These Highlights Do Not Include All The Information Needed To Use Heparin Sodium In Sodium Chloride Injection Safely And Effectively. See Full Prescribing Information For Heparin Sodium In Sodium Chloride Injection.
32e34077-aff1-4ec7-f9b0-b8e240768fb8
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis ( 5.2) 11/2024 Warnings and Precautions, Unresponsiveness to Heparin with Concomitant Use with Andexanet Alfa ( 5.3 ) 11/2025 Warnings and Precautions, Heparin Resistance ( 5.5 ) 11/2024 Warnings and Precautions, Hypersensitivity Reactions ( 5.6 ) 11/2024 Warnings and Precautions, Elevations of Serum Aminotransferases ( 5.9 ) 11/2024
Indications and Usage
Heparin Sodium in Sodium Chloride Injection at a concentration of 2 units/mL is indicated as an anticoagulant to maintain catheter patency.
Dosage and Administration
Infuse through intravenous catheter at a rate of 6 units per hour. ( 2.2 )
Warnings and Precautions
• Hemorrhage: Fatal hemorrhages have occurred. Monitor for signs of bleeding and manage promptly. ( 5.1 ) • HIT and HITT: Monitor for signs and symptoms and discontinue if indicative of HIT or HITT. ( 5.2 ) • Thrombocytopenia: Monitor platelet count during therapy; discontinue heparin if HIT or HITT is suspected. ( 5.4 ) • Heparin Resistance: Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in postsurgical patients. ( 5.5 ) • Hypersensitivity Reactions: Use in patients with prior reactions only in life-threatening situations. ( 5.6 ) • Increased Risk of Bleeding in Older Patients, Especially Women: A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age. ( 5.7 ) • Hyperkalemia: Measure blood potassium in patients at risk of hyperkalemia before starting heparin therapy and periodically in all patients. ( 5.8 ) • Elevations of Serum Aminotransferases: Interpret elevation of these enzymes with caution. ( 5.9 ) • Laboratory Tests: Periodic platelet counts, hematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration. ( 5.10 )
Contraindications
The use of Heparin Sodium in Sodium Chloride Injection is contraindicated in patients with the following conditions: • Uncontrollable active bleeding state, except when this is due to disseminated intravascular coagulation [see Warnings and Precautions (5.1)] • History of heparin-induced thrombocytopenia (HIT) and heparin-induced thrombocytopenia with thrombosis (HITT) [see Warnings and Precautions (5.2) ] • Severe thrombocytopenia [see Warnings and Precautions (5.2 , 5.4) ] • Known hypersensitivity to heparin or pork products (e.g., anaphylactoid reactions) [see Warnings and Precautions (5.6) , Adverse Reactions (6.1) ]
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Hemorrhage [see Warnings and Precautions (5.1) ] • Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis [see Warnings and Precautions (5.2) ] • Thrombocytopenia [see Warnings and Precautions (5.4) ] • Heparin Resistance [see Warnings and Precautions (5.5) ] • Hypersensitivity Reactions [see Warnings and Precautions (5.6) ] • Increased Risk of Bleeding in Older Patients, Especially Women [see Warnings and Precautions (5.7) ] • Hyperkalemia [see Warnings and Precautions (5.8) ] • Elevations of Serum Aminotransferases [see Warnings and Precautions (5.9) ]
Drug Interactions
• Drugs that interfere with platelet aggregation or drugs that counteract coagulation may induce bleeding. ( 7 ) • Andexanet alfa may reduce the efficacy of heparin when used concomitantly. ( 7.3 )
Storage and Handling
Heparin Sodium in 0.9% Sodium Chloride Injection is a clear solution and is available in single-dose containers as follows: Unit of Sale Concentration NDC 0409-7620-03 Case of 18 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-1005-20 Case of 20 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-7620-59 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL) NDC 0409-2222-12 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL)
How Supplied
Heparin Sodium in 0.9% Sodium Chloride Injection is a clear solution and is available in single-dose containers as follows: Unit of Sale Concentration NDC 0409-7620-03 Case of 18 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-1005-20 Case of 20 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-7620-59 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL) NDC 0409-2222-12 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL)
Medication Information
Warnings and Precautions
• Hemorrhage: Fatal hemorrhages have occurred. Monitor for signs of bleeding and manage promptly. ( 5.1 ) • HIT and HITT: Monitor for signs and symptoms and discontinue if indicative of HIT or HITT. ( 5.2 ) • Thrombocytopenia: Monitor platelet count during therapy; discontinue heparin if HIT or HITT is suspected. ( 5.4 ) • Heparin Resistance: Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in postsurgical patients. ( 5.5 ) • Hypersensitivity Reactions: Use in patients with prior reactions only in life-threatening situations. ( 5.6 ) • Increased Risk of Bleeding in Older Patients, Especially Women: A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age. ( 5.7 ) • Hyperkalemia: Measure blood potassium in patients at risk of hyperkalemia before starting heparin therapy and periodically in all patients. ( 5.8 ) • Elevations of Serum Aminotransferases: Interpret elevation of these enzymes with caution. ( 5.9 ) • Laboratory Tests: Periodic platelet counts, hematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration. ( 5.10 )
Indications and Usage
Heparin Sodium in Sodium Chloride Injection at a concentration of 2 units/mL is indicated as an anticoagulant to maintain catheter patency.
Dosage and Administration
Infuse through intravenous catheter at a rate of 6 units per hour. ( 2.2 )
Contraindications
The use of Heparin Sodium in Sodium Chloride Injection is contraindicated in patients with the following conditions: • Uncontrollable active bleeding state, except when this is due to disseminated intravascular coagulation [see Warnings and Precautions (5.1)] • History of heparin-induced thrombocytopenia (HIT) and heparin-induced thrombocytopenia with thrombosis (HITT) [see Warnings and Precautions (5.2) ] • Severe thrombocytopenia [see Warnings and Precautions (5.2 , 5.4) ] • Known hypersensitivity to heparin or pork products (e.g., anaphylactoid reactions) [see Warnings and Precautions (5.6) , Adverse Reactions (6.1) ]
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Hemorrhage [see Warnings and Precautions (5.1) ] • Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis [see Warnings and Precautions (5.2) ] • Thrombocytopenia [see Warnings and Precautions (5.4) ] • Heparin Resistance [see Warnings and Precautions (5.5) ] • Hypersensitivity Reactions [see Warnings and Precautions (5.6) ] • Increased Risk of Bleeding in Older Patients, Especially Women [see Warnings and Precautions (5.7) ] • Hyperkalemia [see Warnings and Precautions (5.8) ] • Elevations of Serum Aminotransferases [see Warnings and Precautions (5.9) ]
Drug Interactions
• Drugs that interfere with platelet aggregation or drugs that counteract coagulation may induce bleeding. ( 7 ) • Andexanet alfa may reduce the efficacy of heparin when used concomitantly. ( 7.3 )
Storage and Handling
Heparin Sodium in 0.9% Sodium Chloride Injection is a clear solution and is available in single-dose containers as follows: Unit of Sale Concentration NDC 0409-7620-03 Case of 18 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-1005-20 Case of 20 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-7620-59 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL) NDC 0409-2222-12 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL)
How Supplied
Heparin Sodium in 0.9% Sodium Chloride Injection is a clear solution and is available in single-dose containers as follows: Unit of Sale Concentration NDC 0409-7620-03 Case of 18 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-1005-20 Case of 20 Single-dose flexible plastic containers 1,000 USP Units/500 mL (2 USP Units/mL) NDC 0409-7620-59 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL) NDC 0409-2222-12 Case of 12 Single-dose flexible plastic containers 2,000 USP Units/1,000 mL (2 USP Units/mL)
Description
Warnings and Precautions, Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis ( 5.2) 11/2024 Warnings and Precautions, Unresponsiveness to Heparin with Concomitant Use with Andexanet Alfa ( 5.3 ) 11/2025 Warnings and Precautions, Heparin Resistance ( 5.5 ) 11/2024 Warnings and Precautions, Hypersensitivity Reactions ( 5.6 ) 11/2024 Warnings and Precautions, Elevations of Serum Aminotransferases ( 5.9 ) 11/2024
Section 42229-5
To Open
Tear outer wrap and remove solution container.
Section 43683-2
|
Warnings and Precautions, Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis (5.2) |
11/2024 |
|
Warnings and Precautions, Unresponsiveness to Heparin with Concomitant Use with Andexanet Alfa (5.3) |
11/2025 |
|
Warnings and Precautions, Heparin Resistance (5.5) |
11/2024 |
|
Warnings and Precautions, Hypersensitivity Reactions (5.6) |
11/2024 |
|
Warnings and Precautions, Elevations of Serum Aminotransferases (5.9) |
11/2024 |
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
10 Overdosage
An overdose requires immediate medical attention and treatment.
Symptoms
Bleeding is the chief sign of heparin overdosage. Easy bruising, petechial formations, nosebleeds, blood in urine or tarry stools may be the first signs or symptoms of a heparin overdose. In the event of symptomatic heparin overdose, consider stopping heparin infusion.
11 Description
Intravenous solutions with heparin sodium (derived from porcine intestinal mucosa) are sterile, nonpyrogenic fluids for intravenous administration. Each 100 mL contains heparin sodium 200 USP Units; sodium chloride, 0.9 g; citric acid, monohydrate, 40 mg and dibasic sodium phosphate, heptahydrate, 434 mg added as buffers. Each liter contains the following electrolytes: Sodium 186.4 mEq; phosphate (as HPO4=) 32.4 mEq, citrate 5.7 mEq and chloride 154 mEq. Osmolar concentration, 378 mOsmol/liter (calc.); pH 7.0 (5.0 – 7.5).
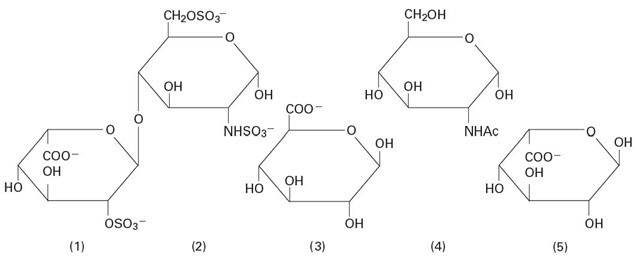
Heparin Sodium, USP is a heterogeneous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans having anticoagulant properties. Although others may be present, the main sugars occurring in heparin are: (1) α-L-iduronic acid 2-sulfate, (2) 2-deoxy-2-sulfamino-α-D-glucose-6-sulfate, (3) β-D-glucuronic acid, (4) 2-acetamido-2-deoxy-α-D-glucose, and (5) α-L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2) > (1) > (4) > (3) > (5), and are joined by glycosidic linkages, forming polymers of varying sizes. Heparin is strongly acidic because of its content of covalently linked sulfate and carboxylic acid groups. In heparin sodium, the acidic protons of the sulfate units are partially replaced by sodium ions. The potency is determined by a biological assay using a USP reference standard based on units of heparin activity per milligram.
Structure of Heparin Sodium (representative subunits):
Sodium Chloride, USP is chemically designated NaCl, a white crystalline compound freely soluble in water.
Dibasic Sodium Phosphate, USP (heptahydrate), is chemically designated (Na2HPO4 ∙ 7H2O), colorless or white granular salt freely soluble in water.
Citric Acid, USP, hydrous (monohydrate) is chemically designated C6H8O7 ∙ H2O, colorless, translucent crystals or white crystalline powder very soluble in water. It has the following structural formula:
Water for Injection, USP is chemically designated H2O.
The flexible plastic container is fabricated from either polyvinylchloride or polyolefin plastic. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions inside the plastic container also can leach out certain of its chemical components in very small amounts before the expiration period is attained. However, the safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers.
5.1 Hemorrhage
Avoid using heparin in the presence of major bleeding, except when the benefits of heparin therapy outweigh the potential risks.
Hemorrhage can occur at virtually any site in patients receiving heparin. Fatal hemorrhages have occurred. An unexplained fall in hematocrit or fall in blood pressure, or any other unexplained symptom should lead to serious consideration of a hemorrhagic event.
Use heparin sodium with caution in disease states in which there is increased risk of hemorrhage, including:
-
•Cardiovascular — Subacute bacterial endocarditis. Severe hypertension.
-
•Surgical — During and immediately following (a) spinal tap or spinal anesthesia or (b) major surgery, especially involving the brain, spinal cord or eye.
-
•Hematologic — Conditions associated with increased bleeding tendencies, such as hemophilia, thrombocytopenia and some vascular purpuras.
-
•Gastrointestinal — Ulcerative lesions and continuous tube drainage of the stomach or small intestine.
-
•Patients with hereditary antithrombin III deficiency receiving concurrent antithrombin III therapy – The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. To reduce the risk of bleeding, reduce the heparin dose during concomitant treatment with antithrombin III (human).
-
•Other — Menstruation, liver disease with impaired hemostasis.
5.8 Hyperkalemia
Heparin can suppress adrenal secretion of aldosterone leading to hyperkalemia, particularly in patients with diabetes mellitus, chronic renal failure, pre-existing metabolic acidosis, a raised plasma potassium, or taking potassium sparing drugs. The risk of hyperkalemia appears to increase with duration of therapy but is usually reversible upon discontinuation of heparin.
Measure blood potassium in patients at risk of hyperkalemia before starting heparin therapy and periodically in all patients treated for more than 5 days or earlier as deemed fit by the clinician.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
A higher incidence of bleeding has been reported in patients over 60 years of age, especially women [see Warnings and Precautions (5.7)].
4 Contraindications
The use of Heparin Sodium in Sodium Chloride Injection is contraindicated in patients with the following conditions:
-
•Uncontrollable active bleeding state, except when this is due to disseminated intravascular coagulation [see Warnings and Precautions (5.1)]
-
•History of heparin-induced thrombocytopenia (HIT) and heparin-induced thrombocytopenia with thrombosis (HITT) [see Warnings and Precautions (5.2)]
-
•Severe thrombocytopenia [see Warnings and Precautions (5.2, 5.4)]
-
•Known hypersensitivity to heparin or pork products (e.g., anaphylactoid reactions) [see Warnings and Precautions (5.6), Adverse Reactions (6.1)]
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Hemorrhage [see Warnings and Precautions (5.1)]
-
•Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis [see Warnings and Precautions (5.2)]
-
•Thrombocytopenia [see Warnings and Precautions (5.4)]
-
•Heparin Resistance [see Warnings and Precautions (5.5)]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.6)]
-
•Increased Risk of Bleeding in Older Patients, Especially Women [see Warnings and Precautions (5.7)]
-
•Hyperkalemia [see Warnings and Precautions (5.8)]
-
•Elevations of Serum Aminotransferases [see Warnings and Precautions (5.9)]
7 Drug Interactions
5.4 Thrombocytopenia
Thrombocytopenia in patients receiving heparin has been reported at frequencies up to 30%. It can occur 2 to 20 days (average 5 to 9) following the onset of heparin therapy. Obtain platelet counts before and periodically during heparin therapy. If the count falls below 100,000/mm3 or if recurrent thrombosis develops, promptly discontinue heparin, evaluate for HIT and HITT, and, if necessary, administer an alternative anticoagulant [see Warnings and Precautions (5.2)].
12.2 Pharmacodynamics
Various times (activated clotting time, activated partial thromboplastin time, prothrombin time, whole blood clotting time) are prolonged by full therapeutic doses of heparin; in most cases, they are not measurably affected by low doses of heparin. Bleeding time is usually unaffected by heparin.
5.10 Laboratory Tests
Periodic platelet counts, hematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration.
5.5 Heparin Resistance
Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer, in postsurgical patients, and patients with antithrombin deficiency. Consider measurement of antithrombin levels if heparin resistance is suspected. Monitor coagulation tests frequently in such patients. It may be necessary to adjust the dose of heparin based on coagulation test monitoring, such as anti-Factor Xa levels and/or partial thromboplastin time.
1 Indications and Usage
Heparin Sodium in Sodium Chloride Injection at a concentration of 2 units/mL is indicated as an anticoagulant to maintain catheter patency.
7.1 Oral Anticoagulants
Heparin sodium may prolong the one-stage prothrombin time. Therefore, when heparin sodium is given with dicumarol or warfarin sodium, a period of at least 5 hours after the last intravenous dose or 24 hours after the last subcutaneous dose should elapse before blood is drawn if a valid prothrombin time is to be obtained.
7.2 Platelet Inhibitors
Drugs such as NSAIDS (including acetylsalicylic acid, ibuprofen, indomethacin, and celecoxib), dextran, phenylbutazone, thienopyridines, dipyridamole, hydroxychloroquine, glycoprotein IIv/IIa antagonists (including abciximab, eptifobatide, and tirofiban), and others that interfere with platelet-aggregation reactions (the main hemostatic defense of heparinized patients) may induce bleeding and should be used with caution in patients receiving heparin sodium. To reduce the risk of bleeding, a reduction in the dose of the antiplatelet agent or heparin is recommended.
12.1 Mechanism of Action
Heparin interacts with the naturally occurring plasma protein, antithrombin III, to induce a conformational change, which markedly enhances the serine protease activity of Antithrombin II, thereby inhibiting the activated coagulation factors involved in the closing sequence, particularly Xa and IIa. Small amounts of heparin inhibit Factor Xa, and larger amounts inhibit thrombin (Factor IIa).
Heparin also prevents the formation of a stable fibrin clot by inhibiting the activation of the fibrin stabilizing factor. Heparin does not have fibrinolytic activity; therefore, it will not lyse existing clots.
5 Warnings and Precautions
-
•Hemorrhage: Fatal hemorrhages have occurred. Monitor for signs of bleeding and manage promptly. (5.1)
-
•HIT and HITT: Monitor for signs and symptoms and discontinue if indicative of HIT or HITT. (5.2)
-
•Thrombocytopenia: Monitor platelet count during therapy; discontinue heparin if HIT or HITT is suspected. (5.4)
-
•Heparin Resistance: Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in postsurgical patients. (5.5)
-
•Hypersensitivity Reactions: Use in patients with prior reactions only in life-threatening situations. (5.6)
-
•Increased Risk of Bleeding in Older Patients, Especially Women: A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age. (5.7)
-
•Hyperkalemia: Measure blood potassium in patients at risk of hyperkalemia before starting heparin therapy and periodically in all patients. (5.8)
-
•Elevations of Serum Aminotransferases: Interpret elevation of these enzymes with caution. (5.9)
-
•Laboratory Tests: Periodic platelet counts, hematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration. (5.10)
2 Dosage and Administration
Infuse through intravenous catheter at a rate of 6 units per hour. (2.2)
3 Dosage Forms and Strengths
Heparin Sodium in 0.9% Sodium Chloride Injection is available as:
-
•Injection: 1,000 USP Units/500 mL (2 USP Units/mL) clear solution in a single-dose flexible plastic container
-
•Injection: 2,000 USP Units/1,000 mL (2 USP Units/mL) clear solution in a single-dose flexible plastic container
6.1 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of heparin sodium. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
5.6 Hypersensitivity Reactions
Hypersensitivity reactions with chills, fever, and urticaria as the most usual manifestations and also asthma, rhinitis, lacrimation, and anaphylactoid reactions have been reported. Patients with documented hypersensitivity to heparin should be given the drug only in clearly life-threatening situations. Because Heparin Sodium in Sodium Chloride Injection is derived from animal tissue, it should be used with caution in patients with a history of allergy to pork products.
2.1 Preparation for Administration
Do not administer unless solution is clear and seal is intact. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Warning: Do not use plastic containers in series connection. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Do not use Heparin Sodium in Sodium Chloride Injection as a “catheter lock flush” product.
Do not admix with other drugs. Discard unused portion.
16 How Supplied/storage and Handling
Heparin Sodium in 0.9% Sodium Chloride Injection is a clear solution and is available in single-dose containers as follows:
|
Unit of Sale |
Concentration |
|
NDC 0409-7620-03 Case of 18 Single-dose flexible plastic containers |
1,000 USP Units/500 mL (2 USP Units/mL) |
|
NDC 0409-1005-20 Case of 20 Single-dose flexible plastic containers |
1,000 USP Units/500 mL (2 USP Units/mL) |
|
NDC 0409-7620-59 Case of 12 Single-dose flexible plastic containers |
2,000 USP Units/1,000 mL (2 USP Units/mL) |
|
NDC 0409-2222-12 Case of 12 Single-dose flexible plastic containers |
2,000 USP Units/1,000 mL (2 USP Units/mL) |
Principal Display Panel Bag Overwrap
TO OPEN TEAR AT NOTCH
2
HDPE
DO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER
FIRMLY. IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE
IMPAIRED. RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID
EXCESSIVE HEAT. PROTECT FROM FREEZING. SEE INSERT.
IM-4498
Not Made With Natural Rubber Latex
14622100
5.9 Elevations of Serum Aminotransferases
Significant elevations of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels have occurred in patients who have received heparin. Elevation of these enzymes in patients receiving heparin should be interpreted with caution. These elevations typically resolve upon heparin discontinuation.
Principal Display Panel 500 Ml Bag 7620
1,000 USP Units/500 mL
(2 USP Units/mL)
500 mL
NDC 0409-7620-13
HEPARIN SODIUM
1,000 USP Units/500 mL
(2 USP Units/mL)
in 0.9% Sodium Chloride Injection
EACH 100 mL CONTAINS HEPARIN SODIUM 200 USP UNITS
(PORCINE INTESTINAL MUCOSA); SODIUM CHLORIDE 0.9 g;
CITRIC ACID, MONOHYDRATE 40 mg AND DIBASIC SODIUM
PHOSPHATE, HEPTAHYDRATE 434 mg ADDED AS BUFFERS.
STERILE. NOT MADE WITH NATURAL RUBBER LATEX.
USUAL DOSAGE: SEE INSERT
ADDITIVES SHOULD NOT BE MADE TO THIS SOLUTION.
SINGLE DOSE CONTAINER. DISCARD UNUSED PORTION. FOR
INTRAVENOUS USE ONLY.
3
V
CONTAINS DEHP
Rx only
DISTRIBUTED BY
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira
14478901
IM-4470
Principal Display Panel 1,000 Ml Bag 7620
LOT NO.
EXP. DATE
2,000 USP Units /1,000 mL
(2 USP Units/mL)
1,000 mL
NDC 0409-7620-49
HEPARIN SODIUM
2,000 USP Units
per 1,000 mL
(2 USP Units/mL)
in 0.9% Sodium Chloride Injection
EACH 100 mL CONTAINS HEPARIN SODIUM
200 USP UNITS (PORCINE INTESTINAL
MUCOSA); SODIUM CHLORIDE 0.9 g; CITRIC
ACID, MONOHYDRATE 40 mg AND DIBASIC
SODIUM PHOSPHATE, HEPTAHYDRATE
434 mg ADDED AS BUFFERS. STERILE.
NOT MADE WITH NATURAL RUBBER LATEX.
USUAL DOSAGE: SEE INSERT
ADDITIVES SHOULD NOT BE MADE
TO THIS SOLUTION.
SINGLE DOSE CONTAINER. DISCARD UNUSED
PORTION. FOR INTRAVENOUS USE ONLY.
Rx only
3
V
CONTAINS DEHP
DISTRIBUTED BY
HOSPIRA, INC., LAKE FOREST,
IL 60045 USA
Hospira
14479101
IM-4471
Principal Display Panel 500 Ml Bag Label 1005
500mL
NDC 0409-1005-01
Heparin Sodium
1,000 USP Units / 500 mL
(2 USP Units / mL)
in 0.9% Sodium Chloride Injection
Each 100 mL contains Heparin Sodium 200 USP Units (Porcine
Intestinal Mucosa); Sodium Chloride 0.9 g;
Citric acid, Monohydrate 40 mg and Dibasic Sodium Phosphate,
Heptahydrate 434 mg added as buffers.
Sterile. Not made with natural rubber latex.
Recommended dosage: See prescribing information.
Additives should not be made to this solution.
Single dose container. Discard unused portion.
For Intravenous Use only. Inspect bag by squeezing firmly.
If leaks are found, discard. Store at 20℃ to 25℃ (68℉ to 77℉).
[See USP Controlled Room Temperature.] Protect from freezing.
Do not remove from overwrap until ready for use.
Rx only
5
PP
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 1000 Ml Bag Label 2222
2,000 USP Units / 1,000 mL
(2 USP Units / mL)
1,000mL
NDC 0409-2222-01
Heparin Sodium
2,000 USP Units / 1,000 mL
(2 USP Units / mL)
in 0.9% Sodium Chloride Injection
Each 100 mL contains Heparin Sodium 200 USP Units
(Porcine Intestinal Mucosa); Sodium Chloride 0.9 g;
Citric acid, Monohydrate 40 mg and Dibasic Sodium
Phosphate, Heptahydrate 434 mg added as buffers.
Sterile. Not made with natural rubber latex.
Recommended dosage: See prescribing information.
Additives should not be made to this solution.
Single dose container. Discard unused portion.
For Intravenous Use only.
Check for minute leaks. If leaks are found, discard.
Store at 20℃ to 25℃ (68℉ to 77℉). [See USP Controlled Room
Temperature.] Protect from freezing.
Do not remove from overwrap until ready for use.
Rx only
5
PP
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
7.4 Other Medications That May Interfere With Heparin
Digitalis, tetracyclines, nicotine or antihistamines may partially counteract the anticoagulant action of heparin sodium. Intravenous nitroglycerin administered to heparinized patients may result in a decrease of the partial thromboplastin time with subsequent rebound effect upon discontinuation of nitroglycerin. Careful monitoring of partial thromboplastin time and adjustment of heparin dosage are recommended during coadministration of heparin and intravenous nitroglycerin.
Antithrombin III (human) – The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. To reduce the risk of bleeding, a reduced dosage of heparin is recommended during treatment with antithrombin III (human).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long‑term studies in animals have been performed to evaluate carcinogenic potential of heparin. Also, no reproduction studies in animals have been performed concerning mutagenesis or impairment of fertility.
2.2 Recommended Dosage for Maintenance of Catheter Patency
The recommended starting dose is 6 units per hour by intravenous infusion through an intravenous catheter to maintain catheter patency.
5.7 Increased Risk of Bleeding in Older Patients, Especially Women
A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age [see Use in Specific Populations (8.5)].
5.3 Unresponsiveness to Heparin With Concomitant Use With Andexanet Alfa
Unresponsiveness to unfractionated heparin leading to non-prolongation of activated clotting times and serious thrombotic events has occurred when unfractionated heparin was administered after use of andexanet alfa for the reversal of direct Factor Xa inhibitors (apixaban and rivaroxaban). Avoid use of heparin after use of andexanet alfa. Use an alternative anticoagulant to heparin [see Drug Interactions (7.3)].
7.3 Unresponsiveness to Heparin With Concomitant Use With Andexanet Alfa
Andexanet binds to heparin-bound antithrombin III (ATIII) and may reduce the anticoagulant effect of heparin. Unresponsiveness to unfractionated heparin may lead to serious and life-threatening thrombotic events. Use of andexanet alfa as an antidote for heparin has not been established. Avoid use of heparin after use of andexanet alfa for the reversal of direct Factor Xa inhibitors (apixaban and rivaroxaban). If anticoagulation is needed, use an alternative anticoagulant to heparin [see Warnings and Precautions (5.3)].
5.2 Heparin Induced Thrombocytopenia and Heparin Induced Thrombocytopenia With Thrombosis
Heparin-induced thrombocytopenia (HIT) is a serious immune-mediated reaction. HIT occurs in patients treated with heparin and is due to the development of antibodies to a platelet Factor-4-heparin complex that induce in vivo platelet aggregation. HIT may progress to the development of venous and arterial thromboses, a condition referred to as heparin-induced thrombocytopenia with thrombosis (HITT). Thrombotic events may also be the initial presentation for HITT. These serious thromboembolic events include deep vein thrombosis, pulmonary embolism, cerebral vein thrombosis, limb ischemia, stroke, myocardial infarction, mesenteric thrombosis, thrombus formation on a prosthetic cardiac valve, renal arterial thrombosis, skin necrosis, gangrene of the extremities that may lead to amputation, and possibly death.
Once HIT or HITT is diagnosed or strongly suspected, discontinue all heparin sources (including heparin flushes) and use an alternative anticoagulant.
Immune-mediated HIT is diagnosed based on clinical findings supplemented by laboratory tests confirming the presence of antibodies to heparin, or platelet activation induced by heparin. Obtain platelet counts at baseline and periodically during heparin administration. A drop in platelet count greater than 50% from baseline is considered indicative of HIT. Platelet counts begin to fall 5 to 10 days after exposure to heparin in heparin–naive individuals and reach a threshold by days 7 to 14. In contrast, “rapid onset” HIT can occur very quickly (within 24 hours following heparin initiation), especially in patients with a recent exposure to heparin (i.e., previous 3 months).
Thrombosis development shortly after documenting thrombocytopenia is a characteristic finding in almost half of all patients with HIT.
If the platelet count falls below 100,000/mm3 or if recurrent thrombosis develops, promptly discontinue heparin, evaluate for HIT and HITT, and, if necessary, administer an alternative anticoagulant. HIT or HITT can occur up to several weeks after the discontinuation of heparin therapy. Patients presenting with thrombocytopenia or thrombosis after discontinuation of heparin sodium should be evaluated for HIT or HITT.
Structured Label Content
Section 42229-5 (42229-5)
To Open
Tear outer wrap and remove solution container.
Section 43683-2 (43683-2)
|
Warnings and Precautions, Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis (5.2) |
11/2024 |
|
Warnings and Precautions, Unresponsiveness to Heparin with Concomitant Use with Andexanet Alfa (5.3) |
11/2025 |
|
Warnings and Precautions, Heparin Resistance (5.5) |
11/2024 |
|
Warnings and Precautions, Hypersensitivity Reactions (5.6) |
11/2024 |
|
Warnings and Precautions, Elevations of Serum Aminotransferases (5.9) |
11/2024 |
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
10 Overdosage (10 OVERDOSAGE)
An overdose requires immediate medical attention and treatment.
Symptoms
Bleeding is the chief sign of heparin overdosage. Easy bruising, petechial formations, nosebleeds, blood in urine or tarry stools may be the first signs or symptoms of a heparin overdose. In the event of symptomatic heparin overdose, consider stopping heparin infusion.
11 Description (11 DESCRIPTION)
Intravenous solutions with heparin sodium (derived from porcine intestinal mucosa) are sterile, nonpyrogenic fluids for intravenous administration. Each 100 mL contains heparin sodium 200 USP Units; sodium chloride, 0.9 g; citric acid, monohydrate, 40 mg and dibasic sodium phosphate, heptahydrate, 434 mg added as buffers. Each liter contains the following electrolytes: Sodium 186.4 mEq; phosphate (as HPO4=) 32.4 mEq, citrate 5.7 mEq and chloride 154 mEq. Osmolar concentration, 378 mOsmol/liter (calc.); pH 7.0 (5.0 – 7.5).
Heparin Sodium, USP is a heterogeneous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans having anticoagulant properties. Although others may be present, the main sugars occurring in heparin are: (1) α-L-iduronic acid 2-sulfate, (2) 2-deoxy-2-sulfamino-α-D-glucose-6-sulfate, (3) β-D-glucuronic acid, (4) 2-acetamido-2-deoxy-α-D-glucose, and (5) α-L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2) > (1) > (4) > (3) > (5), and are joined by glycosidic linkages, forming polymers of varying sizes. Heparin is strongly acidic because of its content of covalently linked sulfate and carboxylic acid groups. In heparin sodium, the acidic protons of the sulfate units are partially replaced by sodium ions. The potency is determined by a biological assay using a USP reference standard based on units of heparin activity per milligram.
Structure of Heparin Sodium (representative subunits):
Sodium Chloride, USP is chemically designated NaCl, a white crystalline compound freely soluble in water.
Dibasic Sodium Phosphate, USP (heptahydrate), is chemically designated (Na2HPO4 ∙ 7H2O), colorless or white granular salt freely soluble in water.
Citric Acid, USP, hydrous (monohydrate) is chemically designated C6H8O7 ∙ H2O, colorless, translucent crystals or white crystalline powder very soluble in water. It has the following structural formula:
Water for Injection, USP is chemically designated H2O.
The flexible plastic container is fabricated from either polyvinylchloride or polyolefin plastic. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions inside the plastic container also can leach out certain of its chemical components in very small amounts before the expiration period is attained. However, the safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers.
5.1 Hemorrhage
Avoid using heparin in the presence of major bleeding, except when the benefits of heparin therapy outweigh the potential risks.
Hemorrhage can occur at virtually any site in patients receiving heparin. Fatal hemorrhages have occurred. An unexplained fall in hematocrit or fall in blood pressure, or any other unexplained symptom should lead to serious consideration of a hemorrhagic event.
Use heparin sodium with caution in disease states in which there is increased risk of hemorrhage, including:
-
•Cardiovascular — Subacute bacterial endocarditis. Severe hypertension.
-
•Surgical — During and immediately following (a) spinal tap or spinal anesthesia or (b) major surgery, especially involving the brain, spinal cord or eye.
-
•Hematologic — Conditions associated with increased bleeding tendencies, such as hemophilia, thrombocytopenia and some vascular purpuras.
-
•Gastrointestinal — Ulcerative lesions and continuous tube drainage of the stomach or small intestine.
-
•Patients with hereditary antithrombin III deficiency receiving concurrent antithrombin III therapy – The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. To reduce the risk of bleeding, reduce the heparin dose during concomitant treatment with antithrombin III (human).
-
•Other — Menstruation, liver disease with impaired hemostasis.
5.8 Hyperkalemia
Heparin can suppress adrenal secretion of aldosterone leading to hyperkalemia, particularly in patients with diabetes mellitus, chronic renal failure, pre-existing metabolic acidosis, a raised plasma potassium, or taking potassium sparing drugs. The risk of hyperkalemia appears to increase with duration of therapy but is usually reversible upon discontinuation of heparin.
Measure blood potassium in patients at risk of hyperkalemia before starting heparin therapy and periodically in all patients treated for more than 5 days or earlier as deemed fit by the clinician.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
A higher incidence of bleeding has been reported in patients over 60 years of age, especially women [see Warnings and Precautions (5.7)].
4 Contraindications (4 CONTRAINDICATIONS)
The use of Heparin Sodium in Sodium Chloride Injection is contraindicated in patients with the following conditions:
-
•Uncontrollable active bleeding state, except when this is due to disseminated intravascular coagulation [see Warnings and Precautions (5.1)]
-
•History of heparin-induced thrombocytopenia (HIT) and heparin-induced thrombocytopenia with thrombosis (HITT) [see Warnings and Precautions (5.2)]
-
•Severe thrombocytopenia [see Warnings and Precautions (5.2, 5.4)]
-
•Known hypersensitivity to heparin or pork products (e.g., anaphylactoid reactions) [see Warnings and Precautions (5.6), Adverse Reactions (6.1)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Hemorrhage [see Warnings and Precautions (5.1)]
-
•Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia with Thrombosis [see Warnings and Precautions (5.2)]
-
•Thrombocytopenia [see Warnings and Precautions (5.4)]
-
•Heparin Resistance [see Warnings and Precautions (5.5)]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.6)]
-
•Increased Risk of Bleeding in Older Patients, Especially Women [see Warnings and Precautions (5.7)]
-
•Hyperkalemia [see Warnings and Precautions (5.8)]
-
•Elevations of Serum Aminotransferases [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.4 Thrombocytopenia
Thrombocytopenia in patients receiving heparin has been reported at frequencies up to 30%. It can occur 2 to 20 days (average 5 to 9) following the onset of heparin therapy. Obtain platelet counts before and periodically during heparin therapy. If the count falls below 100,000/mm3 or if recurrent thrombosis develops, promptly discontinue heparin, evaluate for HIT and HITT, and, if necessary, administer an alternative anticoagulant [see Warnings and Precautions (5.2)].
12.2 Pharmacodynamics
Various times (activated clotting time, activated partial thromboplastin time, prothrombin time, whole blood clotting time) are prolonged by full therapeutic doses of heparin; in most cases, they are not measurably affected by low doses of heparin. Bleeding time is usually unaffected by heparin.
5.10 Laboratory Tests
Periodic platelet counts, hematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration.
5.5 Heparin Resistance
Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer, in postsurgical patients, and patients with antithrombin deficiency. Consider measurement of antithrombin levels if heparin resistance is suspected. Monitor coagulation tests frequently in such patients. It may be necessary to adjust the dose of heparin based on coagulation test monitoring, such as anti-Factor Xa levels and/or partial thromboplastin time.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Heparin Sodium in Sodium Chloride Injection at a concentration of 2 units/mL is indicated as an anticoagulant to maintain catheter patency.
7.1 Oral Anticoagulants
Heparin sodium may prolong the one-stage prothrombin time. Therefore, when heparin sodium is given with dicumarol or warfarin sodium, a period of at least 5 hours after the last intravenous dose or 24 hours after the last subcutaneous dose should elapse before blood is drawn if a valid prothrombin time is to be obtained.
7.2 Platelet Inhibitors
Drugs such as NSAIDS (including acetylsalicylic acid, ibuprofen, indomethacin, and celecoxib), dextran, phenylbutazone, thienopyridines, dipyridamole, hydroxychloroquine, glycoprotein IIv/IIa antagonists (including abciximab, eptifobatide, and tirofiban), and others that interfere with platelet-aggregation reactions (the main hemostatic defense of heparinized patients) may induce bleeding and should be used with caution in patients receiving heparin sodium. To reduce the risk of bleeding, a reduction in the dose of the antiplatelet agent or heparin is recommended.
12.1 Mechanism of Action
Heparin interacts with the naturally occurring plasma protein, antithrombin III, to induce a conformational change, which markedly enhances the serine protease activity of Antithrombin II, thereby inhibiting the activated coagulation factors involved in the closing sequence, particularly Xa and IIa. Small amounts of heparin inhibit Factor Xa, and larger amounts inhibit thrombin (Factor IIa).
Heparin also prevents the formation of a stable fibrin clot by inhibiting the activation of the fibrin stabilizing factor. Heparin does not have fibrinolytic activity; therefore, it will not lyse existing clots.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Hemorrhage: Fatal hemorrhages have occurred. Monitor for signs of bleeding and manage promptly. (5.1)
-
•HIT and HITT: Monitor for signs and symptoms and discontinue if indicative of HIT or HITT. (5.2)
-
•Thrombocytopenia: Monitor platelet count during therapy; discontinue heparin if HIT or HITT is suspected. (5.4)
-
•Heparin Resistance: Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in postsurgical patients. (5.5)
-
•Hypersensitivity Reactions: Use in patients with prior reactions only in life-threatening situations. (5.6)
-
•Increased Risk of Bleeding in Older Patients, Especially Women: A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age. (5.7)
-
•Hyperkalemia: Measure blood potassium in patients at risk of hyperkalemia before starting heparin therapy and periodically in all patients. (5.8)
-
•Elevations of Serum Aminotransferases: Interpret elevation of these enzymes with caution. (5.9)
-
•Laboratory Tests: Periodic platelet counts, hematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Infuse through intravenous catheter at a rate of 6 units per hour. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Heparin Sodium in 0.9% Sodium Chloride Injection is available as:
-
•Injection: 1,000 USP Units/500 mL (2 USP Units/mL) clear solution in a single-dose flexible plastic container
-
•Injection: 2,000 USP Units/1,000 mL (2 USP Units/mL) clear solution in a single-dose flexible plastic container
6.1 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of heparin sodium. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.6 Hypersensitivity Reactions
Hypersensitivity reactions with chills, fever, and urticaria as the most usual manifestations and also asthma, rhinitis, lacrimation, and anaphylactoid reactions have been reported. Patients with documented hypersensitivity to heparin should be given the drug only in clearly life-threatening situations. Because Heparin Sodium in Sodium Chloride Injection is derived from animal tissue, it should be used with caution in patients with a history of allergy to pork products.
2.1 Preparation for Administration
Do not administer unless solution is clear and seal is intact. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Warning: Do not use plastic containers in series connection. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Do not use Heparin Sodium in Sodium Chloride Injection as a “catheter lock flush” product.
Do not admix with other drugs. Discard unused portion.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Heparin Sodium in 0.9% Sodium Chloride Injection is a clear solution and is available in single-dose containers as follows:
|
Unit of Sale |
Concentration |
|
NDC 0409-7620-03 Case of 18 Single-dose flexible plastic containers |
1,000 USP Units/500 mL (2 USP Units/mL) |
|
NDC 0409-1005-20 Case of 20 Single-dose flexible plastic containers |
1,000 USP Units/500 mL (2 USP Units/mL) |
|
NDC 0409-7620-59 Case of 12 Single-dose flexible plastic containers |
2,000 USP Units/1,000 mL (2 USP Units/mL) |
|
NDC 0409-2222-12 Case of 12 Single-dose flexible plastic containers |
2,000 USP Units/1,000 mL (2 USP Units/mL) |
Principal Display Panel Bag Overwrap (PRINCIPAL DISPLAY PANEL - Bag Overwrap)
TO OPEN TEAR AT NOTCH
2
HDPE
DO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER
FIRMLY. IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE
IMPAIRED. RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID
EXCESSIVE HEAT. PROTECT FROM FREEZING. SEE INSERT.
IM-4498
Not Made With Natural Rubber Latex
14622100
5.9 Elevations of Serum Aminotransferases
Significant elevations of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels have occurred in patients who have received heparin. Elevation of these enzymes in patients receiving heparin should be interpreted with caution. These elevations typically resolve upon heparin discontinuation.
Principal Display Panel 500 Ml Bag 7620 (PRINCIPAL DISPLAY PANEL - 500 mL Bag - 7620)
1,000 USP Units/500 mL
(2 USP Units/mL)
500 mL
NDC 0409-7620-13
HEPARIN SODIUM
1,000 USP Units/500 mL
(2 USP Units/mL)
in 0.9% Sodium Chloride Injection
EACH 100 mL CONTAINS HEPARIN SODIUM 200 USP UNITS
(PORCINE INTESTINAL MUCOSA); SODIUM CHLORIDE 0.9 g;
CITRIC ACID, MONOHYDRATE 40 mg AND DIBASIC SODIUM
PHOSPHATE, HEPTAHYDRATE 434 mg ADDED AS BUFFERS.
STERILE. NOT MADE WITH NATURAL RUBBER LATEX.
USUAL DOSAGE: SEE INSERT
ADDITIVES SHOULD NOT BE MADE TO THIS SOLUTION.
SINGLE DOSE CONTAINER. DISCARD UNUSED PORTION. FOR
INTRAVENOUS USE ONLY.
3
V
CONTAINS DEHP
Rx only
DISTRIBUTED BY
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira
14478901
IM-4470
Principal Display Panel 1,000 Ml Bag 7620 (PRINCIPAL DISPLAY PANEL - 1,000 mL Bag - 7620)
LOT NO.
EXP. DATE
2,000 USP Units /1,000 mL
(2 USP Units/mL)
1,000 mL
NDC 0409-7620-49
HEPARIN SODIUM
2,000 USP Units
per 1,000 mL
(2 USP Units/mL)
in 0.9% Sodium Chloride Injection
EACH 100 mL CONTAINS HEPARIN SODIUM
200 USP UNITS (PORCINE INTESTINAL
MUCOSA); SODIUM CHLORIDE 0.9 g; CITRIC
ACID, MONOHYDRATE 40 mg AND DIBASIC
SODIUM PHOSPHATE, HEPTAHYDRATE
434 mg ADDED AS BUFFERS. STERILE.
NOT MADE WITH NATURAL RUBBER LATEX.
USUAL DOSAGE: SEE INSERT
ADDITIVES SHOULD NOT BE MADE
TO THIS SOLUTION.
SINGLE DOSE CONTAINER. DISCARD UNUSED
PORTION. FOR INTRAVENOUS USE ONLY.
Rx only
3
V
CONTAINS DEHP
DISTRIBUTED BY
HOSPIRA, INC., LAKE FOREST,
IL 60045 USA
Hospira
14479101
IM-4471
Principal Display Panel 500 Ml Bag Label 1005 (PRINCIPAL DISPLAY PANEL - 500 mL Bag Label - 1005)
500mL
NDC 0409-1005-01
Heparin Sodium
1,000 USP Units / 500 mL
(2 USP Units / mL)
in 0.9% Sodium Chloride Injection
Each 100 mL contains Heparin Sodium 200 USP Units (Porcine
Intestinal Mucosa); Sodium Chloride 0.9 g;
Citric acid, Monohydrate 40 mg and Dibasic Sodium Phosphate,
Heptahydrate 434 mg added as buffers.
Sterile. Not made with natural rubber latex.
Recommended dosage: See prescribing information.
Additives should not be made to this solution.
Single dose container. Discard unused portion.
For Intravenous Use only. Inspect bag by squeezing firmly.
If leaks are found, discard. Store at 20℃ to 25℃ (68℉ to 77℉).
[See USP Controlled Room Temperature.] Protect from freezing.
Do not remove from overwrap until ready for use.
Rx only
5
PP
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 1000 Ml Bag Label 2222 (PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - 2222)
2,000 USP Units / 1,000 mL
(2 USP Units / mL)
1,000mL
NDC 0409-2222-01
Heparin Sodium
2,000 USP Units / 1,000 mL
(2 USP Units / mL)
in 0.9% Sodium Chloride Injection
Each 100 mL contains Heparin Sodium 200 USP Units
(Porcine Intestinal Mucosa); Sodium Chloride 0.9 g;
Citric acid, Monohydrate 40 mg and Dibasic Sodium
Phosphate, Heptahydrate 434 mg added as buffers.
Sterile. Not made with natural rubber latex.
Recommended dosage: See prescribing information.
Additives should not be made to this solution.
Single dose container. Discard unused portion.
For Intravenous Use only.
Check for minute leaks. If leaks are found, discard.
Store at 20℃ to 25℃ (68℉ to 77℉). [See USP Controlled Room
Temperature.] Protect from freezing.
Do not remove from overwrap until ready for use.
Rx only
5
PP
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
7.4 Other Medications That May Interfere With Heparin (7.4 Other Medications that May Interfere With Heparin)
Digitalis, tetracyclines, nicotine or antihistamines may partially counteract the anticoagulant action of heparin sodium. Intravenous nitroglycerin administered to heparinized patients may result in a decrease of the partial thromboplastin time with subsequent rebound effect upon discontinuation of nitroglycerin. Careful monitoring of partial thromboplastin time and adjustment of heparin dosage are recommended during coadministration of heparin and intravenous nitroglycerin.
Antithrombin III (human) – The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. To reduce the risk of bleeding, a reduced dosage of heparin is recommended during treatment with antithrombin III (human).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long‑term studies in animals have been performed to evaluate carcinogenic potential of heparin. Also, no reproduction studies in animals have been performed concerning mutagenesis or impairment of fertility.
2.2 Recommended Dosage for Maintenance of Catheter Patency
The recommended starting dose is 6 units per hour by intravenous infusion through an intravenous catheter to maintain catheter patency.
5.7 Increased Risk of Bleeding in Older Patients, Especially Women
A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age [see Use in Specific Populations (8.5)].
5.3 Unresponsiveness to Heparin With Concomitant Use With Andexanet Alfa (5.3 Unresponsiveness to Heparin with Concomitant Use With Andexanet Alfa)
Unresponsiveness to unfractionated heparin leading to non-prolongation of activated clotting times and serious thrombotic events has occurred when unfractionated heparin was administered after use of andexanet alfa for the reversal of direct Factor Xa inhibitors (apixaban and rivaroxaban). Avoid use of heparin after use of andexanet alfa. Use an alternative anticoagulant to heparin [see Drug Interactions (7.3)].
7.3 Unresponsiveness to Heparin With Concomitant Use With Andexanet Alfa (7.3 Unresponsiveness to Heparin with Concomitant Use With Andexanet Alfa)
Andexanet binds to heparin-bound antithrombin III (ATIII) and may reduce the anticoagulant effect of heparin. Unresponsiveness to unfractionated heparin may lead to serious and life-threatening thrombotic events. Use of andexanet alfa as an antidote for heparin has not been established. Avoid use of heparin after use of andexanet alfa for the reversal of direct Factor Xa inhibitors (apixaban and rivaroxaban). If anticoagulation is needed, use an alternative anticoagulant to heparin [see Warnings and Precautions (5.3)].
5.2 Heparin Induced Thrombocytopenia and Heparin Induced Thrombocytopenia With Thrombosis (5.2 Heparin-Induced Thrombocytopenia and Heparin-Induced Thrombocytopenia With Thrombosis)
Heparin-induced thrombocytopenia (HIT) is a serious immune-mediated reaction. HIT occurs in patients treated with heparin and is due to the development of antibodies to a platelet Factor-4-heparin complex that induce in vivo platelet aggregation. HIT may progress to the development of venous and arterial thromboses, a condition referred to as heparin-induced thrombocytopenia with thrombosis (HITT). Thrombotic events may also be the initial presentation for HITT. These serious thromboembolic events include deep vein thrombosis, pulmonary embolism, cerebral vein thrombosis, limb ischemia, stroke, myocardial infarction, mesenteric thrombosis, thrombus formation on a prosthetic cardiac valve, renal arterial thrombosis, skin necrosis, gangrene of the extremities that may lead to amputation, and possibly death.
Once HIT or HITT is diagnosed or strongly suspected, discontinue all heparin sources (including heparin flushes) and use an alternative anticoagulant.
Immune-mediated HIT is diagnosed based on clinical findings supplemented by laboratory tests confirming the presence of antibodies to heparin, or platelet activation induced by heparin. Obtain platelet counts at baseline and periodically during heparin administration. A drop in platelet count greater than 50% from baseline is considered indicative of HIT. Platelet counts begin to fall 5 to 10 days after exposure to heparin in heparin–naive individuals and reach a threshold by days 7 to 14. In contrast, “rapid onset” HIT can occur very quickly (within 24 hours following heparin initiation), especially in patients with a recent exposure to heparin (i.e., previous 3 months).
Thrombosis development shortly after documenting thrombocytopenia is a characteristic finding in almost half of all patients with HIT.
If the platelet count falls below 100,000/mm3 or if recurrent thrombosis develops, promptly discontinue heparin, evaluate for HIT and HITT, and, if necessary, administer an alternative anticoagulant. HIT or HITT can occur up to several weeks after the discontinuation of heparin therapy. Patients presenting with thrombocytopenia or thrombosis after discontinuation of heparin sodium should be evaluated for HIT or HITT.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:52.030266 · Updated: 2026-03-14T22:38:43.060348