These Highlights Do Not Include All The Information Needed To Use Emgality Safely And Effectively. See Full Prescribing Information For Emgality.
33a147be-233a-40e8-a55e-e40936e28db0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.2 , 5.3 ) 03/2025
Indications and Usage
EMGALITY ® is a calcitonin-gene related peptide antagonist indicated in adults for the: preventive treatment of migraine. ( 1.1 ) treatment of episodic cluster headache. ( 1.2 )
Dosage and Administration
For subcutaneous use only. ( 2.1 , 2.2 , 2.3 ) Migraine recommended dosage: 240 mg loading dose (administered as two consecutive injections of 120 mg each), followed by monthly doses of 120 mg. ( 2.1 ) Episodic cluster headache recommended dosage: 300 mg (administered as three consecutive injections of 100 mg each) at the onset of the cluster period, and then monthly until the end of the cluster period. ( 2.2 ) Administer in the abdomen, thigh, back of the upper arm, or buttocks subcutaneously. ( 2.3 )
Warnings and Precautions
Hypersensitivity Reactions: If a serious hypersensitivity reaction occurs, discontinue administration of EMGALITY and initiate appropriate therapy. Hypersensitivity reactions can occur days after administration, and may be prolonged. ( 5.1 ) Hypertension: New-onset or worsening of pre-existing hypertension may occur. ( 5.2 ) Raynaud's Phenomenon: New-onset or worsening of pre-existing Raynaud's phenomenon may occur. ( 5.3 )
Contraindications
EMGALITY is contraindicated in patients with serious hypersensitivity to galcanezumab-gnlm or to any of the excipients [see Warnings and Precautions ( 5.1 )] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Contraindications ( 4 ) and Warnings and Precautions ( 5.1 )] Hypertension [see Warnings and Precautions ( 5.2 )] Raynaud's Phenomenon [see Warnings and Precautions ( 5.3 )]
Storage and Handling
Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect EMGALITY from light until use. Do not freeze. Do not shake. EMGALITY may be stored out of refrigeration in the original carton at temperatures up to 30°C (86°F) for up to 7 days. Once stored out of refrigeration, do not place back in the refrigerator. If these conditions are exceeded, EMGALITY must be discarded. Discard the EMGALITY single-dose prefilled pen or syringe after use in a puncture-resistant container.
How Supplied
EMGALITY (galcanezumab-gnlm) injection is a sterile, preservative-free, clear to opalescent, colorless to slightly yellow to slightly brown solution for subcutaneous administration. EMGALITY is not made with natural rubber latex. EMGALITY is supplied as follows: Pack Size NDC Prefilled pen 120 mg/mL single-dose Carton of 1 0002-1436-11 120 mg/mL single-dose Carton of 2 0002-1436-27 Prefilled syringe 100 mg/mL single-dose Carton of 3 0002-3115-09 120 mg/mL single-dose Carton of 1 0002-2377-11 120 mg/mL single-dose Carton of 2 0002-2377-27
Medication Information
Warnings and Precautions
Hypersensitivity Reactions: If a serious hypersensitivity reaction occurs, discontinue administration of EMGALITY and initiate appropriate therapy. Hypersensitivity reactions can occur days after administration, and may be prolonged. ( 5.1 ) Hypertension: New-onset or worsening of pre-existing hypertension may occur. ( 5.2 ) Raynaud's Phenomenon: New-onset or worsening of pre-existing Raynaud's phenomenon may occur. ( 5.3 )
Indications and Usage
EMGALITY ® is a calcitonin-gene related peptide antagonist indicated in adults for the: preventive treatment of migraine. ( 1.1 ) treatment of episodic cluster headache. ( 1.2 )
Dosage and Administration
For subcutaneous use only. ( 2.1 , 2.2 , 2.3 ) Migraine recommended dosage: 240 mg loading dose (administered as two consecutive injections of 120 mg each), followed by monthly doses of 120 mg. ( 2.1 ) Episodic cluster headache recommended dosage: 300 mg (administered as three consecutive injections of 100 mg each) at the onset of the cluster period, and then monthly until the end of the cluster period. ( 2.2 ) Administer in the abdomen, thigh, back of the upper arm, or buttocks subcutaneously. ( 2.3 )
Contraindications
EMGALITY is contraindicated in patients with serious hypersensitivity to galcanezumab-gnlm or to any of the excipients [see Warnings and Precautions ( 5.1 )] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Contraindications ( 4 ) and Warnings and Precautions ( 5.1 )] Hypertension [see Warnings and Precautions ( 5.2 )] Raynaud's Phenomenon [see Warnings and Precautions ( 5.3 )]
Storage and Handling
Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect EMGALITY from light until use. Do not freeze. Do not shake. EMGALITY may be stored out of refrigeration in the original carton at temperatures up to 30°C (86°F) for up to 7 days. Once stored out of refrigeration, do not place back in the refrigerator. If these conditions are exceeded, EMGALITY must be discarded. Discard the EMGALITY single-dose prefilled pen or syringe after use in a puncture-resistant container.
How Supplied
EMGALITY (galcanezumab-gnlm) injection is a sterile, preservative-free, clear to opalescent, colorless to slightly yellow to slightly brown solution for subcutaneous administration. EMGALITY is not made with natural rubber latex. EMGALITY is supplied as follows: Pack Size NDC Prefilled pen 120 mg/mL single-dose Carton of 1 0002-1436-11 120 mg/mL single-dose Carton of 2 0002-1436-27 Prefilled syringe 100 mg/mL single-dose Carton of 3 0002-3115-09 120 mg/mL single-dose Carton of 1 0002-2377-11 120 mg/mL single-dose Carton of 2 0002-2377-27
Description
Warnings and Precautions ( 5.2 , 5.3 ) 03/2025
Section 42229-5
Migraine
The safety of EMGALITY has been evaluated in 2586 patients with migraine who received at least one dose of EMGALITY, representing 1487 patient-years of exposure. Of these, 1920 patients were exposed to EMGALITY once monthly for at least 6 months, and 526 patients were exposed for 12 months.
In placebo-controlled clinical studies (Studies 1, 2, and 3), 705 patients received at least one dose of EMGALITY 120 mg once monthly, and 1451 patients received placebo, during 3 months or 6 months of double-blind treatment [see Clinical Studies (14.1)]. Of the EMGALITY-treated patients, approximately 85% were female, 77% were white, and the mean age was 41 years at study entry.
The most common adverse reaction was injection site reactions. In Studies 1, 2, and 3, 1.8% of patients discontinued double-blind treatment because of adverse events. Table 1 summarizes the adverse reactions that occurred within up to 6 months of treatment in the migraine studies.
|
a Injection site reactions include multiple related adverse event terms, such as injection site pain, injection site reaction, injection site erythema, and injection site pruritus. |
||
|
Adverse Reaction |
EMGALITY 120 mg
Monthly (N=705) % |
Placebo
Monthly (N=1451) % |
| Injection site reactionsa | 18 | 13 |
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration |
Revised: 10/2025 |
|
PATIENT INFORMATION
EMGALITY® (em-GAL-it-ē) (galcanezumab-gnlm) injection, for subcutaneous use |
|
|
What is EMGALITY?
EMGALITY is a prescription medicine used in adults for the:
|
|
| It is not known if EMGALITY is safe and effective in children. | |
|
Who should not use EMGALITY?
Do not use EMGALITY if you are allergic to galcanezumab-gnlm or any of the ingredients in EMGALITY. See the end of this Patient Information for a complete list of ingredients in EMGALITY. |
|
Before you use EMGALITY, tell your healthcare provider if you:
|
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist when you get a new medicine. | |
How should I use EMGALITY?
|
|
|
What are the possible side effects of EMGALITY?
EMGALITY may cause serious side effects, including:
|
|
The most common side effects of EMGALITY include:
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of EMGALITY. For more information, ask your healthcare provider or pharmacist. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store EMGALITY?
|
|
| Keep EMGALITY and all medicines out of the reach of children. | |
|
General information about the safe and effective use of EMGALITY.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not use EMGALITY for a condition for which it was not prescribed. Do not give EMGALITY to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about EMGALITY that is written for health professionals. |
|
|
What are the ingredients in EMGALITY?
Active ingredient: galcanezumab-gnlm Inactive ingredients: L-histidine, L-histidine hydrochloride monohydrate, Polysorbate 80, Sodium Chloride, and Water for Injection, USP. EMGALITY prefilled pen and prefilled syringes are not made with natural rubber latex. EMGALITY® is a registered trademark of Eli Lilly and Company. Eli Lilly and Company Indianapolis, IN 46285, USA US License Number 1891 Copyright © 2018, 2025, Eli Lilly and Company. All rights reserved. EMG-0007-PPI-20251021 For more information, call 1-800-LillyRx (1-800-545-5979) or go to www.emgality.com. |
Section 51945-4
PACKAGE CARTON – EMGALITY Autoinjector 120 mg
NDC 0002-1436-11
EMGALITY®
(galcanezumab-gnlm) injection
120 mg/mL
1 x 120 mg/mL Single-Dose prefilled pen
For Subcutaneous Use Only
Single-Dose Only
Rx only
Lilly
Section 59845-8
| INSTRUCTIONS FOR USE |
| EMGALITY® [em-GAL-it-ē] |
| (galcanezumab-gnlm) |
| injection, for subcutaneous use |
| Prefilled Pen |
| This Instructions for Use contains information on how to inject EMGALITY |
|
|
| This Instructions for Use is for patients with migraine. |
| For subcutaneous injection only. |
| Important Information You Need to Know Before Injecting EMGALITY |
|
|
|
|
|
|
|
| INSTRUCTIONS FOR USE |
| Before you use the EMGALITY Pen, read and carefully follow all the step-by-step instructions. |
| Parts of the EMGALITY Pen |
|
|
| Step 1 Preparing to Inject EMGALITY | |
|
Step 1a Take the Pen from the refrigerator Check your prescription.
Put the original package with any unused Pens back in the refrigerator. Leave the base cap on until you are ready to inject. Leave the Pen at room temperature for 30 minutes before injecting. Do not microwave the Pen, run hot water over it, or leave it in direct sunlight. Do not shake. |
|
|
Step 1b Gather Supplies For each injection you will need:
|
|
|
Step 1c Inspect the Pen and the medicine Make sure you have the right medicine. The medicine inside should be clear. Its color may be colorless to slightly yellow to slightly brown. Do not use the Pen, and throw away (dispose of) as directed by your healthcare provider or pharmacist if:
|
|
|
Figure A
|
|
|
Step 1d Prepare for injection Wash your hands with soap and water before you inject your EMGALITY. Make sure a sharps disposal container is close by. |
|
|
Step 1e Choose your injection site Your healthcare provider can help you choose the injection site that is best for you. |
|
|
Figure B
|
|
|
|
|
|
|
|
|
|
|
| Step 2 Injecting EMGALITY | ||
| Step 2a Uncap the Pen |
Figure C
|
|
|
|
Make sure the Pen is locked. Leave the base cap on until you are ready to inject. |
|
|
||
|
||
|
||
| Step 2b Place and Unlock |
Figure D
|
|
|
||
|
|
Turn the lock ring to the unlock position. |
|
| Step 2c Press and Hold for 10 Seconds |
Figure E
|
|
|
||
|
||
|
Figure F
|
|
Step 3 Disposing of EMGALITY |
|
| Step 3a Throw away the used Pen | |
|
Figure G
|
|
|
| Commonly Asked Questions | |
| Q. | What if I see bubbles in the Pen? |
| A. | It is normal to have air bubbles in the Pen. EMGALITY is injected under your skin (subcutaneous injection), so these air bubbles will not harm you. |
| Q. | What if there is a drop of liquid on the tip of the needle when I remove the base cap? |
| A. | It is okay to see a drop of liquid on the tip of the needle. |
| Q. | What if I unlocked the Pen and pressed the teal injection button before I twisted off the base cap? |
| A. | Do not remove the base cap. Dispose of the Pen and get a new one. |
| Q. | Do I need to hold the injection button down until the injection is complete? |
| A. | This is not necessary, but it may help you keep the Pen steady and firm against your skin. |
| Q. | What if the needle did not retract after my injection? |
| A. | Do not touch the needle or replace the base cap. Store in a safe place to avoid an accidental needlestick and contact 1-800-545-5979 for instructions on how to return the Pen. |
| Q. | What if there is a drop of liquid or blood on my skin after my injection? |
| A. | This is normal. Press a cotton ball or gauze over the injection site. Do not rub the injection site. |
| Q. | What if I heard more than 2 clicks during my injection – 2 loud clicks and a soft one. Did I get my complete injection? |
| A. | Some patients may hear a soft click right before the second loud click. That is the normal operation of the Pen. Do not remove the Pen from your skin until you hear the second loud click. |
| Q. | How can I tell if my injection is complete? |
| A. | After you press the teal injection button, you will hear 2 loud clicks. The second click tells you that your injection is complete. You will also see the gray plunger at the top of the clear base. |
| If you have more questions about how to use the EMGALITY Pen: | |
|
|
|
Storing EMGALITY |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Read the full Prescribing Information and Patient Information for EMGALITY inside this box to learn more about your medicine. |
|
Eli Lilly and Company Indianapolis, IN 46285, USA |
| US License Number 1891 |
|
EMGALITY® is a registered trademark of Eli Lilly and Company. |
|
Copyright © 2018, 2025, Eli Lilly and Company. All rights reserved. |
|
The Pen meets the current dose accuracy and functional requirements of ISO 11608-1 and 11608-5. This Instructions for Use has been approved by the U.S. Food and Drug Administration. Revised: 11/2025 |
|
EMG-0006-AI-120MG-IFU-20251119 |
1.1 Migraine
EMGALITY is indicated for the preventive treatment of migraine in adults.
14.1 Migraine
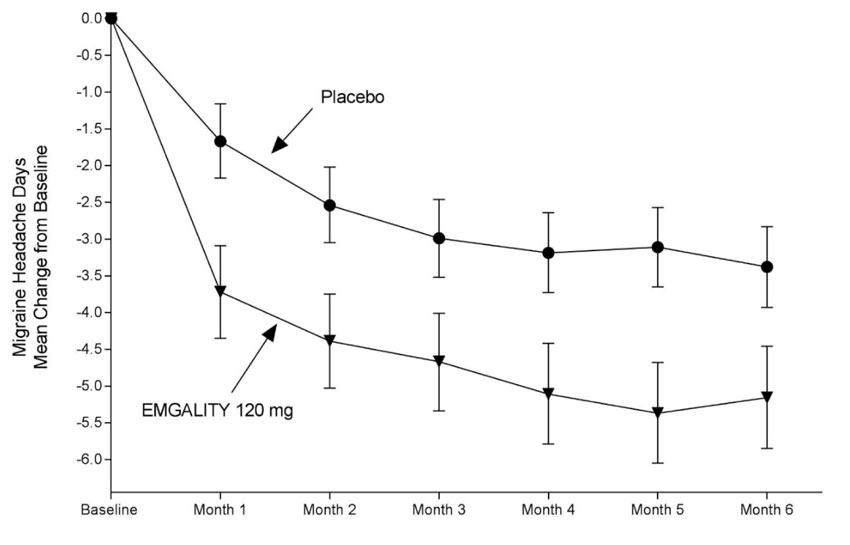
The efficacy of EMGALITY was evaluated as a preventive treatment of episodic or chronic migraine in three multicenter, randomized, double-blind, placebo-controlled studies: two 6-month studies in patients with episodic migraine (Studies 1 and 2) and one 3-month study in patients with chronic migraine (Study 3).
11 Description
Galcanezumab-gnlm is a humanized IgG4 monoclonal antibody specific for calcitonin-gene related peptide (CGRP) ligand. Galcanezumab-gnlm is produced in Chinese Hamster Ovary (CHO) cells by recombinant DNA technology. Galcanezumab-gnlm is composed of two identical immunoglobulin kappa light chains and two identical immunoglobulin gamma heavy chains and has an overall molecular weight of approximately 147 kDa.
EMGALITY (galcanezumab-gnlm) injection is a sterile, preservative-free, clear to opalescent and colorless to slightly yellow to slightly brown solution, for subcutaneous use. EMGALITY is supplied in a 1 mL single-dose prefilled pen to deliver 120 mg of galcanezumab-gnlm or a 1 mL single-dose prefilled syringe to deliver 100 mg or 120 mg of galcanezumab-gnlm. Each mL of solution contains 100 mg or 120 mg of galcanezumab-gnlm; L-histidine (0.5 mg); L-histidine hydrochloride monohydrate (1.5 mg); Polysorbate 80 (0.5 mg); Sodium Chloride (8.8 mg); Water for Injection, USP. The pH range is 5.3 - 6.3.
5.2 Hypertension
Development of hypertension and worsening of pre-existing hypertension have been reported following the use of CGRP antagonists, including EMGALITY, in the postmarketing setting. Some of the patients who developed new-onset hypertension had risk factors for hypertension. There were cases requiring initiation of pharmacological treatment for hypertension and, in some cases, hospitalization. Hypertension may occur at any time during treatment but was most frequently reported within 7 days of therapy initiation. EMGALITY was discontinued in many of the reported cases.
Monitor patients treated with EMGALITY for new-onset hypertension or worsening of pre-existing hypertension, and consider whether discontinuation of EMGALITY is warranted if evaluation fails to establish an alternative etiology or blood pressure is inadequately controlled.
16.1 How Supplied
EMGALITY (galcanezumab-gnlm) injection is a sterile, preservative-free, clear to opalescent, colorless to slightly yellow to slightly brown solution for subcutaneous administration.
EMGALITY is not made with natural rubber latex.
EMGALITY is supplied as follows:
| Pack Size | NDC | |
| Prefilled pen | ||
| 120 mg/mL single-dose | Carton of 1 | 0002-1436-11 |
| 120 mg/mL single-dose | Carton of 2 | 0002-1436-27 |
| Prefilled syringe | ||
| 100 mg/mL single-dose | Carton of 3 | 0002-3115-09 |
| 120 mg/mL single-dose | Carton of 1 | 0002-2377-11 |
| 120 mg/mL single-dose | Carton of 2 | 0002-2377-27 |
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of EMGALITY did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients.
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease.
For these reasons, comparison of the incidence of antibodies to galcanezumab-gnlm in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
The immunogenicity of EMGALITY has been evaluated using an in vitro immunoassay for the detection of binding anti-galcanezumab-gnlm antibodies. For patients whose sera tested positive in the screening immunoassay, an in vitro ligand-binding immunoassay was performed to detect neutralizing antibodies.
In controlled studies with EMGALITY up to 6 months (Study 1, Study 2, and Study 3), the incidence of anti-galcanezumab-gnlm antibody development was 4.8% (33/688) in patients receiving EMGALITY once monthly (32 out of 33 of whom had in vitro neutralizing activity). With 12 months of treatment in an open-label study, up to 12.5% (16/128) of EMGALITY-treated patients developed anti-galcanezumab-gnlm antibodies, most of whom tested positive for neutralizing antibodies.
Although anti-galcanezumab-gnlm antibody development was not found to affect the pharmacokinetics, safety or efficacy of EMGALITY in these patients, the available data are too limited to make definitive conclusions.
4 Contraindications
EMGALITY is contraindicated in patients with serious hypersensitivity to galcanezumab-gnlm or to any of the excipients [see Warnings and Precautions (5.1)].
6 Adverse Reactions
12.2 Pharmacodynamics
There are no relevant data on the pharmacodynamic effects of galcanezumab-gnlm.
12.3 Pharmacokinetics
Galcanezumab-gnlm exhibits linear pharmacokinetics and exposure increases proportionally with doses between 1 and 600 mg.
A loading dose of 240 mg achieved the serum galcanezumab-gnlm steady-state concentration after the first dose. A dose of 300 mg monthly would achieve steady-state concentration after the fourth dose. The time to maximum concentration is 5 days, and the elimination half-life is 27 days.
There was no difference in pharmacokinetic parameters between healthy volunteers, patients with episodic or chronic migraine, and patients with episodic cluster headache.
1 Indications and Usage
12.1 Mechanism of Action
Galcanezumab-gnlm is a humanized monoclonal antibody that binds to calcitonin gene-related peptide (CGRP) ligand and blocks its binding to the receptor.
5.3 Raynaud's Phenomenon
Development of Raynaud’s phenomenon and recurrence or worsening of pre-existing Raynaud’s phenomenon have been reported in the postmarketing setting following the use of CGRP antagonists, including EMGALITY. In reported cases with monoclonal antibody CGRP antagonists, symptom onset occurred after a median of 71 days following dosing. Many of the cases reported serious outcomes, including hospitalizations and disability, generally related to debilitating pain. In most reported cases, discontinuation of the CGRP antagonist resulted in resolution of symptoms.
EMGALITY should be discontinued if signs or symptoms of Raynaud’s phenomenon develop, and patients should be evaluated by a healthcare provider if symptoms do not resolve. Patients with a history of Raynaud’s phenomenon should be monitored for, and informed about the possibility of, worsening or recurrence of signs and symptoms.
16.2 Storage and Handling
- Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect EMGALITY from light until use.
- Do not freeze.
- Do not shake.
- EMGALITY may be stored out of refrigeration in the original carton at temperatures up to 30°C (86°F) for up to 7 days. Once stored out of refrigeration, do not place back in the refrigerator.
- If these conditions are exceeded, EMGALITY must be discarded.
- Discard the EMGALITY single-dose prefilled pen or syringe after use in a puncture-resistant container.
5 Warnings and Precautions
- Hypersensitivity Reactions: If a serious hypersensitivity reaction occurs, discontinue administration of EMGALITY and initiate appropriate therapy. Hypersensitivity reactions can occur days after administration, and may be prolonged. (5.1)
- Hypertension: New-onset or worsening of pre-existing hypertension may occur. (5.2)
- Raynaud's Phenomenon: New-onset or worsening of pre-existing Raynaud's phenomenon may occur. (5.3)
2 Dosage and Administration
- For subcutaneous use only. (2.1, 2.2, 2.3)
- Migraine recommended dosage: 240 mg loading dose (administered as two consecutive injections of 120 mg each), followed by monthly doses of 120 mg. (2.1)
- Episodic cluster headache recommended dosage: 300 mg (administered as three consecutive injections of 100 mg each) at the onset of the cluster period, and then monthly until the end of the cluster period. (2.2)
- Administer in the abdomen, thigh, back of the upper arm, or buttocks subcutaneously. (2.3)
3 Dosage Forms and Strengths
EMGALITY is a sterile clear to opalescent, colorless to slightly yellow to slightly brown solution available as follows:
- Injection: 120 mg/mL in a single-dose prefilled pen
- Injection: 120 mg/mL in a single-dose prefilled syringe
- Injection: 100 mg/mL in a single-dose prefilled syringe
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of EMGALITY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to EMGALITY exposure.
Immune System Disorders — Anaphylaxis, angioedema [see Contraindications (4) and Warnings and Precautions (5.1)].
Skin and Subcutaneous Tissue Disorders — Rash.
Vascular Disorders — Hypertension [see Warnings and Precautions (5.2)], Raynaud's Phenomenon [see Warnings and Precautions (5.3)]
1.2 Episodic Cluster Headache
EMGALITY is indicated for the treatment of episodic cluster headache in adults.
14.2 Episodic Cluster Headache
The efficacy of EMGALITY was evaluated for the treatment of episodic cluster headache in a randomized, 8-week, double-blind, placebo-controlled study (Study 4).
Study 4 (NCT02397473) included adults who met the International Classification of Headache Disorders 3rd edition (beta version) diagnostic criteria for episodic cluster headache and had a maximum of 8 attacks per day, a minimum of one attack every other day, and at least 4 attacks during the prospective 7-day baseline period. All patients were randomized in a 1:1 ratio to receive once-monthly subcutaneous injections of EMGALITY 300 mg or placebo. Patients were allowed to use certain specified acute/abortive cluster headache treatments, including triptans, oxygen, acetaminophen, and NSAIDs during the study.
The study excluded patients on other treatments intended to reduce the frequency of cluster headache attacks; patients with medication overuse headache; patients with ECG abnormalities compatible with an acute cardiovascular event or conduction delay; and patients with a history of myocardial infarction, unstable angina, percutaneous coronary intervention, coronary artery bypass grafting, deep vein thrombosis, or pulmonary embolism within 6 months of screening. In addition, patients with any history of stroke, intracranial or carotid aneurysm, intracranial hemorrhage, or vasospastic angina; clinical evidence of peripheral vascular disease; or diagnosis of Raynaud’s disease were excluded.
The primary efficacy endpoint for Study 4 was the mean change from baseline in weekly cluster headache attack frequency across Weeks 1 to 3. A secondary endpoint was the percentage of patients who achieved a response (defined as a reduction from baseline of 50% or greater in the weekly cluster headache attack frequency) at Week 3.
In Study 4, a total of 106 patients (88 males, 18 females) ranging in age from 19 to 65 years were randomized and treated. A total of 90 patients completed the 8-week double-blind phase. In the prospective baseline phase, the mean number of weekly cluster headache attacks was 17.5, and was similar across treatment groups.
EMGALITY 300 mg demonstrated statistically significant improvements for efficacy endpoints compared to placebo, as summarized in Table 4.
|
EMGALITY
300 mg N = 49 |
Placebo
N = 57 |
|
| Mean Reduction in Weekly Cluster Headache Attack Frequency (over Weeks 1 to 3) | ||
| Prospective Baseline Cluster Headache Attack Frequency | 17.8 | 17.3 |
| Mean change from baseline | -8.7 | -5.2 |
| Difference from placebo | -3.5 | |
| p-value | 0.036 | |
| ≥50% Weekly Cluster Headache Attack Frequency Responders (at Week 3) | ||
| % Responders | 71.4% | 52.6% |
| Difference from placebo | 18.8% | |
| p-value | 0.046 |
Figure 7: Mean Change in Weekly Cluster Headache Attack Frequency over Weeks 1 to 3 in Study 4a
a Abbreviations: BL = baseline; LS = least square; SE = standard error.
Figure 8 shows the distribution of the average percent change from baseline in weekly cluster headache attack frequency across Weeks 1 to 3 in bins of 25%, by treatment group, in Study 4.
Figure 8: Distribution of the Average Percent Change from Baseline in Weekly Cluster Headache Attack Frequency over Weeks 1 to 3 in Study 4 a
a N = number of intent to treat patients with non-missing average percentage change from baseline in weekly cluster headache attack frequency over weeks 1 to 3.
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including dyspnea, urticaria, and rash, have occurred with EMGALITY in clinical studies and the postmarketing setting. Cases of anaphylaxis and angioedema have also been reported in the postmarketing setting. If a serious or severe hypersensitivity reaction occurs, discontinue administration of EMGALITY and initiate appropriate therapy [see Contraindications (4), Adverse Reactions (6.1), and Patient Counseling Information (17)]. Hypersensitivity reactions can occur days after administration and may be prolonged.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Instructions on Self-Administration: Provide guidance to patients and/or caregivers on proper subcutaneous injection technique, including aseptic technique, and how to use the prefilled pen or prefilled syringe correctly [see Instructions for Use]. Instruct patients and/or caregivers to read and follow the Instructions for Use each time they use EMGALITY.
Hypersensitivity Reactions:
Inform patients about the signs and symptoms of hypersensitivity reactions and that these reactions can occur with EMGALITY. Advise patients to seek immediate medical attention if they experience any symptoms of serious or severe hypersensitivity reactions [see Warnings and Precautions (5.1)].
Hypertension:
Inform patients that hypertension can develop or pre-existing hypertension can worsen with EMGALITY, and that they should contact their healthcare provider if they experience elevation in their blood pressure [see Warnings and Precautions (5.2)].
Raynaud's Phenomenon:
Inform patients that Raynaud's phenomenon can develop or worsen with EMGALITY. Advise patients to discontinue EMGALITY and contact their healthcare provider if they experience signs or symptoms of Raynaud's phenomenon [see Warnings and Precautions (5.3)].
Pregnancy Exposure Registry: Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to EMGALITY during pregnancy [see Use in Specific Populations (8.1)].
For more information go to www.emgality.com or call 1-800-LillyRx (1-800-545-5979).
Literature revised: 10/2025
Eli Lilly and Company, Indianapolis, IN 46285, USA
US License Number 1891
Copyright © 2018, 2025, Eli Lilly and Company. All rights reserved.
Pat.: www.lilly.com/patents
EMG-0009-USPI-20251021
2.1 Recommended Dosing for Migraine
The recommended dosage of EMGALITY is 240 mg (two consecutive subcutaneous injections of 120 mg each) once as a loading dose, followed by monthly doses of 120 mg injected subcutaneously.
If a dose of EMGALITY is missed, administer as soon as possible. Thereafter, EMGALITY can be scheduled monthly from the date of the last dose.
2.3 Important Administration Instructions
EMGALITY is for subcutaneous use only.
EMGALITY is intended for patient self-administration. Prior to use, provide proper training to patients and/or caregivers on how to prepare and administer EMGALITY using the single-dose prefilled pen or single-dose prefilled syringe, including aseptic technique [see How Supplied/Storage and Handling (16.2) and Instructions for Use]:
- Protect EMGALITY from direct sunlight.
- Prior to subcutaneous administration, allow EMGALITY to sit at room temperature for 30 minutes. Do not warm by using a heat source such as hot water or a microwave.
- Do not shake the product.
- Inspect EMGALITY visually for particulate matter and discoloration prior to administration, whenever solution and container permit [see Dosage Forms and Strengths (3) and How Supplied/Storage and Handling (16.1)]. Do not use EMGALITY if it is cloudy or there are visible particles.
- Administer EMGALITY in the abdomen, thigh, back of the upper arm, or buttocks subcutaneously. Do not inject into areas where the skin is tender, bruised, red, or hard.
- Both the prefilled pen and prefilled syringe are single-dose and deliver the entire contents.
2.2 Recommended Dosing for Episodic Cluster Headache
The recommended dosage of EMGALITY is 300 mg (three consecutive subcutaneous injections of 100 mg each) at the onset of the cluster period, and then monthly until the end of the cluster period.
If a dose of EMGALITY is missed during a cluster period, administer as soon as possible. Thereafter, EMGALITY can be scheduled monthly from the date of the last dose until the end of the cluster period.
Structured Label Content
Section 42229-5 (42229-5)
Migraine
The safety of EMGALITY has been evaluated in 2586 patients with migraine who received at least one dose of EMGALITY, representing 1487 patient-years of exposure. Of these, 1920 patients were exposed to EMGALITY once monthly for at least 6 months, and 526 patients were exposed for 12 months.
In placebo-controlled clinical studies (Studies 1, 2, and 3), 705 patients received at least one dose of EMGALITY 120 mg once monthly, and 1451 patients received placebo, during 3 months or 6 months of double-blind treatment [see Clinical Studies (14.1)]. Of the EMGALITY-treated patients, approximately 85% were female, 77% were white, and the mean age was 41 years at study entry.
The most common adverse reaction was injection site reactions. In Studies 1, 2, and 3, 1.8% of patients discontinued double-blind treatment because of adverse events. Table 1 summarizes the adverse reactions that occurred within up to 6 months of treatment in the migraine studies.
|
a Injection site reactions include multiple related adverse event terms, such as injection site pain, injection site reaction, injection site erythema, and injection site pruritus. |
||
|
Adverse Reaction |
EMGALITY 120 mg
Monthly (N=705) % |
Placebo
Monthly (N=1451) % |
| Injection site reactionsa | 18 | 13 |
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration |
Revised: 10/2025 |
|
PATIENT INFORMATION
EMGALITY® (em-GAL-it-ē) (galcanezumab-gnlm) injection, for subcutaneous use |
|
|
What is EMGALITY?
EMGALITY is a prescription medicine used in adults for the:
|
|
| It is not known if EMGALITY is safe and effective in children. | |
|
Who should not use EMGALITY?
Do not use EMGALITY if you are allergic to galcanezumab-gnlm or any of the ingredients in EMGALITY. See the end of this Patient Information for a complete list of ingredients in EMGALITY. |
|
Before you use EMGALITY, tell your healthcare provider if you:
|
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist when you get a new medicine. | |
How should I use EMGALITY?
|
|
|
What are the possible side effects of EMGALITY?
EMGALITY may cause serious side effects, including:
|
|
The most common side effects of EMGALITY include:
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of EMGALITY. For more information, ask your healthcare provider or pharmacist. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store EMGALITY?
|
|
| Keep EMGALITY and all medicines out of the reach of children. | |
|
General information about the safe and effective use of EMGALITY.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not use EMGALITY for a condition for which it was not prescribed. Do not give EMGALITY to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about EMGALITY that is written for health professionals. |
|
|
What are the ingredients in EMGALITY?
Active ingredient: galcanezumab-gnlm Inactive ingredients: L-histidine, L-histidine hydrochloride monohydrate, Polysorbate 80, Sodium Chloride, and Water for Injection, USP. EMGALITY prefilled pen and prefilled syringes are not made with natural rubber latex. EMGALITY® is a registered trademark of Eli Lilly and Company. Eli Lilly and Company Indianapolis, IN 46285, USA US License Number 1891 Copyright © 2018, 2025, Eli Lilly and Company. All rights reserved. EMG-0007-PPI-20251021 For more information, call 1-800-LillyRx (1-800-545-5979) or go to www.emgality.com. |
Section 51945-4 (51945-4)
PACKAGE CARTON – EMGALITY Autoinjector 120 mg
NDC 0002-1436-11
EMGALITY®
(galcanezumab-gnlm) injection
120 mg/mL
1 x 120 mg/mL Single-Dose prefilled pen
For Subcutaneous Use Only
Single-Dose Only
Rx only
Lilly
Section 59845-8 (59845-8)
| INSTRUCTIONS FOR USE |
| EMGALITY® [em-GAL-it-ē] |
| (galcanezumab-gnlm) |
| injection, for subcutaneous use |
| Prefilled Pen |
| This Instructions for Use contains information on how to inject EMGALITY |
|
|
| This Instructions for Use is for patients with migraine. |
| For subcutaneous injection only. |
| Important Information You Need to Know Before Injecting EMGALITY |
|
|
|
|
|
|
|
| INSTRUCTIONS FOR USE |
| Before you use the EMGALITY Pen, read and carefully follow all the step-by-step instructions. |
| Parts of the EMGALITY Pen |
|
|
| Step 1 Preparing to Inject EMGALITY | |
|
Step 1a Take the Pen from the refrigerator Check your prescription.
Put the original package with any unused Pens back in the refrigerator. Leave the base cap on until you are ready to inject. Leave the Pen at room temperature for 30 minutes before injecting. Do not microwave the Pen, run hot water over it, or leave it in direct sunlight. Do not shake. |
|
|
Step 1b Gather Supplies For each injection you will need:
|
|
|
Step 1c Inspect the Pen and the medicine Make sure you have the right medicine. The medicine inside should be clear. Its color may be colorless to slightly yellow to slightly brown. Do not use the Pen, and throw away (dispose of) as directed by your healthcare provider or pharmacist if:
|
|
|
Figure A
|
|
|
Step 1d Prepare for injection Wash your hands with soap and water before you inject your EMGALITY. Make sure a sharps disposal container is close by. |
|
|
Step 1e Choose your injection site Your healthcare provider can help you choose the injection site that is best for you. |
|
|
Figure B
|
|
|
|
|
|
|
|
|
|
|
| Step 2 Injecting EMGALITY | ||
| Step 2a Uncap the Pen |
Figure C
|
|
|
|
Make sure the Pen is locked. Leave the base cap on until you are ready to inject. |
|
|
||
|
||
|
||
| Step 2b Place and Unlock |
Figure D
|
|
|
||
|
|
Turn the lock ring to the unlock position. |
|
| Step 2c Press and Hold for 10 Seconds |
Figure E
|
|
|
||
|
||
|
Figure F
|
|
Step 3 Disposing of EMGALITY |
|
| Step 3a Throw away the used Pen | |
|
Figure G
|
|
|
| Commonly Asked Questions | |
| Q. | What if I see bubbles in the Pen? |
| A. | It is normal to have air bubbles in the Pen. EMGALITY is injected under your skin (subcutaneous injection), so these air bubbles will not harm you. |
| Q. | What if there is a drop of liquid on the tip of the needle when I remove the base cap? |
| A. | It is okay to see a drop of liquid on the tip of the needle. |
| Q. | What if I unlocked the Pen and pressed the teal injection button before I twisted off the base cap? |
| A. | Do not remove the base cap. Dispose of the Pen and get a new one. |
| Q. | Do I need to hold the injection button down until the injection is complete? |
| A. | This is not necessary, but it may help you keep the Pen steady and firm against your skin. |
| Q. | What if the needle did not retract after my injection? |
| A. | Do not touch the needle or replace the base cap. Store in a safe place to avoid an accidental needlestick and contact 1-800-545-5979 for instructions on how to return the Pen. |
| Q. | What if there is a drop of liquid or blood on my skin after my injection? |
| A. | This is normal. Press a cotton ball or gauze over the injection site. Do not rub the injection site. |
| Q. | What if I heard more than 2 clicks during my injection – 2 loud clicks and a soft one. Did I get my complete injection? |
| A. | Some patients may hear a soft click right before the second loud click. That is the normal operation of the Pen. Do not remove the Pen from your skin until you hear the second loud click. |
| Q. | How can I tell if my injection is complete? |
| A. | After you press the teal injection button, you will hear 2 loud clicks. The second click tells you that your injection is complete. You will also see the gray plunger at the top of the clear base. |
| If you have more questions about how to use the EMGALITY Pen: | |
|
|
|
Storing EMGALITY |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Read the full Prescribing Information and Patient Information for EMGALITY inside this box to learn more about your medicine. |
|
Eli Lilly and Company Indianapolis, IN 46285, USA |
| US License Number 1891 |
|
EMGALITY® is a registered trademark of Eli Lilly and Company. |
|
Copyright © 2018, 2025, Eli Lilly and Company. All rights reserved. |
|
The Pen meets the current dose accuracy and functional requirements of ISO 11608-1 and 11608-5. This Instructions for Use has been approved by the U.S. Food and Drug Administration. Revised: 11/2025 |
|
EMG-0006-AI-120MG-IFU-20251119 |
1.1 Migraine
EMGALITY is indicated for the preventive treatment of migraine in adults.
14.1 Migraine
The efficacy of EMGALITY was evaluated as a preventive treatment of episodic or chronic migraine in three multicenter, randomized, double-blind, placebo-controlled studies: two 6-month studies in patients with episodic migraine (Studies 1 and 2) and one 3-month study in patients with chronic migraine (Study 3).
11 Description (11 DESCRIPTION)
Galcanezumab-gnlm is a humanized IgG4 monoclonal antibody specific for calcitonin-gene related peptide (CGRP) ligand. Galcanezumab-gnlm is produced in Chinese Hamster Ovary (CHO) cells by recombinant DNA technology. Galcanezumab-gnlm is composed of two identical immunoglobulin kappa light chains and two identical immunoglobulin gamma heavy chains and has an overall molecular weight of approximately 147 kDa.
EMGALITY (galcanezumab-gnlm) injection is a sterile, preservative-free, clear to opalescent and colorless to slightly yellow to slightly brown solution, for subcutaneous use. EMGALITY is supplied in a 1 mL single-dose prefilled pen to deliver 120 mg of galcanezumab-gnlm or a 1 mL single-dose prefilled syringe to deliver 100 mg or 120 mg of galcanezumab-gnlm. Each mL of solution contains 100 mg or 120 mg of galcanezumab-gnlm; L-histidine (0.5 mg); L-histidine hydrochloride monohydrate (1.5 mg); Polysorbate 80 (0.5 mg); Sodium Chloride (8.8 mg); Water for Injection, USP. The pH range is 5.3 - 6.3.
5.2 Hypertension
Development of hypertension and worsening of pre-existing hypertension have been reported following the use of CGRP antagonists, including EMGALITY, in the postmarketing setting. Some of the patients who developed new-onset hypertension had risk factors for hypertension. There were cases requiring initiation of pharmacological treatment for hypertension and, in some cases, hospitalization. Hypertension may occur at any time during treatment but was most frequently reported within 7 days of therapy initiation. EMGALITY was discontinued in many of the reported cases.
Monitor patients treated with EMGALITY for new-onset hypertension or worsening of pre-existing hypertension, and consider whether discontinuation of EMGALITY is warranted if evaluation fails to establish an alternative etiology or blood pressure is inadequately controlled.
16.1 How Supplied
EMGALITY (galcanezumab-gnlm) injection is a sterile, preservative-free, clear to opalescent, colorless to slightly yellow to slightly brown solution for subcutaneous administration.
EMGALITY is not made with natural rubber latex.
EMGALITY is supplied as follows:
| Pack Size | NDC | |
| Prefilled pen | ||
| 120 mg/mL single-dose | Carton of 1 | 0002-1436-11 |
| 120 mg/mL single-dose | Carton of 2 | 0002-1436-27 |
| Prefilled syringe | ||
| 100 mg/mL single-dose | Carton of 3 | 0002-3115-09 |
| 120 mg/mL single-dose | Carton of 1 | 0002-2377-11 |
| 120 mg/mL single-dose | Carton of 2 | 0002-2377-27 |
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of EMGALITY did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients.
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease.
For these reasons, comparison of the incidence of antibodies to galcanezumab-gnlm in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
The immunogenicity of EMGALITY has been evaluated using an in vitro immunoassay for the detection of binding anti-galcanezumab-gnlm antibodies. For patients whose sera tested positive in the screening immunoassay, an in vitro ligand-binding immunoassay was performed to detect neutralizing antibodies.
In controlled studies with EMGALITY up to 6 months (Study 1, Study 2, and Study 3), the incidence of anti-galcanezumab-gnlm antibody development was 4.8% (33/688) in patients receiving EMGALITY once monthly (32 out of 33 of whom had in vitro neutralizing activity). With 12 months of treatment in an open-label study, up to 12.5% (16/128) of EMGALITY-treated patients developed anti-galcanezumab-gnlm antibodies, most of whom tested positive for neutralizing antibodies.
Although anti-galcanezumab-gnlm antibody development was not found to affect the pharmacokinetics, safety or efficacy of EMGALITY in these patients, the available data are too limited to make definitive conclusions.
4 Contraindications (4 CONTRAINDICATIONS)
EMGALITY is contraindicated in patients with serious hypersensitivity to galcanezumab-gnlm or to any of the excipients [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
12.2 Pharmacodynamics
There are no relevant data on the pharmacodynamic effects of galcanezumab-gnlm.
12.3 Pharmacokinetics
Galcanezumab-gnlm exhibits linear pharmacokinetics and exposure increases proportionally with doses between 1 and 600 mg.
A loading dose of 240 mg achieved the serum galcanezumab-gnlm steady-state concentration after the first dose. A dose of 300 mg monthly would achieve steady-state concentration after the fourth dose. The time to maximum concentration is 5 days, and the elimination half-life is 27 days.
There was no difference in pharmacokinetic parameters between healthy volunteers, patients with episodic or chronic migraine, and patients with episodic cluster headache.
1 Indications and Usage (1 INDICATIONS AND USAGE)
12.1 Mechanism of Action
Galcanezumab-gnlm is a humanized monoclonal antibody that binds to calcitonin gene-related peptide (CGRP) ligand and blocks its binding to the receptor.
5.3 Raynaud's Phenomenon
Development of Raynaud’s phenomenon and recurrence or worsening of pre-existing Raynaud’s phenomenon have been reported in the postmarketing setting following the use of CGRP antagonists, including EMGALITY. In reported cases with monoclonal antibody CGRP antagonists, symptom onset occurred after a median of 71 days following dosing. Many of the cases reported serious outcomes, including hospitalizations and disability, generally related to debilitating pain. In most reported cases, discontinuation of the CGRP antagonist resulted in resolution of symptoms.
EMGALITY should be discontinued if signs or symptoms of Raynaud’s phenomenon develop, and patients should be evaluated by a healthcare provider if symptoms do not resolve. Patients with a history of Raynaud’s phenomenon should be monitored for, and informed about the possibility of, worsening or recurrence of signs and symptoms.
16.2 Storage and Handling
- Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect EMGALITY from light until use.
- Do not freeze.
- Do not shake.
- EMGALITY may be stored out of refrigeration in the original carton at temperatures up to 30°C (86°F) for up to 7 days. Once stored out of refrigeration, do not place back in the refrigerator.
- If these conditions are exceeded, EMGALITY must be discarded.
- Discard the EMGALITY single-dose prefilled pen or syringe after use in a puncture-resistant container.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: If a serious hypersensitivity reaction occurs, discontinue administration of EMGALITY and initiate appropriate therapy. Hypersensitivity reactions can occur days after administration, and may be prolonged. (5.1)
- Hypertension: New-onset or worsening of pre-existing hypertension may occur. (5.2)
- Raynaud's Phenomenon: New-onset or worsening of pre-existing Raynaud's phenomenon may occur. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For subcutaneous use only. (2.1, 2.2, 2.3)
- Migraine recommended dosage: 240 mg loading dose (administered as two consecutive injections of 120 mg each), followed by monthly doses of 120 mg. (2.1)
- Episodic cluster headache recommended dosage: 300 mg (administered as three consecutive injections of 100 mg each) at the onset of the cluster period, and then monthly until the end of the cluster period. (2.2)
- Administer in the abdomen, thigh, back of the upper arm, or buttocks subcutaneously. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
EMGALITY is a sterile clear to opalescent, colorless to slightly yellow to slightly brown solution available as follows:
- Injection: 120 mg/mL in a single-dose prefilled pen
- Injection: 120 mg/mL in a single-dose prefilled syringe
- Injection: 100 mg/mL in a single-dose prefilled syringe
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of EMGALITY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to EMGALITY exposure.
Immune System Disorders — Anaphylaxis, angioedema [see Contraindications (4) and Warnings and Precautions (5.1)].
Skin and Subcutaneous Tissue Disorders — Rash.
Vascular Disorders — Hypertension [see Warnings and Precautions (5.2)], Raynaud's Phenomenon [see Warnings and Precautions (5.3)]
1.2 Episodic Cluster Headache
EMGALITY is indicated for the treatment of episodic cluster headache in adults.
14.2 Episodic Cluster Headache
The efficacy of EMGALITY was evaluated for the treatment of episodic cluster headache in a randomized, 8-week, double-blind, placebo-controlled study (Study 4).
Study 4 (NCT02397473) included adults who met the International Classification of Headache Disorders 3rd edition (beta version) diagnostic criteria for episodic cluster headache and had a maximum of 8 attacks per day, a minimum of one attack every other day, and at least 4 attacks during the prospective 7-day baseline period. All patients were randomized in a 1:1 ratio to receive once-monthly subcutaneous injections of EMGALITY 300 mg or placebo. Patients were allowed to use certain specified acute/abortive cluster headache treatments, including triptans, oxygen, acetaminophen, and NSAIDs during the study.
The study excluded patients on other treatments intended to reduce the frequency of cluster headache attacks; patients with medication overuse headache; patients with ECG abnormalities compatible with an acute cardiovascular event or conduction delay; and patients with a history of myocardial infarction, unstable angina, percutaneous coronary intervention, coronary artery bypass grafting, deep vein thrombosis, or pulmonary embolism within 6 months of screening. In addition, patients with any history of stroke, intracranial or carotid aneurysm, intracranial hemorrhage, or vasospastic angina; clinical evidence of peripheral vascular disease; or diagnosis of Raynaud’s disease were excluded.
The primary efficacy endpoint for Study 4 was the mean change from baseline in weekly cluster headache attack frequency across Weeks 1 to 3. A secondary endpoint was the percentage of patients who achieved a response (defined as a reduction from baseline of 50% or greater in the weekly cluster headache attack frequency) at Week 3.
In Study 4, a total of 106 patients (88 males, 18 females) ranging in age from 19 to 65 years were randomized and treated. A total of 90 patients completed the 8-week double-blind phase. In the prospective baseline phase, the mean number of weekly cluster headache attacks was 17.5, and was similar across treatment groups.
EMGALITY 300 mg demonstrated statistically significant improvements for efficacy endpoints compared to placebo, as summarized in Table 4.
|
EMGALITY
300 mg N = 49 |
Placebo
N = 57 |
|
| Mean Reduction in Weekly Cluster Headache Attack Frequency (over Weeks 1 to 3) | ||
| Prospective Baseline Cluster Headache Attack Frequency | 17.8 | 17.3 |
| Mean change from baseline | -8.7 | -5.2 |
| Difference from placebo | -3.5 | |
| p-value | 0.036 | |
| ≥50% Weekly Cluster Headache Attack Frequency Responders (at Week 3) | ||
| % Responders | 71.4% | 52.6% |
| Difference from placebo | 18.8% | |
| p-value | 0.046 |
Figure 7: Mean Change in Weekly Cluster Headache Attack Frequency over Weeks 1 to 3 in Study 4a
a Abbreviations: BL = baseline; LS = least square; SE = standard error.
Figure 8 shows the distribution of the average percent change from baseline in weekly cluster headache attack frequency across Weeks 1 to 3 in bins of 25%, by treatment group, in Study 4.
Figure 8: Distribution of the Average Percent Change from Baseline in Weekly Cluster Headache Attack Frequency over Weeks 1 to 3 in Study 4 a
a N = number of intent to treat patients with non-missing average percentage change from baseline in weekly cluster headache attack frequency over weeks 1 to 3.
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including dyspnea, urticaria, and rash, have occurred with EMGALITY in clinical studies and the postmarketing setting. Cases of anaphylaxis and angioedema have also been reported in the postmarketing setting. If a serious or severe hypersensitivity reaction occurs, discontinue administration of EMGALITY and initiate appropriate therapy [see Contraindications (4), Adverse Reactions (6.1), and Patient Counseling Information (17)]. Hypersensitivity reactions can occur days after administration and may be prolonged.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Instructions on Self-Administration: Provide guidance to patients and/or caregivers on proper subcutaneous injection technique, including aseptic technique, and how to use the prefilled pen or prefilled syringe correctly [see Instructions for Use]. Instruct patients and/or caregivers to read and follow the Instructions for Use each time they use EMGALITY.
Hypersensitivity Reactions:
Inform patients about the signs and symptoms of hypersensitivity reactions and that these reactions can occur with EMGALITY. Advise patients to seek immediate medical attention if they experience any symptoms of serious or severe hypersensitivity reactions [see Warnings and Precautions (5.1)].
Hypertension:
Inform patients that hypertension can develop or pre-existing hypertension can worsen with EMGALITY, and that they should contact their healthcare provider if they experience elevation in their blood pressure [see Warnings and Precautions (5.2)].
Raynaud's Phenomenon:
Inform patients that Raynaud's phenomenon can develop or worsen with EMGALITY. Advise patients to discontinue EMGALITY and contact their healthcare provider if they experience signs or symptoms of Raynaud's phenomenon [see Warnings and Precautions (5.3)].
Pregnancy Exposure Registry: Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to EMGALITY during pregnancy [see Use in Specific Populations (8.1)].
For more information go to www.emgality.com or call 1-800-LillyRx (1-800-545-5979).
Literature revised: 10/2025
Eli Lilly and Company, Indianapolis, IN 46285, USA
US License Number 1891
Copyright © 2018, 2025, Eli Lilly and Company. All rights reserved.
Pat.: www.lilly.com/patents
EMG-0009-USPI-20251021
2.1 Recommended Dosing for Migraine
The recommended dosage of EMGALITY is 240 mg (two consecutive subcutaneous injections of 120 mg each) once as a loading dose, followed by monthly doses of 120 mg injected subcutaneously.
If a dose of EMGALITY is missed, administer as soon as possible. Thereafter, EMGALITY can be scheduled monthly from the date of the last dose.
2.3 Important Administration Instructions
EMGALITY is for subcutaneous use only.
EMGALITY is intended for patient self-administration. Prior to use, provide proper training to patients and/or caregivers on how to prepare and administer EMGALITY using the single-dose prefilled pen or single-dose prefilled syringe, including aseptic technique [see How Supplied/Storage and Handling (16.2) and Instructions for Use]:
- Protect EMGALITY from direct sunlight.
- Prior to subcutaneous administration, allow EMGALITY to sit at room temperature for 30 minutes. Do not warm by using a heat source such as hot water or a microwave.
- Do not shake the product.
- Inspect EMGALITY visually for particulate matter and discoloration prior to administration, whenever solution and container permit [see Dosage Forms and Strengths (3) and How Supplied/Storage and Handling (16.1)]. Do not use EMGALITY if it is cloudy or there are visible particles.
- Administer EMGALITY in the abdomen, thigh, back of the upper arm, or buttocks subcutaneously. Do not inject into areas where the skin is tender, bruised, red, or hard.
- Both the prefilled pen and prefilled syringe are single-dose and deliver the entire contents.
2.2 Recommended Dosing for Episodic Cluster Headache
The recommended dosage of EMGALITY is 300 mg (three consecutive subcutaneous injections of 100 mg each) at the onset of the cluster period, and then monthly until the end of the cluster period.
If a dose of EMGALITY is missed during a cluster period, administer as soon as possible. Thereafter, EMGALITY can be scheduled monthly from the date of the last dose until the end of the cluster period.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:32.268971 · Updated: 2026-03-14T22:37:42.596050