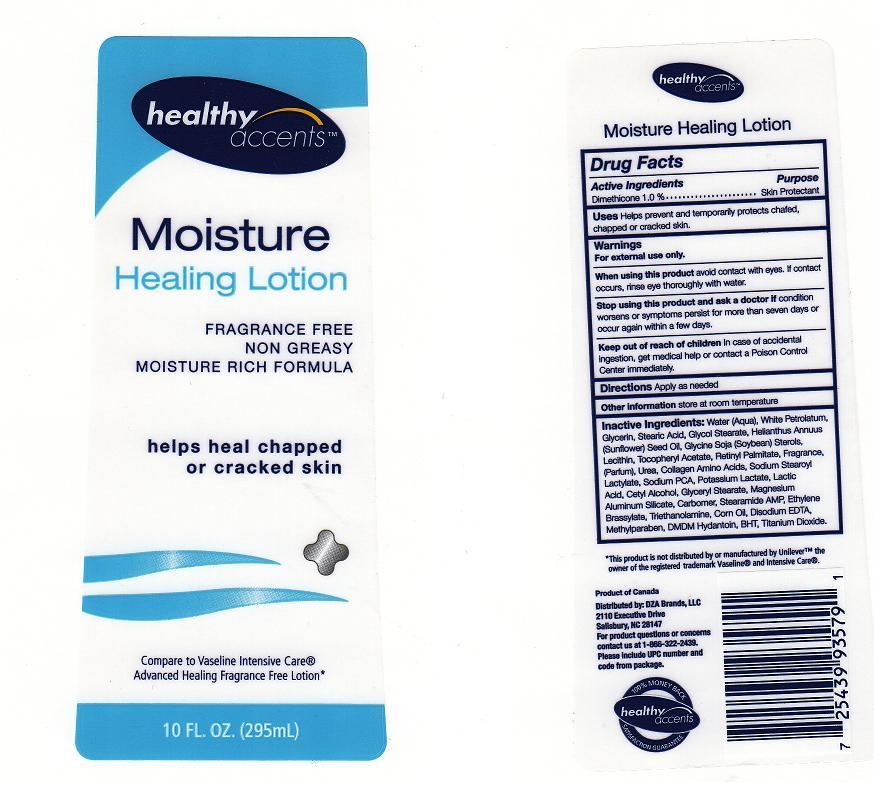
Back Label - Drug Facts Box
3334f923-ed7d-4209-aad4-849d8d559569
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
DIMETHICONE 1.0%
Purpose
Skin Protectant
Medication Information
Purpose
Description
DIMETHICONE 1.0%
Uses
Warning
FOR EXTERNAL USE ONLY.
Directions
Active Ingredient
DIMETHICONE 1.0%
Inactive Ingredients
Water, White Petrolatum, Glycerin, Stearic Acid, Glycol Stearate, Helianthus Annuus (Sunflower) Seed Oil, Glycine Soja (Soybean) Sterols, Lecithin, Tocopheryl Acetate, Retinyl Palmitate, Fragrance, Urea, Collagen Amino Acids, Sodium Stearoyl Lactylate, Sodium PCA, Potassium Lactate, Lactic Acid, Cetyl Alcohol, Glyceryl Stearate, Magnesium Aluminum Silicate, Carbomer, Stearamide AMP, Ethylene Brassylate, Triethanolamine, Corn Oil, Disodium EDTA, Methylparaben, DMDM Hydantoin, BHT, Titanium Dioxide
When Using This Product
AVOID CONTACT WITH EYES, IF CONTACT OCCURS, RINSE EYE THROUGHLY WITH WATER.
Keep Out of Reach of Children
IN CASE OF ACCIDENTAL INGESTION, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER IMMEDIATELY.
Package Front and Back Labels
10 OZ Front and Back Labels: hac10.jpg
Stop Using This Product and Ask A Doctor If
CONDITION WORSENS OR SYMPTOMS PERSIST FOR MORE THAN SEVEN DAYS OR OCCUR AGAIN WITHIN A FEW DAYS.
Structured Label Content
Uses
Purpose
Warning (WARNING)
FOR EXTERNAL USE ONLY.
Directions
Active Ingredient (ACTIVE INGREDIENT)
DIMETHICONE 1.0%
Inactive Ingredients
Water, White Petrolatum, Glycerin, Stearic Acid, Glycol Stearate, Helianthus Annuus (Sunflower) Seed Oil, Glycine Soja (Soybean) Sterols, Lecithin, Tocopheryl Acetate, Retinyl Palmitate, Fragrance, Urea, Collagen Amino Acids, Sodium Stearoyl Lactylate, Sodium PCA, Potassium Lactate, Lactic Acid, Cetyl Alcohol, Glyceryl Stearate, Magnesium Aluminum Silicate, Carbomer, Stearamide AMP, Ethylene Brassylate, Triethanolamine, Corn Oil, Disodium EDTA, Methylparaben, DMDM Hydantoin, BHT, Titanium Dioxide
When Using This Product (When using this product)
AVOID CONTACT WITH EYES, IF CONTACT OCCURS, RINSE EYE THROUGHLY WITH WATER.
Keep Out of Reach of Children (KEEP OUT OF REACH OF CHILDREN)
IN CASE OF ACCIDENTAL INGESTION, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER IMMEDIATELY.
Package Front and Back Labels
10 OZ Front and Back Labels: hac10.jpg
Stop Using This Product and Ask A Doctor If (STOP USING THIS PRODUCT AND ASK A DOCTOR IF)
CONDITION WORSENS OR SYMPTOMS PERSIST FOR MORE THAN SEVEN DAYS OR OCCUR AGAIN WITHIN A FEW DAYS.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:08.648859 · Updated: 2026-03-14T22:52:29.225907