These Highlights Do Not Include All The Information Needed To Use Lybalvi®
32ffddd1-4e2b-45d9-9b36-bb730167ec80
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions ( 5.1 )] .
Indications and Usage
LYBALVI is indicated for the treatment of: Schizophrenia in adults Bipolar I disorder in adults Acute treatment of manic or mixed episodes as monotherapy and as adjunct to lithium or valproate Maintenance monotherapy treatment
Dosage and Administration
Indication Recommended Starting Dose (olanzapine/samidorphan) Recommended Dose (olanzapine/samidorphan) Schizophrenia ( 2.2 ) 5 mg/10 mg or 10 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg Bipolar I disorder (manic or mixed episodes) ( 2.3 ) 10 mg/10 mg or 15 mg/10 mg 5 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg Bipolar I disorder adjunct to lithium or valproate ( 2.3 ) 10 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg See the full prescribing information for the recommended titration and maximum recommended dosage. ( 2.2 , 2.3 ) Administer LYBALVI once daily with or without food. Do not divide tablets or combine strengths. ( 2.4 ) Recommended starting dosage is 5 mg/10 mg once daily in patients who have a predisposition to hypotensive reactions, have potential for slower metabolism of olanzapine, or may be more pharmacodynamically sensitive to olanzapine. ( 2.5 )
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis : Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). ( 5.2 ) Precipitation of Opioid Withdrawal in Patients Who are Dependent on Opioids : LYBALVI can precipitate opioid withdrawal in patients who are dependent on opioids. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from the last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids to avoid precipitation of opioid withdrawal. ( 2.1 , 5.3 ) Vulnerability to Life-Threatening Opioid Overdose : Risk of Opioid Overdose from Attempts to Overcome LYBALVI Opioid Blockade: Attempts to overcome LYBALVI opioid blockade with high or repeated doses of opioids may lead to fatal opioid intoxication, particularly if LYBALVI therapy is interrupted or discontinued. ( 5.4 ) Risk of Resuming Opioids in Patients with Prior Opioid Use : Patients with a history of chronic opioid use prior to LYBALVI treatment may have decreased opioid tolerance if LYBALVI therapy is interrupted or discontinued. ( 5.4 ) Neuroleptic Malignant Syndrome : Manage with immediate discontinuation and close monitoring. ( 5.5 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) : Discontinue if DRESS is suspected. ( 5.6 ) Metabolic Changes : Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.7 ) Tardive Dyskinesia : Discontinue if clinically appropriate. ( 5.8 ) Orthostatic Hypotension and Syncope : Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. ( 5.9 ) Leukopenia, Neutropenia, and Agranulocytosis : Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors. ( 5.11 ) Seizures : Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.13 ) Potential for Cognitive and Motor Impairment : Use caution when operating machinery. ( 5.14 ) Anticholinergic (Antimuscarinic) Effects: Use with caution with other anticholinergic drugs and in patients with urinary retention, prostatic hypertrophy, constipation, paralytic ileus or related conditions. ( 5.16 ) Hyperprolactinemia : May elevate prolactin levels. ( 5.17 )
Contraindications
LYBALVI is contraindicated in patients: who are using opioids [ see Warnings and Precautions ( 5.3 , 5.4 ), Drug Interactions ( 7.3 )] . who are undergoing acute opioid withdrawal [see Warnings and Precautions ( 5.3 , 5.4 ), Drug Interactions ( 7.3 )] . If LYBALVI is administered with lithium or valproate, refer to the lithium or valproate Prescribing Information for the contraindications for these products [see Warnings and Precautions ( 5.18 )] .
Adverse Reactions
Cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack), including fatalities, were reported in patients in trials of olanzapine in elderly patients with dementia-related psychosis. In placebo-controlled trials, there was a significantly higher incidence of cerebrovascular adverse reactions in patients treated with olanzapine compared to patients treated with placebo. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions ( 5.1 )] .
Drug Interactions
Strong CYP3A4 Inducers : Not recommended. ( 7.1 ) Strong CYP1A2 Inhibitors : Consider dosage reduction of olanzapine component of LYBALVI. ( 7.1 ) CYP1A2 Inducer : Consider dosage increase of the olanzapine component of LYBALVI. ( 7.1 ) CNS Acting Drugs : May potentiate orthostatic hypotension. ( 7.1 ) Anticholinergic Drugs: Can increase risk for severe gastrointestinal adverse reactions. ( 7.1 ) Antihypertensive Agents : Monitor blood pressure. ( 7.2 ) Levodopa and Dopamine Agonists : Not recommended. ( 7.2 )
Medication Information
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis : Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). ( 5.2 ) Precipitation of Opioid Withdrawal in Patients Who are Dependent on Opioids : LYBALVI can precipitate opioid withdrawal in patients who are dependent on opioids. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from the last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids to avoid precipitation of opioid withdrawal. ( 2.1 , 5.3 ) Vulnerability to Life-Threatening Opioid Overdose : Risk of Opioid Overdose from Attempts to Overcome LYBALVI Opioid Blockade: Attempts to overcome LYBALVI opioid blockade with high or repeated doses of opioids may lead to fatal opioid intoxication, particularly if LYBALVI therapy is interrupted or discontinued. ( 5.4 ) Risk of Resuming Opioids in Patients with Prior Opioid Use : Patients with a history of chronic opioid use prior to LYBALVI treatment may have decreased opioid tolerance if LYBALVI therapy is interrupted or discontinued. ( 5.4 ) Neuroleptic Malignant Syndrome : Manage with immediate discontinuation and close monitoring. ( 5.5 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) : Discontinue if DRESS is suspected. ( 5.6 ) Metabolic Changes : Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.7 ) Tardive Dyskinesia : Discontinue if clinically appropriate. ( 5.8 ) Orthostatic Hypotension and Syncope : Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. ( 5.9 ) Leukopenia, Neutropenia, and Agranulocytosis : Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors. ( 5.11 ) Seizures : Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.13 ) Potential for Cognitive and Motor Impairment : Use caution when operating machinery. ( 5.14 ) Anticholinergic (Antimuscarinic) Effects: Use with caution with other anticholinergic drugs and in patients with urinary retention, prostatic hypertrophy, constipation, paralytic ileus or related conditions. ( 5.16 ) Hyperprolactinemia : May elevate prolactin levels. ( 5.17 )
Indications and Usage
LYBALVI is indicated for the treatment of: Schizophrenia in adults Bipolar I disorder in adults Acute treatment of manic or mixed episodes as monotherapy and as adjunct to lithium or valproate Maintenance monotherapy treatment
Dosage and Administration
Indication Recommended Starting Dose (olanzapine/samidorphan) Recommended Dose (olanzapine/samidorphan) Schizophrenia ( 2.2 ) 5 mg/10 mg or 10 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg Bipolar I disorder (manic or mixed episodes) ( 2.3 ) 10 mg/10 mg or 15 mg/10 mg 5 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg Bipolar I disorder adjunct to lithium or valproate ( 2.3 ) 10 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg See the full prescribing information for the recommended titration and maximum recommended dosage. ( 2.2 , 2.3 ) Administer LYBALVI once daily with or without food. Do not divide tablets or combine strengths. ( 2.4 ) Recommended starting dosage is 5 mg/10 mg once daily in patients who have a predisposition to hypotensive reactions, have potential for slower metabolism of olanzapine, or may be more pharmacodynamically sensitive to olanzapine. ( 2.5 )
Contraindications
LYBALVI is contraindicated in patients: who are using opioids [ see Warnings and Precautions ( 5.3 , 5.4 ), Drug Interactions ( 7.3 )] . who are undergoing acute opioid withdrawal [see Warnings and Precautions ( 5.3 , 5.4 ), Drug Interactions ( 7.3 )] . If LYBALVI is administered with lithium or valproate, refer to the lithium or valproate Prescribing Information for the contraindications for these products [see Warnings and Precautions ( 5.18 )] .
Adverse Reactions
Cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack), including fatalities, were reported in patients in trials of olanzapine in elderly patients with dementia-related psychosis. In placebo-controlled trials, there was a significantly higher incidence of cerebrovascular adverse reactions in patients treated with olanzapine compared to patients treated with placebo. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions ( 5.1 )] .
Drug Interactions
Strong CYP3A4 Inducers : Not recommended. ( 7.1 ) Strong CYP1A2 Inhibitors : Consider dosage reduction of olanzapine component of LYBALVI. ( 7.1 ) CYP1A2 Inducer : Consider dosage increase of the olanzapine component of LYBALVI. ( 7.1 ) CNS Acting Drugs : May potentiate orthostatic hypotension. ( 7.1 ) Anticholinergic Drugs: Can increase risk for severe gastrointestinal adverse reactions. ( 7.1 ) Antihypertensive Agents : Monitor blood pressure. ( 7.2 ) Levodopa and Dopamine Agonists : Not recommended. ( 7.2 )
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions ( 5.1 )] .
Section 42229-5
Monotherapy: Initiate LYBALVI at 10 mg/10 mg or 15 mg/10 mg once daily. The recommended dosage is 10 mg/10 mg, 15 mg/10 mg, or 20 mg/10 mg once daily. The maximum recommended dosage is 20 mg/10 mg once daily.
Dosage adjustments should occur at intervals of not less than 24 hours. When dosage adjustments are necessary, dose increments/decrements of 5 mg (based on the olanzapine component of LYBALVI) are recommended.
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised: 1/2024 |
|
|
MEDICATION GUIDE
LYBALVI® (lee bawl' vee) (olanzapine and samidorphan) tablets, for oral use |
||
| If your healthcare provider prescribes LYBALVI in combination with valproate or lithium, also read the Medication Guide that comes with those medicines. | ||
|
What is the most important information I should know about LYBALVI?
|
||
|
What is LYBALVI?
LYBALVI is a prescription medicine which contains 2 medicines (olanzapine and samidorphan) used in adults:
|
||
| It is not known if LYBALVI is safe or effective in children. | ||
| Do not take LYBALVI if you are taking opioids or are experiencing acute opioid withdrawal. See “What are the possible side effects of LYBALVI.” | ||
Before taking LYBALVI, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. LYBALVI and other medicines may affect each other causing possible serious side effects. LYBALVI may affect the way other medicines work, and other medicines may affect how LYBALVI works. Especially tell your healthcare provider if you:
|
||
| Your healthcare provider can tell you if it is safe to take LYBALVI with your other medicines. Do not start or stop any medicines while taking LYBALVI without first talking to your healthcare provider. Tell your healthcare provider if you take a urine drug screening test because LYBALVI may affect your test results. Tell those giving the drug screening test that you are taking LYBALVI. Know the medicines you take. Keep a list of your medicines to show your healthcare provider and pharmacist when you get a new medicine. |
||
How should I take LYBALVI?
|
||
|
What should I avoid while taking LYBALVI?
|
||
|
What are the possible side effects of LYBALVI?
|
||
|
|
|
|
||
|
|
|
|
||
|
||
| The most common side effects of LYBALVI when used to treat people with schizophrenia include: | ||
|
|
|
|
The most common side effects of LYBALVI when used alone to treat people with mixed or manic episodes that happen with bipolar I disorder include:
|
||
|
|
|
|
The most common side effects of LYBALVI when used in combination with lithium or valproate to treat people with mixed or manic episodes that happen with bipolar I disorder include:
|
||
|
|
|
| These are not all the possible side effects of LYBALVI. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store LYBALVI?
|
||
| Keep LYBALVI and all medicines out of the reach of children. | ||
|
General information about the safe and effective use of LYBALVI
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use LYBALVI for a condition for which it is not prescribed. Do not give LYBALVI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LYBALVI that is written for healthcare providers. |
||
|
What are the ingredients in LYBALVI?
Active ingredients: olanzapine and samidorphan Inactive ingredients: colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate and microcrystalline cellulose. The film coating ingredients include hypromellose, titanium dioxide, triacetin and iron oxide yellow (5 mg/10 mg); iron oxide yellow and iron oxide red (10 mg/10 mg); FD&C Blue No. 2/indigo carmine aluminum lake (15 mg/10 mg); iron oxide red (20 mg/10 mg) Manufactured and marketed by: Alkermes, Inc., Waltham, MA 02451 For more information, go to www.LYBALVI.com or call 1-844-LYBALVI (1-844-592-2584). |
Section 44425-7
Storage and Handling
Store at room temperature 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Keep tightly closed and protect from moisture.
Section 51945-4
Principal Display Panel - 5 mg/10 mg 30 Tablets Carton Label
NDC 65757-651-42 Rx Only
LYBALVI ®
(olanzapine/samidorphan)
tablets
5 mg/10 mg
Dispense the accompanying
Medication Guide to each patient.
30 Tablets
5.10 Falls
Antipsychotics, including LYBALVI, may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
7.3 Opioids
LYBALVI is contraindicated in patients who are using opioids or undergoing acute opioid withdrawal [see Contraindications (4)].
LYBALVI increases the risk of precipitating acute opioid withdrawal in patients who are dependent on opioids. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from the last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids [see Dosage and Administration (2.1), Warnings and Precautions (5.3)].
In emergency situations, if a LYBALVI-treated patient requires opioid treatment for anesthesia or analgesia, discontinue LYBALVI. The opioid should be administered by properly trained individual(s), and the patient should be properly monitored in a setting equipped and staffed for cardiopulmonary resuscitation [see Warnings and Precautions (5.4)].
In non-emergency situations, if a LYBALVI-treated patient is expected to require opioid treatment (e.g., for analgesia during or after an elective surgical procedure) discontinue LYBALVI at least 5 days before opioid treatment and start olanzapine or another antipsychotic, if needed.
Given that LYBALVI contains samidorphan, an opioid antagonist, opioid treatment may be less effective or ineffective shortly after LYBALVI discontinuation because of the presence of samidorphan.
5.13 Seizures
Like other antipsychotic drugs, LYBALVI may cause seizures. This risk is greatest in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in older patients.
11 Description
LYBALVI is a combination of olanzapine, an atypical antipsychotic, and samidorphan (as samidorphan L-malate), an opioid antagonist.
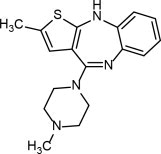
Olanzapine is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b][1,5]benzodiazepine. The molecular formula of olanzapine is: C17H20N4S and the molecular weight is 312.44 g/mol. It is a yellow crystalline powder and has pKa values of 7.80 and 5.44. The chemical structure is:
Samidorphan L-malate is morphinan-3-carboxamide, 17-(cyclopropylmethyl)-4, 14-dihydroxy-6-oxo-, (2S)-2-hydroxybutanedioate. The molecular formula of samidorphan L-malate is C21H26N2O4 • C4H6O5 and the molecular weight is 504.54 g/mol. It is a white to off-white crystalline powder and has pKa values of 8.3 (amine) and 10.1 (phenol). The chemical structure is:
LYBALVI is intended for oral administration and is available as film-coated, bilayer tablets in the following strengths: 5 mg/10 mg, 10 mg/10 mg, 15 mg/10 mg, and 20 mg/10 mg of olanzapine and samidorphan (equivalent to 13.6 mg of samidorphan L-malate).
Inactive ingredients include colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. The film coating ingredients include hypromellose, titanium dioxide, triacetin, and color additives [iron oxide yellow (5 mg/10 mg); iron oxide yellow and iron oxide red (10 mg/10 mg); FD&C Blue No. 2/ indigo carmine aluminum lake (15 mg/10 mg); iron oxide red (20 mg/10 mg)].
5.12 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Antipsychotic drugs, including LYBALVI, should be used cautiously in patients at risk for aspiration.
8.4 Pediatric Use
The safety and effectiveness of LYBALVI have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of LYBALVI did not include sufficient numbers of patients 65 years of age and older to determine whether they responded differently than younger adult patients.
14.1 Schizophrenia
The efficacy of LYBALVI in the treatment of schizophrenia in adults is based, in part, upon adequate and well-controlled studies of orally administered olanzapine. Efficacy of LYBALVI was also evaluated in a 4-week, randomized, double-blind, placebo- and active-controlled study (Study 1).
In Study 1 (NCT02634346), adult patients who met DSM-5 criteria for schizophrenia were randomized in a 1:1:1 ratio to LYBALVI, olanzapine, or placebo for 4 weeks of daily dosing. Dosing was flexible based on clinical response and tolerability for the first 2 weeks of the study; doses thereafter were fixed. Patients assigned to LYBALVI could receive either 10 mg/10 mg or 20 mg/10 mg, and patients assigned to olanzapine could receive either 10 mg or 20 mg. The study was designed to compare LYBALVI with placebo, not with olanzapine.
Eligible patients were 18 to 70 years of age, with a body mass index (BMI) of 18.0–40.0 kg/m2, Positive and Negative Syndrome Scale (PANSS) total score of ≥80, and a score of ≥4 on at least 3 of the selected Positive Scale items. Patients were also required to have a Clinical Global Impression-Severity (CGI-S) score ≥4.
The primary efficacy endpoint was change from baseline in PANSS total score at Week 4. The PANSS is a 30-item scale that measures positive symptoms of schizophrenia (7 items), negative symptoms of schizophrenia (7 items), and general psychopathology (16 items), each rated on a scale of 1 (absent) to 7 (extreme). Total PANSS scores range from 30 to 210, with a higher score reflecting greater symptom severity. The secondary efficacy endpoint was defined as the change from baseline in CGI-S score at Week 4. The CGI-S is a validated scale that requires clinicians to rate a patient's current illness severity and overall clinical state based on experience with the illness population. Scores range from 1 (normal, not at all ill) to 7 (extremely ill).
Compared with patients on placebo, a statistically significant improvement in the change from baseline in PANSS total score at Week 4 was observed in patients treated with LYBALVI (Table 7). The inclusion of samidorphan in LYBALVI did not appear to negatively impact the antipsychotic efficacy of olanzapine.
| Treatment Group | Total PANSS Score | ||
|---|---|---|---|
| Baseline Mean Score (SD) | LS Mean Change from Baseline (SE) | Placebo-subtracted Difference a (95% CI) | |
|
Abbreviations: CI: confidence interval; LS: least squares; SD: standard deviation; SE: standard error. |
|||
|
a Difference (drug minus placebo) in least squares mean change from baseline. A negative value for the placebo subtracted difference represents improvement. |
|||
| LYBALVI (10 mg/10 mg, 20 mg/10 mg) (N=132) |
101.8 (11.6) |
-23.9 (1.3) |
-6.4 (-10.0, -2.8) |
| Placebo (N=133) |
102.7 (11.9) |
-17.5 (1.3) |
-- |
| Olanzapine (10 mg, 20 mg) (N=132) |
100.6 (12.1) |
-22.8 (1.3) |
-5.3 (-8.9, -1.7) |
The change from baseline in PANSS total score in Study 1 is displayed in Figure 4.
Figure 4: Change from Baseline in PANSS Total Score by Time (Week) in Patients with Schizophrenia (Study 1)
Error bars represent standard error. The numbers under the figure indicate the number of patients at each timepoint.
Compared with patients on placebo, a statistically significant improvement in CGI-S score at Week 4 was observed in patients treated with LYBALVI.
4 Contraindications
LYBALVI is contraindicated in patients:
- who are using opioids [see Warnings and Precautions ( 5.3 , 5.4), Drug Interactions ( 7.3)].
- who are undergoing acute opioid withdrawal [see Warnings and Precautions (5.3, 5.4), Drug Interactions (7.3)].
If LYBALVI is administered with lithium or valproate, refer to the lithium or valproate Prescribing Information for the contraindications for these products [see Warnings and Precautions (5.18)].
6 Adverse Reactions
The following adverse reactions are discussed in detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-related Psychosis [see Boxed Warning, Warnings and Precautions (5.1)]
- Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis [see Warnings and Precautions (5.2)]
- Precipitation of Opioid Withdrawal in Patients Who Are Dependent on Opioids [see Warnings and Precautions (5.3)]
- Vulnerability to Life-Threatening Opioid Overdose [see Warnings and Precautions (5.4)]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.5)]
- Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions (5.6)]
- Metabolic Changes [see Warnings and Precautions (5.7)]
- Tardive Dyskinesia [see Warnings and Precautions (5.8)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.9)]
- Falls [see Warnings and Precautions (5.10)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.11)]
- Dysphagia [see Warnings and Precautions (5.12)]
- Seizures [see Warnings and Precautions (5.13)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.14)]
- Body Temperature Regulation [see Warnings and Precautions (5.15)]
- Anticholinergic (Antimuscarinic) Effects [see Warnings and Precautions (5.16)]
- Hyperprolactinemia [see Warnings and Precautions (5.17)]
- Risks Associated with Combination Treatment with Lithium or Valproate [see Warnings and Precautions (5.18)]
7 Drug Interactions
- Strong CYP3A4 Inducers: Not recommended. (7.1)
- Strong CYP1A2 Inhibitors: Consider dosage reduction of olanzapine component of LYBALVI. (7.1)
- CYP1A2 Inducer: Consider dosage increase of the olanzapine component of LYBALVI. (7.1)
- CNS Acting Drugs: May potentiate orthostatic hypotension. (7.1)
- Anticholinergic Drugs: Can increase risk for severe gastrointestinal adverse reactions. (7.1)
- Antihypertensive Agents: Monitor blood pressure. (7.2)
- Levodopa and Dopamine Agonists: Not recommended. (7.2)
8.7 Renal Impairment
Plasma exposure to olanzapine and samidorphan was higher in patients with severe renal impairment (eGFR 15 to 29 mL/minute/1.73 m2) compared to those with normal renal function [see Clinical Pharmacology (12.3)]. No dose adjustment of LYBALVI is needed in patients with mild (eGFR 60 to 89 mL/minute/1.73 m2), moderate (eGFR 30 to 59 mL/minute/1.73 m2), or severe renal impairment (eGFR 15 to 29 mL/minute/1.73 m2).
The effect of LYBALVI in patients with end-stage renal disease was not studied. LYBALVI is not recommended for patients with end-stage renal disease (eGFR of <15 mL/minute/1.73 m2).
12.3 Pharmacokinetics
The pharmacokinetics of both olanzapine and samidorphan are linear over the clinical dose range and there is no PK interaction between olanzapine and samidorphan after oral administration of LYBALVI. Steady-state concentrations of olanzapine and samidorphan are reached within 7 days of commencement of once-daily administration of LYBALVI. The primary pharmacological activities of LYBALVI are due to the parent drugs, olanzapine and samidorphan.
Following a single dose administration of LYBALVI (10 mg olanzapine/10 mg samidorphan), the mean AUC0-inf and Cmax of olanzapine was 628 ng·h/mL and 16 ng/mL, respectively. The mean AUC0-inf and Cmax of olanzapine after 10 mg single dose administration of olanzapine tablet was 610 ng·h/mL and 16 ng/mL, respectively.
Pharmacokinetic properties of the components of LYBALVI are provided in Table 6.
| Parameters | Olanzapine | Samidorphan |
|---|---|---|
|
Abbreviations: AUC24h=area under the concentration-time curve over the 24-hour dosing interval; Cmax=maximum plasma concentration; CYP=cytochrome P450; NA=not applicable; Tmax=time to Cmax; t½=terminal elimination half-life; UGT=Uridine 5'-diphospho-glucuronosyltransferase. |
||
|
a Presented as arithmetic mean (standard deviation). |
||
|
b Presented as range of the median across multiple studies. |
||
|
c Geometric mean ratio (90% confidence interval) [high fat meal/fasting]. High fat meal defined as meal containing approximately 900-1000 calories and 50% fat content. No clinically relevant food effect. |
||
|
d Presented as range of the mean across multiple studies. |
||
| General | ||
| Steady State Exposure (20 mg olanzapine/10 mg samidorphan) | ||
| Cmax (ng/mL)a | 64.6 (28.9) | 45.1 (11.4) |
| AUC24h (ng∙hr/mL)a | 1086 (556) | 364 (112) |
| Time to Reach Steady State | 7 days | 5 days |
| Accumulation at Steady State | 2-fold | 1.3-fold |
| Absorption | ||
| Absolute Oral Bioavailability | NA | 69% |
| Tmax (h)b | 4.5–7 | 1–2 |
| Effect of Food | ||
| Cmax Ratioc | 0.88 (0.82, 0.95) | 0.85 (0.76, 0.94) |
| AUC Ratioc | 0.93 (0.91, 0.96) | 1.03 (1.00, 1.05) |
| Distribution | ||
| Plasma Protein Binding | 93% | 23% - 33% |
| Blood-to-Plasma Ratio | Not Determined | 0.8 |
| Elimination | ||
| t1/2 (h)d | 35–52 | 7–11 |
| CL/F (L/h)d | 15–22 | 35–45 |
| Metabolism | ||
| Primary Pathway(s) | UGT1A4, CYP1A2 | CYP3A4 |
| Minor Pathway(s) | CYP2D6 | CYP3A5, CYP2C19, CYP2C8 |
| Major Circulating Metabolites | 10-N-glucuronide and 4′-N-desmethyl-olanzapine. Both metabolites lack pharmacological activity at the therapeutic concentrations |
N-dealkylated and cis-N-oxide metabolites. Neither metabolite contributes to the pharmacological effects of samidorphan |
| Excretion | ||
| Primary Route of Elimination | Metabolism | Metabolism |
| Urine (Unchanged) | 7% | 18% |
| Urine (Unchanged + metabolites) | 57% | 67% |
| Feces (Unchanged + metabolites) | 30% | 16% |
5.7 Metabolic Changes
Atypical antipsychotic drugs, including LYBALVI, have been associated with metabolic changes that include hyperglycemia, diabetes mellitus, dyslipidemia, and body weight gain [see Adverse Reactions (6.1)]. While all drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Any patient treated with LYBALVI should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with LYBALVI should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required anti-diabetic treatment despite discontinuation of the suspect drug. Patients starting treatment with LYBALVI should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment.
Antipsychotics have caused adverse alterations in lipids. Patients starting treatment with LYBALVI should undergo fasting lipid profile testing at the beginning of treatment and periodically during treatment.
Weight gain has been observed with use of antipsychotics. Monitor weight prior to initiating LYBALVI and frequently thereafter.
5.8 Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. The risk appears to be highest among the elderly, especially elderly women, but it is not possible to predict which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible increases with the duration of treatment and the cumulative dose. The syndrome can develop after a relatively brief treatment period, even at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, LYBALVI should be prescribed in a manner that is most likely to reduce the risk of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients: 1) who suffer from a chronic illness that is known to respond to antipsychotic drugs; and 2) for whom alternative, effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, use the lowest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. Periodically reassess the need for continued treatment.
If signs and symptoms of tardive dyskinesia appear in a patient on LYBALVI, drug discontinuation should be considered. However, some patients may require treatment with LYBALVI despite the presence of the syndrome.
8.6 Hepatic Impairment
Olanzapine and samidorphan plasma exposures were found to be higher in subjects with moderate hepatic impairment than in subjects with normal hepatic function. The effect of severe hepatic impairment was not studied. The higher plasma exposure in patients with moderate hepatic impairment was not expected to be clinically relevant. No dose adjustment of LYBALVI is needed in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage
LYBALVI is indicated for the treatment of:
- Schizophrenia in adults
- Bipolar I disorder in adults
- Acute treatment of manic or mixed episodes as monotherapy and as adjunct to lithium or valproate
- Maintenance monotherapy treatment
14.2 Bipolar I Disorder
The efficacy of LYBALVI in the treatment of adult patients with bipolar I disorder has been established based on adequate and well-controlled studies of orally administered olanzapine. The information below describes the results of adequate and well-controlled studies of olanzapine in patients with bipolar I disorder.
5.17 Hyperprolactinemia
As with other drugs that antagonize dopamine D2 receptors, olanzapine, a component of LYBALVI, elevates prolactin levels, and the elevation can persist during chronic administration. Hyperprolactinemia may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotropin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects.
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if the prescription of these drugs is considered in a patient with previously-detected breast cancer. As is common with compounds which increase prolactin release, an increase in mammary gland neoplasia was observed in the olanzapine carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)]. Published epidemiologic studies have shown inconsistent results when exploring the potential association between hyperprolactinemia and breast cancer.
In the 4-week placebo-controlled trial, shifts from normal to high prolactin values (>30 ng/mL for females; >20 ng/mL for males) occurred in 41.4% of females and 32.9% of males treated with LYBALVI, in 56.1% of females and 37.1% of males treated with olanzapine, and in 10% of females and 4.8% of males treated with placebo [see Use in Specific Populations (8.3)].
In the 24-week, olanzapine-controlled study, shifts from normal to high prolactin values occurred in 32.9% of females and 22.5% of males treated with LYBALVI, and in 41.7% of females and 28.5% of males treated with olanzapine.
12.1 Mechanism of Action
The mechanism of action of olanzapine is unclear; however, its efficacy in the treatment of schizophrenia or bipolar I disorder could be mediated through a combination of dopamine and serotonin type 2 (5HT2) antagonism.
The mechanism of action of samidorphan could be mediated through opioid receptor antagonism.
5 Warnings and Precautions
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). (5.2)
- Precipitation of Opioid Withdrawal in Patients Who are Dependent on Opioids: LYBALVI can precipitate opioid withdrawal in patients who are dependent on opioids. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from the last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids to avoid precipitation of opioid withdrawal. (2.1, 5.3)
-
Vulnerability to Life-Threatening Opioid Overdose:
- Risk of Opioid Overdose from Attempts to Overcome LYBALVI Opioid Blockade: Attempts to overcome LYBALVI opioid blockade with high or repeated doses of opioids may lead to fatal opioid intoxication, particularly if LYBALVI therapy is interrupted or discontinued. (5.4)
- Risk of Resuming Opioids in Patients with Prior Opioid Use: Patients with a history of chronic opioid use prior to LYBALVI treatment may have decreased opioid tolerance if LYBALVI therapy is interrupted or discontinued. (5.4)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring. (5.5)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue if DRESS is suspected. (5.6)
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. (5.7)
- Tardive Dyskinesia: Discontinue if clinically appropriate. (5.8)
- Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. (5.9)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors. (5.11)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. (5.13)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery. (5.14)
- Anticholinergic (Antimuscarinic) Effects: Use with caution with other anticholinergic drugs and in patients with urinary retention, prostatic hypertrophy, constipation, paralytic ileus or related conditions. (5.16)
- Hyperprolactinemia: May elevate prolactin levels. (5.17)
2 Dosage and Administration
| Indication |
Recommended Starting Dose (olanzapine/samidorphan) |
Recommended Dose (olanzapine/samidorphan) |
| Schizophrenia (2.2) | 5 mg/10 mg or 10 mg/10 mg |
10 mg/10 mg 15 mg/10 mg 20 mg/10 mg |
| Bipolar I disorder (manic or mixed episodes) (2.3) |
10 mg/10 mg or 15 mg/10 mg |
5 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg |
| Bipolar I disorder adjunct to lithium or valproate (2.3) | 10 mg/10 mg | 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg |
- See the full prescribing information for the recommended titration and maximum recommended dosage. (2.2, 2.3)
- Administer LYBALVI once daily with or without food. Do not divide tablets or combine strengths. (2.4)
- Recommended starting dosage is 5 mg/10 mg once daily in patients who have a predisposition to hypotensive reactions, have potential for slower metabolism of olanzapine, or may be more pharmacodynamically sensitive to olanzapine. (2.5)
3 Dosage Forms and Strengths
LYBALVI tablets are available in four strengths (Table 1).
| Tablet Strength | Tablet Color/Shape | Tablet Markings |
|---|---|---|
| 5 mg/10 mg (olanzapine/samidorphan) | Yellow, capsule-shaped | “OS” and “5” |
| 10 mg/10 mg (olanzapine/samidorphan) | Orange, capsule-shaped | “OS” and “10” |
| 15 mg/10 mg (olanzapine/samidorphan) | Blue, capsule-shaped | “OS” and “15” |
| 20 mg/10 mg (olanzapine/samidorphan) | Pink, capsule-shaped | “OS” and “20” |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of olanzapine. Because these reactions are reported voluntarily from a population of uncertain size, it is difficult to reliably estimate their frequency or evaluate a causal relationship to drug exposure.
- allergic reactions (e.g., anaphylactoid reaction, angioedema, pruritus or urticaria)
- cholestatic or mixed liver injury, hepatitis, jaundice
- diabetic coma, diabetic ketoacidosis
- discontinuation reaction (diaphoresis, nausea or vomiting)
- Drug reaction with eosinophilia and systemic symptoms (DRESS)
- fecal incontinence
- hyperlipidemia (random cholesterol levels of ≥240 mg/dL and random triglyceride levels of ≥1000 mg/dL have been reported)
- neutropenia
- pancreatitis
- priapism
- rash
- restless legs syndrome
- rhabdomyolysis
- salivary hypersecretion
- syndrome of inappropriate antidiuretic hormone secretion (SIADH)
- somnambulism
- stutteringStuttering was only studied in oral and long acting injection (LAI) formulations.
- venous thromboembolic events (including pulmonary embolism and deep venous thrombosis)
8 Use in Specific Populations
2.4 Administration Information
Administer LYBALVI orally once daily with or without food as a single tablet.
Do not divide tablets or combine strengths.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.5 Neuroleptic Malignant Syndrome
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with administration of antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, delirium, and autonomic instability. Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue LYBALVI and provide intensive symptomatic treatment and monitoring.
5.15 Body Temperature Dysregulation
Atypical antipsychotics may disrupt the body's ability to reduce core body temperature. Strenuous exercise, exposure to extreme heat, dehydration, and anticholinergic medications may contribute to an elevation in core body temperature; use LYBALVI with caution in patients who may experience these conditions.
7.1 Effects of Other Drugs On Lybalvi
Table 4 describes clinically significant drug interactions where the concomitant use of other drugs affects LYBALVI.
| Strong CYP3A4 Inducer | |
| Clinical Implication: | Coadministration of LYBALVI with a strong CYP3A4 inducer decreases AUCinf of olanzapine and samidorphan [see Clinical Pharmacology (12.3)], which may reduce LYBALVI efficacy. |
| Prevention or Management: | Concomitant use of LYBALVI with strong CYP3A4 inducers is not recommended. |
| Strong CYP1A2 Inhibitor | |
| Clinical Implication: | Concomitant use of LYBALVI with a strong CYP1A2 inhibitor increases olanzapine AUC and Cmax [see Clinical Pharmacology (12.3)], which may increase the risk of LYBALVI adverse reactions. |
| Prevention or Management: | Consider reducing the dosage of the olanzapine component in LYBALVI when used concomitantly with strong CYP1A2 inhibitors. |
| CYP1A2 Inducer | |
| Clinical Implication: | Concomitant use of LYBALVI with CYP1A2 inducers decreases olanzapine exposure [see Clinical Pharmacology (12.3)], which may reduce LYBALVI efficacy. |
| Prevention or Management: | Consider increasing the dosage of the olanzapine component in LYBALVI when used concomitantly with CYP1A2 inducers. |
| Diazepam, Alcohol, and Other CNS Acting Drugs | |
| Clinical Implication: | Concomitant use of diazepam, alcohol, or other CNS acting drugs with LYBALVI may potentiate the orthostatic hypotension observed with olanzapine [see Warnings and Precautions (5.9)]. |
| Prevention or Management: | LYBALVI should be used with caution in patients receiving concomitantly diazepam or other CNS acting drugs, or using alcohol. |
| Anticholinergic Drugs | |
| Clinical Implication: | Concomitant treatment with olanzapine and other drugs with anticholinergic activity can increase the risk for severe gastrointestinal adverse reactions related to hypomotility. |
| Prevention or Management: | LYBALVI should be used with caution in patients receiving medications having anticholinergic (antimuscarinic) effects [see Warnings and Precautions (5.16)]. |
7.2 Effects of Lybalvi On Other Drugs
Table 5 describes clinically significant drug interactions where concomitant use of LYBALVI affects other drugs.
| Antihypertensive Agents | |
| Clinical Implication: | LYBALVI may enhance the effects of certain antihypertensive agents. |
| Prevention or Management: | Monitor blood pressure and reduce dosage of antihypertensive drug in accordance with its approved product labeling. |
| Levodopa and Dopamine Agonists | |
| Clinical Implication: | LYBALVI may antagonize the effects of levodopa and dopamine agonists. |
| Prevention or Management: | Concomitant use of LYBALVI is not recommended with levodopa and dopamine agonists. |
7.4 Interference With Laboratory Tests
Interference with Laboratory Tests for Opioid Detection
Because LYBALVI contains samidorphan, an opioid antagonist, LYBALVI may be cross-reactive with urinary immunoassay methods used for detecting opioids, resulting in false positive results. Use an alternative analytical technique (e.g., chromatographic methods) to confirm positive opioid urine drug screen results [see Contraindications (4) and Warnings and Precautions (5.19)].
2.2 Recommended Dosage in Schizophrenia
Initiate LYBALVI at 5 mg/10 mg (contains 5 mg of olanzapine and 10 mg of samidorphan) or 10 mg/10 mg (contains 10 mg of olanzapine and 10 mg of samidorphan) orally once daily. The recommended dosage is 10 mg/10 mg, 15 mg/10 mg (contains 15 mg of olanzapine and 10 mg of samidorphan), or 20 mg/10 mg (contains 20 mg of olanzapine and 10 mg of samidorphan) once daily.
Dosage may be adjusted at weekly intervals of 5 mg (based on the olanzapine component of LYBALVI) depending upon clinical response and tolerability, up to the maximum recommended dosage of 20 mg/10 mg once daily.
5.9 Orthostatic Hypotension and Syncope
Atypical antipsychotics cause orthostatic hypotension and syncope. Generally, the risk is greatest during initial dose titration and when increasing the dose. In the 4-week, placebo-controlled study, from analysis of the vital signs data, rates of orthostatic hypotension were less than 2% in LYBALVI- and placebo-, and olanzapine-treated patients. In the 24-week, olanzapine-controlled study, from analysis of the vital signs data, rates of orthostatic hypotension in LYBALVI-treated patients were 3.7%, compared to 0.4% in olanzapine-treated patients.
Monitor orthostatic vital signs in patients who are vulnerable to hypotension (e.g., elderly patients, patients with dehydration, hypovolemia, concomitant treatment with antihypertensive medications or CNS depressants [see Drug Interactions (7.1, 7.2)], patients with known cardiovascular disease (history of myocardial infarction, ischemic heart disease, heart failure, or conduction abnormalities), and patients with cerebrovascular disease. LYBALVI has not been evaluated in patients with a recent history of myocardial infarction or unstable cardiovascular disease. Such patients were excluded from the premarketing clinical trials.
5.16 Anticholinergic (antimuscarinic) Effects
Olanzapine, a component of LYBALVI, exhibits in vitro muscarinic receptor affinity [see Clinical Pharmacology (12.2)]. In premarketing clinical trials with oral olanzapine, olanzapine was associated with constipation, dry mouth, and tachycardia, all adverse reactions possibly related to cholinergic antagonism. Such adverse reactions were not often the basis for discontinuations, but LYBALVI should be used with caution in patients with a current diagnosis or prior history of urinary retention, clinically significant prostatic hypertrophy, constipation, or a history of paralytic ileus or related conditions. In postmarketing experience, the risk for severe adverse reactions (including fatalities) was increased with concomitant use of anticholinergic medications [see Drug Interactions (7.1)].
2.1 Lybalvi Initiation in Patients Using Opioids
LYBALVI is contraindicated in patients using opioids or undergoing acute opioid withdrawal.
In patients who use opioids, delay initiation of LYBALVI for a minimum of 7 days after last use of short-acting opioids and 14 days after last use of long-acting opioids [see Warnings and Precautions ( 5.3)].
5.11 Leukopenia, Neutropenia, and Agranulocytosis
Leukopenia and neutropenia have been reported during treatment with antipsychotic agents, including LYBALVI [see Adverse Reactions (6)]. Agranulocytosis (including fatal cases) has been reported with other agents in this class.
Possible risk factors for leukopenia and neutropenia include pre-existing low white blood cell count (WBC) or absolute neutrophil count (ANC) and history of drug-induced leukopenia or neutropenia. In patients with a pre-existing low WBC or ANC or a history of drug-induced leukopenia or neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of LYBALVI at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue LYBALVI in patients with severe neutropenia (absolute neutrophil count <1000/mm3) and follow their WBC until recovery.
5.14 Potential for Cognitive and Motor Impairment
LYBALVI, like other antipsychotics, may cause somnolence and has the potential to impair judgment, thinking, or motor skills. In a LYBALVI placebo-controlled study, somnolence occurred in 9% of LYBALVI-treated patients compared to 2.2% in patients treated with placebo.
Patients should be cautioned about operating hazardous machinery, including motor vehicles, until they are reasonably certain that LYBALVI therapy does not affect them adversely.
2.5 Dosage Recommendations in Specific Populations
The recommended starting dosage of LYBALVI is 5 mg/10 mg once daily in patients who have a higher risk of hypotensive reactions, are at risk of slower olanzapine metabolism, or may be more pharmacodynamically sensitive to olanzapine [see Warnings and Precautions (5.9), Drug Interactions (7.2), Use in Specific Populations (8.5), and Clinical Pharmacology (12.3)]. If dose escalation is necessary, increase the dosage slowly in these patients.
5.6 Drug Reaction With Eosinophilia and Systemic Symptoms
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported with exposure to olanzapine, a component of LYBALVI [see Adverse Reactions (6.2)]. DRESS may present with a cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, and/or lymphadenopathy with systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and/or pericarditis. DRESS is sometimes fatal. Discontinue LYBALVI if DRESS is suspected.
5.19 Interference With Laboratory Tests for Opioid Detection
5.18 Risks Associated With Combination Treatment With Lithium Or Valproate
If LYBALVI is administered with lithium or valproate, refer to the lithium or valproate Prescribing Information for a description of the risks for these products including, but not limited to, the warnings and precautions for lithium or valproate [see Contraindications (4)].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. In placebo-controlled clinical trials of elderly patients with dementia-related psychosis, the incidence of death in olanzapine-treated patients was significantly greater than in placebo-treated patients (3.5% vs 1.5%, respectively). Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in the drug-treated patients of between 1.6 to 1.7 times that seen in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.2)].
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
5.3 Precipitation of Severe Opioid Withdrawal in Patients Who Are Physiologically Dependent On Opioids
Samidorphan, an opioid antagonist that is a component of LYBALVI, can precipitate opioid withdrawal in patients who are dependent on opioids, which can lead to an opioid withdrawal syndrome, sometimes requiring hospitalization. Therefore, LYBALVI is contraindicated in patients who are using opioids or undergoing acute opioid withdrawal. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids. Explain the risks associated with precipitated withdrawal and the importance of giving an accurate account of last opioid use to patients and caregivers [see Dosage and Administration (2.1), Drug Interactions (7.3)].
5.2 Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients With Dementia Related Psychosis
Cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack), including fatalities, were reported in patients in trials of olanzapine in elderly patients with dementia-related psychosis. In placebo-controlled trials, there was a significantly higher incidence of cerebrovascular adverse reactions in patients treated with olanzapine compared to patients treated with placebo. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
Structured Label Content
Section 42229-5 (42229-5)
Monotherapy: Initiate LYBALVI at 10 mg/10 mg or 15 mg/10 mg once daily. The recommended dosage is 10 mg/10 mg, 15 mg/10 mg, or 20 mg/10 mg once daily. The maximum recommended dosage is 20 mg/10 mg once daily.
Dosage adjustments should occur at intervals of not less than 24 hours. When dosage adjustments are necessary, dose increments/decrements of 5 mg (based on the olanzapine component of LYBALVI) are recommended.
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised: 1/2024 |
|
|
MEDICATION GUIDE
LYBALVI® (lee bawl' vee) (olanzapine and samidorphan) tablets, for oral use |
||
| If your healthcare provider prescribes LYBALVI in combination with valproate or lithium, also read the Medication Guide that comes with those medicines. | ||
|
What is the most important information I should know about LYBALVI?
|
||
|
What is LYBALVI?
LYBALVI is a prescription medicine which contains 2 medicines (olanzapine and samidorphan) used in adults:
|
||
| It is not known if LYBALVI is safe or effective in children. | ||
| Do not take LYBALVI if you are taking opioids or are experiencing acute opioid withdrawal. See “What are the possible side effects of LYBALVI.” | ||
Before taking LYBALVI, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. LYBALVI and other medicines may affect each other causing possible serious side effects. LYBALVI may affect the way other medicines work, and other medicines may affect how LYBALVI works. Especially tell your healthcare provider if you:
|
||
| Your healthcare provider can tell you if it is safe to take LYBALVI with your other medicines. Do not start or stop any medicines while taking LYBALVI without first talking to your healthcare provider. Tell your healthcare provider if you take a urine drug screening test because LYBALVI may affect your test results. Tell those giving the drug screening test that you are taking LYBALVI. Know the medicines you take. Keep a list of your medicines to show your healthcare provider and pharmacist when you get a new medicine. |
||
How should I take LYBALVI?
|
||
|
What should I avoid while taking LYBALVI?
|
||
|
What are the possible side effects of LYBALVI?
|
||
|
|
|
|
||
|
|
|
|
||
|
||
| The most common side effects of LYBALVI when used to treat people with schizophrenia include: | ||
|
|
|
|
The most common side effects of LYBALVI when used alone to treat people with mixed or manic episodes that happen with bipolar I disorder include:
|
||
|
|
|
|
The most common side effects of LYBALVI when used in combination with lithium or valproate to treat people with mixed or manic episodes that happen with bipolar I disorder include:
|
||
|
|
|
| These are not all the possible side effects of LYBALVI. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store LYBALVI?
|
||
| Keep LYBALVI and all medicines out of the reach of children. | ||
|
General information about the safe and effective use of LYBALVI
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use LYBALVI for a condition for which it is not prescribed. Do not give LYBALVI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LYBALVI that is written for healthcare providers. |
||
|
What are the ingredients in LYBALVI?
Active ingredients: olanzapine and samidorphan Inactive ingredients: colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate and microcrystalline cellulose. The film coating ingredients include hypromellose, titanium dioxide, triacetin and iron oxide yellow (5 mg/10 mg); iron oxide yellow and iron oxide red (10 mg/10 mg); FD&C Blue No. 2/indigo carmine aluminum lake (15 mg/10 mg); iron oxide red (20 mg/10 mg) Manufactured and marketed by: Alkermes, Inc., Waltham, MA 02451 For more information, go to www.LYBALVI.com or call 1-844-LYBALVI (1-844-592-2584). |
Section 44425-7 (44425-7)
Storage and Handling
Store at room temperature 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Keep tightly closed and protect from moisture.
Section 51945-4 (51945-4)
Principal Display Panel - 5 mg/10 mg 30 Tablets Carton Label
NDC 65757-651-42 Rx Only
LYBALVI ®
(olanzapine/samidorphan)
tablets
5 mg/10 mg
Dispense the accompanying
Medication Guide to each patient.
30 Tablets
5.10 Falls
Antipsychotics, including LYBALVI, may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
7.3 Opioids
LYBALVI is contraindicated in patients who are using opioids or undergoing acute opioid withdrawal [see Contraindications (4)].
LYBALVI increases the risk of precipitating acute opioid withdrawal in patients who are dependent on opioids. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from the last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids [see Dosage and Administration (2.1), Warnings and Precautions (5.3)].
In emergency situations, if a LYBALVI-treated patient requires opioid treatment for anesthesia or analgesia, discontinue LYBALVI. The opioid should be administered by properly trained individual(s), and the patient should be properly monitored in a setting equipped and staffed for cardiopulmonary resuscitation [see Warnings and Precautions (5.4)].
In non-emergency situations, if a LYBALVI-treated patient is expected to require opioid treatment (e.g., for analgesia during or after an elective surgical procedure) discontinue LYBALVI at least 5 days before opioid treatment and start olanzapine or another antipsychotic, if needed.
Given that LYBALVI contains samidorphan, an opioid antagonist, opioid treatment may be less effective or ineffective shortly after LYBALVI discontinuation because of the presence of samidorphan.
5.13 Seizures
Like other antipsychotic drugs, LYBALVI may cause seizures. This risk is greatest in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in older patients.
11 Description (11 DESCRIPTION)
LYBALVI is a combination of olanzapine, an atypical antipsychotic, and samidorphan (as samidorphan L-malate), an opioid antagonist.
Olanzapine is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b][1,5]benzodiazepine. The molecular formula of olanzapine is: C17H20N4S and the molecular weight is 312.44 g/mol. It is a yellow crystalline powder and has pKa values of 7.80 and 5.44. The chemical structure is:
Samidorphan L-malate is morphinan-3-carboxamide, 17-(cyclopropylmethyl)-4, 14-dihydroxy-6-oxo-, (2S)-2-hydroxybutanedioate. The molecular formula of samidorphan L-malate is C21H26N2O4 • C4H6O5 and the molecular weight is 504.54 g/mol. It is a white to off-white crystalline powder and has pKa values of 8.3 (amine) and 10.1 (phenol). The chemical structure is:
LYBALVI is intended for oral administration and is available as film-coated, bilayer tablets in the following strengths: 5 mg/10 mg, 10 mg/10 mg, 15 mg/10 mg, and 20 mg/10 mg of olanzapine and samidorphan (equivalent to 13.6 mg of samidorphan L-malate).
Inactive ingredients include colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. The film coating ingredients include hypromellose, titanium dioxide, triacetin, and color additives [iron oxide yellow (5 mg/10 mg); iron oxide yellow and iron oxide red (10 mg/10 mg); FD&C Blue No. 2/ indigo carmine aluminum lake (15 mg/10 mg); iron oxide red (20 mg/10 mg)].
5.12 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Antipsychotic drugs, including LYBALVI, should be used cautiously in patients at risk for aspiration.
8.4 Pediatric Use
The safety and effectiveness of LYBALVI have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of LYBALVI did not include sufficient numbers of patients 65 years of age and older to determine whether they responded differently than younger adult patients.
14.1 Schizophrenia
The efficacy of LYBALVI in the treatment of schizophrenia in adults is based, in part, upon adequate and well-controlled studies of orally administered olanzapine. Efficacy of LYBALVI was also evaluated in a 4-week, randomized, double-blind, placebo- and active-controlled study (Study 1).
In Study 1 (NCT02634346), adult patients who met DSM-5 criteria for schizophrenia were randomized in a 1:1:1 ratio to LYBALVI, olanzapine, or placebo for 4 weeks of daily dosing. Dosing was flexible based on clinical response and tolerability for the first 2 weeks of the study; doses thereafter were fixed. Patients assigned to LYBALVI could receive either 10 mg/10 mg or 20 mg/10 mg, and patients assigned to olanzapine could receive either 10 mg or 20 mg. The study was designed to compare LYBALVI with placebo, not with olanzapine.
Eligible patients were 18 to 70 years of age, with a body mass index (BMI) of 18.0–40.0 kg/m2, Positive and Negative Syndrome Scale (PANSS) total score of ≥80, and a score of ≥4 on at least 3 of the selected Positive Scale items. Patients were also required to have a Clinical Global Impression-Severity (CGI-S) score ≥4.
The primary efficacy endpoint was change from baseline in PANSS total score at Week 4. The PANSS is a 30-item scale that measures positive symptoms of schizophrenia (7 items), negative symptoms of schizophrenia (7 items), and general psychopathology (16 items), each rated on a scale of 1 (absent) to 7 (extreme). Total PANSS scores range from 30 to 210, with a higher score reflecting greater symptom severity. The secondary efficacy endpoint was defined as the change from baseline in CGI-S score at Week 4. The CGI-S is a validated scale that requires clinicians to rate a patient's current illness severity and overall clinical state based on experience with the illness population. Scores range from 1 (normal, not at all ill) to 7 (extremely ill).
Compared with patients on placebo, a statistically significant improvement in the change from baseline in PANSS total score at Week 4 was observed in patients treated with LYBALVI (Table 7). The inclusion of samidorphan in LYBALVI did not appear to negatively impact the antipsychotic efficacy of olanzapine.
| Treatment Group | Total PANSS Score | ||
|---|---|---|---|
| Baseline Mean Score (SD) | LS Mean Change from Baseline (SE) | Placebo-subtracted Difference a (95% CI) | |
|
Abbreviations: CI: confidence interval; LS: least squares; SD: standard deviation; SE: standard error. |
|||
|
a Difference (drug minus placebo) in least squares mean change from baseline. A negative value for the placebo subtracted difference represents improvement. |
|||
| LYBALVI (10 mg/10 mg, 20 mg/10 mg) (N=132) |
101.8 (11.6) |
-23.9 (1.3) |
-6.4 (-10.0, -2.8) |
| Placebo (N=133) |
102.7 (11.9) |
-17.5 (1.3) |
-- |
| Olanzapine (10 mg, 20 mg) (N=132) |
100.6 (12.1) |
-22.8 (1.3) |
-5.3 (-8.9, -1.7) |
The change from baseline in PANSS total score in Study 1 is displayed in Figure 4.
Figure 4: Change from Baseline in PANSS Total Score by Time (Week) in Patients with Schizophrenia (Study 1)
Error bars represent standard error. The numbers under the figure indicate the number of patients at each timepoint.
Compared with patients on placebo, a statistically significant improvement in CGI-S score at Week 4 was observed in patients treated with LYBALVI.
4 Contraindications (4 CONTRAINDICATIONS)
LYBALVI is contraindicated in patients:
- who are using opioids [see Warnings and Precautions ( 5.3 , 5.4), Drug Interactions ( 7.3)].
- who are undergoing acute opioid withdrawal [see Warnings and Precautions (5.3, 5.4), Drug Interactions (7.3)].
If LYBALVI is administered with lithium or valproate, refer to the lithium or valproate Prescribing Information for the contraindications for these products [see Warnings and Precautions (5.18)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-related Psychosis [see Boxed Warning, Warnings and Precautions (5.1)]
- Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis [see Warnings and Precautions (5.2)]
- Precipitation of Opioid Withdrawal in Patients Who Are Dependent on Opioids [see Warnings and Precautions (5.3)]
- Vulnerability to Life-Threatening Opioid Overdose [see Warnings and Precautions (5.4)]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.5)]
- Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions (5.6)]
- Metabolic Changes [see Warnings and Precautions (5.7)]
- Tardive Dyskinesia [see Warnings and Precautions (5.8)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.9)]
- Falls [see Warnings and Precautions (5.10)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.11)]
- Dysphagia [see Warnings and Precautions (5.12)]
- Seizures [see Warnings and Precautions (5.13)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.14)]
- Body Temperature Regulation [see Warnings and Precautions (5.15)]
- Anticholinergic (Antimuscarinic) Effects [see Warnings and Precautions (5.16)]
- Hyperprolactinemia [see Warnings and Precautions (5.17)]
- Risks Associated with Combination Treatment with Lithium or Valproate [see Warnings and Precautions (5.18)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Strong CYP3A4 Inducers: Not recommended. (7.1)
- Strong CYP1A2 Inhibitors: Consider dosage reduction of olanzapine component of LYBALVI. (7.1)
- CYP1A2 Inducer: Consider dosage increase of the olanzapine component of LYBALVI. (7.1)
- CNS Acting Drugs: May potentiate orthostatic hypotension. (7.1)
- Anticholinergic Drugs: Can increase risk for severe gastrointestinal adverse reactions. (7.1)
- Antihypertensive Agents: Monitor blood pressure. (7.2)
- Levodopa and Dopamine Agonists: Not recommended. (7.2)
8.7 Renal Impairment
Plasma exposure to olanzapine and samidorphan was higher in patients with severe renal impairment (eGFR 15 to 29 mL/minute/1.73 m2) compared to those with normal renal function [see Clinical Pharmacology (12.3)]. No dose adjustment of LYBALVI is needed in patients with mild (eGFR 60 to 89 mL/minute/1.73 m2), moderate (eGFR 30 to 59 mL/minute/1.73 m2), or severe renal impairment (eGFR 15 to 29 mL/minute/1.73 m2).
The effect of LYBALVI in patients with end-stage renal disease was not studied. LYBALVI is not recommended for patients with end-stage renal disease (eGFR of <15 mL/minute/1.73 m2).
12.3 Pharmacokinetics
The pharmacokinetics of both olanzapine and samidorphan are linear over the clinical dose range and there is no PK interaction between olanzapine and samidorphan after oral administration of LYBALVI. Steady-state concentrations of olanzapine and samidorphan are reached within 7 days of commencement of once-daily administration of LYBALVI. The primary pharmacological activities of LYBALVI are due to the parent drugs, olanzapine and samidorphan.
Following a single dose administration of LYBALVI (10 mg olanzapine/10 mg samidorphan), the mean AUC0-inf and Cmax of olanzapine was 628 ng·h/mL and 16 ng/mL, respectively. The mean AUC0-inf and Cmax of olanzapine after 10 mg single dose administration of olanzapine tablet was 610 ng·h/mL and 16 ng/mL, respectively.
Pharmacokinetic properties of the components of LYBALVI are provided in Table 6.
| Parameters | Olanzapine | Samidorphan |
|---|---|---|
|
Abbreviations: AUC24h=area under the concentration-time curve over the 24-hour dosing interval; Cmax=maximum plasma concentration; CYP=cytochrome P450; NA=not applicable; Tmax=time to Cmax; t½=terminal elimination half-life; UGT=Uridine 5'-diphospho-glucuronosyltransferase. |
||
|
a Presented as arithmetic mean (standard deviation). |
||
|
b Presented as range of the median across multiple studies. |
||
|
c Geometric mean ratio (90% confidence interval) [high fat meal/fasting]. High fat meal defined as meal containing approximately 900-1000 calories and 50% fat content. No clinically relevant food effect. |
||
|
d Presented as range of the mean across multiple studies. |
||
| General | ||
| Steady State Exposure (20 mg olanzapine/10 mg samidorphan) | ||
| Cmax (ng/mL)a | 64.6 (28.9) | 45.1 (11.4) |
| AUC24h (ng∙hr/mL)a | 1086 (556) | 364 (112) |
| Time to Reach Steady State | 7 days | 5 days |
| Accumulation at Steady State | 2-fold | 1.3-fold |
| Absorption | ||
| Absolute Oral Bioavailability | NA | 69% |
| Tmax (h)b | 4.5–7 | 1–2 |
| Effect of Food | ||
| Cmax Ratioc | 0.88 (0.82, 0.95) | 0.85 (0.76, 0.94) |
| AUC Ratioc | 0.93 (0.91, 0.96) | 1.03 (1.00, 1.05) |
| Distribution | ||
| Plasma Protein Binding | 93% | 23% - 33% |
| Blood-to-Plasma Ratio | Not Determined | 0.8 |
| Elimination | ||
| t1/2 (h)d | 35–52 | 7–11 |
| CL/F (L/h)d | 15–22 | 35–45 |
| Metabolism | ||
| Primary Pathway(s) | UGT1A4, CYP1A2 | CYP3A4 |
| Minor Pathway(s) | CYP2D6 | CYP3A5, CYP2C19, CYP2C8 |
| Major Circulating Metabolites | 10-N-glucuronide and 4′-N-desmethyl-olanzapine. Both metabolites lack pharmacological activity at the therapeutic concentrations |
N-dealkylated and cis-N-oxide metabolites. Neither metabolite contributes to the pharmacological effects of samidorphan |
| Excretion | ||
| Primary Route of Elimination | Metabolism | Metabolism |
| Urine (Unchanged) | 7% | 18% |
| Urine (Unchanged + metabolites) | 57% | 67% |
| Feces (Unchanged + metabolites) | 30% | 16% |
5.7 Metabolic Changes
Atypical antipsychotic drugs, including LYBALVI, have been associated with metabolic changes that include hyperglycemia, diabetes mellitus, dyslipidemia, and body weight gain [see Adverse Reactions (6.1)]. While all drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Any patient treated with LYBALVI should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with LYBALVI should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required anti-diabetic treatment despite discontinuation of the suspect drug. Patients starting treatment with LYBALVI should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment.
Antipsychotics have caused adverse alterations in lipids. Patients starting treatment with LYBALVI should undergo fasting lipid profile testing at the beginning of treatment and periodically during treatment.
Weight gain has been observed with use of antipsychotics. Monitor weight prior to initiating LYBALVI and frequently thereafter.
5.8 Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. The risk appears to be highest among the elderly, especially elderly women, but it is not possible to predict which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible increases with the duration of treatment and the cumulative dose. The syndrome can develop after a relatively brief treatment period, even at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, LYBALVI should be prescribed in a manner that is most likely to reduce the risk of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients: 1) who suffer from a chronic illness that is known to respond to antipsychotic drugs; and 2) for whom alternative, effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, use the lowest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. Periodically reassess the need for continued treatment.
If signs and symptoms of tardive dyskinesia appear in a patient on LYBALVI, drug discontinuation should be considered. However, some patients may require treatment with LYBALVI despite the presence of the syndrome.
8.6 Hepatic Impairment
Olanzapine and samidorphan plasma exposures were found to be higher in subjects with moderate hepatic impairment than in subjects with normal hepatic function. The effect of severe hepatic impairment was not studied. The higher plasma exposure in patients with moderate hepatic impairment was not expected to be clinically relevant. No dose adjustment of LYBALVI is needed in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
LYBALVI is indicated for the treatment of:
- Schizophrenia in adults
- Bipolar I disorder in adults
- Acute treatment of manic or mixed episodes as monotherapy and as adjunct to lithium or valproate
- Maintenance monotherapy treatment
14.2 Bipolar I Disorder
The efficacy of LYBALVI in the treatment of adult patients with bipolar I disorder has been established based on adequate and well-controlled studies of orally administered olanzapine. The information below describes the results of adequate and well-controlled studies of olanzapine in patients with bipolar I disorder.
5.17 Hyperprolactinemia
As with other drugs that antagonize dopamine D2 receptors, olanzapine, a component of LYBALVI, elevates prolactin levels, and the elevation can persist during chronic administration. Hyperprolactinemia may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotropin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects.
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if the prescription of these drugs is considered in a patient with previously-detected breast cancer. As is common with compounds which increase prolactin release, an increase in mammary gland neoplasia was observed in the olanzapine carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)]. Published epidemiologic studies have shown inconsistent results when exploring the potential association between hyperprolactinemia and breast cancer.
In the 4-week placebo-controlled trial, shifts from normal to high prolactin values (>30 ng/mL for females; >20 ng/mL for males) occurred in 41.4% of females and 32.9% of males treated with LYBALVI, in 56.1% of females and 37.1% of males treated with olanzapine, and in 10% of females and 4.8% of males treated with placebo [see Use in Specific Populations (8.3)].
In the 24-week, olanzapine-controlled study, shifts from normal to high prolactin values occurred in 32.9% of females and 22.5% of males treated with LYBALVI, and in 41.7% of females and 28.5% of males treated with olanzapine.
12.1 Mechanism of Action
The mechanism of action of olanzapine is unclear; however, its efficacy in the treatment of schizophrenia or bipolar I disorder could be mediated through a combination of dopamine and serotonin type 2 (5HT2) antagonism.
The mechanism of action of samidorphan could be mediated through opioid receptor antagonism.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). (5.2)
- Precipitation of Opioid Withdrawal in Patients Who are Dependent on Opioids: LYBALVI can precipitate opioid withdrawal in patients who are dependent on opioids. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from the last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids to avoid precipitation of opioid withdrawal. (2.1, 5.3)
-
Vulnerability to Life-Threatening Opioid Overdose:
- Risk of Opioid Overdose from Attempts to Overcome LYBALVI Opioid Blockade: Attempts to overcome LYBALVI opioid blockade with high or repeated doses of opioids may lead to fatal opioid intoxication, particularly if LYBALVI therapy is interrupted or discontinued. (5.4)
- Risk of Resuming Opioids in Patients with Prior Opioid Use: Patients with a history of chronic opioid use prior to LYBALVI treatment may have decreased opioid tolerance if LYBALVI therapy is interrupted or discontinued. (5.4)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring. (5.5)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue if DRESS is suspected. (5.6)
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. (5.7)
- Tardive Dyskinesia: Discontinue if clinically appropriate. (5.8)
- Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. (5.9)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors. (5.11)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. (5.13)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery. (5.14)
- Anticholinergic (Antimuscarinic) Effects: Use with caution with other anticholinergic drugs and in patients with urinary retention, prostatic hypertrophy, constipation, paralytic ileus or related conditions. (5.16)
- Hyperprolactinemia: May elevate prolactin levels. (5.17)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
| Indication |
Recommended Starting Dose (olanzapine/samidorphan) |
Recommended Dose (olanzapine/samidorphan) |
| Schizophrenia (2.2) | 5 mg/10 mg or 10 mg/10 mg |
10 mg/10 mg 15 mg/10 mg 20 mg/10 mg |
| Bipolar I disorder (manic or mixed episodes) (2.3) |
10 mg/10 mg or 15 mg/10 mg |
5 mg/10 mg 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg |
| Bipolar I disorder adjunct to lithium or valproate (2.3) | 10 mg/10 mg | 10 mg/10 mg 15 mg/10 mg 20 mg/10 mg |
- See the full prescribing information for the recommended titration and maximum recommended dosage. (2.2, 2.3)
- Administer LYBALVI once daily with or without food. Do not divide tablets or combine strengths. (2.4)
- Recommended starting dosage is 5 mg/10 mg once daily in patients who have a predisposition to hypotensive reactions, have potential for slower metabolism of olanzapine, or may be more pharmacodynamically sensitive to olanzapine. (2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
LYBALVI tablets are available in four strengths (Table 1).
| Tablet Strength | Tablet Color/Shape | Tablet Markings |
|---|---|---|
| 5 mg/10 mg (olanzapine/samidorphan) | Yellow, capsule-shaped | “OS” and “5” |
| 10 mg/10 mg (olanzapine/samidorphan) | Orange, capsule-shaped | “OS” and “10” |
| 15 mg/10 mg (olanzapine/samidorphan) | Blue, capsule-shaped | “OS” and “15” |
| 20 mg/10 mg (olanzapine/samidorphan) | Pink, capsule-shaped | “OS” and “20” |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of olanzapine. Because these reactions are reported voluntarily from a population of uncertain size, it is difficult to reliably estimate their frequency or evaluate a causal relationship to drug exposure.
- allergic reactions (e.g., anaphylactoid reaction, angioedema, pruritus or urticaria)
- cholestatic or mixed liver injury, hepatitis, jaundice
- diabetic coma, diabetic ketoacidosis
- discontinuation reaction (diaphoresis, nausea or vomiting)
- Drug reaction with eosinophilia and systemic symptoms (DRESS)
- fecal incontinence
- hyperlipidemia (random cholesterol levels of ≥240 mg/dL and random triglyceride levels of ≥1000 mg/dL have been reported)
- neutropenia
- pancreatitis
- priapism
- rash
- restless legs syndrome
- rhabdomyolysis
- salivary hypersecretion
- syndrome of inappropriate antidiuretic hormone secretion (SIADH)
- somnambulism
- stutteringStuttering was only studied in oral and long acting injection (LAI) formulations.
- venous thromboembolic events (including pulmonary embolism and deep venous thrombosis)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
2.4 Administration Information
Administer LYBALVI orally once daily with or without food as a single tablet.
Do not divide tablets or combine strengths.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.5 Neuroleptic Malignant Syndrome
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with administration of antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, delirium, and autonomic instability. Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue LYBALVI and provide intensive symptomatic treatment and monitoring.
5.15 Body Temperature Dysregulation
Atypical antipsychotics may disrupt the body's ability to reduce core body temperature. Strenuous exercise, exposure to extreme heat, dehydration, and anticholinergic medications may contribute to an elevation in core body temperature; use LYBALVI with caution in patients who may experience these conditions.
7.1 Effects of Other Drugs On Lybalvi (7.1 Effects of Other Drugs on LYBALVI)
Table 4 describes clinically significant drug interactions where the concomitant use of other drugs affects LYBALVI.
| Strong CYP3A4 Inducer | |
| Clinical Implication: | Coadministration of LYBALVI with a strong CYP3A4 inducer decreases AUCinf of olanzapine and samidorphan [see Clinical Pharmacology (12.3)], which may reduce LYBALVI efficacy. |
| Prevention or Management: | Concomitant use of LYBALVI with strong CYP3A4 inducers is not recommended. |
| Strong CYP1A2 Inhibitor | |
| Clinical Implication: | Concomitant use of LYBALVI with a strong CYP1A2 inhibitor increases olanzapine AUC and Cmax [see Clinical Pharmacology (12.3)], which may increase the risk of LYBALVI adverse reactions. |
| Prevention or Management: | Consider reducing the dosage of the olanzapine component in LYBALVI when used concomitantly with strong CYP1A2 inhibitors. |
| CYP1A2 Inducer | |
| Clinical Implication: | Concomitant use of LYBALVI with CYP1A2 inducers decreases olanzapine exposure [see Clinical Pharmacology (12.3)], which may reduce LYBALVI efficacy. |
| Prevention or Management: | Consider increasing the dosage of the olanzapine component in LYBALVI when used concomitantly with CYP1A2 inducers. |
| Diazepam, Alcohol, and Other CNS Acting Drugs | |
| Clinical Implication: | Concomitant use of diazepam, alcohol, or other CNS acting drugs with LYBALVI may potentiate the orthostatic hypotension observed with olanzapine [see Warnings and Precautions (5.9)]. |
| Prevention or Management: | LYBALVI should be used with caution in patients receiving concomitantly diazepam or other CNS acting drugs, or using alcohol. |
| Anticholinergic Drugs | |
| Clinical Implication: | Concomitant treatment with olanzapine and other drugs with anticholinergic activity can increase the risk for severe gastrointestinal adverse reactions related to hypomotility. |
| Prevention or Management: | LYBALVI should be used with caution in patients receiving medications having anticholinergic (antimuscarinic) effects [see Warnings and Precautions (5.16)]. |
7.2 Effects of Lybalvi On Other Drugs (7.2 Effects of LYBALVI on Other Drugs)
Table 5 describes clinically significant drug interactions where concomitant use of LYBALVI affects other drugs.
| Antihypertensive Agents | |
| Clinical Implication: | LYBALVI may enhance the effects of certain antihypertensive agents. |
| Prevention or Management: | Monitor blood pressure and reduce dosage of antihypertensive drug in accordance with its approved product labeling. |
| Levodopa and Dopamine Agonists | |
| Clinical Implication: | LYBALVI may antagonize the effects of levodopa and dopamine agonists. |
| Prevention or Management: | Concomitant use of LYBALVI is not recommended with levodopa and dopamine agonists. |
7.4 Interference With Laboratory Tests (7.4 Interference with Laboratory Tests)
Interference with Laboratory Tests for Opioid Detection
Because LYBALVI contains samidorphan, an opioid antagonist, LYBALVI may be cross-reactive with urinary immunoassay methods used for detecting opioids, resulting in false positive results. Use an alternative analytical technique (e.g., chromatographic methods) to confirm positive opioid urine drug screen results [see Contraindications (4) and Warnings and Precautions (5.19)].
2.2 Recommended Dosage in Schizophrenia
Initiate LYBALVI at 5 mg/10 mg (contains 5 mg of olanzapine and 10 mg of samidorphan) or 10 mg/10 mg (contains 10 mg of olanzapine and 10 mg of samidorphan) orally once daily. The recommended dosage is 10 mg/10 mg, 15 mg/10 mg (contains 15 mg of olanzapine and 10 mg of samidorphan), or 20 mg/10 mg (contains 20 mg of olanzapine and 10 mg of samidorphan) once daily.
Dosage may be adjusted at weekly intervals of 5 mg (based on the olanzapine component of LYBALVI) depending upon clinical response and tolerability, up to the maximum recommended dosage of 20 mg/10 mg once daily.
5.9 Orthostatic Hypotension and Syncope
Atypical antipsychotics cause orthostatic hypotension and syncope. Generally, the risk is greatest during initial dose titration and when increasing the dose. In the 4-week, placebo-controlled study, from analysis of the vital signs data, rates of orthostatic hypotension were less than 2% in LYBALVI- and placebo-, and olanzapine-treated patients. In the 24-week, olanzapine-controlled study, from analysis of the vital signs data, rates of orthostatic hypotension in LYBALVI-treated patients were 3.7%, compared to 0.4% in olanzapine-treated patients.
Monitor orthostatic vital signs in patients who are vulnerable to hypotension (e.g., elderly patients, patients with dehydration, hypovolemia, concomitant treatment with antihypertensive medications or CNS depressants [see Drug Interactions (7.1, 7.2)], patients with known cardiovascular disease (history of myocardial infarction, ischemic heart disease, heart failure, or conduction abnormalities), and patients with cerebrovascular disease. LYBALVI has not been evaluated in patients with a recent history of myocardial infarction or unstable cardiovascular disease. Such patients were excluded from the premarketing clinical trials.
5.16 Anticholinergic (antimuscarinic) Effects (5.16 Anticholinergic (Antimuscarinic) Effects)
Olanzapine, a component of LYBALVI, exhibits in vitro muscarinic receptor affinity [see Clinical Pharmacology (12.2)]. In premarketing clinical trials with oral olanzapine, olanzapine was associated with constipation, dry mouth, and tachycardia, all adverse reactions possibly related to cholinergic antagonism. Such adverse reactions were not often the basis for discontinuations, but LYBALVI should be used with caution in patients with a current diagnosis or prior history of urinary retention, clinically significant prostatic hypertrophy, constipation, or a history of paralytic ileus or related conditions. In postmarketing experience, the risk for severe adverse reactions (including fatalities) was increased with concomitant use of anticholinergic medications [see Drug Interactions (7.1)].
2.1 Lybalvi Initiation in Patients Using Opioids (2.1 LYBALVI Initiation In Patients Using Opioids)
LYBALVI is contraindicated in patients using opioids or undergoing acute opioid withdrawal.
In patients who use opioids, delay initiation of LYBALVI for a minimum of 7 days after last use of short-acting opioids and 14 days after last use of long-acting opioids [see Warnings and Precautions ( 5.3)].
5.11 Leukopenia, Neutropenia, and Agranulocytosis
Leukopenia and neutropenia have been reported during treatment with antipsychotic agents, including LYBALVI [see Adverse Reactions (6)]. Agranulocytosis (including fatal cases) has been reported with other agents in this class.
Possible risk factors for leukopenia and neutropenia include pre-existing low white blood cell count (WBC) or absolute neutrophil count (ANC) and history of drug-induced leukopenia or neutropenia. In patients with a pre-existing low WBC or ANC or a history of drug-induced leukopenia or neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of LYBALVI at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue LYBALVI in patients with severe neutropenia (absolute neutrophil count <1000/mm3) and follow their WBC until recovery.
5.14 Potential for Cognitive and Motor Impairment
LYBALVI, like other antipsychotics, may cause somnolence and has the potential to impair judgment, thinking, or motor skills. In a LYBALVI placebo-controlled study, somnolence occurred in 9% of LYBALVI-treated patients compared to 2.2% in patients treated with placebo.
Patients should be cautioned about operating hazardous machinery, including motor vehicles, until they are reasonably certain that LYBALVI therapy does not affect them adversely.
2.5 Dosage Recommendations in Specific Populations
The recommended starting dosage of LYBALVI is 5 mg/10 mg once daily in patients who have a higher risk of hypotensive reactions, are at risk of slower olanzapine metabolism, or may be more pharmacodynamically sensitive to olanzapine [see Warnings and Precautions (5.9), Drug Interactions (7.2), Use in Specific Populations (8.5), and Clinical Pharmacology (12.3)]. If dose escalation is necessary, increase the dosage slowly in these patients.
5.6 Drug Reaction With Eosinophilia and Systemic Symptoms (5.6 Drug Reaction with Eosinophilia and Systemic Symptoms)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported with exposure to olanzapine, a component of LYBALVI [see Adverse Reactions (6.2)]. DRESS may present with a cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, and/or lymphadenopathy with systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and/or pericarditis. DRESS is sometimes fatal. Discontinue LYBALVI if DRESS is suspected.
5.19 Interference With Laboratory Tests for Opioid Detection (5.19 Interference with Laboratory Tests for Opioid Detection)
5.18 Risks Associated With Combination Treatment With Lithium Or Valproate (5.18 Risks Associated with Combination Treatment with Lithium or Valproate)
If LYBALVI is administered with lithium or valproate, refer to the lithium or valproate Prescribing Information for a description of the risks for these products including, but not limited to, the warnings and precautions for lithium or valproate [see Contraindications (4)].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis (5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. In placebo-controlled clinical trials of elderly patients with dementia-related psychosis, the incidence of death in olanzapine-treated patients was significantly greater than in placebo-treated patients (3.5% vs 1.5%, respectively). Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in the drug-treated patients of between 1.6 to 1.7 times that seen in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.2)].
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis (WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
5.3 Precipitation of Severe Opioid Withdrawal in Patients Who Are Physiologically Dependent On Opioids (5.3 Precipitation of Severe Opioid Withdrawal in Patients Who Are Physiologically Dependent on Opioids)
Samidorphan, an opioid antagonist that is a component of LYBALVI, can precipitate opioid withdrawal in patients who are dependent on opioids, which can lead to an opioid withdrawal syndrome, sometimes requiring hospitalization. Therefore, LYBALVI is contraindicated in patients who are using opioids or undergoing acute opioid withdrawal. Prior to initiating LYBALVI, there should be at least a 7-day opioid-free interval from last use of short-acting opioids, and at least a 14-day opioid-free interval from the last use of long-acting opioids. Explain the risks associated with precipitated withdrawal and the importance of giving an accurate account of last opioid use to patients and caregivers [see Dosage and Administration (2.1), Drug Interactions (7.3)].
5.2 Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients With Dementia Related Psychosis (5.2 Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis)
Cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack), including fatalities, were reported in patients in trials of olanzapine in elderly patients with dementia-related psychosis. In placebo-controlled trials, there was a significantly higher incidence of cerebrovascular adverse reactions in patients treated with olanzapine compared to patients treated with placebo. LYBALVI is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:52.035901 · Updated: 2026-03-14T22:50:04.032953