Lisinopril Tablets, Usp
32eb18bc-a80d-4de1-8f38-e591d8f66870
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: FETAL TOXICITY See full prescribing information for complete boxed warning. • When pregnancy is detected, discontinue lisinopril as soon as possible. • Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity.
Contraindications
Lisinopril is contraindicated in patients who are hypersensitive to this product and in patients with a history of angioedema related to previous treatment with an angiotensin-converting enzyme inhibitor and in patients with hereditary or idiopathic angioedema.
Adverse Reactions
Lisinopril has been found to be generally well tolerated in controlled clinical trials involving 1969 patients with hypertension or heart failure. For the most part, adverse experiences were mild and transient.
How Supplied
2.5 mg Tablets: White, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “417” on the other side are supplied in bottle of 30 tablets (NDC 76282-417-30), bottle of 45 tablets (NDC 76282-417-45) bottle of 90 tablets (NDC 76282-417-90) bottle of 100 tablets (NDC 76282-417-01), bottle of 180 tablets (NDC 76282-417-18), bottle of 500 tablets (NDC 76282-417-05) and 1000 tablets (NDC 76282-417-10). 5 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘I’ on the left side of functional score and ‘G’ on the right side of functional score, and “418” on the other side are supplied in bottles of 30 tablets (NDC 76282-418-30), bottle of 45 tablets (NDC 76282-418-45) bottle of 90 tablets (NDC 76282-418-90) bottle of 100 tablets (NDC 76282-418-01), bottle of 180 tablets (NDC 76282-418-18), bottle of 500 tablets (NDC 76282-418-05) and 1000 tablets (NDC 76282-418-10). 10 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “419” on the other side are supplied in bottles of 30 tablets (NDC 76282-419-30), bottle of 45 tablets (NDC 76282-419-45) bottle of 90 tablets (NDC 76282-419-90) bottle of 100 tablets (NDC 76282-419-01) bottle of 180 tablets (NDC 76282-419-18), bottle of 500 tablets (NDC 76282-419-05), and 1000 tablets (NDC 76282-419-10). 20 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “420” on the other side are supplied in bottles of 30 tablets (NDC 76282-420-30), bottle of 45 tablets (NDC 76282-420-45) bottle of 90 tablets (NDC 76282-420-90) bottle of 100 tablets (NDC 76282-420-01), bottle of 180 tablets (NDC 76282-420-18), bottle of 500 tablets (NDC 76282-420-05) and 1000 tablets (NDC 76282-420-10). 30 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “421” on the other side are supplied in bottles of 30 tablets (NDC 76282-421-30), bottle of 45 tablets (NDC 76282-421-45) bottle of 90 tablets (NDC 76282-421-90) bottle of 100 tablets (NDC 76282-421-01), bottle of 180 tablets (NDC 76282-421-18), bottle of 500 tablets (NDC 76282-421-05) and 1000 tablets (NDC 76282-421-10). 40 mg Tablets: Yellow, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “422” on the other side are supplied in bottles of 30 tablets (NDC 76282-422-30), bottle of 45 tablets (NDC 76282-422-45) bottle of 90 tablets (NDC 76282-422-90) bottle of 100 tablets (NDC 76282-422-01), bottle of 180 tablets (NDC 76282-422-18), bottle of 500 tablets (NDC 76282-422-05) and 1000 tablets (NDC 76282-422-10). Storage Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature]. Protect from moisture, freezing and excessive heat. Dispense in a tight container. *AN69 is a registered trademark of Hospal Ltd All brand names listed are the registered trademarks of their respective owners and are not trademarks of Exelan Pharmaceuticals, Inc. Manufactured for: Exelan Pharmaceuticals, Inc. Lawrenceville, GA 30046 Manufactured by: InvaGen Pharmaceuticals, Inc. Hauppauge, NY 11788 Rev: 11/13 Barcode: 422-11-2013
Medication Information
Recent Major Changes
WARNING: FETAL TOXICITY
See full prescribing information for complete boxed warning.
-
•When pregnancy is detected, discontinue lisinopril as soon as possible.
-
•Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity.
Contraindications
Lisinopril is contraindicated in patients who are hypersensitive to this product and in patients with a history of angioedema related to previous treatment with an angiotensin-converting enzyme inhibitor and in patients with hereditary or idiopathic angioedema.
Adverse Reactions
Lisinopril has been found to be generally well tolerated in controlled clinical trials involving 1969 patients with hypertension or heart failure. For the most part, adverse experiences were mild and transient.
How Supplied
2.5 mg Tablets: White, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “417” on the other side are supplied in bottle of 30 tablets (NDC 76282-417-30), bottle of 45 tablets (NDC 76282-417-45) bottle of 90 tablets (NDC 76282-417-90) bottle of 100 tablets (NDC 76282-417-01), bottle of 180 tablets (NDC 76282-417-18), bottle of 500 tablets (NDC 76282-417-05) and 1000 tablets (NDC 76282-417-10).
5 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘I’ on the left side of functional score and ‘G’ on the right side of functional score, and “418” on the other side are supplied in bottles of 30 tablets (NDC 76282-418-30), bottle of 45 tablets (NDC 76282-418-45) bottle of 90 tablets (NDC 76282-418-90) bottle of 100 tablets (NDC 76282-418-01), bottle of 180 tablets (NDC 76282-418-18), bottle of 500 tablets (NDC 76282-418-05) and 1000 tablets (NDC 76282-418-10).
10 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “419” on the other side are supplied in bottles of 30 tablets (NDC 76282-419-30), bottle of 45 tablets (NDC 76282-419-45) bottle of 90 tablets (NDC 76282-419-90) bottle of 100 tablets (NDC 76282-419-01) bottle of 180 tablets (NDC 76282-419-18), bottle of 500 tablets (NDC 76282-419-05), and 1000 tablets (NDC 76282-419-10).
20 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “420” on the other side are supplied in bottles of 30 tablets (NDC 76282-420-30), bottle of 45 tablets (NDC 76282-420-45) bottle of 90 tablets (NDC 76282-420-90) bottle of 100 tablets (NDC 76282-420-01), bottle of 180 tablets (NDC 76282-420-18), bottle of 500 tablets (NDC 76282-420-05) and 1000 tablets (NDC 76282-420-10).
30 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “421” on the other side are supplied in bottles of 30 tablets (NDC 76282-421-30), bottle of 45 tablets (NDC 76282-421-45) bottle of 90 tablets (NDC 76282-421-90) bottle of 100 tablets (NDC 76282-421-01), bottle of 180 tablets (NDC 76282-421-18), bottle of 500 tablets (NDC 76282-421-05) and 1000 tablets (NDC 76282-421-10).
40 mg Tablets: Yellow, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “422” on the other side are supplied in bottles of 30 tablets (NDC 76282-422-30), bottle of 45 tablets (NDC 76282-422-45) bottle of 90 tablets (NDC 76282-422-90) bottle of 100 tablets (NDC 76282-422-01), bottle of 180 tablets (NDC 76282-422-18), bottle of 500 tablets (NDC 76282-422-05) and 1000 tablets (NDC 76282-422-10).
Storage
Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature]. Protect from moisture, freezing and excessive heat. Dispense in a tight container.
*AN69 is a registered trademark of Hospal Ltd
All brand names listed are the registered trademarks of their respective owners and are not trademarks of Exelan Pharmaceuticals, Inc.
Manufactured for:
Exelan Pharmaceuticals, Inc.
Lawrenceville, GA 30046
Manufactured by:
InvaGen Pharmaceuticals, Inc.
Hauppauge, NY 11788
Rev: 11/13
Barcode: 422-11-2013
Description
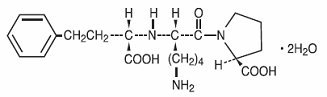
Lisinopril, USP is an oral long-acting angiotensin converting enzyme inhibitor. Lisinopril, USP a synthetic peptide derivative, is chemically described as (S)-1-[N2-(1-carboxy-3-phenylpropyl)-L-lysyl]-L- proline dihydrate. Its molecular formula is C21H31N3O5 • 2H2O and its structural formula is:
Lisinopril, USP is a white to off-white, crystalline powder, with a molecular weight of 441.53. It is soluble in water and sparingly soluble in methanol and practically insoluble in ethanol.
Lisinopril tablets, USP are supplied as 2.5 mg, 5 mg, 10 mg, 20 mg, 30 mg and 40 mg tablets for oral administration.
Inactive Ingredients:
2.5 mg tablets – lactose monohydrate, maize starch, colloidal silicon dioxide, magnesium stearate.
5 mg, 10 mg, 20 mg and 30 mg tablets – lactose monohydrate, maize starch, colloidal silicon dioxide, magnesium stearate, iron oxide red.
40 mg tablets - lactose monohydrate, maize starch, colloidal silicon dioxide, magnesium stearate, iron oxide yellow.
Section 42229-5
Adult Patients: Administration of lisinopril to patients with hypertension results in a reduction of both supine and standing blood pressure to about the same extent with no compensatory tachycardia. Symptomatic postural hypotension is usually not observed although it can occur and should be anticipated in volume and/or salt-depleted patients (See WARNINGS ). When given together with thiazide-type diuretics, the blood pressure lowering effects of the two drugs are approximately additive.
In most patients studied, onset of antihypertensive activity was seen at one hour after oral administration of an individual dose of lisinopril, with peak reduction of blood pressure achieved by 6 hours. Although an antihypertensive effect was observed 24 hours after dosing with recommended single daily doses, the effect was more consistent and the mean effect was considerably larger in some studies with doses of 20 mg or more than with lower doses; however, at all doses studied, the mean antihypertensive effect was substantially smaller 24 hours after dosing than it was 6 hours after dosing.
In some patients achievement of optimal blood pressure reduction may require two to four weeks of therapy.
The antihypertensive effects of lisinopril are maintained during long-term therapy. Abrupt withdrawal of lisinopril has not been associated with a rapid increase in blood pressure, or a significant increase in blood pressure compared to pretreatment levels.
Two dose-response studies utilizing a once-daily regimen were conducted in 438 mild to moderate hypertensive patients not on a diuretic. Blood pressure was measured 24 hours after dosing. An antihypertensive effect of lisinopril was seen with 5 mg in some patients. However, in both studies blood pressure reduction occurred sooner and was greater in patients treated with 10 mg, 20 mg or 80 mg of lisinopril. In controlled clinical studies, lisinopril 20 to 80 mg has been compared in patients with mild to moderate hypertension to hydrochlorothiazide 12.5 to 50 mg and with atenolol 50 to 200 mg; and in patients with moderate to severe hypertension to metoprolol 100 to 200 mg. It was superior to hydrochlorothiazide in effects on systolic and diastolic pressure in a population that was 3/4 Caucasian. Lisinopril was approximately equivalent to atenolol and metoprolol in effects on diastolic blood pressure, and had somewhat greater effects on systolic blood pressure.
Lisinopril had similar effectiveness and adverse effects in younger and older (> 65 years) patients. It was less effective in Blacks than in Caucasians.
In hemodynamic studies in patients with essential hypertension, blood pressure reduction was accompanied by a reduction in peripheral arterial resistance with little or no change in cardiac output and in heart rate. In a study in nine hypertensive patients, following administration of lisinopril, there was an increase in mean renal blood flow that was not significant. Data from several small studies are inconsistent with respect to the effect of lisinopril on glomerular filtration rate in hypertensive patients with normal renal function, but suggest that changes, if any, are not large.
In patients with renovascular hypertension lisinopril has been shown to be well tolerated and effective in controlling blood pressure (See PRECAUTIONS ).
Overdosage
Following a single oral dose of 20 g/kg no lethality occurred in rats, and death occurred in one of 20 mice receiving the same dose. The most likely manifestation of overdosage would be hypotension, for which the usual treatment would be intravenous infusion of normal saline solution.
Lisinopril can be removed by hemodialysis (See WARNINGS, Anaphylactoid Reactions During Membrane Exposure).
Hypertension
In clinical trials in patients with hypertension treated with lisinopril, discontinuation of therapy due to clinical adverse experiences occurred in 5.7% of patients. The overall frequency of adverse experiences could not be related to total daily dosage within the recommended therapeutic dosage range.
For adverse experiences occurring in greater than 1% of patients with hypertension treated with lisinopril or lisinopril plus hydrochlorothiazide in controlled clinical trials, and more frequently with lisinopril and/or lisinopril plus hydrochlorothiazide than placebo, comparative incidence data are listed in the table below:
|
LISINOPRIL (n=1349) Incidence (discontinuation) |
LISINOPRIL/ Hydrochlorothiazide (n=629) Incidence (discontinuation) |
PLACEBO (n=207) Incidence (discontinuation) |
|
|
Body as a Whole |
|||
|
Fatigue |
2.5 (0.3) |
4.0 (0.5) |
1.0 (0.0) |
|
Asthenia |
1.3 (0.5) |
2.1 (0.2) |
1.0 (0.0) |
|
Orthostatic Effects |
1.2 (0.0) |
3.5 (0.2) |
1.0 (0.0) |
|
Cardiovascular |
|||
|
Hypotension |
1.2 (0.5) |
1.6 (0.5) |
0.5 (0.5) |
|
Digestive |
|||
|
Diarrhea |
2.7 (0.2) |
2.7 (0.3) |
2.4 (0.0) |
|
Nausea |
2.0 (0.4) |
2.5 (0.2) |
2.4 (0.0) |
|
Vomiting |
1.1 (0.2) |
1.4 (0.1) |
0.5 (0.0) |
|
Dyspepsia |
0.9 (0.0) |
1.9 (0.0) |
0.0 (0.0) |
|
Musculoskeletal |
|||
|
Muscle Cramps |
0.5 (0.0) |
2.9 (0.8) |
0.5 (0.0) |
|
Nervous/Psychiatric |
|||
|
Headache |
5.7 (0.2) |
4.5 (0.5) |
1.9 (0.0) |
|
Dizziness |
5.4 (0.4) |
9.2 (1.0) |
1.9 (0.0) |
|
Paresthesia |
0.8 (0.1) |
2.1 (0.2) |
0.0 (0.0) |
|
Decreased Libido |
0.4 (0.1) |
1.3 (0.1) |
0.0 (0.0) |
|
Vertigo |
0.2 (0.1) |
1.1 (0.2) |
0.0 (0.0) |
|
Respiratory |
|||
|
Cough |
3.5 (0.7) |
4.6 (0.8) |
1.0 (0.0) |
|
Upper Respiratory Infection |
2.1 (0.1) |
2.7 (0.1) |
0.0 (0.0) |
|
Common Cold |
1.1 (0.1) |
1.3 (0.1) |
0.0 (0.0) |
|
Nasal Congestion |
0.4 (0.1) |
1.3 (0.1) |
0.0 (0.0) |
|
Influenza |
0.3 (0.1) |
1.1 (0.1) |
0.0 (0.0) |
|
Skin |
|||
|
Rash |
1.3 (0.4) |
1.6 (0.2) |
0.5 (0.5) |
|
Urogenital |
|||
|
Impotence |
1.0 (0.4) |
1.6 (0.5) |
0.0 (0.0) |
Chest pain and back pain were also seen, but were more common on placebo than lisinopril.
Geriatric Use
Clinical studies of lisinopril in patients with hypertension did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other clinical experience in this population has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
In the ATLAS trial of lisinopril in patients with congestive heart failure, 1,596 (50%) were 65 and over, while 437 (14%) were 75 and over. In a clinical study of lisinopril in patients with myocardial infarctions 4,413 (47%) were 65 and over, while 1,656 (18%) were 75 and over. In these studies, no overall differences in safety or effectiveness were observed between elderly and younger patients, and other reported clinical experiences has not identified differences in responses between the elderly and younger patients (See CLINICAL PHARMACOLOGY, Pharmacodynamics and Clinical Effects, Heart Failure and CLINICAL PHARMACOLOGY, Pharmacodynamics and Clinical Effects, Acute Myocardial Infarction).
Other reported clinical experience has not identified differences in responses between elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Pharmacokinetic studies indicate that maximum blood levels and area under the plasma concentration time curve (AUC) are doubled in older patients (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism).
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection. Evaluation of patients with hypertension, congestive heart failure, or myocardial infarction should always include assessment of renal function (See DOSAGE AND ADMINISTRATION ).
Heart Failure
Lisinopril tablets, USP are indicated as adjunctive therapy in the management of heart failure in patients who are not responding adequately to diuretics and digitalis.
Hypotension
Excessive hypotension is rare in patients with uncomplicated hypertension treated with lisinopril alone.
Patients with heart failure given lisinopril commonly have some reduction in blood pressure, with peak blood pressure reduction occurring 6 to 8 hours post dose. Evidence from the two-dose ATLAS trial suggested that incidence of hypotension may increase with dose of lisinopril in heart failure patients. Discontinuation of therapy because of continuing symptomatic hypotension usually is not necessary when dosing instructions are followed; caution should be observed when initiating therapy (See DOSAGE AND ADMINISTRATION).
Patients at risk of excessive hypotension, sometimes associated with oliguria and/or progressive azotemia, and rarely with acute renal failure and/or death, include those with the following conditions or characteristics: heart failure with systolic blood pressure below 100 mmHg, hyponatremia, high dose diuretic therapy, recent intensive diuresis or increase in diuretic dose, renal dialysis, or severe volume and/or salt depletion of any etiology. It may be advisable to eliminate the diuretic (except in patients with heart failure), reduce the diuretic dose or increase salt intake cautiously before initiating therapy with lisinopril in patients at risk for excessive hypotension who are able to tolerate such adjustments (See PRECAUTIONS, Drug Interactions and ADVERSE REACTIONS ).
Patients with acute myocardial infarction in the GISSI-3 trial had a higher (9.0% versus 3.7%) incidence of persistent hypotension (systolic blood pressure <90 mmHg for more than 1 hour) when treated with lisinopril. Treatment with lisinopril must not be initiated in acute myocardial infarction patients at risk of further serious hemodynamic deterioration after treatment with a vasodilator (e.g., systolic blood pressure of 100 mmHg or lower) or cardiogenic shock.
In patients at risk of excessive hypotension, therapy should be started under very close medical supervision and such patients should be followed closely for the first two weeks of treatment and whenever the dose of lisinopril and/or diuretic is increased. Similar considerations may apply to patients with ischemic heart or cerebrovascular disease, or in patients with acute myocardial infarction, in whom an excessive fall in blood pressure could result in a myocardial infarction or cerebrovascular accident.
If excessive hypotension occurs, the patient should be placed in the supine position and, if necessary, receive an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further doses of lisinopril which usually can be given without difficulty once the blood pressure has stabilized. If symptomatic hypotension develops, a dose reduction or discontinuation of lisinopril or concomitant diuretic may be necessary.
Pediatric Use
Neonates with a history of in utero exposure to lisinopril:
If oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and/or substituting for disordered renal function. Lisinopril, which crosses the placenta, has been removed from neonatal circulation by peritoneal dialysis with some clinical benefit, and theoretically may be removed by exchange transfusion, although there is no experience with the latter procedure.
Antihypertensive effects of lisinopril have been established in hypertensive pediatric patients aged 6 to 16 years.
There are no data on the effect of lisinopril on blood pressure in pediatric patients under the age 6 or in pediatric patients with glomerular filtration rate < 30 mL/min/1.73 m2. (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism and Pharmacodynamics and Clinical Effects, and DOSAGE AND ADMINISTRATION ).
Hypertension
Lisinopril Tablets, USP are indicated for the treatment of hypertension to lower blood pressure. Lowering blood pressure lowers the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including lisinopril.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than 1 drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (eg, on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Lisinopril may be administered alone or with other antihypertensive agents.
Use in Elderly
In general, the clinical response was similar in younger and older patients given similar doses of lisinopril. Pharmacokinetic studies, however indicate that maximum blood levels and area under the plasma concentration time curve (AUC) are doubled in older patients, so that dosage adjustments should be made with particular caution.
Hepatic Failure
Rarely, ACE inhibitors have been associated with a syndrome that starts with cholestatic jaundice or hepatitis and progresses to fulminant hepatic necrosis and (sometimes) death. The mechanism of this syndrome is not understood. Patients receiving ACE inhibitors who develop jaundice or marked elevations of hepatic enzymes should discontinue the ACE inhibitor and receive appropriate medical follow-up.
Nursing Mothers
Milk of lactating rats contains radioactivity following administration of 14C lisinopril. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from ACE inhibitors, a decision should be made whether to discontinue nursing or discontinue lisinopril, taking into account the importance of the drug to the mother.
Mechanism of Action
Lisinopril inhibits angiotensin-converting enzyme (ACE) in human subjects and animals. ACE is a peptidyl dipeptidase that catalyzes the conversion of angiotensin I to the vasoconstrictor substance, angiotensin II. Angiotensin II also stimulates aldosterone secretion by the adrenal cortex. The beneficial effects of lisinopril in hypertension and heart failure appear to result primarily from suppression of the renin-angiotensin-aldosterone system. Inhibition of ACE results in decreased plasma angiotensin II which leads to decreased vasopressor activity and to decreased aldosterone secretion. The latter decrease may result in a small increase of serum potassium. In hypertensive patients with normal renal function treated with lisinopril alone for up to 24 weeks, the mean increase in serum potassium was approximately 0.1 mEq/L; however, approximately 15% of patients had increases greater than 0.5 mEq/L and approximately 6% had a decrease greater than 0.5 mEq/L. In the same study, patients treated with lisinopril and hydrochlorothiazide for up to 24 weeks had a mean decrease in serum potassium of 0.1 mEq/L; approximately 4% of patients had increases greater than 0.5 mEq/L and approximately 12% had a decrease greater than 0.5 mEq/L (See PRECAUTIONS ). Removal of angiotensin II negative feedback on renin secretion leads to increased plasma renin activity.
ACE is identical to kininase, an enzyme that degrades bradykinin. Whether increased levels of bradykinin, a potent vasodepressor peptide, play a role in the therapeutic effects of lisinopril remains to be elucidated.
While the mechanism through which lisinopril lowers blood pressure is believed to be primarily suppression of the renin-angiotensin-aldosterone system, lisinopril is antihypertensive even in patients with low-renin hypertension. Although lisinopril was antihypertensive in all races studied, Black hypertensive patients (usually a low-renin hypertensive population) had a smaller average response to monotherapy than non-Black patients.
Concomitant administration of lisinopril and hydrochlorothiazide further reduced blood pressure in Black and non-Black patients and any racial differences in blood pressure response were no longer evident.
Pregnancy Category D
Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue lisinopril as soon as possible. These adverse outcomes are usually associated with use of these drugs in the second and third trimester of pregnancy. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. Appropriate management of maternal hypertension during pregnancy is important to optimize outcomes for both mother and fetus.
In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus. Perform serial ultrasound examinations to assess the intra-amniotic environment. If oligohydramnios is observed, discontinue lisinopril, unless it is considered lifesaving for the mother. Fetal testing may be appropriate, based on the week of pregnancy. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to lisinopril for hypotension, oliguria, and hyperkalemia (see Precautions, Pediatric Use).
No teratogenic effects of lisinopril were seen in studies of pregnant rats, mice, and rabbits. On a mg/kg basis, the doses used were up to 625 times (in mice), 188 times (in rats), and 0.6 times (in rabbits) the maximum recommended human dose.
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 2.5mg |
| 90 | 43353-270-90 |
| Count | 5mg |
| 30 | 43353-298-30 |
| 45 | 43353-298-45 |
| Count | 10mg |
| 30 | 43353-301-30 |
| 45 | 43353-301-45 |
| 90 | 43353-301-60 |
| Count | 20mg |
| 30 | 43353-271-30 |
| 45 | 43353-271-45 |
| 90 | 43353-271-60 |
| 180 | 43353-271-80 |
| Count | 40mg |
| 30 | 43353-009-30 |
| 45 | 43353-009-45 |
| 90 | 43353-009-60 |
| 180 | 43353-009-80 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20170629JH
Acute Myocardial Infarction
Lisinopril tablets, USP are indicated for the treatment of hemodynamically stable patients within 24 hours of acute myocardial infarction, to improve survival. Patients should receive, as appropriate, the standard recommended treatments such as thrombolytics, aspirin and beta-blockers.
In using lisinopril, consideration should be given to the fact that another angiotensin-converting enzyme inhibitor, captopril, has caused agranulocytosis, particularly in patients with renal impairment or collagen vascular disease, and that available data are insufficient to show that lisinopril does not have a similar risk (See WARNINGS ).
In considering the use of lisinopril, it should be noted that in controlled clinical trials ACE inhibitors have an effect on blood pressure that is less in Black patients than in non-Blacks. In addition, ACE inhibitors have been associated with a higher rate of angioedema in Black than in non-Black patients (See WARNINGS, Anaphylactoid and Possibly Related Reactions ).
Package/label Display Panel
NDC 43353-270-90
Lisinopril Tablets, USP
2.5 mg
Rx Only 90 Tablets
Pharmacokinetics and Metabolism
Adult Patients: Following oral administration of lisinopril, peak serum concentrations of lisinopril occur within about 7 hours, although there was a trend to a small delay in time taken to reach peak serum concentrations in acute myocardial infarction patients. Declining serum concentrations exhibit a prolonged terminal phase which does not contribute to drug accumulation. This terminal phase probably represents saturable binding to ACE and is not proportional to dose.
Lisinopril does not appear to be bound to other serum proteins. Lisinopril does not undergo metabolism and is excreted unchanged entirely in the urine. Based on urinary recovery, the mean extent of absorption of lisinopril is approximately 25%, with large intersubject variability (6% to 60%) at all doses tested (5 mg to 80 mg). Lisinopril absorption is not influenced by the presence of food in the gastrointestinal tract. The absolute bioavailability of lisinopril is reduced to 16% in patients with stable NYHA Class II-IV congestive heart failure, and the volume of distribution appears to be slightly smaller than that in normal subjects. The oral bioavailability of lisinopril in patients with acute myocardial infarction is similar to that in healthy volunteers.
Upon multiple dosing, lisinopril exhibits an effective half-life of accumulation of 12 hours.
Impaired renal function decreases elimination of lisinopril, which is excreted principally through the kidneys, but this decrease becomes clinically important only when the glomerular filtration rate is below 30 mL/min. Above this glomerular filtration rate, the elimination half-life is little changed. With greater impairment, however, peak and trough lisinopril levels increase, time to peak concentration increases and time to attain steady state is prolonged. Older patients, on average, have (approximately doubled) higher blood levels and area under the plasma concentration time curve (AUC) than younger patients (See DOSAGE AND ADMINISTRATION). Lisinopril can be removed by hemodialysis.
Studies in rats indicate that lisinopril crosses the blood-brain barrier poorly. Multiple doses of lisinopril in rats do not result in accumulation in any tissues. Milk of lactating rats contains radioactivity following administration of 14C lisinopril. By whole body autoradiography, radioactivity was found in the placenta following administration of labeled drug to pregnant rats, but none was found in the fetuses.
Pediatric Patients: The pharmacokinetics of lisinopril were studied in 29 pediatric hypertensive patients between 6 years and 16 years with glomerular filtration rate > 30 mL/min/1.73 m2. After doses of 0.1 to 0.2 mg/kg, steady state peak plasma concentrations of lisinopril occurred within 6 hours and the extent of absorption based on urinary recovery was about 28%. These values are similar to those obtained previously in adults. The typical value of lisinopril oral clearance (systemic clearance/absolute bioavailability) in a child weighing 30 kg is 10 L/h, which increases in proportion to renal function.
Leukopenia/neutropenia/agranulocytosis
Another angiotensin-converting enzyme inhibitor, captopril, has been shown to cause agranulocytosis and bone marrow depression, rarely in uncomplicated patients but more frequently in patients with renal impairment especially if they also have a collagen vascular disease. Available data from clinical trials of lisinopril are insufficient to show that lisinopril does not cause agranulocytosis at similar rates. Marketing experience has revealed rare cases of leukopenia/neutropenia and bone marrow depression in which a causal relationship to lisinopril cannot be excluded. Periodic monitoring of white blood cell counts in patients with collagen vascular disease and renal disease should be considered.
Anaphylactoid and Possibly Related Reactions
Presumably because angiotensin-converting enzyme inhibitors affect the metabolism of eicosanoids and polypeptides, including endogenous bradykinin, patients receiving ACE inhibitors (including lisinopril) may be subject to a variety of adverse reactions, some of them serious.
Pediatric Hypertensive Patients ≥ 6 Years of Age
The usual recommended starting dose is 0.07 mg/kg once daily (up to 5 mg total). Dosage should be adjusted according to blood pressure response. Doses above 0.61 mg/kg (or in excess of 40 mg) have not been studied in pediatric patients (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism and Pharmacodynamics and Clinical Effects).
Lisinopril is not recommended in pediatric patients < 6 years or in pediatric patients with glomerular filtration rate < 30 mL/min/1.73 m2 (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism and Pharmacodynamics and Clinical Effects and PRECAUTIONS).
Dual Blockade of the Renin Angiotensin System (ras)
Dual blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Closely monitor blood pressure, renal function and electrolytes in patients on lisinopril and other agents that affect the RAS.
Do not co-administer aliskiren with lisinopril in patients with diabetes. Avoid use of aliskiren with lisinopril in patients with renal impairment (GFR <60 ml/min).
Carcinogenesis, Mutagenesis, Impairment of Fertility
There was no evidence of a tumorigenic effect when lisinopril was administered for 105 weeks to male and female rats at doses up to 90 mg/kg/day (about 56 or 9 times* the maximum recommended daily human dose, based on body weight and body surface area, respectively). There was no evidence of carcinogenicity when lisinopril was administered for 92 weeks to (male and female) mice at doses up to 135 mg/kg/day (about 84 times* the maximum recommended daily human dose). This dose was 6.8 times the maximum human dose based on body surface area in mice.
Lisinopril was not mutagenic in the Ames microbial mutagen test with or without metabolic activation. It was also negative in a forward mutation assay using Chinese hamster lung cells. Lisinopril did not produce single strand DNA breaks in an in vitro alkaline elution rat hepatocyte assay. In addition, lisinopril did not produce increases in chromosomal aberrations in an in vitro test in Chinese hamster ovary cells or in an in vivo study in mouse bone marrow.
There were no adverse effects on reproductive performance in male and female rats treated with up to 300 mg/kg/day of lisinopril. This dose is 188 times and 30 times the maximum human dose when based on mg/kg and mg/m2, respectively.
*Calculations assume a human weight of 50 kg and human body surface area of 1.62 m2.
Preparation of Suspension (for 200 Ml of A 1.0 Mg/ml Suspension):
Add 10 mL of Purified Water USP to a polyethylene terephthalate (PET) bottle containing ten 20 mg tablets of lisinopril and shake for at least one minute. Add 30 mL of Bicitra ®† diluent and 160 mL of Ora-Sweet SF™†† to the concentrate in the PET bottle and gently shake for several seconds to disperse the ingredients. The suspension should be stored at or below 25°C (77°F) and can be stored for up to four weeks. Shake the suspension before each use.
† Registered trademark of Alza Corporation
†† Trademark of Paddock Laboratories, Inc.
Structured Label Content
Recent Major Changes (34066-1)
WARNING: FETAL TOXICITY
See full prescribing information for complete boxed warning.
-
•When pregnancy is detected, discontinue lisinopril as soon as possible.
-
•Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity.
Section 42229-5 (42229-5)
Adult Patients: Administration of lisinopril to patients with hypertension results in a reduction of both supine and standing blood pressure to about the same extent with no compensatory tachycardia. Symptomatic postural hypotension is usually not observed although it can occur and should be anticipated in volume and/or salt-depleted patients (See WARNINGS ). When given together with thiazide-type diuretics, the blood pressure lowering effects of the two drugs are approximately additive.
In most patients studied, onset of antihypertensive activity was seen at one hour after oral administration of an individual dose of lisinopril, with peak reduction of blood pressure achieved by 6 hours. Although an antihypertensive effect was observed 24 hours after dosing with recommended single daily doses, the effect was more consistent and the mean effect was considerably larger in some studies with doses of 20 mg or more than with lower doses; however, at all doses studied, the mean antihypertensive effect was substantially smaller 24 hours after dosing than it was 6 hours after dosing.
In some patients achievement of optimal blood pressure reduction may require two to four weeks of therapy.
The antihypertensive effects of lisinopril are maintained during long-term therapy. Abrupt withdrawal of lisinopril has not been associated with a rapid increase in blood pressure, or a significant increase in blood pressure compared to pretreatment levels.
Two dose-response studies utilizing a once-daily regimen were conducted in 438 mild to moderate hypertensive patients not on a diuretic. Blood pressure was measured 24 hours after dosing. An antihypertensive effect of lisinopril was seen with 5 mg in some patients. However, in both studies blood pressure reduction occurred sooner and was greater in patients treated with 10 mg, 20 mg or 80 mg of lisinopril. In controlled clinical studies, lisinopril 20 to 80 mg has been compared in patients with mild to moderate hypertension to hydrochlorothiazide 12.5 to 50 mg and with atenolol 50 to 200 mg; and in patients with moderate to severe hypertension to metoprolol 100 to 200 mg. It was superior to hydrochlorothiazide in effects on systolic and diastolic pressure in a population that was 3/4 Caucasian. Lisinopril was approximately equivalent to atenolol and metoprolol in effects on diastolic blood pressure, and had somewhat greater effects on systolic blood pressure.
Lisinopril had similar effectiveness and adverse effects in younger and older (> 65 years) patients. It was less effective in Blacks than in Caucasians.
In hemodynamic studies in patients with essential hypertension, blood pressure reduction was accompanied by a reduction in peripheral arterial resistance with little or no change in cardiac output and in heart rate. In a study in nine hypertensive patients, following administration of lisinopril, there was an increase in mean renal blood flow that was not significant. Data from several small studies are inconsistent with respect to the effect of lisinopril on glomerular filtration rate in hypertensive patients with normal renal function, but suggest that changes, if any, are not large.
In patients with renovascular hypertension lisinopril has been shown to be well tolerated and effective in controlling blood pressure (See PRECAUTIONS ).
Overdosage (OVERDOSAGE)
Following a single oral dose of 20 g/kg no lethality occurred in rats, and death occurred in one of 20 mice receiving the same dose. The most likely manifestation of overdosage would be hypotension, for which the usual treatment would be intravenous infusion of normal saline solution.
Lisinopril can be removed by hemodialysis (See WARNINGS, Anaphylactoid Reactions During Membrane Exposure).
Description (DESCRIPTION)
Lisinopril, USP is an oral long-acting angiotensin converting enzyme inhibitor. Lisinopril, USP a synthetic peptide derivative, is chemically described as (S)-1-[N2-(1-carboxy-3-phenylpropyl)-L-lysyl]-L- proline dihydrate. Its molecular formula is C21H31N3O5 • 2H2O and its structural formula is:
Lisinopril, USP is a white to off-white, crystalline powder, with a molecular weight of 441.53. It is soluble in water and sparingly soluble in methanol and practically insoluble in ethanol.
Lisinopril tablets, USP are supplied as 2.5 mg, 5 mg, 10 mg, 20 mg, 30 mg and 40 mg tablets for oral administration.
Inactive Ingredients:
2.5 mg tablets – lactose monohydrate, maize starch, colloidal silicon dioxide, magnesium stearate.
5 mg, 10 mg, 20 mg and 30 mg tablets – lactose monohydrate, maize starch, colloidal silicon dioxide, magnesium stearate, iron oxide red.
40 mg tablets - lactose monohydrate, maize starch, colloidal silicon dioxide, magnesium stearate, iron oxide yellow.
How Supplied (HOW SUPPLIED)
2.5 mg Tablets: White, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “417” on the other side are supplied in bottle of 30 tablets (NDC 76282-417-30), bottle of 45 tablets (NDC 76282-417-45) bottle of 90 tablets (NDC 76282-417-90) bottle of 100 tablets (NDC 76282-417-01), bottle of 180 tablets (NDC 76282-417-18), bottle of 500 tablets (NDC 76282-417-05) and 1000 tablets (NDC 76282-417-10).
5 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘I’ on the left side of functional score and ‘G’ on the right side of functional score, and “418” on the other side are supplied in bottles of 30 tablets (NDC 76282-418-30), bottle of 45 tablets (NDC 76282-418-45) bottle of 90 tablets (NDC 76282-418-90) bottle of 100 tablets (NDC 76282-418-01), bottle of 180 tablets (NDC 76282-418-18), bottle of 500 tablets (NDC 76282-418-05) and 1000 tablets (NDC 76282-418-10).
10 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “419” on the other side are supplied in bottles of 30 tablets (NDC 76282-419-30), bottle of 45 tablets (NDC 76282-419-45) bottle of 90 tablets (NDC 76282-419-90) bottle of 100 tablets (NDC 76282-419-01) bottle of 180 tablets (NDC 76282-419-18), bottle of 500 tablets (NDC 76282-419-05), and 1000 tablets (NDC 76282-419-10).
20 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “420” on the other side are supplied in bottles of 30 tablets (NDC 76282-420-30), bottle of 45 tablets (NDC 76282-420-45) bottle of 90 tablets (NDC 76282-420-90) bottle of 100 tablets (NDC 76282-420-01), bottle of 180 tablets (NDC 76282-420-18), bottle of 500 tablets (NDC 76282-420-05) and 1000 tablets (NDC 76282-420-10).
30 mg Tablets: Red, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “421” on the other side are supplied in bottles of 30 tablets (NDC 76282-421-30), bottle of 45 tablets (NDC 76282-421-45) bottle of 90 tablets (NDC 76282-421-90) bottle of 100 tablets (NDC 76282-421-01), bottle of 180 tablets (NDC 76282-421-18), bottle of 500 tablets (NDC 76282-421-05) and 1000 tablets (NDC 76282-421-10).
40 mg Tablets: Yellow, round, biconvex, bevel-edged tablets debossed with ‘IG’ on one side and “422” on the other side are supplied in bottles of 30 tablets (NDC 76282-422-30), bottle of 45 tablets (NDC 76282-422-45) bottle of 90 tablets (NDC 76282-422-90) bottle of 100 tablets (NDC 76282-422-01), bottle of 180 tablets (NDC 76282-422-18), bottle of 500 tablets (NDC 76282-422-05) and 1000 tablets (NDC 76282-422-10).
Storage
Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature]. Protect from moisture, freezing and excessive heat. Dispense in a tight container.
*AN69 is a registered trademark of Hospal Ltd
All brand names listed are the registered trademarks of their respective owners and are not trademarks of Exelan Pharmaceuticals, Inc.
Manufactured for:
Exelan Pharmaceuticals, Inc.
Lawrenceville, GA 30046
Manufactured by:
InvaGen Pharmaceuticals, Inc.
Hauppauge, NY 11788
Rev: 11/13
Barcode: 422-11-2013
Hypertension
In clinical trials in patients with hypertension treated with lisinopril, discontinuation of therapy due to clinical adverse experiences occurred in 5.7% of patients. The overall frequency of adverse experiences could not be related to total daily dosage within the recommended therapeutic dosage range.
For adverse experiences occurring in greater than 1% of patients with hypertension treated with lisinopril or lisinopril plus hydrochlorothiazide in controlled clinical trials, and more frequently with lisinopril and/or lisinopril plus hydrochlorothiazide than placebo, comparative incidence data are listed in the table below:
|
LISINOPRIL (n=1349) Incidence (discontinuation) |
LISINOPRIL/ Hydrochlorothiazide (n=629) Incidence (discontinuation) |
PLACEBO (n=207) Incidence (discontinuation) |
|
|
Body as a Whole |
|||
|
Fatigue |
2.5 (0.3) |
4.0 (0.5) |
1.0 (0.0) |
|
Asthenia |
1.3 (0.5) |
2.1 (0.2) |
1.0 (0.0) |
|
Orthostatic Effects |
1.2 (0.0) |
3.5 (0.2) |
1.0 (0.0) |
|
Cardiovascular |
|||
|
Hypotension |
1.2 (0.5) |
1.6 (0.5) |
0.5 (0.5) |
|
Digestive |
|||
|
Diarrhea |
2.7 (0.2) |
2.7 (0.3) |
2.4 (0.0) |
|
Nausea |
2.0 (0.4) |
2.5 (0.2) |
2.4 (0.0) |
|
Vomiting |
1.1 (0.2) |
1.4 (0.1) |
0.5 (0.0) |
|
Dyspepsia |
0.9 (0.0) |
1.9 (0.0) |
0.0 (0.0) |
|
Musculoskeletal |
|||
|
Muscle Cramps |
0.5 (0.0) |
2.9 (0.8) |
0.5 (0.0) |
|
Nervous/Psychiatric |
|||
|
Headache |
5.7 (0.2) |
4.5 (0.5) |
1.9 (0.0) |
|
Dizziness |
5.4 (0.4) |
9.2 (1.0) |
1.9 (0.0) |
|
Paresthesia |
0.8 (0.1) |
2.1 (0.2) |
0.0 (0.0) |
|
Decreased Libido |
0.4 (0.1) |
1.3 (0.1) |
0.0 (0.0) |
|
Vertigo |
0.2 (0.1) |
1.1 (0.2) |
0.0 (0.0) |
|
Respiratory |
|||
|
Cough |
3.5 (0.7) |
4.6 (0.8) |
1.0 (0.0) |
|
Upper Respiratory Infection |
2.1 (0.1) |
2.7 (0.1) |
0.0 (0.0) |
|
Common Cold |
1.1 (0.1) |
1.3 (0.1) |
0.0 (0.0) |
|
Nasal Congestion |
0.4 (0.1) |
1.3 (0.1) |
0.0 (0.0) |
|
Influenza |
0.3 (0.1) |
1.1 (0.1) |
0.0 (0.0) |
|
Skin |
|||
|
Rash |
1.3 (0.4) |
1.6 (0.2) |
0.5 (0.5) |
|
Urogenital |
|||
|
Impotence |
1.0 (0.4) |
1.6 (0.5) |
0.0 (0.0) |
Chest pain and back pain were also seen, but were more common on placebo than lisinopril.
Geriatric Use
Clinical studies of lisinopril in patients with hypertension did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other clinical experience in this population has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
In the ATLAS trial of lisinopril in patients with congestive heart failure, 1,596 (50%) were 65 and over, while 437 (14%) were 75 and over. In a clinical study of lisinopril in patients with myocardial infarctions 4,413 (47%) were 65 and over, while 1,656 (18%) were 75 and over. In these studies, no overall differences in safety or effectiveness were observed between elderly and younger patients, and other reported clinical experiences has not identified differences in responses between the elderly and younger patients (See CLINICAL PHARMACOLOGY, Pharmacodynamics and Clinical Effects, Heart Failure and CLINICAL PHARMACOLOGY, Pharmacodynamics and Clinical Effects, Acute Myocardial Infarction).
Other reported clinical experience has not identified differences in responses between elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Pharmacokinetic studies indicate that maximum blood levels and area under the plasma concentration time curve (AUC) are doubled in older patients (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism).
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection. Evaluation of patients with hypertension, congestive heart failure, or myocardial infarction should always include assessment of renal function (See DOSAGE AND ADMINISTRATION ).
Heart Failure
Lisinopril tablets, USP are indicated as adjunctive therapy in the management of heart failure in patients who are not responding adequately to diuretics and digitalis.
Hypotension
Excessive hypotension is rare in patients with uncomplicated hypertension treated with lisinopril alone.
Patients with heart failure given lisinopril commonly have some reduction in blood pressure, with peak blood pressure reduction occurring 6 to 8 hours post dose. Evidence from the two-dose ATLAS trial suggested that incidence of hypotension may increase with dose of lisinopril in heart failure patients. Discontinuation of therapy because of continuing symptomatic hypotension usually is not necessary when dosing instructions are followed; caution should be observed when initiating therapy (See DOSAGE AND ADMINISTRATION).
Patients at risk of excessive hypotension, sometimes associated with oliguria and/or progressive azotemia, and rarely with acute renal failure and/or death, include those with the following conditions or characteristics: heart failure with systolic blood pressure below 100 mmHg, hyponatremia, high dose diuretic therapy, recent intensive diuresis or increase in diuretic dose, renal dialysis, or severe volume and/or salt depletion of any etiology. It may be advisable to eliminate the diuretic (except in patients with heart failure), reduce the diuretic dose or increase salt intake cautiously before initiating therapy with lisinopril in patients at risk for excessive hypotension who are able to tolerate such adjustments (See PRECAUTIONS, Drug Interactions and ADVERSE REACTIONS ).
Patients with acute myocardial infarction in the GISSI-3 trial had a higher (9.0% versus 3.7%) incidence of persistent hypotension (systolic blood pressure <90 mmHg for more than 1 hour) when treated with lisinopril. Treatment with lisinopril must not be initiated in acute myocardial infarction patients at risk of further serious hemodynamic deterioration after treatment with a vasodilator (e.g., systolic blood pressure of 100 mmHg or lower) or cardiogenic shock.
In patients at risk of excessive hypotension, therapy should be started under very close medical supervision and such patients should be followed closely for the first two weeks of treatment and whenever the dose of lisinopril and/or diuretic is increased. Similar considerations may apply to patients with ischemic heart or cerebrovascular disease, or in patients with acute myocardial infarction, in whom an excessive fall in blood pressure could result in a myocardial infarction or cerebrovascular accident.
If excessive hypotension occurs, the patient should be placed in the supine position and, if necessary, receive an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further doses of lisinopril which usually can be given without difficulty once the blood pressure has stabilized. If symptomatic hypotension develops, a dose reduction or discontinuation of lisinopril or concomitant diuretic may be necessary.
Pediatric Use
Neonates with a history of in utero exposure to lisinopril:
If oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and/or substituting for disordered renal function. Lisinopril, which crosses the placenta, has been removed from neonatal circulation by peritoneal dialysis with some clinical benefit, and theoretically may be removed by exchange transfusion, although there is no experience with the latter procedure.
Antihypertensive effects of lisinopril have been established in hypertensive pediatric patients aged 6 to 16 years.
There are no data on the effect of lisinopril on blood pressure in pediatric patients under the age 6 or in pediatric patients with glomerular filtration rate < 30 mL/min/1.73 m2. (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism and Pharmacodynamics and Clinical Effects, and DOSAGE AND ADMINISTRATION ).
Hypertension
Lisinopril Tablets, USP are indicated for the treatment of hypertension to lower blood pressure. Lowering blood pressure lowers the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including lisinopril.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than 1 drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (eg, on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Lisinopril may be administered alone or with other antihypertensive agents.
Use in Elderly
In general, the clinical response was similar in younger and older patients given similar doses of lisinopril. Pharmacokinetic studies, however indicate that maximum blood levels and area under the plasma concentration time curve (AUC) are doubled in older patients, so that dosage adjustments should be made with particular caution.
Hepatic Failure
Rarely, ACE inhibitors have been associated with a syndrome that starts with cholestatic jaundice or hepatitis and progresses to fulminant hepatic necrosis and (sometimes) death. The mechanism of this syndrome is not understood. Patients receiving ACE inhibitors who develop jaundice or marked elevations of hepatic enzymes should discontinue the ACE inhibitor and receive appropriate medical follow-up.
Nursing Mothers
Milk of lactating rats contains radioactivity following administration of 14C lisinopril. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from ACE inhibitors, a decision should be made whether to discontinue nursing or discontinue lisinopril, taking into account the importance of the drug to the mother.
Adverse Reactions (ADVERSE REACTIONS)
Lisinopril has been found to be generally well tolerated in controlled clinical trials involving 1969 patients with hypertension or heart failure. For the most part, adverse experiences were mild and transient.
Contraindications (CONTRAINDICATIONS)
Lisinopril is contraindicated in patients who are hypersensitive to this product and in patients with a history of angioedema related to previous treatment with an angiotensin-converting enzyme inhibitor and in patients with hereditary or idiopathic angioedema.
Mechanism of Action
Lisinopril inhibits angiotensin-converting enzyme (ACE) in human subjects and animals. ACE is a peptidyl dipeptidase that catalyzes the conversion of angiotensin I to the vasoconstrictor substance, angiotensin II. Angiotensin II also stimulates aldosterone secretion by the adrenal cortex. The beneficial effects of lisinopril in hypertension and heart failure appear to result primarily from suppression of the renin-angiotensin-aldosterone system. Inhibition of ACE results in decreased plasma angiotensin II which leads to decreased vasopressor activity and to decreased aldosterone secretion. The latter decrease may result in a small increase of serum potassium. In hypertensive patients with normal renal function treated with lisinopril alone for up to 24 weeks, the mean increase in serum potassium was approximately 0.1 mEq/L; however, approximately 15% of patients had increases greater than 0.5 mEq/L and approximately 6% had a decrease greater than 0.5 mEq/L. In the same study, patients treated with lisinopril and hydrochlorothiazide for up to 24 weeks had a mean decrease in serum potassium of 0.1 mEq/L; approximately 4% of patients had increases greater than 0.5 mEq/L and approximately 12% had a decrease greater than 0.5 mEq/L (See PRECAUTIONS ). Removal of angiotensin II negative feedback on renin secretion leads to increased plasma renin activity.
ACE is identical to kininase, an enzyme that degrades bradykinin. Whether increased levels of bradykinin, a potent vasodepressor peptide, play a role in the therapeutic effects of lisinopril remains to be elucidated.
While the mechanism through which lisinopril lowers blood pressure is believed to be primarily suppression of the renin-angiotensin-aldosterone system, lisinopril is antihypertensive even in patients with low-renin hypertension. Although lisinopril was antihypertensive in all races studied, Black hypertensive patients (usually a low-renin hypertensive population) had a smaller average response to monotherapy than non-Black patients.
Concomitant administration of lisinopril and hydrochlorothiazide further reduced blood pressure in Black and non-Black patients and any racial differences in blood pressure response were no longer evident.
Pregnancy Category D (Pregnancy category D)
Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue lisinopril as soon as possible. These adverse outcomes are usually associated with use of these drugs in the second and third trimester of pregnancy. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. Appropriate management of maternal hypertension during pregnancy is important to optimize outcomes for both mother and fetus.
In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus. Perform serial ultrasound examinations to assess the intra-amniotic environment. If oligohydramnios is observed, discontinue lisinopril, unless it is considered lifesaving for the mother. Fetal testing may be appropriate, based on the week of pregnancy. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to lisinopril for hypotension, oliguria, and hyperkalemia (see Precautions, Pediatric Use).
No teratogenic effects of lisinopril were seen in studies of pregnant rats, mice, and rabbits. On a mg/kg basis, the doses used were up to 625 times (in mice), 188 times (in rats), and 0.6 times (in rabbits) the maximum recommended human dose.
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 2.5mg |
| 90 | 43353-270-90 |
| Count | 5mg |
| 30 | 43353-298-30 |
| 45 | 43353-298-45 |
| Count | 10mg |
| 30 | 43353-301-30 |
| 45 | 43353-301-45 |
| 90 | 43353-301-60 |
| Count | 20mg |
| 30 | 43353-271-30 |
| 45 | 43353-271-45 |
| 90 | 43353-271-60 |
| 180 | 43353-271-80 |
| Count | 40mg |
| 30 | 43353-009-30 |
| 45 | 43353-009-45 |
| 90 | 43353-009-60 |
| 180 | 43353-009-80 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20170629JH
Acute Myocardial Infarction
Lisinopril tablets, USP are indicated for the treatment of hemodynamically stable patients within 24 hours of acute myocardial infarction, to improve survival. Patients should receive, as appropriate, the standard recommended treatments such as thrombolytics, aspirin and beta-blockers.
In using lisinopril, consideration should be given to the fact that another angiotensin-converting enzyme inhibitor, captopril, has caused agranulocytosis, particularly in patients with renal impairment or collagen vascular disease, and that available data are insufficient to show that lisinopril does not have a similar risk (See WARNINGS ).
In considering the use of lisinopril, it should be noted that in controlled clinical trials ACE inhibitors have an effect on blood pressure that is less in Black patients than in non-Blacks. In addition, ACE inhibitors have been associated with a higher rate of angioedema in Black than in non-Black patients (See WARNINGS, Anaphylactoid and Possibly Related Reactions ).
Package/label Display Panel (Package/Label Display Panel)
NDC 43353-270-90
Lisinopril Tablets, USP
2.5 mg
Rx Only 90 Tablets
Pharmacokinetics and Metabolism
Adult Patients: Following oral administration of lisinopril, peak serum concentrations of lisinopril occur within about 7 hours, although there was a trend to a small delay in time taken to reach peak serum concentrations in acute myocardial infarction patients. Declining serum concentrations exhibit a prolonged terminal phase which does not contribute to drug accumulation. This terminal phase probably represents saturable binding to ACE and is not proportional to dose.
Lisinopril does not appear to be bound to other serum proteins. Lisinopril does not undergo metabolism and is excreted unchanged entirely in the urine. Based on urinary recovery, the mean extent of absorption of lisinopril is approximately 25%, with large intersubject variability (6% to 60%) at all doses tested (5 mg to 80 mg). Lisinopril absorption is not influenced by the presence of food in the gastrointestinal tract. The absolute bioavailability of lisinopril is reduced to 16% in patients with stable NYHA Class II-IV congestive heart failure, and the volume of distribution appears to be slightly smaller than that in normal subjects. The oral bioavailability of lisinopril in patients with acute myocardial infarction is similar to that in healthy volunteers.
Upon multiple dosing, lisinopril exhibits an effective half-life of accumulation of 12 hours.
Impaired renal function decreases elimination of lisinopril, which is excreted principally through the kidneys, but this decrease becomes clinically important only when the glomerular filtration rate is below 30 mL/min. Above this glomerular filtration rate, the elimination half-life is little changed. With greater impairment, however, peak and trough lisinopril levels increase, time to peak concentration increases and time to attain steady state is prolonged. Older patients, on average, have (approximately doubled) higher blood levels and area under the plasma concentration time curve (AUC) than younger patients (See DOSAGE AND ADMINISTRATION). Lisinopril can be removed by hemodialysis.
Studies in rats indicate that lisinopril crosses the blood-brain barrier poorly. Multiple doses of lisinopril in rats do not result in accumulation in any tissues. Milk of lactating rats contains radioactivity following administration of 14C lisinopril. By whole body autoradiography, radioactivity was found in the placenta following administration of labeled drug to pregnant rats, but none was found in the fetuses.
Pediatric Patients: The pharmacokinetics of lisinopril were studied in 29 pediatric hypertensive patients between 6 years and 16 years with glomerular filtration rate > 30 mL/min/1.73 m2. After doses of 0.1 to 0.2 mg/kg, steady state peak plasma concentrations of lisinopril occurred within 6 hours and the extent of absorption based on urinary recovery was about 28%. These values are similar to those obtained previously in adults. The typical value of lisinopril oral clearance (systemic clearance/absolute bioavailability) in a child weighing 30 kg is 10 L/h, which increases in proportion to renal function.
Leukopenia/neutropenia/agranulocytosis (Leukopenia/Neutropenia/Agranulocytosis)
Another angiotensin-converting enzyme inhibitor, captopril, has been shown to cause agranulocytosis and bone marrow depression, rarely in uncomplicated patients but more frequently in patients with renal impairment especially if they also have a collagen vascular disease. Available data from clinical trials of lisinopril are insufficient to show that lisinopril does not cause agranulocytosis at similar rates. Marketing experience has revealed rare cases of leukopenia/neutropenia and bone marrow depression in which a causal relationship to lisinopril cannot be excluded. Periodic monitoring of white blood cell counts in patients with collagen vascular disease and renal disease should be considered.
Anaphylactoid and Possibly Related Reactions
Presumably because angiotensin-converting enzyme inhibitors affect the metabolism of eicosanoids and polypeptides, including endogenous bradykinin, patients receiving ACE inhibitors (including lisinopril) may be subject to a variety of adverse reactions, some of them serious.
Pediatric Hypertensive Patients ≥ 6 Years of Age (Pediatric Hypertensive Patients ≥ 6 years of age)
The usual recommended starting dose is 0.07 mg/kg once daily (up to 5 mg total). Dosage should be adjusted according to blood pressure response. Doses above 0.61 mg/kg (or in excess of 40 mg) have not been studied in pediatric patients (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism and Pharmacodynamics and Clinical Effects).
Lisinopril is not recommended in pediatric patients < 6 years or in pediatric patients with glomerular filtration rate < 30 mL/min/1.73 m2 (See CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism and Pharmacodynamics and Clinical Effects and PRECAUTIONS).
Dual Blockade of the Renin Angiotensin System (ras) (Dual Blockade of the Renin-Angiotensin System (RAS))
Dual blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Closely monitor blood pressure, renal function and electrolytes in patients on lisinopril and other agents that affect the RAS.
Do not co-administer aliskiren with lisinopril in patients with diabetes. Avoid use of aliskiren with lisinopril in patients with renal impairment (GFR <60 ml/min).
Carcinogenesis, Mutagenesis, Impairment of Fertility
There was no evidence of a tumorigenic effect when lisinopril was administered for 105 weeks to male and female rats at doses up to 90 mg/kg/day (about 56 or 9 times* the maximum recommended daily human dose, based on body weight and body surface area, respectively). There was no evidence of carcinogenicity when lisinopril was administered for 92 weeks to (male and female) mice at doses up to 135 mg/kg/day (about 84 times* the maximum recommended daily human dose). This dose was 6.8 times the maximum human dose based on body surface area in mice.
Lisinopril was not mutagenic in the Ames microbial mutagen test with or without metabolic activation. It was also negative in a forward mutation assay using Chinese hamster lung cells. Lisinopril did not produce single strand DNA breaks in an in vitro alkaline elution rat hepatocyte assay. In addition, lisinopril did not produce increases in chromosomal aberrations in an in vitro test in Chinese hamster ovary cells or in an in vivo study in mouse bone marrow.
There were no adverse effects on reproductive performance in male and female rats treated with up to 300 mg/kg/day of lisinopril. This dose is 188 times and 30 times the maximum human dose when based on mg/kg and mg/m2, respectively.
*Calculations assume a human weight of 50 kg and human body surface area of 1.62 m2.
Preparation of Suspension (for 200 Ml of A 1.0 Mg/ml Suspension): (Preparation of Suspension (for 200 mL of a 1.0 mg/mL suspension):)
Add 10 mL of Purified Water USP to a polyethylene terephthalate (PET) bottle containing ten 20 mg tablets of lisinopril and shake for at least one minute. Add 30 mL of Bicitra ®† diluent and 160 mL of Ora-Sweet SF™†† to the concentrate in the PET bottle and gently shake for several seconds to disperse the ingredients. The suspension should be stored at or below 25°C (77°F) and can be stored for up to four weeks. Shake the suspension before each use.
† Registered trademark of Alza Corporation
†† Trademark of Paddock Laboratories, Inc.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:15.148404 · Updated: 2026-03-14T21:45:41.180903