992
3389e947-b7d1-6caf-e063-6294a90a2238
34390-5
HUMAN OTC DRUG LABEL
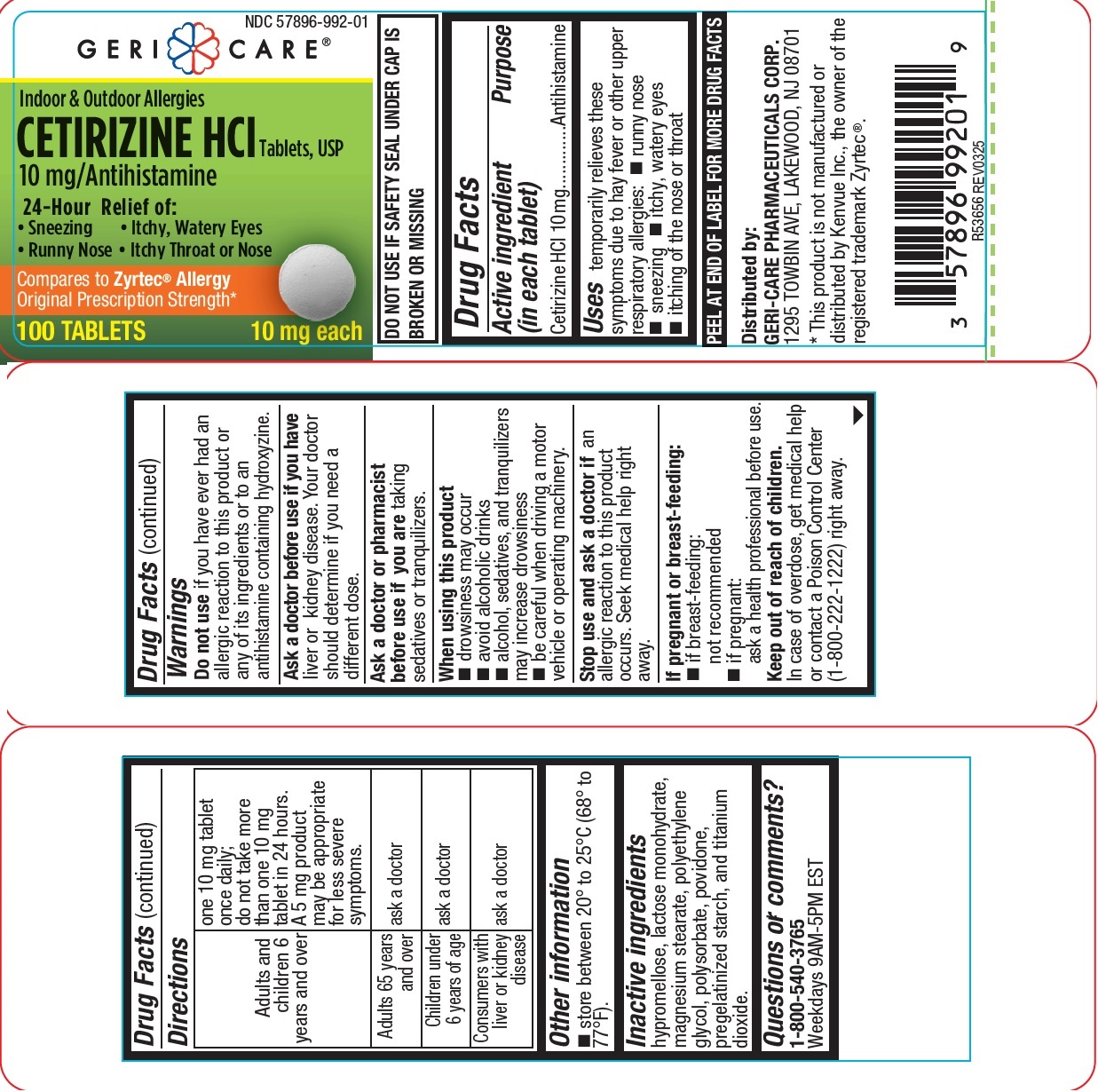
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cetirizine HCl 10 mg
Medication Information
Dosage and Administration
| Adults and
children 6 years and over |
one 10 mg tablet
once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| Adults 65 years
and over |
ask a doctor |
| Children under
6 years of age |
ask a doctor |
| Consumers with
liver or kidney disease |
ask a doctor |
Description
Cetirizine HCl 10 mg
Uses
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing,
- itchy, watery eyes
- itching of the nose or throat
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 51727-6
hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, polysorbate, povidone,
pregelatinized starch, and titanium dioxide.
Section 51945-4
Section 55105-1
Antihistamine
Section 55106-9
Cetirizine HCl 10 mg
Warnings
Do not use if you have ever had an allergic reaction to this product or any of its ingredients or an antihistamine containing hydroxyzine
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
- drowsiness may occur
- Avoid alcoholic drinks
- Alcohol, sedatives and tranquilizers may increase drowsiness
- Be careful when driving a motor vehicle or operating machinery.
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding:
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Other Information
store between 20° to 25°C (68° to 77°F).
Questions Or Comments?
1-800-540-3765
Weekdays 9AM-5PM EST
Structured Label Content
Uses
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing,
- itchy, watery eyes
- itching of the nose or throat
Dosage and Administration (34068-7)
| Adults and
children 6 years and over |
one 10 mg tablet
once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| Adults 65 years
and over |
ask a doctor |
| Children under
6 years of age |
ask a doctor |
| Consumers with
liver or kidney disease |
ask a doctor |
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 51727-6 (51727-6)
hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, polysorbate, povidone,
pregelatinized starch, and titanium dioxide.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Antihistamine
Section 55106-9 (55106-9)
Cetirizine HCl 10 mg
Warnings
Do not use if you have ever had an allergic reaction to this product or any of its ingredients or an antihistamine containing hydroxyzine
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
- drowsiness may occur
- Avoid alcoholic drinks
- Alcohol, sedatives and tranquilizers may increase drowsiness
- Be careful when driving a motor vehicle or operating machinery.
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding:
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Other Information (Other information)
store between 20° to 25°C (68° to 77°F).
Questions Or Comments? (Questions or comments?)
1-800-540-3765
Weekdays 9AM-5PM EST
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:58.798391 · Updated: 2026-03-14T23:08:27.251685