrozole tablets. Symptoms of a severe allergic reaction to anastrozole tablets include: swelling of the face, lips, tongue or throat, trouble breathing or swallowing, hives and itching.
What should I tell my healthcare provider before taking anastrozole tablets?
Before you take anastrozole tablets, tell your healthcare provider if you:
- are still having menstrual periods (are not past menopause). Talk to your healthcare provider if you are not sure.
- have or had a heart problem
- have been told you have bone thinning or weakness (osteoporosis)
- have high cholesterol
- have any other medical conditions
- are pregnant or plan to become pregnant. Taking anastrozole tablets during pregnancy or within 3 weeks of becoming pregnant may harm your unborn baby.
- Females who are able to become pregnant should have a pregnancy test before starting treatment with anastrozole tablets.
- Females who are able to become pregnant should use effective birth control (contraceptive) during treatment with anastrozole tablets and for 3 weeks after your last dose of anastrozole tablets. Tell your healthcare provider right away if you become pregnant or think you may be pregnant.
- are breastfeeding or plan to breastfeed. It is not known if anastrozole tablets passes into breast milk. Do not breastfeed during treatment with anastrozole tablets and for 2 weeks after your last dose of anastrozole tablets.
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take:
- tamoxifen.You should not take anastrozole tabletsif you take tamoxifen. Taking anastrozole tablets with tamoxifen may lower the amount of anastrozole tablets in your blood and may cause anastrozole tablets not to work as well.
-
Medicines that contain estrogen.Anastrozole tablets may not work if taken with any of these medicines:
- hormone replacement therapy
- birth control pills
- estrogen creams
- vaginal rings
- vaginal suppositories
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take anastrozole tablets?
- Take anastrozole tablets exactly as your healthcare provider tells you to take it.
- Continue taking anastrozole tablets until your healthcare provider tells you to stop.
- Anastrozole tablets can be taken with or without food.
- If you miss a dose, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Take your next regularly scheduled dose. Do not take two doses at the same time.
If you take too much anastrozole tablets, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of anastrozole tablets?
Anastrozole tablets may cause serious side effects including:
- See " What is the most important information I should know about anastrozole tablets?"
- bone thinning or weakness (osteoporosis).Anastrozole tablets lowers estrogen in your body, which may cause your bones to become thinner and weaker. This may increase your risk of fractures, especially of your spine, hip and wrist. Your healthcare provider may order a bone mineral density test before you start and during treatment with anastrozole tablets to check you for bone changes.
- increased blood cholesterol (fat in the blood).Your healthcare provider may do blood tests to check your cholesterol while you are taking anastrozole tablets.
- skin reactions.Stop taking anastrozole tablets and call your healthcare provider right away if you get any skin lesions, ulcers, or blisters.
-
severe allergic reactions.Get medical help right away if you get:
- swelling of your face, lips, tongue, or throat
- trouble swallowing or breathing
- liver problems.Anastrozole tablets can cause inflammation of your liver and changes in liver function blood tests. Your healthcare provider may check you for this.
Stop taking anastrozole tablets and call your healthcare provider right awayif you have any of these signs or symptoms of a liver problem:
- a general feeling of not being well
- yellowing of your skin or whites of your eyes
- pain on the right side of your stomach-area (abdomen)
Common side effects in women taking anastrozole tablets include:
- hot flashes
- weakness
- joint aches
- joint pain, stiffness or swelling (arthritis)
- pain
- sore throat
- high blood pressure
- depression
- nausea and vomiting
- rash
- back pain
- sleep problems
- bone pain
- headache
- swelling of your legs, ankles, or feet
- increased cough
- shortness of breath
- build up of lymph fluid in the tissues of your affected arm (lymphedema)
Anastrozole tablets may also cause you to have tickling, tingling or numbness of your skin.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of anastrozole tablets. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store anastrozole tablets?
- Store anastrozole tablets at room temperature between 68°F to 77°F (20°C to 25°C).
Keep anastrozole tablets and all medicines out of the reach of children.
General information about the safe and effective use of anastrozole tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not take anastrozole tablets for a condition for which it was not prescribed. Do not give anastrozole tablets to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about anastrozole tablets that is written for health professionals. For more information, call 1-800-367-3395 or go to www.breckenridgepharma.com.
What are the ingredients in anastrozole tablets?
Active ingredient:anastrozole, USP
Inactive ingredients:hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone, sodium starch glycolate, and titanium dioxide.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Natco Pharma Limited.
Visakhapatnam - 531019
AP, India.
Distributed by:
Breckenridge Pharmaceutical, Inc.
Berkeley Heights, NJ 07922
XXXXXXXX
Revised: 10/2024
2.1 Recommended Dose
The dose of anastrozole tablets are one 1 mg tablet taken once a day. For patients with advanced breast cancer, anastrozole tablets should be continued until tumor progression. Anastrozole tablets can be taken with or without food.
For adjuvant treatment of early breast cancer in postmenopausal women, the optimal duration of therapy is unknown. In the ATAC trial, anastrozole tablets were administered for five years [see Clinical Studies (14.1)] .
No dosage adjustment is necessary for patients with renal impairment or for elderly patients [see Use in Specific Populations (8.6)].
8.6 Renal Impairment
Since only about 10% of anastrozole is excreted unchanged in the urine, the renal impairment does not influence the total body clearance. Dosage adjustment in patients with renal impairment is not necessary [see Dosage and Administration (2.1)and Clinical Pharmacology (12.3)].
1.1 Adjuvant Treatment
Anastrozole tablets are indicated for adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer.
8.7 Hepatic Impairment
The plasma anastrozole concentrations in the subjects with hepatic cirrhosis were within the range of concentrations seen in normal subjects across all clinical trials. Therefore, dosage adjustment is also not necessary in patients with stable hepatic cirrhosis. Anastrozole tablets have not been studied in patients with severe hepatic impairment [see Dosage and Administration (2.2)and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Anastrozole tablets are an aromatase inhibitor indicated for:
- Adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer ( 1.1)
- First-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer ( 1.2)
- Treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets ( 1.3)
1.2 First Line Treatment (1.2 First-Line Treatment)
Anastrozole tablets are indicated for the first-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer.
12.1 Mechanism of Action
The growth of many cancers of the breast is stimulated or maintained by estrogens.
In postmenopausal women, estrogens are mainly derived from the action of the aromatase enzyme, which converts adrenal androgens (primarily androstenedione and testosterone) to estrone and estradiol. The suppression of estrogen biosynthesis in peripheral tissues and in the cancer tissue itself can therefore be achieved by specifically inhibiting the aromatase enzyme.
Anastrozole is a selective non-steroidal aromatase inhibitor. It significantly lowers serum estradiol concentrations and has no detectable effect on formation of adrenal corticosteroids or aldosterone.
1.3 Second Line Treatment (1.3 Second-Line Treatment)
Anastrozole tablets are indicated for the treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to anastrozole tablets.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, anastrozole tablets can cause fetal harm when administered to a pregnant woman. Anastrozole caused embryo-fetal toxicities in rats at maternal exposure that were 9 times the human clinical exposure, based on area under the curve (AUC). In rabbits, anastrozole caused pregnancy failure at doses equal to or greater than 16 times the recommended human dose on a mg/m 2basis. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during therapy with anastrozole tablets and for at least 3 weeks after the last dose [see Use in Specific Populations (8.1, 8.3)and Clinical Pharmacology (12.1)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- In women with pre-existing ischemic heart disease, an increased incidence of ischemic cardiovascular events occurred with anastrozole tablet use compared to tamoxifen use. Consider risks and benefits. ( 5.1, 6.1)
- Decreases in bone mineral density may occur. Consider bone mineral density monitoring. ( 5.2, 6.1)
- Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.3, 6.1)
- Embryo-Fetal Toxicity: Anastrozole tablets may cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.4, 8.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
One 1 mg tablet taken once daily ( 2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
The tablets are white to off-white, round, film-coated containing 1 mg of anastrozole. The tablets are debossed with "AN" and "1" on one side and plain surface on the other side.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
These adverse reactions are reported voluntarily from a population of uncertain size. Therefore, it is not always possible to estimate reliably their frequency or establish a causal relationship to drug exposure. The following have been reported in post-approval use of anastrozole tablets:
- Hepatobiliary events including increases in alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, gamma-GT, and bilirubin; hepatitis
- Rash including cases of mucocutaneous disorders such as erythema multiforme and Stevens-Johnson syndrome
- Cases of allergic reactions including angioedema, urticaria and anaphylaxis [see Contraindications (4)]
- Myalgia, trigger finger and hypercalcemia (with or without an increase in parathyroid hormone)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Do not breastfeed. ( 8.2)
- Females and Males of Reproductive Potential: Verify pregnancy status prior to initiation of anastrozole tablets. ( 8.3)
- Pediatric patients: Efficacy has not been demonstrated for pubertal boys of adolescent age with gynecomastia or girls with McCune-Albright Syndrome and progressive precocious puberty. ( 8.4)
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA approved patient labeling ( Patient Information).
5.1 Ischemic Cardiovascular Events
In women with pre-existing ischemic heart disease, an increased incidence of ischemic cardiovascular events was observed with anastrozole tablets in the ATAC trial (17% of patients on anastrozole tablets and 10% of patients on tamoxifen). Consider risk and benefits of anastrozole tablets therapy in patients with pre-existing ischemic heart disease [see Adverse Reactions (6.1)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
These tablets are supplied in bottles 90 tablets (NDC 680701-3838-9)
2.2 Patients With Hepatic Impairment (2.2 Patients with Hepatic Impairment)
No changes in dose are recommended for patients with mild-to-moderate hepatic impairment. Anastrozole tablets have not been studied in patients with severe hepatic impairment [see Use in Specific Populations (8.7)].
Principal Display Panel 1 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label)
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A conventional carcinogenesis study in rats at doses of 1.0 to 25 mg/kg/day (about 10 to 243 times the daily maximum recommended human dose on a mg/m 2basis) administered by oral gavage for up to 2 years revealed an increase in the incidence of hepatocellular adenoma and carcinoma and uterine stromal polyps in females and thyroid adenoma in males at the high dose. A dose-related increase was observed in the incidence of ovarian and uterine hyperplasia in females. At 25 mg/kg/day, plasma AUC 0-24 hrlevels in rats were 110 to 125 times higher than the level exhibited in postmenopausal volunteers at the recommended dose. A separate carcinogenicity study in mice at oral doses of 5 to 50 mg/kg/day (about 24 to 243 times the daily maximum recommended human dose on a mg/m 2basis) for up to 2 years produced an increase in the incidence of benign ovarian stromal, epithelial and granulosa cell tumors at all dose levels. A dose-related increase in the incidence of ovarian hyperplasia was also observed in female mice. These ovarian changes are considered to be rodent-specific effects of aromatase inhibition and are of questionable significance to humans. The incidence of lymphosarcoma was increased in males and females at the high dose. At 50 mg/kg/day, plasma AUC levels in mice were 35 to 40 times higher than the level exhibited in postmenopausal volunteers at the recommended dose.
Anastrozole tablets have not been shown to be mutagenic in in vitrotests (Ames and E. coli bacterial tests, CHO-K1 gene mutation assay) or clastogenic either in vitro(chromosome aberrations in human lymphocytes) or in vivo(micronucleus test in rats).
Oral administration of anastrozole to female rats (from 2 weeks before mating to pregnancy day 7) produced significant incidence of infertility and reduced numbers of viable pregnancies at 1 mg/kg/day (about 10 times the recommended human dose on a mg/m 2basis and 9 times higher than the AUC 0-24 hrfound in postmenopausal volunteers at the recommended dose). Pre-implantation loss of ova or fetus was increased at doses equal to or greater than 0.02 mg/kg/day (about one-fifth the recommended human dose on a mg/m 2basis). Recovery of fertility was observed following a 5-week non-dosing period which followed 3 weeks of dosing. It is not known whether these effects observed in female rats are indicative of impaired fertility in humans.
Multiple-dose studies in rats administered anastrozole for 6 months at doses equal to or greater than 1 mg/kg/day (which produced plasma anastrozole C ssmaxand AUC 0-24 hrthat were 19 and 9 times higher than the respective values found in postmenopausal volunteers at the recommended dose) resulted in hypertrophy of the ovaries and the presence of follicular cysts. In addition, hyperplastic uteri were observed in 6-month studies in female dogs administered doses equal to or greater than 1 mg/kg/day (which produced plasma anastrozole C ssmaxand AUC 0-24 hrthat were 22 times and 16 times higher than the respective values found in postmenopausal women at the recommended dose). It is not known whether these effects on the reproductive organs of animals are associated with impaired fertility in premenopausal women.
14.1 Adjuvant Treatment of Breast Cancer in Postmenopausal Women
A multicenter, double-blind trial (ATAC) randomized 9,366 postmenopausal women with operable breast cancer to adjuvant treatment with anastrozole tablets 1 mg daily, tamoxifen 20 mg daily, or a combination of the two treatments for five years or until recurrence of the disease.
The primary endpoint of the trial was disease-free survival (i.e., time to occurrence of a distant or local recurrence, or contralateral breast cancer or death from any cause). Secondary endpoints of the trial included distant disease-free survival, the incidence of contralateral breast cancer and overall survival. At a median follow-up of 33 months, the combination of anastrozole tablets and tamoxifen did not demonstrate any efficacy benefit when compared with tamoxifen in all patients as well as in the hormone receptor positive subpopulation. This treatment arm was discontinued from the trial. Based on clinical and pharmacokinetic results from the ATAC trial, tamoxifen should not be administered with anastrozole [see Drug Interactions (7.1)].
Demographic and other baseline characteristics were similar among the three treatment groups (see Table 7).
| Demographic Characteristic | Anastrozole tablets
1 mg (N N=Number of patients randomized to the treatment =3125)
|
Tamoxifen
20 mg (N =3116) |
Anastrozole tablets 1 mg plus Tamoxifen
The combination arm was discontinued due to lack of efficacy benefit at 33 months of follow-up 20 mg
(N =3125) |
|---|---|---|---|
| Mean age (yrs.) | 64.1 | 64.1 | 64.3 |
| Age Range (yrs.) | 38.1 - 92.8 | 32.8 - 94.9 | 37.0 – 92.2 |
| Age Distribution (%) | |||
| <45 yrs. | 0.7 | 0.4 | 0.5 |
| 45-60 yrs. | 34.6 | 35.0 | 34.5 |
| >60 <70 yrs. | 38.0 | 37.1 | 37.7 |
| >70 yrs. | 26.7 | 27.4 | 27.3 |
| Mean Weight (kg) | 70.8 | 71.1 | 71.3 |
| Receptor Status (%) | |||
| Positive
Includes patients who were estrogen receptor (ER) positive or progesterone receptor (PgR) positive, or both positive
|
83.5 | 83.1 | 84.0 |
| Negative
Includes patients with both ER negative and PgR negative receptor status
|
7.4 | 8.0 | 7.0 |
| Other
Includes all other combinations of ER and PgR receptor status unknown
|
8.8 | 8.6 | 9.0 |
| Other Treatment (%) prior to Randomization | |||
| Mastectomy | 47.8 | 47.3 | 48.1 |
| Breast conservation
Among the patients who had breast conservation, radiotherapy was administered to 95.0% of patients in the anastrozole tablets arm, 94.1% in the tamoxifen arm and 94.5% in the anastrozole tablets plus tamoxifen arm.
|
52.3 | 52.8 | 51.9 |
| Axillary surgery | 95.5 | 95.7 | 95.2 |
| Radiotherapy | 63.3 | 62.5 | 61.9 |
| Chemotherapy | 22.3 | 20.8 | 20.8 |
| Neoadjuvant Tamoxifen | 1.6 | 1.6 | 1.7 |
| Primary Tumor Size (%) | |||
| T1 (≤2 cm) | 63.9 | 62.9 | 64.1 |
| T2 (>2 cm and ≤5 cm) | 32.6 | 34.2 | 32.9 |
| T3 (>5 cm) | 2.7 | 2.2 | 2.3 |
| Nodal Status (%) | |||
| Node positive | 34.9 | 33.6 | 33.5 |
| 1-3 (# of nodes) | 24.4 | 24.4 | 24.3 |
| 4-9 | 7.5 | 6.4 | 6.8 |
| >9 | 2.9 | 2.7 | 2.3 |
| Tumor Grade (%) | |||
| Well-differentiated | 20.8 | 20.5 | 21.2 |
| Moderately differentiated | 46.8 | 47.8 | 46.5 |
| Poorly/undifferentiated | 23.7 | 23.3 | 23.7 |
| Not assessed/recorded | 8.7 | 8.4 | 8.5 |
Patients in the two monotherapy arms of the ATAC trial were treated for a median of 60 months (5 years) and followed for a median of 68 months. Disease-free survival in the intent-to-treat population was statistically significantly improved [Hazard Ratio (HR) = 0.87, 95% CI: 0.78, 0.97, p=0.0127] in the anastrozole tablets arm compared to the tamoxifen arm. In the hormone receptor-positive subpopulation representing about 84% of the trial patients, disease-free survival was also statistically significantly improved (HR =0.83, 95% CI: 0.73, 0.94, p=0.0049) in the anastrozole tablets arm compared to the tamoxifen arm.
Figure 1 – Disease-Free Survival Kaplan Meier Survival Curve for all Patients Randomized to Anastrozole Tablets or Tamoxifen Monotherapy in the ATAC Trial (Intent-to-Treat)
Figure 2 – Disease-Free Survival for Hormone Receptor-Positive Subpopulation of Patients Randomized to Anastrozole Tablets or Tamoxifen Monotherapy in the ATAC Trial
The survival data with 68 months follow-up is presented in Table 9.
In the group of patients who had previous adjuvant chemotherapy (N=698 for anastrozole tablets and N=647 for tamoxifen), the hazard ratio for disease-free survival was 0.91 (95% CI: 0.73 to 1.13) in the anastrozole tablets arm compared to the tamoxifen arm.
The frequency of individual events in the intent-to-treat population and the hormone receptor-positive subpopulation are described in Table 8.
| Intent-To-Treat Population
Patients may fall into more than one category.
|
Hormone Receptor-Positive Subpopulation | |||
|---|---|---|---|---|
| Anastrozole tablets
1 mg (N N=Number of patients randomized =3125)
|
Tamoxifen
20 mg (N =3116) |
Anastrozole tablets
1 mg (N =2618) |
Tamoxifen
20 mg (N =2598) |
|
| Median Duration of Therapy (mo) | 60 | 60 | 60 | 60 |
| Median Efficacy Follow-up (mo) | 68 | 68 | 68 | 68 |
| Loco-regional recurrence | 119 (3.8) | 149 (4.8) | 76 (2.9) | 101 (3.9) |
| Contralateral breast cancer | 35 (1.1) | 59 (1.9) | 26 (1.0) | 54 (2.1) |
| Invasive | 27 (0.9) | 52 (1.7) | 21 (0.8) | 48 (1.8) |
| Ductal carcinoma in situ | 8 (0.3) | 6 (0.2) | 5 (0.2) | 5 (0.2) |
| Unknown | 0 | 1 (<0.1) | 0 | 1 (<0.1) |
| Distant recurrence | 324 (10.4) | 375 (12.0) | 226 (8.6) | 265 (10.2) |
| Death from Any Cause | 411 (13.2) | 420 (13.5) | 296 (11.3) | 301 (11.6) |
| Death breast cancer | 218 (7.0) | 248 (8.0) | 138 (5.3) | 160 (6.2) |
| Death other reason (including unknown) | 193 (6.2) | 172 (5.5) | 158 (6.0) | 141 (5.4) |
A summary of the study efficacy results is provided in Table 9.
| Intent-To-Treat Population | Hormone Receptor-Positive Subpopulation | |||
|---|---|---|---|---|
| Anastrozole tablets
1 mg (N=3125) |
Tamoxifen
20 mg (N=3116) |
Anastrozole tablets
1 mg (N=2618) |
Tamoxifen
20 mg (N=2598) |
|
| Number of Events | Number of Events | |||
| Disease-free Survival | 575 | 651 | 424 | 497 |
| Hazard ratio | 0.87 | 0.83 | ||
| 2-sided 95% CI | 0.78 to 0.97 | 0.73 to 0.94 | ||
| p-value | 0.0127 | 0.0049 | ||
| Distant Disease-free Survival | 500 | 530 | 370 | 394 |
| Hazard ratio | 0.94 | 0.93 | ||
| 2-sided 95% CI | 0.83 to 1.06 | 0.80 to 1.07 | ||
| Overall Survival | 411 | 420 | 296 | 301 |
| Hazard ratio | 0.97 | 0.97 | ||
| 2-sided 95% CI | 0.85 to 1.12 | 0.83 to 1.14 |
14.2 First Line Therapy in Postmenopausal Women With Advanced Breast Cancer (14.2 First-Line Therapy in Postmenopausal Women with Advanced Breast Cancer)
Two double-blind, controlled clinical studies of similar design (0030, a North American study and 0027, a predominately European study) were conducted to assess the efficacy of anastrozole tablets compared with tamoxifen as first-line therapy for hormone receptor positive or hormone receptor unknown locally advanced or metastatic breast cancer in postmenopausal women. A total of 1021 patients between the ages of 30 and 92 years old were randomized to receive trial treatment. Patients were randomized to receive 1 mg of anastrozole tablets once daily or 20 mg of tamoxifen once daily. The primary end points for both trials were time to tumor progression, objective tumor response rate, and safety.
Demographics and other baseline characteristics, including patients who had measurable and no measurable disease, patients who were given previous adjuvant therapy, the site of metastatic disease and ethnic origin were similar for the two treatment groups for both trials. The following table summarizes the hormone receptor status at entry for all randomized patients in trials 0030 and 0027.
| Number (%) of subjects | ||||
|---|---|---|---|---|
| Trial 0030 | Trial 0027 | |||
| Receptor status | Anastrozole tablets
1 mg (N=171) |
Tamoxifen
20 mg (N=182) |
Anastrozole tablets
1 mg (N=340) |
Tamoxifen
20 mg (N=328) |
| ER
ER=Estrogen receptor and/or PgR
PgR=Progesterone receptor
|
151 (88.3) | 162 (89.0) | 154 (45.3) | 144 (43.9) |
| ER unknown, PgR Unknown | 19 (11.1) | 20 (11.0) | 185 (54.4) | 183 (55.8) |
For the primary endpoints, trial 0030 showed that anastrozole tablets had a statistically significant advantage over tamoxifen (p=0.006) for time to tumor progression; objective tumor response rates were similar for anastrozole tablets and tamoxifen. Trial 0027 showed that anastrozole tablets and tamoxifen had similar objective tumor response rates and time to tumor progression (see Table 12and Figures 5and 6).
Table 12 below summarizes the results of trial 0030 and trial 0027 for the primary efficacy endpoints.
| Endpoint | Trial 0030 | Trial 0027 | ||
|---|---|---|---|---|
| Anastrozole tablets
1 mg (N=171) |
Tamoxifen
20 mg (N=182) |
Anastrozole tablets
1 mg (N=340) |
Tamoxifen
20 mg (N=328) |
|
| Time to progression (TTP) | ||||
| Median TTP (months) | 11.1 | 5.6 | 8.2 | 8.3 |
| Number (%) of subjects who progressed | 114 (67%) | 138 (76%) | 249 (73%) | 247 (75%) |
| Hazard ratio (LCL
LCL=Lower Confidence Limit )
Tamoxifen:Anastrozole tablets
|
1.42 (1.15) | 1.01 (0.87) | ||
| 2-sided 95% CI
CI=Confidence Interval
|
(1.11, 1.82) | (0.85, 1.20) | ||
| p-value
Two-sided Log Rank
|
0.006 | 0.920 | ||
| Best objective response rate | ||||
| Number (%) of subjects with CR
CR=Complete Response + PR
PR=Partial Response
|
36 (21.1%) | 31 (17.0%) | 112 (32.9%) | 107 (32.6%) |
| Odds Ratio (LCL
)
Anastrozole tablets:Tamoxifen
|
1.30 (0.83) | 1.01 (0.77) |
Figure 5 — Kaplan-Meier probability of time to disease progression for all randomized patients (intent-to-treat) in Trial 0030
Figure 6 — Kaplan-Meier probability of time to progression for all randomized patients (intent-to-treat) in Trial 0027
Results from the secondary endpoints were supportive of the results of the primary efficacy endpoints. There were too few deaths occurring across treatment groups of both trials to draw conclusions on overall survival differences.
14.3 Second Line Therapy in Postmenopausal Women With Advanced Breast Cancer Who Had Disease Progression Following Tamoxifen Therapy (14.3 Second-Line Therapy in Postmenopausal Women with Advanced Breast Cancer who had Disease Progression following Tamoxifen Therapy)
Anastrozole was studied in two controlled clinical trials (0004, a North American study; 0005, a predominately European study) in postmenopausal women with advanced breast cancer who had disease progression following tamoxifen therapy for either advanced or early breast cancer. Some of the patients had also received previous cytotoxic treatment. Most patients were ER-positive; a smaller fraction were ER-unknown or ER-negative; the ER-negative patients were eligible only if they had a positive response to tamoxifen. Eligible patients with measurable and non-measurable disease were randomized to receive either a single daily dose of 1 mg or 10 mg of anastrozole tablets or megestrol acetate 40 mg four times a day. The studies were double-blinded with respect to anastrozole tablets. Time to progression and objective response (only patients with measurable disease could be considered partial responders) rates were the primary efficacy variables. Objective response rates were calculated based on the Union Internationale Contre le Cancer (UICC) criteria. The rate of prolonged (more than 24 weeks) stable disease, the rate of progression, and survival were also calculated.
Both trials included over 375 patients; demographics and other baseline characteristics were similar for the three treatment groups in each trial. Patients in the 0005 trial had responded better to prior tamoxifen treatment. Of the patients entered who had prior tamoxifen therapy for advanced disease (58% in Trial 0004; 57% in Trial 0005), 18% of these patients in Trial 0004 and 42% in Trial 0005 were reported by the primary investigator to have responded. In Trial 0004, 81% of patients were ER-positive, 13% were ER-unknown, and 6% were ER-negative. In Trial 0005, 58% of patients were ER-positive, 37% were ER-unknown, and 5% were ER-negative. In Trial 0004, 62% of patients had measurable disease compared to 79% in Trial 0005. The sites of metastatic disease were similar among treatment groups for each trial. On average, 40% of the patients had soft tissue metastases; 60% had bone metastases; and 40% had visceral (15% liver) metastases.
Efficacy results from the two studies were similar as presented in Table 13. In both studies there were no significant differences between treatment arms with respect to any of the efficacy parameters listed in the table below.
| Anastrozole tablets
1 mg |
Anastrozole tablets
10 mg |
Megestrol Acetate
160 mg |
|
|---|---|---|---|
| Trial 0004 | |||
| ( N. America) | (N=128) | (N=130) | (N=128) |
| Median Follow-up (months)
Surviving Patients
|
31.3 | 30.9 | 32.9 |
| Median Time to Death (months) | 29.6 | 25.7 | 26.7 |
| 2 Year Survival Probability (%) | 62.0 | 58.0 | 53.1 |
| Median Time to Progression (months) | 5.7 | 5.3 | 5.1 |
| Objective Response (all patients ) (%) | 12.5 | 10.0 | 10.2 |
| Stable Disease for >24 weeks (%) | 35.2 | 29.2 | 32.8 |
| Progression (%) | 86.7 | 85.4 | 90.6 |
| Trial 0005 | |||
| ( Europe, Australia, S. Africa) | (N=135) | (N=118) | (N=125) |
| Median Follow-up (months) | 31.0 | 30.9 | 31.5 |
| Median Time to Death (months) | 24.3 | 24.8 | 19.8 |
| 2 Year Survival Probability (%) | 50.5 | 50.9 | 39.1 |
| Median Time to Progression (months) | 4.4 | 5.3 | 3.9 |
| Objective Response (all patients) (%) | 12.6 | 15.3 | 14.4 |
| Stable Disease for >24 weeks (%) | 24.4 | 25.4 | 23.2 |
| Progression (%) | 91.9 | 89.8 | 92.0 |
When data from the two controlled trials are pooled, the objective response rates and median times to progression and death were similar for patients randomized to anastrozole tablets 1 mg and megestrol acetate. There is, in this data, no indication that anastrozole tablets 10 mg are superior to anastrozole tablets 1 mg.
| Trials 0004 & 0005
(Pooled Data) |
Anastrozole tablets
1 mg N=263 |
Anastrozole tablets
10 mg N=248 |
Megestrol Acetate
160 mg N=253 |
|---|---|---|---|
| Median Time to Death (months) | 26.7 | 25.5 | 22.5 |
| 2 Year Survival Probability (%) | 56.1 | 54.6 | 46.3 |
| Median Time to Progression | 4.8 | 5.3 | 4.6 |
| Objective Response (all patients) (%) | 12.5 | 12.5 | 12.3 |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:54.857838 · Updated: 2026-03-14T22:25:56.100461