180 Readybath Luxe (scented)
32bc07dc-7a9c-63ed-e054-00144ff8d46c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzalkonium chloride 0.12%
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Benzalkonium chloride 0.12%
Uses
- for body cleansing to decrease bacteria on the skin
- helps kill germs that can cause odor
Warnings
For external use only
Directions
- gently pull label tab until package opening is exposed
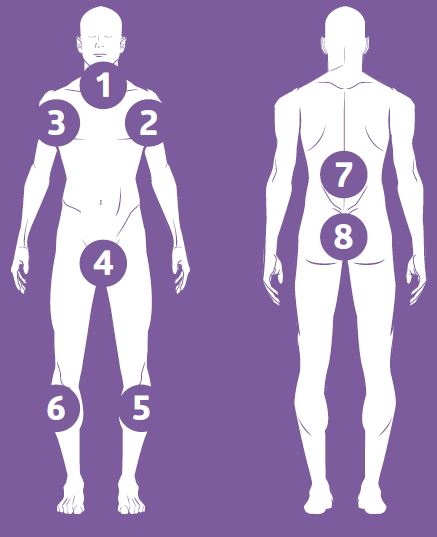
- remove cloths one at a time to cleanse body parts in the order of the diagram
- allow skin to dry
- dispose of cloth in waste receptacle
- do not flush cloths in toilet
Do Not Use
- in the eyes
Package Label
Active Ingredient
Benzalkonium chloride 0.12%
Other Information
- avoid excessive heat, protect from freezing
- washcloth: rayon/polyester
Inactive Ingredients
water (aqua), cocamidopropyl PG-dimonium chloride phosphate, glycerin, phenoxyethanol, benzoic acid, dehydroacetic acid, ethylhexylglycerin, disodium EDTA, polysorbate 20, sodium citrate, fragrance, simethicone, aloe barbadensis leaf juice
Manufacturing Information
Manufactured by:
Medline Industries, LP
Three Lakes Drive, Northfield, IL 60093 USA
Made in USA with domestic and foreign materials
www.medline.com
1-800-MEDLINE (633-5463)
REF: MSC095100
V1 RK21DYN
Stop Use and Ask A Doctor If
- irritation and redness develop
- the condition persists for more than 72 hours or gets worse
Keep Out of Reach of Children.
If swallowed get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have
- deep or puncture wounds
- animal bites
- serious burns
Structured Label Content
Uses
- for body cleansing to decrease bacteria on the skin
- helps kill germs that can cause odor
Purpose
Antiseptic
Warnings
For external use only
Directions
- gently pull label tab until package opening is exposed
- remove cloths one at a time to cleanse body parts in the order of the diagram
- allow skin to dry
- dispose of cloth in waste receptacle
- do not flush cloths in toilet
Do Not Use (Do not use)
- in the eyes
Package Label
Active Ingredient (Active ingredient)
Benzalkonium chloride 0.12%
Other Information (Other information)
- avoid excessive heat, protect from freezing
- washcloth: rayon/polyester
Inactive Ingredients (Inactive ingredients)
water (aqua), cocamidopropyl PG-dimonium chloride phosphate, glycerin, phenoxyethanol, benzoic acid, dehydroacetic acid, ethylhexylglycerin, disodium EDTA, polysorbate 20, sodium citrate, fragrance, simethicone, aloe barbadensis leaf juice
Manufacturing Information
Manufactured by:
Medline Industries, LP
Three Lakes Drive, Northfield, IL 60093 USA
Made in USA with domestic and foreign materials
www.medline.com
1-800-MEDLINE (633-5463)
REF: MSC095100
V1 RK21DYN
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- irritation and redness develop
- the condition persists for more than 72 hours or gets worse
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- deep or puncture wounds
- animal bites
- serious burns
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:17.853814 · Updated: 2026-03-14T23:08:59.288880