Beef Tallow Sunscreen
3380f1ac-0320-35f2-e063-6394a90a97eb
34390-5
HUMAN OTC DRUG LABEL
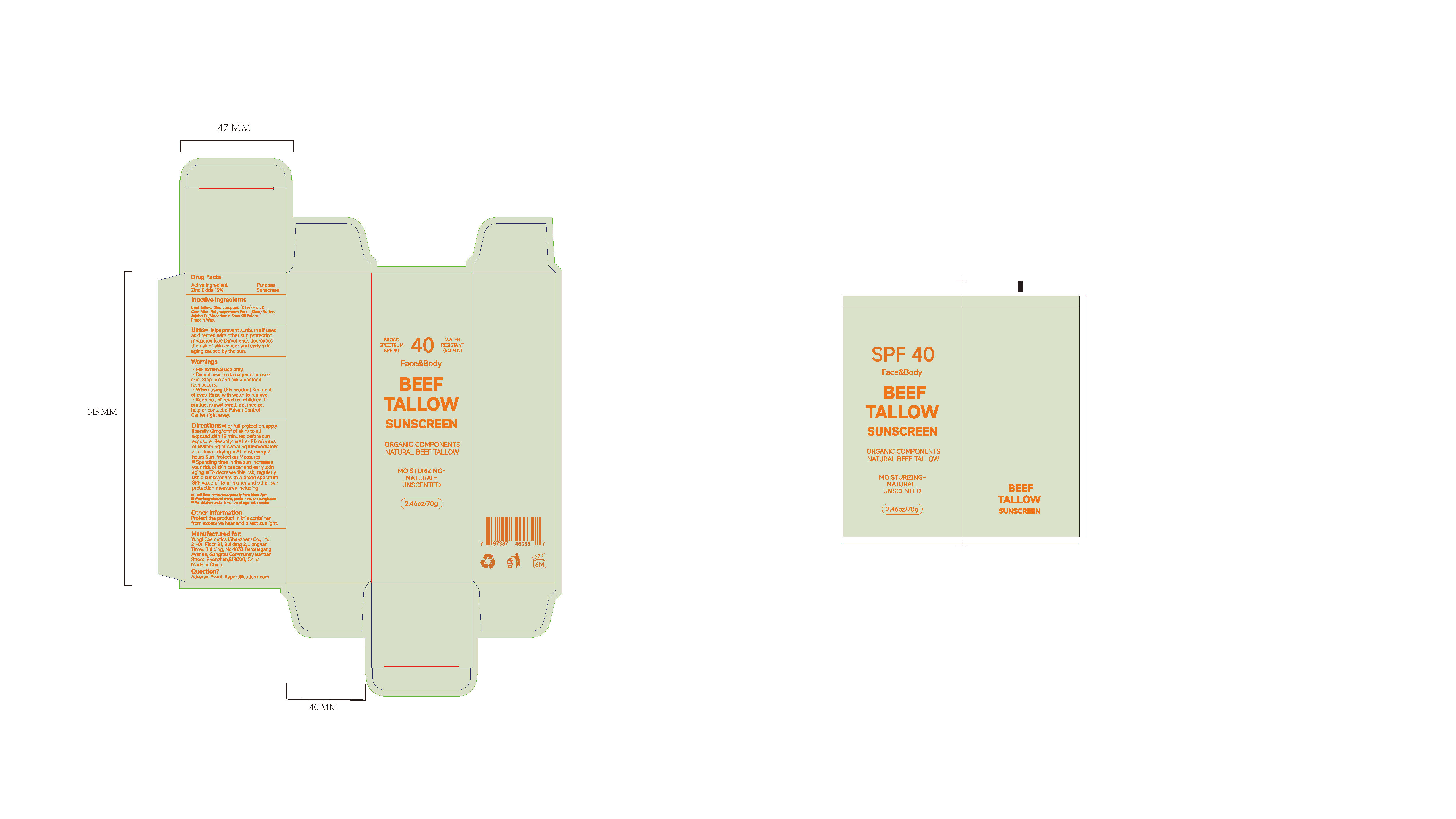
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Zinc Oxide
Medication Information
Warnings and Precautions
For exteral use onby
Indications and Usage
Uses.Halps prevent sunburnm lf usedas directed with oter sun protectionmeasures (see Directions)decreasesthe risk of skin cancer and early skinaging caused by the sun.
Dosage and Administration
Directions .For full protection,applyliberally(2mg/cm of skin) to allexposed skin 15 minutes before sunexposure.Reapply:After 80 minutesof swlmming or sweating.lmmedlatelyafter towel drying .At least every 2hours Sun Protection Measures:Spending time in the sun increasesyour risk of skin cancer and early skinaging .To decrease this risk, regularlyuse a sunscreen with a broad spectumSPF value of 15 or higher and cther sunprotection measures including
Lmit tma In the sun,especlally tom 10em 2pm
Wesr lon aleeved chirtss, pants, hats, and runglaeses
For chilcren under b manths of aoet aek a cloctor
Description
Zinc Oxide
Section 50565-1
keep out or reach of chlldren. lfproduct is swallowed, get medicalhelp ar contact a Poison ContolCenter night away
Section 50566-9
Stop use and ask a doctor if rash occurs.
Section 50567-7
When using thls preduct keep outof eves. Rinse with water to remove.
Section 50570-1
Do not use on damaged or brokenskin. Stop use and ask a doctor if rash occurs
Section 51727-6
Beef Tallow
Olea Europaea (Olive)Fruit Oil
Cera Alba
Butyrospermum Parkii (Shea) Butter
Jojoba Oil/Macadamia Seed Oil Esters
Propolis Wax
Section 51945-4
Section 55105-1
Sunscreen
Section 55106-9
Zinc Oxide
Structured Label Content
Indications and Usage (34067-9)
Uses.Halps prevent sunburnm lf usedas directed with oter sun protectionmeasures (see Directions)decreasesthe risk of skin cancer and early skinaging caused by the sun.
Dosage and Administration (34068-7)
Directions .For full protection,applyliberally(2mg/cm of skin) to allexposed skin 15 minutes before sunexposure.Reapply:After 80 minutesof swlmming or sweating.lmmedlatelyafter towel drying .At least every 2hours Sun Protection Measures:Spending time in the sun increasesyour risk of skin cancer and early skinaging .To decrease this risk, regularlyuse a sunscreen with a broad spectumSPF value of 15 or higher and cther sunprotection measures including
Lmit tma In the sun,especlally tom 10em 2pm
Wesr lon aleeved chirtss, pants, hats, and runglaeses
For chilcren under b manths of aoet aek a cloctor
Warnings and Precautions (34071-1)
For exteral use onby
Section 50565-1 (50565-1)
keep out or reach of chlldren. lfproduct is swallowed, get medicalhelp ar contact a Poison ContolCenter night away
Section 50566-9 (50566-9)
Stop use and ask a doctor if rash occurs.
Section 50567-7 (50567-7)
When using thls preduct keep outof eves. Rinse with water to remove.
Section 50570-1 (50570-1)
Do not use on damaged or brokenskin. Stop use and ask a doctor if rash occurs
Section 51727-6 (51727-6)
Beef Tallow
Olea Europaea (Olive)Fruit Oil
Cera Alba
Butyrospermum Parkii (Shea) Butter
Jojoba Oil/Macadamia Seed Oil Esters
Propolis Wax
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Sunscreen
Section 55106-9 (55106-9)
Zinc Oxide
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:58.788248 · Updated: 2026-03-14T23:08:25.015175