These Highlights Do Not Include All The Information Needed To Use Tenofovir Disoproxil Fumarate Tablets Safely And Effectively. See Full Prescribing Information For Tenofovir Disoproxil Fumarate Tablets. Tenofovir Disoproxil Fumarate Tablets, For Oral Use Initial U.s. Approval: 2001
3285f345-e58d-4831-a6a8-31094c8c546d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Severe acute exacerbations of hepatitis B virus (HBV) have been reported in HBV-infected patients who have discontinued anti-hepatitis B therapy, including tenofovir disoproxil fumarate. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in HBV-infected patients who discontinue anti-hepatitis B therapy, including tenofovir disoproxil fumarate. If appropriate, resumption of anti-hepatitis B therapy may be warranted [See Warnings and Precautions (5.1) ] .
Indications and Usage
Tenofovir disoproxil fumarate is a nucleotide analog HIV-1 reverse transcriptase inhibitor and an HBV reverse transcriptase inhibitor and is indicated: in combination with other antiretroviral agents for the treatment of HIV-1 infection in adults and pediatric patients 2 years of age and older weighing at least 35 kg. ( 1.1 ) for the treatment of chronic hepatitis B in adults and pediatric patients 2 years and older weighing at least 35 kg. ( 1.2 )
Dosage and Administration
Testing: Prior to or when initiating tenofovir disoproxil fumarate test for hepatitis B virus infection and HIV-1 infection. Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorous. ( 2.1 ) Recommended tablet dosage in adults and pediatric patients weighing at least 35 kg: One tenofovir disoproxil fumarate 300 mg tablet once daily taken orally without regard to food. ( 2.2 ) Recommended dosage in pediatric patients at least 2 years of age and adults: For patients weighing at least 35 kg who can swallow an intact tablet, one tenofovir disoproxil fumarate tablet (300 mg based on body weight) once daily taken orally without regard to food. ( 2.2 ) Recommended dosage in renally impaired adult patients: Creatinine clearance (CrCl) 30-49 mL/min: 300 mg every 48 hours. ( 2.4 ) CrCl 10-29 mL/min: 300 mg every 72 to 96 hours. ( 2.4 ) Hemodialysis: 300 mg every 7 days or after approximately 12 hours of dialysis. ( 2.4 )
Warnings and Precautions
New onset or worsening renal impairment: Can include acute renal failure and Fanconi syndrome. Avoid administering tenofovir disoproxil fumarate with concurrent or recent use of nephrotoxic drugs.( 5.2 ) HIV testing: HIV antibody testing should be offered to all HBV-infected patients before initiating therapy with tenofovir disoproxil fumarate. Tenofovir disoproxil fumarate should only be used as part of an appropriate antiretroviral combination regimen in HIV-infected patients with or without HBV coinfection. ( 5.3 ) Immune reconstitution syndrome: May necessitate further evaluation and treatment. ( 5.4 ) Decreases in bone mineral density (BMD): Consider assessment of BMD in patients with a history of pathologic fracture or other risk factors for osteoporosis or bone loss. ( 5.5 ) Lactic acidosis/severe hepatomegaly with steatosis: Discontinue treatment in patients who develop symptoms or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity. ( 5.6 )
Contraindications
None.
Adverse Reactions
The concomitant use of tenofovir disoproxil fumarate and other drugs may result in known or potentially significant drug interactions, some of which may lead to possible clinically significant adverse reactions from greater exposures of concomitant drugs [see Drug Interactions (7.2) ] . See Table 12 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during therapy with tenofovir disoproxil fumarate; review concomitant medications during therapy with tenofovir disoproxil fumarate; and monitor for adverse reactions associated with the concomitant drugs.
Drug Interactions
The concomitant use of tenofovir disoproxil fumarate and other drugs may result in known or potentially significant drug interactions, some of which may lead to possible clinically significant adverse reactions from greater exposures of concomitant drugs [see Drug Interactions (7.2) ] . See Table 12 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during therapy with tenofovir disoproxil fumarate; review concomitant medications during therapy with tenofovir disoproxil fumarate; and monitor for adverse reactions associated with the concomitant drugs.
Storage and Handling
Tenofovir disoproxil fumarate tablets, 300 mg, are almond-shaped, blue, film-coated tablets containing 300 mg of tenofovir DF, which is equivalent to 245 mg of tenofovir disoproxil, and are debossed with “32” on one side. NDC: 71335-2364-1: 30 TABLETs in a BOTTLE NDC: 71335-2364-2: 15 TABLETs in a BOTTLE Store tenofovir disoproxil fumarate tablets at 25°C (77°F), excursions permitted to 15-30°C(59-86°F) (see USP Controlled Room Temperature). • Keep container tightly closed. • Dispense only in original container. • Do not use if seal over bottle opening is broken or missing. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
How Supplied
Tenofovir disoproxil fumarate tablets, 300 mg, are almond-shaped, blue, film-coated tablets containing 300 mg of tenofovir DF, which is equivalent to 245 mg of tenofovir disoproxil, and are debossed with “32” on one side. NDC: 71335-2364-1: 30 TABLETs in a BOTTLE NDC: 71335-2364-2: 15 TABLETs in a BOTTLE Store tenofovir disoproxil fumarate tablets at 25°C (77°F), excursions permitted to 15-30°C(59-86°F) (see USP Controlled Room Temperature). • Keep container tightly closed. • Dispense only in original container. • Do not use if seal over bottle opening is broken or missing. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Medication Information
Warnings and Precautions
New onset or worsening renal impairment: Can include acute renal failure and Fanconi syndrome. Avoid administering tenofovir disoproxil fumarate with concurrent or recent use of nephrotoxic drugs.( 5.2 ) HIV testing: HIV antibody testing should be offered to all HBV-infected patients before initiating therapy with tenofovir disoproxil fumarate. Tenofovir disoproxil fumarate should only be used as part of an appropriate antiretroviral combination regimen in HIV-infected patients with or without HBV coinfection. ( 5.3 ) Immune reconstitution syndrome: May necessitate further evaluation and treatment. ( 5.4 ) Decreases in bone mineral density (BMD): Consider assessment of BMD in patients with a history of pathologic fracture or other risk factors for osteoporosis or bone loss. ( 5.5 ) Lactic acidosis/severe hepatomegaly with steatosis: Discontinue treatment in patients who develop symptoms or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity. ( 5.6 )
Indications and Usage
Tenofovir disoproxil fumarate is a nucleotide analog HIV-1 reverse transcriptase inhibitor and an HBV reverse transcriptase inhibitor and is indicated: in combination with other antiretroviral agents for the treatment of HIV-1 infection in adults and pediatric patients 2 years of age and older weighing at least 35 kg. ( 1.1 ) for the treatment of chronic hepatitis B in adults and pediatric patients 2 years and older weighing at least 35 kg. ( 1.2 )
Dosage and Administration
Testing: Prior to or when initiating tenofovir disoproxil fumarate test for hepatitis B virus infection and HIV-1 infection. Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorous. ( 2.1 ) Recommended tablet dosage in adults and pediatric patients weighing at least 35 kg: One tenofovir disoproxil fumarate 300 mg tablet once daily taken orally without regard to food. ( 2.2 ) Recommended dosage in pediatric patients at least 2 years of age and adults: For patients weighing at least 35 kg who can swallow an intact tablet, one tenofovir disoproxil fumarate tablet (300 mg based on body weight) once daily taken orally without regard to food. ( 2.2 ) Recommended dosage in renally impaired adult patients: Creatinine clearance (CrCl) 30-49 mL/min: 300 mg every 48 hours. ( 2.4 ) CrCl 10-29 mL/min: 300 mg every 72 to 96 hours. ( 2.4 ) Hemodialysis: 300 mg every 7 days or after approximately 12 hours of dialysis. ( 2.4 )
Contraindications
None.
Adverse Reactions
The concomitant use of tenofovir disoproxil fumarate and other drugs may result in known or potentially significant drug interactions, some of which may lead to possible clinically significant adverse reactions from greater exposures of concomitant drugs [see Drug Interactions (7.2) ] . See Table 12 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during therapy with tenofovir disoproxil fumarate; review concomitant medications during therapy with tenofovir disoproxil fumarate; and monitor for adverse reactions associated with the concomitant drugs.
Drug Interactions
The concomitant use of tenofovir disoproxil fumarate and other drugs may result in known or potentially significant drug interactions, some of which may lead to possible clinically significant adverse reactions from greater exposures of concomitant drugs [see Drug Interactions (7.2) ] . See Table 12 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during therapy with tenofovir disoproxil fumarate; review concomitant medications during therapy with tenofovir disoproxil fumarate; and monitor for adverse reactions associated with the concomitant drugs.
Storage and Handling
Tenofovir disoproxil fumarate tablets, 300 mg, are almond-shaped, blue, film-coated tablets containing 300 mg of tenofovir DF, which is equivalent to 245 mg of tenofovir disoproxil, and are debossed with “32” on one side. NDC: 71335-2364-1: 30 TABLETs in a BOTTLE NDC: 71335-2364-2: 15 TABLETs in a BOTTLE Store tenofovir disoproxil fumarate tablets at 25°C (77°F), excursions permitted to 15-30°C(59-86°F) (see USP Controlled Room Temperature). • Keep container tightly closed. • Dispense only in original container. • Do not use if seal over bottle opening is broken or missing. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
How Supplied
Tenofovir disoproxil fumarate tablets, 300 mg, are almond-shaped, blue, film-coated tablets containing 300 mg of tenofovir DF, which is equivalent to 245 mg of tenofovir disoproxil, and are debossed with “32” on one side. NDC: 71335-2364-1: 30 TABLETs in a BOTTLE NDC: 71335-2364-2: 15 TABLETs in a BOTTLE Store tenofovir disoproxil fumarate tablets at 25°C (77°F), excursions permitted to 15-30°C(59-86°F) (see USP Controlled Room Temperature). • Keep container tightly closed. • Dispense only in original container. • Do not use if seal over bottle opening is broken or missing. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Description
Severe acute exacerbations of hepatitis B virus (HBV) have been reported in HBV-infected patients who have discontinued anti-hepatitis B therapy, including tenofovir disoproxil fumarate. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in HBV-infected patients who discontinue anti-hepatitis B therapy, including tenofovir disoproxil fumarate. If appropriate, resumption of anti-hepatitis B therapy may be warranted [See Warnings and Precautions (5.1) ] .
Section 34077-8
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to tenofovir disoproxil fumarate during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Section 42229-5
The recommended dosage of tenofovir disoproxil fumarate in adults and pediatric patients weighing at least 35 kg is one 300 mg tablet taken orally once daily without regard to food. The dosage for tenofovir disoproxil fumarate is the same for both HIV and HBV indications.
The recommended dosage of tenofovir disoproxil fumarate tablet in adults and pediatric patients 2 years and older weighing at least 35 kg is 8 mg of tenofovir disoproxil fumarate (TDF) per kg of body weight (up to a maximum of 300 mg) once daily. Dosage for pediatric patients 2 years and older weighing at least 35 kg and able to swallow an intact tablet is provided in Table 1. Weight should be monitored periodically and the tenofovir disoproxil fumarate dose adjusted accordingly.
| Body Weight (kg) | Dosing of Tenofovir Disoproxil Fumarate Tablets |
|---|---|
| at least 35 | one 300 mg tablet once daily |
Section 42230-3
| Patient Information
Tenofovir Disoproxil Fumarate (ten-OF-oh-vir dye-soe-PROX-il FUE-ma-rate) Tablets |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: March 2023 |
| Read this Patient Information before you start taking tenofovir disoproxil fumarate tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. | |
|
What is the most important information I should know about tenofovir disoproxil fumarate tablets? |
|
| Tenofovir disoproxil fumarate tablets can cause serious side effects, including: | |
|
|
| For more information about side effects, see “ What are the possible side effects of tenofovir disoproxil fumarate tablets?” | |
| What are tenofovir disoproxil fumarate tablets? | |
| Tenofovir disoproxil fumarate tablets are a prescription medicine that is used to: | |
|
|
| What should I tell my healthcare provider before taking tenofovir disoproxil fumarate tablets? | |
| Before you take tenofovir disoproxil fumarate tablets, tell your healthcare provider about all of your medical conditions, including if you: | |
|
|
| Tell your healthcare provider about all the medicines you take,including prescription and non-prescription medicines, vitamins, and herbal supplements. | |
| Some medicines may interact with tenofovir disoproxil fumarate tablets. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine. | |
|
|
| How should I take tenofovir disoproxil fumarate tablets? | |
|
|
| What are the possible side effects of tenofovir disoproxil fumarate tablets? | |
| Tenofovir disoproxil fumarate tablets may cause serious side effects, including: | |
|
|
| The most common side effects in all people taking tenofovir disoproxil fumarate tablets are: | |
|
|
| In some people with advanced HBV-infection, other common side effects may include: | |
|
|
|
These are not all the possible side effects of tenofovir disoproxil fumarate tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
How should I store tenofovir disoproxil fumarate tablets?
Keep tenofovir disoproxil fumarate tablets and all medicines out of the reach of children. |
|
| General information about the safe and effective use of tenofovir disoproxil fumarate tablets: | |
|
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use tenofovir disoproxil fumarate tablets for a condition for which it was not prescribed. Do not give tenofovir disoproxil fumarate tablets to other people, even if they have the same condition you have. It may harm them. You can ask your pharmacist or healthcare provider for information about tenofovir disoproxil fumarate tablets that is written for health professionals. A vaccine is available to protect people at risk for becoming infected with HBV. You can ask your healthcare provider for information about this vaccine. |
|
| What are the ingredients in tenofovir disoproxil fumarate tablets? | |
| Active ingredient:tenofovir disoproxil fumarate | |
| Inactive ingredients: | |
| Tenofovir disoproxil fumarate tablets: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and pregelatinized starch. | |
| Tablet coating: | |
| Tenofovir disoproxil fumarate tablets 300 mg: Opadry II 32K605004, which contains FD&C blue #2 aluminum lake, hypromellose 2910, lactose monohydrate, titanium dioxide, and triacetin. | |
|
Manufactured by:
Manufactured for:
|
Section 51945-4
Tenofovir Disoproxil Fumarate Tablets
Animal Data
TDF was administered orally to pregnant rats (at 0, 50, 150, or 450 mg/kg/day) and rabbits (at 0, 30, 100, or 300 mg/kg/day) through organogenesis (on gestation days 7 through 17, and 6 through 18, respectively). No significant toxicological effects were observed in embryo-fetal toxicity studies performed with TDF in rats at doses up to 14 times the human dose based on body surface area comparisons and in rabbits at doses up to 19 times the human dose based on body surface area comparisons. In a pre/postnatal development study in rats, TDF was administered orally through lactation at doses up to 600 mg/kg/day; no adverse effects were observed in the offspring at tenofovir exposures of approximately 2.7 times higher than human exposures at the recommended daily dose of tenofovir disoproxil fumarate.
Mutagenesis
Tenofovir disoproxil fumarate was mutagenic in the in vitro mouse lymphoma assay and negative in an in vitro bacterial mutagenicity test (Ames test). In an in vivo mouse micronucleus assay, TDF was negative when administered to male mice.
10 Overdosage
If overdose occurs, the patient must be monitored for evidence of toxicity, and standard supportive treatment applied as necessary.
Tenofovir is efficiently removed by hemodialysis with an extraction coefficient of approximately 54%. Following a single 300 mg dose of tenofovir disoproxil fumarate, a four-hour hemodialysis session removed approximately 10% of the administered tenofovir dose.
8.2 Lactation
Risk Summary
Based on published data, tenofovir has been shown to be present in human breast milk (see Data). It is not known if tenofovir affects milk production or has effects on the breastfed child.
Treatment of HIV-1 infection:
The Centers for Disease Control and Prevention recommend that HIV-1 infected mothers not breastfeed their infants to avoid risking postnatal transmission of HIV-1.
Because of the potential for: (1) HIV transmission (in HIV-negative infants); (2) developing viral resistance (in HIV-positive infants); and (3) adverse reactions in a breastfed infant similar to those seen in adults, instruct mothers not to breastfeed if they are taking tenofovir disoproxil fumarate for the treatment of HIV-1.
Treatment of HBV infection:
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for tenofovir disoproxil fumarate and any potential adverse effects on the breastfed infant from tenofovir disoproxil fumarate or from the underlying maternal condition.
Data
In a study of 50 HIV-uninfected, breastfeeding women on a tenofovir-containing regimen initiated between 1 and 24 weeks postpartum (median 13 weeks), tenofovir was undetectable in the plasma of most infants after 7 days of treatment in mothers. There were no serious adverse events in mothers or infants.
11 Description
Tenofovir disoproxil fumarate (TDF) (a prodrug of tenofovir) is a fumaric acid salt of bis-isopropoxycarbonyloxymethyl ester derivative of tenofovir. TDF is converted in vivo to tenofovir, an acyclic nucleoside phosphonate (nucleotide) analog of adenosine 5’-monophosphate. Tenofovir exhibits activity against HIV-1 reverse transcriptase.
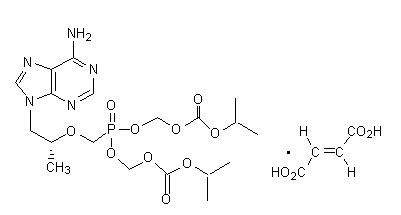
The chemical name of TDF is 9-[( R)-2-[[bis[[(isopropoxycarbonyl)oxy]methoxy]phosphinyl]methoxy]propyl]adenine fumarate (1:1). It has a molecular formula of C 19H 30N 5O 10P • C 4H 4O 4and a molecular weight of 635.52. It has the following structural formula:
Tenofovir disoproxil fumarate is a white to off-white crystalline powder with a solubility of 13.4 mg/mL in distilled water at 25 °C. It has an octanol/phosphate buffer (pH 6.5) partition coefficient (log p) of 1.25 at 25 °C.
Tenofovir disoproxil fumarate is available as tablets.
Tenofovir disoproxil fumarate tablets are for oral administration and are available in the strength of 300 mg of TDF (equivalent to 245 mg of tenofovir disoproxil).
Tenofovir disoproxil fumarate tablets contain the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The 300 mg strength tablets are coated with Opadry II 32K605004, which contains FD&C blue #2 aluminum lake, hypromellose 2910, lactose monohydrate, titanium dioxide, and triacetin.
In this insert, all dosages are expressed in terms of TDF except where otherwise noted.
Carcinogenesis
Long-term oral carcinogenicity studies of TDF in mice and rats were carried out at exposures up to approximately 16 times (mice) and 5 times (rats) those observed in humans at the therapeutic dose for HIV-1 infection. At the high dose in female mice, liver adenomas were increased at exposures 16 times that in humans. In rats, the study was negative for carcinogenic findings at exposures up to 5 times that observed in humans at the therapeutic dose.
8.5 Geriatric Use
Clinical trials of tenofovir disoproxil fumarate did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for the elderly patient should be cautious, keeping in mind the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
1.1 Hiv 1 Infection
Tenofovir disoproxil fumarate is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in adults and pediatric patients 2 years of age and older weighing at least 35 kg.
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling:
- Severe Acute Exacerbation of Hepatitis B in Patients with HBV Infection [see Warnings and Precautions (5.1)].
- New Onset or Worsening Renal Impairment [see Warnings and Precautions (5.2)] .
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.4)] .
- Bone Loss and Mineralization Defects [see Warnings and Precautions (5.5)] .
- Lactic Acidosis/Severe Hepatomegaly with Steatosis [see Warnings and Precautions (5.6)] .
7 Drug Interactions
- Tenofovir disoproxil fumarate increases didanosine concentrations. Dose reduction and close monitoring for didanosine toxicity are warranted. ( 7.2)
- Coadministration decreases atazanavir concentrations. When coadministered with tenofovir disoproxil fumarate, use atazanavir given with ritonavir. ( 7.2)
- Coadministration of tenofovir disoproxil fumarate with certain HIV-1 protease inhibitors or certain drugs to treat HCV increases tenofovir concentrations. Monitor for evidence of tenofovir toxicity. ( 7.2)
- Consult Full Prescribing Information prior to and during treatment for important drug interactions. ( 7.2)
12.3 Pharmacokinetics
The pharmacokinetics of TDF have been evaluated in healthy volunteers and HIV-1 infected individuals. Tenofovir pharmacokinetics are similar between these populations.
8.6 renal Impairment
The dosing interval for tenofovir disoproxil fumarate should be modified in adult patients with estimated creatinine clearance below 50 mL/min or in patients with end stage renal disease requiring dialysis [see Dosage and Administration (2.4)and Clinical Pharmacology (12.3)] .
1 Indications and Usage
Tenofovir disoproxil fumarate is a nucleotide analog HIV-1 reverse transcriptase inhibitor and an HBV reverse transcriptase inhibitor and is indicated:
1.2 Chronic Hepatitis B
Tenofovir disoproxil fumarate is indicated for the treatment of chronic hepatitis B virus (HBV) in adults and pediatric patients 2 years of age and older weighing at least 35 kg.
Impairment of Fertility
There were no effects on fertility, mating performance or early embryonic development when TDF was administered to male rats at a dose equivalent to 10 times the human dose based on body surface area comparisons for 28 days prior to mating and to female rats for 15 days prior to mating through day seven of gestation. There was, however, an alteration of the estrous cycle in female rats.
12.1 Mechanism of Action
Tenofovir disoproxil fumarate is an antiviral drug [See Microbiology (12.4)].
5 Warnings and Precautions
- New onset or worsening renal impairment: Can include acute renal failure and Fanconi syndrome. Avoid administering tenofovir disoproxil fumarate with concurrent or recent use of nephrotoxic drugs.( 5.2)
- HIV testing: HIV antibody testing should be offered to all HBV-infected patients before initiating therapy with tenofovir disoproxil fumarate. Tenofovir disoproxil fumarate should only be used as part of an appropriate antiretroviral combination regimen in HIV-infected patients with or without HBV coinfection. ( 5.3)
- Immune reconstitution syndrome: May necessitate further evaluation and treatment. ( 5.4)
- Decreases in bone mineral density (BMD): Consider assessment of BMD in patients with a history of pathologic fracture or other risk factors for osteoporosis or bone loss. ( 5.5)
- Lactic acidosis/severe hepatomegaly with steatosis: Discontinue treatment in patients who develop symptoms or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity. ( 5.6)
2 Dosage and Administration
- Testing: Prior to or when initiating tenofovir disoproxil fumarate test for hepatitis B virus infection and HIV-1 infection. Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorous. ( 2.1)
- Recommended tablet dosage in adults and pediatric patients weighing at least 35 kg: One tenofovir disoproxil fumarate 300 mg tablet once daily taken orally without regard to food. ( 2.2)
- Recommended dosage in pediatric patients at least 2 years of age and adults:
- For patients weighing at least 35 kg who can swallow an intact tablet, one tenofovir disoproxil fumarate tablet (300 mg based on body weight) once daily taken orally without regard to food. ( 2.2)
- Recommended dosage in renally impaired adult patients:
3 Dosage Forms and Strengths
Tenofovir disoproxil fumarate is available as tablets.
• 300 mg Tablets: 300 mg of TDF (equivalent to 245 mg of tenofovir disoproxil): almond-shaped, blue, film coated, debossed with “32” on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of tenofovir disoproxil fumarate. Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders
allergic reaction, including angioedema
Metabolism and Nutrition Disorders
lactic acidosis, hypokalemia, hypophosphatemia
Respiratory, Thoracic,
and Mediastinal Disorders
dyspnea
Gastrointestinal Disorders
pancreatitis, increased amylase, abdominal pain
Hepatobiliary Disorders
hepatic steatosis, hepatitis, increased liver enzymes (most commonly AST, ALT gamma GT)
Skin and Subcutaneous Tissue Disorders
rash
Musculoskeletal and Connective Tissue Disorders
rhabdomyolysis, osteomalacia (manifested as bone pain and which may contribute to fractures), muscular weakness, myopathy
Renal and Urinary Disorders
acute renal failure, renal failure, acute tubular necrosis, Fanconi syndrome, proximal renal tubulopathy, interstitial nephritis (including acute cases), nephrogenic diabetes insipidus, renal insufficiency, increased creatinine, proteinuria, polyuria
General Disorders and Administration Site Conditions
asthenia
The following adverse reactions, listed under the body system headings above, may occur as a consequence of proximal renal tubulopathy: rhabdomyolysis, osteomalacia, hypokalemia, muscular weakness, myopathy, hypophosphatemia.
8 Use in Specific Populations
Lactation: Breastfeeding in HIV-1 infected mothers is not recommended due to the potential for HIV-1 transmission. ( 8.2)
6.1 clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
14.1 Overview of Clinical Trials
The efficacy and safety of tenofovir disoproxil fumarate in adults and pediatric subjects were evaluated in the trials summarized in Table 19.
| Trial | Population | Study Arms (N)
Randomized and dosed.
|
Timepoint
(Week) |
|---|---|---|---|
| Trial 903
Randomized, double-blind, active-controlled trial.
( NCT00158821) |
HIV-1 treatment-naïve adults | Tenofovir disoproxil fumarate+lamivudine+efavirenz (299)
stavudine+lamivudine+efavirenz (301) |
144 |
| Trial 934
Randomized, open-label active-controlled trial.
( NCT00112047) |
emtricitabine+tenofovir disoproxil fumarate+efavirenz (257)
zidovudine/lamivudine+efavirenz (254) |
144 | |
| Trial 907
Randomized, double-blind, placebo-controlled trial.
( NCT00002450) |
HIV-1 treatment-experienced adults | Tenofovir disoproxil fumarate (368)
Placebo (182) |
24 |
| Trial 0102
( NCT00117676) |
HBeAg-negative adults with chronic HBV | Tenofovir disoproxil fumarate (250)
HEPSERA (125) |
48 |
| Trial 0103
( NCT00116805) |
HBeAg-positive adults with chronic HBV | Tenofovir disoproxil fumarate (176)
HEPSERA (90) |
48 |
| Trial 121
( NCT00737568) |
Adults with lamivudine-resistant chronic HBV | Tenofovir disoproxil fumarate (141) | 96 |
| Trial 0108
( NCT00298363) |
Adults with chronic HBV and decompensated liver disease | Tenofovir disoproxil fumarate (45) | 48 |
| Trial 352
( NCT00528957) |
HIV-1 treatment experienced pediatric subjects 2 years to <12 years | Tenofovir disoproxil fumarate (44)
stavudine or zidovudine (48) |
48 |
| Trial 321
( NCT00352053) |
HIV-1 treatment-experienced pediatric subjects 12 years to <18 years | Tenofovir disoproxil fumarate (45)
Placebo (42) |
48 |
| Trial 115
( NCT00734162) |
Pediatric subjects 12 years to <18 years with chronic HBV | Tenofovir disoproxil fumarate (52)
Placebo (54) |
72 |
|
Trial 144 § ( NCT01651403) |
Pediatric subjects 2 years to <12 years with chronic HBV | Tenofovir disoproxil fumarate (60)
Placebo (29) |
48 |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Severe Acute Exacerbation of Hepatitis B in Patients Infected with HBV
Inform patients that severe acute exacerbations of hepatitis B have been reported in patients infected with hepatitis B virus (HBV) and have discontinued tenofovir disoproxil fumarate. Advise patients not to discontinue tenofovir disoproxil fumarate without first informing their healthcare provider. All patients should be tested for HBV infection before or when starting tenofovir disoproxil fumarate and those who are infected with HBV need close medical follow-up for several months after stopping tenofovir disoproxil fumarate to monitor for exacerbations of hepatitis [ see Warnings and Precautions (5.1) ].
New Onset or Worsening Renal Impairment
Inform patients that renal impairment, including cases of acute renal failure and Fanconi syndrome, has been reported in association with the use of tenofovir disoproxil fumarate. Advise patients to avoid tenofovir disoproxil fumarate with concurrent or recent use of a nephrotoxic agent (e.g., high-dose or multiple NSAIDs) [ see Warnings and Precautions (5.2) ]. The dosing interval of tenofovir disoproxil fumarate may need adjustment in HIV-1 infected patients with renal impairment.
Immune Reconstitution Syndrome
Inform patients that in some patients with advanced HIV infection (AIDS) signs and symptoms of inflammation from previous infections may occur soon after anti-HIV treatment is started. It is believed that these symptoms are due to an improvement in the body’s immune response, enabling the body to fight infections that may have been present with no obvious symptoms. Advise patients to inform their healthcare provider immediately of any symptoms of infection [ see Warnings and Precautions (5.4) ].
Bone Loss and Mineralization Defects
Inform patients that decreases in bone mineral density have been observed with the use of tenofovir disoproxil fumarate. Consider bone monitoring in patients who have a history of pathologic bone fracture or at risk for osteopenia [ see Warnings and Precautions (5.5) ].
Lactic Acidosis and Severe Hepatomegaly
Inform patients that lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported. Treatment with tenofovir disoproxil fumarate should be suspended in any patient who develops clinical symptoms suggestive of lactic acidosis or pronounced hepatotoxicity [ see Warnings and Precautions (5.6) ].
Drug Interactions
Advise patients that tenofovir disoproxil fumarate may interact with many drugs; therefore, advise patients to report to their healthcare provider the use of any other medication, including other HIV drugs and drugs for treatment of hepatitis C virus [ see Warnings and Precautions (5.7)and Drug Interactions (7) ].
Dosing Recommendations
Inform patients that it is important to take tenofovir disoproxil fumarate on a regular dosing schedule with or without food and to avoid missing doses as it can result in development of resistance [ see Dosage and Administration (2) ].
Pregnancy Registry
Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant women exposed to tenofovir disoproxil fumarate [ see Use in Specific Populations (8.1) ].
Lactation
Instruct mothers not to breastfeed if they are taking tenofovir disoproxil fumarate for the treatment of HIV-1 infection because of the risk of passing the HIV-1 virus to the baby [ see Use in Specific Populations (8.2) ].
Treatment Duration
Advise patients that in the treatment of chronic hepatitis B, the optimal duration of treatment is unknown. The relationship between response and long-term prevention of outcomes such as hepatocellular carcinoma is not known.
Manufactured by:
Qilu Pharmaceutical Co., Ltd.
Jinan, 250101, China
Manufactured for:
Quinn Pharmaceuticals
Boca Raton, FL, USA
Code number: 34040051111I
Revised: March 2023
All brand names listed are the registered trademarks of their respective owners and are not trademarks of Qilu Pharmaceutical Co., Ltd..
7.1 Drugs Affecting Renal Function
Tenofovir is primarily eliminated by the kidneys [ see Clinical Pharmacology (12.3) ]. Coadministration of tenofovir disoproxil fumarate with drugs that are eliminated by active tubular secretion may increase concentrations of tenofovir and/or the coadministered drug. Some examples include, but are not limited to, acyclovir, cidofovir, ganciclovir, valacyclovir, valganciclovir, aminoglycosides (e.g., gentamicin), and high-dose or multiple NSAIDs [ see Warnings and Precautions (5.2) ]. Drugs that decrease renal function may increase concentrations of tenofovir.
In the treatment of chronic hepatitis B, tenofovir disoproxil fumarate should not be administered in combination with HEPSERA (adefovir dipivoxil).
5.4 immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in HIV-1 infected patients treated with combination antiretroviral therapy, including tenofovir disoproxil fumarate. During the initial phase of combination antiretroviral treatment, HIV-1 infected patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium aviuminfection, cytomegalovirus, Pneumocystis jiroveciipneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves’ disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
16 How Supplied/storage and Handling
Tenofovir disoproxil fumarate tablets, 300 mg, are almond-shaped, blue, film-coated tablets containing 300 mg of tenofovir DF, which is equivalent to 245 mg of tenofovir disoproxil, and are debossed with “32” on one side.
- NDC: 71335-2364-1: 30 TABLETs in a BOTTLE
- NDC: 71335-2364-2: 15 TABLETs in a BOTTLE
Store tenofovir disoproxil fumarate tablets at 25°C (77°F), excursions permitted to 15-30°C(59-86°F) (see USP Controlled Room Temperature).
• Keep container tightly closed.
• Dispense only in original container.
• Do not use if seal over bottle opening is broken or missing.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
5.5 bone Loss and Mineralization Defects
Bone Mineral Density
In clinical trials in HIV-1 infected adults, tenofovir disoproxil fumarate was associated with slightly greater decreases in bone mineral density (BMD) and increases in biochemical markers of bone metabolism, suggesting increased bone turnover relative to comparators [see Adverse Reactions (6.1)] . Serum parathyroid hormone levels and 1,25 Vitamin D levels were also higher in subjects receiving tenofovir disoproxil fumarate.
Clinical trials evaluating tenofovir disoproxil fumarate in pediatric subjects were conducted. Under normal circumstances, BMD increases rapidly in pediatric patients. In HIV-1 infected subjects 2 years to less than 18 years of age, bone effects were similar to those observed in adult subjects and suggest increased bone turnover. Total body BMD gain was less in the tenofovir disoproxil fumarate-treated HIV-1 infected pediatric subjects as compared to the control groups. Similar trends were observed in chronic HBV-infected pediatric subjects 2 years to less than 18 years of age. In all pediatric trials, normal skeletal growth (height) was not affected for the duration of the clinical trials [see Adverse Reactions (6.1)] .
The effects of tenofovir disoproxil fumarate-associated changes in BMD and biochemical markers on long-term bone health and future fracture risk in adults and pediatric subjects 2 years and older are unknown. The long-term effect of lower spine and total body BMD on skeletal growth in pediatric patients, and in particular, the effects of long-duration exposure in younger children is unknown.
Although the effect of supplementation with calcium and vitamin D was not studied, such supplementation may be beneficial. Assessment of BMD should be considered for adult and pediatric patients who have a history of pathologic bone fracture or other risk factors for osteoporosis or bone loss. If bone abnormalities are suspected, appropriate consultation should be obtained.
Mineralization Defects
Cases of osteomalacia associated with proximal renal tubulopathy, manifested as bone pain or pain in extremities and which may contribute to fractures, have been reported in association with tenofovir disoproxil fumarate use [see Adverse Reactions (6.2)] . Arthralgia and muscle pain or weakness have also been reported in cases of proximal renal tubulopathy. Hypophosphatemia and osteomalacia secondary to proximal renal tubulopathy should be considered in patients at risk of renal dysfunction who present with persistent or worsening bone or muscle symptoms while receiving TDF-containing products [see Warnings and Precautions (5.2)] .
13.2 Animal Toxicology And/or Pharmacology
Tenofovir and TDF administered in toxicology studies to rats, dogs, and monkeys at exposures (based on AUCs) greater than or equal to 6 fold those observed in humans caused bone toxicity. In monkeys the bone toxicity was diagnosed as osteomalacia. Osteomalacia observed in monkeys appeared to be reversible upon dose reduction or discontinuation of tenofovir. In rats and dogs, the bone toxicity manifested as reduced bone mineral density. The mechanism(s) underlying bone toxicity is unknown.
Evidence of renal toxicity was noted in 4 animal species. Increases in serum creatinine, BUN, glycosuria, proteinuria, phosphaturia, and/or calciuria and decreases in serum phosphate were observed to varying degrees in these animals. These toxicities were noted at exposures (based on AUCs) 2–20 times higher than those observed in humans. The relationship of the renal abnormalities, particularly the phosphaturia, to the bone toxicity is not known.
5.2 New Onset Or Worsening Renal Impairment
Tenofovir is principally eliminated by the kidney. Renal impairment, including cases of acute renal failure and Fanconi syndrome (renal tubular injury with severe hypophosphatemia), has been reported with the use of tenofovir disoproxil fumarate [see Adverse Reactions (6.2)] .
Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorus.
Dosing interval adjustment of tenofovir disoproxil fumarate and close monitoring of renal function are recommended in all patients with creatinine clearance below 50 mL/min [see Dosage and Administration (2.4)] . No safety or efficacy data are available in patients with renal impairment who received tenofovir disoproxil fumarate using these dosing guidelines, so the potential benefit of tenofovir disoproxil fumarate therapy should be assessed against the potential risk of renal toxicity.
Tenofovir disoproxil fumarate should be avoided with concurrent or recent use of a nephrotoxic agent (e.g., high-dose or multiple non-steroidal anti-inflammatory drugs (NSAIDs)) [see Drug Interactions (7.1)] . Cases of acute renal failure after initiation of high-dose or multiple NSAIDs have been reported in HIV-infected patients with risk factors for renal dysfunction who appeared stable on TDF. Some patients required hospitalization and renal replacement therapy. Alternatives to NSAIDs should be considered, if needed, in patients at risk for renal dysfunction.
Persistent or worsening bone pain, pain in extremities, fractures and/or muscular pain or weakness may be manifestations of proximal renal tubulopathy and should prompt an evaluation of renal function in patients at risk of renal dysfunction.
5.3 patients Coinfected With Hiv 1 and Hbv
Due to the risk of development of HIV-1 resistance, tenofovir disoproxil fumarate should only be used in HIV-1 and HBV coinfected patients as part of an appropriate antiretroviral combination regimen.
HIV-1 antibody testing should be offered to all HBV-infected patients before initiating therapy with tenofovir disoproxil fumarate. It is also recommended that all patients with HIV-1 be tested for the presence of chronic hepatitis B before initiating treatment with tenofovir disoproxil fumarate.
7.2 Established and Significant Interactions
Table 12 provides a listing of established or clinically significant drug interactions. The drug interactions described are based on studies conducted with TDF [see Clinical Pharmacology (12.3)].
| Concomitant Drug Class: Drug Name |
Effect on Concentration
=Increase, ↓=Decrease
|
Clinical Comment |
|
NRTI:
didanosine |
↑ didanosine |
Patients receiving tenofovir disoproxil fumarate and didanosine should be monitored closely for didanosine-associated adverse reactions. Discontinue didanosine in patients who develop didanosine-associated adverse reactions. Higher didanosine concentrations could potentiate didanosine-associated adverse reactions, including pancreatitis, and neuropathy. Suppression of CD4+ cell counts has been observed in patients receiving tenofovir disoproxil fumarate with didanosine 400 mg daily. In patients weighing greater than 60 kg, reduce the didanosine dose to 250 mg when it is coadministered with tenofovir disoproxil fumarate. In patients weighing less than 60 kg, reduce the didanosine dose to 200 mg when it is coadministered with tenofovir disoproxil fumarate. When coadministered, tenofovir disoproxil fumarate and Videx EC may be taken under fasted conditions or with a light meal (less than 400 kcal, 20% fat). |
|
HIV-1 Protease
Inhibitors:
lopinavir/ritonavir atazanavir/ritonavir darunavir/ritonavir |
↓ atazanavir ↑ tenofovir |
When coadministered with tenofovir disoproxil fumarate, atazanavir 300 mg should be given with ritonavir 100 mg. Monitor patients receiving tenofovir disoproxil fumarate concomitantly with lopinavir/ritonavir, ritonavir-boosted atazanavir, or ritonavir boosted darunavir for TDF-associated adverse reactions. Discontinue tenofovir disoproxil fumarate in patients who develop TDF-associated adverse reactions. |
|
Hepatitis C Antiviral Agents: sofosbuvir/velpatasvir sofosbuvir/velpatasvir/ voxilaprevir
|
↑ tenofovir |
Monitor patients receiving tenofovir disoproxil fumarate concomitantly with EPCLUSA ®(sofosbuvir/velpatasvir) for adverse reactions associated with TDF. Monitor patients receiving tenofovir disoproxil fumarate concomitantly with HARVONI ®(ledipasvir/sofosbuvir) without an HIV-1 protease inhibitor/ritonavir or an HIV-1 protease inhibitor/cobicistat combination, for adverse reactions associated with TDF. In patients receiving tenofovir disoproxil fumarate concomitantly with HARVONI and an HIV-1 protease inhibitor/ritonavir or an HIV-1 protease inhibitor/cobicistat combination, consider an alternative HCV or antiretroviral therapy, as the safety of increased tenofovir concentrations in this setting has not been established. If coadministration is necessary, monitor for adverse reactions associated with TDF. |
5.7 risk of Adverse Reactions Due to Drug Interactions
The concomitant use of tenofovir disoproxil fumarate and other drugs may result in known or potentially significant drug interactions, some of which may lead to possible clinically significant adverse reactions from greater exposures of concomitant drugs [see Drug Interactions (7.2)] .
See Table 12 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during therapy with tenofovir disoproxil fumarate; review concomitant medications during therapy with tenofovir disoproxil fumarate; and monitor for adverse reactions associated with the concomitant drugs.
2.4 dosage Adjustment in Patients With Renal Impairment
Significant increase in drug exposures occurred when tenofovir disoproxil fumarate was administered to subjects with moderate to severe renal impairment (creatinine clearance below 50 mL/min). Table 3 provides dosage interval adjustment for patients with renal impairment. No dosage adjustment of tenofovir disoproxil fumarate tablets 300 mg is necessary for patients with mild renal impairment (creatinine clearance 50–80 mL/min) [see Warnings and Precautions (5.3), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)] .
| Creatinine Clearance
(mL/min) Calculated using ideal (lean) body weight.
|
||||
|---|---|---|---|---|
| 50 or greater | 30-49 | 10-29 | Hemodialysis Patients | |
| Recommended 300 mg Dosing Interval | Every 24 hours | Every 48 hours | Every 72 to 96 hours | Every 7 days or after a total of approximately 12 hours of dialysis
Generally once weekly assuming 3 hemodialysis sessions a week of approximately 4 hours' duration. Tenofovir disoproxil fumarate should be administered following completion of dialysis.
|
No data are available to make dosage recommendations in patients with creatinine clearance below 10 mL/min who are not on hemodialysis.
No data are available to make dosage recommendations in pediatric patients with renal impairment.
Warning: Posttreatment Acute Exacerbation of Hepatitis B
Severe acute exacerbations of hepatitis B virus (HBV) have been reported in HBV-infected patients who have discontinued anti-hepatitis B therapy, including tenofovir disoproxil fumarate. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in HBV-infected patients who discontinue anti-hepatitis B therapy, including tenofovir disoproxil fumarate. If appropriate, resumption of anti-hepatitis B therapy may be warranted [See Warnings and Precautions (5.1)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14.3 Clinical Trial Results in Pediatric Subjects With Hiv 1 Infection
In Trial 352, 92 treatment-experienced subjects 2 years to less than 12 years of age with stable, virologic suppression on a stavudine (d4T)- or zidovudine (AZT)-containing regimen were randomized to either replace d4T or AZT with tenofovir disoproxil fumarate (N=44) or continue their original regimen (N=48) for 48 weeks. Five additional subjects over the age of 12 years were enrolled and randomized (tenofovir disoproxil fumarate N=4, original regimen N=1) but are not included in the efficacy analysis. After 48 weeks, all eligible subjects were allowed to continue in the trial receiving open-label tenofovir disoproxil fumarate. At Week 48, 89% of subjects in the tenofovir disoproxil fumarate treatment group and 90% of subjects in the d4T or AZT treatment group had HIV-1 RNA concentrations <400 copies/mL. During the 48-week randomized phase of the trial, 1 subject in the tenofovir disoproxil fumarate group discontinued the trial prematurely because of virologic failure/lack of efficacy and 3 subjects (2 subjects in the tenofovir disoproxil fumarate group and 1 subject in the d4T or AZT group) discontinued for other reasons.
In Trial 321, 87 treatment-experienced subjects 12 years to less than 18 years of age were treated with tenofovir disoproxil fumarate (N=45) or placebo (N=42) in combination with an optimized background regimen (OBR) for 48 weeks. The mean baseline CD4 cell count was 374 cells/mm 3and the mean baseline plasma HIV-1 RNA was 4.6 log 10copies/mL. At baseline, 90% of subjects harbored NRTI resistance-associated substitutions in their HIV-1 isolates. Overall, the trial failed to show a difference in virologic response between the tenofovir disoproxil fumarate and placebo groups. Subgroup analyses suggest the lack of difference in virologic response may be attributable to imbalances between treatment arms in baseline viral susceptibility to tenofovir disoproxil fumarate and OBR.
Although changes in HIV-1 RNA in these highly treatment-experienced subjects were less than anticipated, the comparability of the pharmacokinetic and safety data to that observed in adults supports the use of tenofovir disoproxil fumarate in pediatric patients 12 years and older who weigh at least 35 kg and whose HIV-1 isolate is expected to be sensitive to tenofovir disoproxil fumarate [see Warnings and Precautions (5.5), Adverse Reactions (6.1), and Clinical Pharmacology (12.3)].
5.1 severe Acute Exacerbation of Hepatitis B in Patients With Hbv Infection
All patients should be tested for the presence of chronic hepatitis B virus (HBV) before or when initiating tenofovir disoproxil fumarate [see Dosage and Administration (2.1)] .
Discontinuation of anti-HBV therapy, including tenofovir disoproxil fumarate, may be associated with severe acute exacerbations of hepatitis B. Patients infected with HBV who discontinue tenofovir disoproxil fumarate should be closely monitored with both clinical and laboratory follow-up for at least several months after stopping treatment. If appropriate, resumption of anti-hepatitis B therapy may be warranted, especially in patients with advanced liver disease or cirrhosis, since posttreatment exacerbation of hepatitis may lead to hepatic decompensation and liver failure.
2.1 testing Prior to Initiation of Tenofovir Disoproxil Fumarate for Treatment of Hiv 1 Infection Or Chronic Hepatitis B
Prior to or when initiating tenofovir disoproxil fumarate, test patients for HBV infection and HIV-1 infection. Tenofovir disoproxil fumarate alone should not be used in patients with HIV-1 infection [see Warnings and Precautions (5.3)] .
Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorus [see Warnings and Precautions (5.2)] .
Structured Label Content
Section 34077-8 (34077-8)
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to tenofovir disoproxil fumarate during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Section 42229-5 (42229-5)
The recommended dosage of tenofovir disoproxil fumarate in adults and pediatric patients weighing at least 35 kg is one 300 mg tablet taken orally once daily without regard to food. The dosage for tenofovir disoproxil fumarate is the same for both HIV and HBV indications.
The recommended dosage of tenofovir disoproxil fumarate tablet in adults and pediatric patients 2 years and older weighing at least 35 kg is 8 mg of tenofovir disoproxil fumarate (TDF) per kg of body weight (up to a maximum of 300 mg) once daily. Dosage for pediatric patients 2 years and older weighing at least 35 kg and able to swallow an intact tablet is provided in Table 1. Weight should be monitored periodically and the tenofovir disoproxil fumarate dose adjusted accordingly.
| Body Weight (kg) | Dosing of Tenofovir Disoproxil Fumarate Tablets |
|---|---|
| at least 35 | one 300 mg tablet once daily |
Section 42230-3 (42230-3)
| Patient Information
Tenofovir Disoproxil Fumarate (ten-OF-oh-vir dye-soe-PROX-il FUE-ma-rate) Tablets |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: March 2023 |
| Read this Patient Information before you start taking tenofovir disoproxil fumarate tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. | |
|
What is the most important information I should know about tenofovir disoproxil fumarate tablets? |
|
| Tenofovir disoproxil fumarate tablets can cause serious side effects, including: | |
|
|
| For more information about side effects, see “ What are the possible side effects of tenofovir disoproxil fumarate tablets?” | |
| What are tenofovir disoproxil fumarate tablets? | |
| Tenofovir disoproxil fumarate tablets are a prescription medicine that is used to: | |
|
|
| What should I tell my healthcare provider before taking tenofovir disoproxil fumarate tablets? | |
| Before you take tenofovir disoproxil fumarate tablets, tell your healthcare provider about all of your medical conditions, including if you: | |
|
|
| Tell your healthcare provider about all the medicines you take,including prescription and non-prescription medicines, vitamins, and herbal supplements. | |
| Some medicines may interact with tenofovir disoproxil fumarate tablets. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine. | |
|
|
| How should I take tenofovir disoproxil fumarate tablets? | |
|
|
| What are the possible side effects of tenofovir disoproxil fumarate tablets? | |
| Tenofovir disoproxil fumarate tablets may cause serious side effects, including: | |
|
|
| The most common side effects in all people taking tenofovir disoproxil fumarate tablets are: | |
|
|
| In some people with advanced HBV-infection, other common side effects may include: | |
|
|
|
These are not all the possible side effects of tenofovir disoproxil fumarate tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
How should I store tenofovir disoproxil fumarate tablets?
Keep tenofovir disoproxil fumarate tablets and all medicines out of the reach of children. |
|
| General information about the safe and effective use of tenofovir disoproxil fumarate tablets: | |
|
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use tenofovir disoproxil fumarate tablets for a condition for which it was not prescribed. Do not give tenofovir disoproxil fumarate tablets to other people, even if they have the same condition you have. It may harm them. You can ask your pharmacist or healthcare provider for information about tenofovir disoproxil fumarate tablets that is written for health professionals. A vaccine is available to protect people at risk for becoming infected with HBV. You can ask your healthcare provider for information about this vaccine. |
|
| What are the ingredients in tenofovir disoproxil fumarate tablets? | |
| Active ingredient:tenofovir disoproxil fumarate | |
| Inactive ingredients: | |
| Tenofovir disoproxil fumarate tablets: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and pregelatinized starch. | |
| Tablet coating: | |
| Tenofovir disoproxil fumarate tablets 300 mg: Opadry II 32K605004, which contains FD&C blue #2 aluminum lake, hypromellose 2910, lactose monohydrate, titanium dioxide, and triacetin. | |
|
Manufactured by:
Manufactured for:
|
Section 51945-4 (51945-4)
Tenofovir Disoproxil Fumarate Tablets
Animal Data
TDF was administered orally to pregnant rats (at 0, 50, 150, or 450 mg/kg/day) and rabbits (at 0, 30, 100, or 300 mg/kg/day) through organogenesis (on gestation days 7 through 17, and 6 through 18, respectively). No significant toxicological effects were observed in embryo-fetal toxicity studies performed with TDF in rats at doses up to 14 times the human dose based on body surface area comparisons and in rabbits at doses up to 19 times the human dose based on body surface area comparisons. In a pre/postnatal development study in rats, TDF was administered orally through lactation at doses up to 600 mg/kg/day; no adverse effects were observed in the offspring at tenofovir exposures of approximately 2.7 times higher than human exposures at the recommended daily dose of tenofovir disoproxil fumarate.
Mutagenesis
Tenofovir disoproxil fumarate was mutagenic in the in vitro mouse lymphoma assay and negative in an in vitro bacterial mutagenicity test (Ames test). In an in vivo mouse micronucleus assay, TDF was negative when administered to male mice.
10 Overdosage (10 OVERDOSAGE)
If overdose occurs, the patient must be monitored for evidence of toxicity, and standard supportive treatment applied as necessary.
Tenofovir is efficiently removed by hemodialysis with an extraction coefficient of approximately 54%. Following a single 300 mg dose of tenofovir disoproxil fumarate, a four-hour hemodialysis session removed approximately 10% of the administered tenofovir dose.
8.2 Lactation
Risk Summary
Based on published data, tenofovir has been shown to be present in human breast milk (see Data). It is not known if tenofovir affects milk production or has effects on the breastfed child.
Treatment of HIV-1 infection:
The Centers for Disease Control and Prevention recommend that HIV-1 infected mothers not breastfeed their infants to avoid risking postnatal transmission of HIV-1.
Because of the potential for: (1) HIV transmission (in HIV-negative infants); (2) developing viral resistance (in HIV-positive infants); and (3) adverse reactions in a breastfed infant similar to those seen in adults, instruct mothers not to breastfeed if they are taking tenofovir disoproxil fumarate for the treatment of HIV-1.
Treatment of HBV infection:
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for tenofovir disoproxil fumarate and any potential adverse effects on the breastfed infant from tenofovir disoproxil fumarate or from the underlying maternal condition.
Data
In a study of 50 HIV-uninfected, breastfeeding women on a tenofovir-containing regimen initiated between 1 and 24 weeks postpartum (median 13 weeks), tenofovir was undetectable in the plasma of most infants after 7 days of treatment in mothers. There were no serious adverse events in mothers or infants.
11 Description (11 DESCRIPTION)
Tenofovir disoproxil fumarate (TDF) (a prodrug of tenofovir) is a fumaric acid salt of bis-isopropoxycarbonyloxymethyl ester derivative of tenofovir. TDF is converted in vivo to tenofovir, an acyclic nucleoside phosphonate (nucleotide) analog of adenosine 5’-monophosphate. Tenofovir exhibits activity against HIV-1 reverse transcriptase.
The chemical name of TDF is 9-[( R)-2-[[bis[[(isopropoxycarbonyl)oxy]methoxy]phosphinyl]methoxy]propyl]adenine fumarate (1:1). It has a molecular formula of C 19H 30N 5O 10P • C 4H 4O 4and a molecular weight of 635.52. It has the following structural formula:
Tenofovir disoproxil fumarate is a white to off-white crystalline powder with a solubility of 13.4 mg/mL in distilled water at 25 °C. It has an octanol/phosphate buffer (pH 6.5) partition coefficient (log p) of 1.25 at 25 °C.
Tenofovir disoproxil fumarate is available as tablets.
Tenofovir disoproxil fumarate tablets are for oral administration and are available in the strength of 300 mg of TDF (equivalent to 245 mg of tenofovir disoproxil).
Tenofovir disoproxil fumarate tablets contain the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The 300 mg strength tablets are coated with Opadry II 32K605004, which contains FD&C blue #2 aluminum lake, hypromellose 2910, lactose monohydrate, titanium dioxide, and triacetin.
In this insert, all dosages are expressed in terms of TDF except where otherwise noted.
Carcinogenesis
Long-term oral carcinogenicity studies of TDF in mice and rats were carried out at exposures up to approximately 16 times (mice) and 5 times (rats) those observed in humans at the therapeutic dose for HIV-1 infection. At the high dose in female mice, liver adenomas were increased at exposures 16 times that in humans. In rats, the study was negative for carcinogenic findings at exposures up to 5 times that observed in humans at the therapeutic dose.
8.5 Geriatric Use
Clinical trials of tenofovir disoproxil fumarate did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for the elderly patient should be cautious, keeping in mind the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
1.1 Hiv 1 Infection (1.1 HIV-1 Infection)
Tenofovir disoproxil fumarate is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in adults and pediatric patients 2 years of age and older weighing at least 35 kg.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in other sections of the labeling:
- Severe Acute Exacerbation of Hepatitis B in Patients with HBV Infection [see Warnings and Precautions (5.1)].
- New Onset or Worsening Renal Impairment [see Warnings and Precautions (5.2)] .
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.4)] .
- Bone Loss and Mineralization Defects [see Warnings and Precautions (5.5)] .
- Lactic Acidosis/Severe Hepatomegaly with Steatosis [see Warnings and Precautions (5.6)] .
7 Drug Interactions (7 DRUG INTERACTIONS)
- Tenofovir disoproxil fumarate increases didanosine concentrations. Dose reduction and close monitoring for didanosine toxicity are warranted. ( 7.2)
- Coadministration decreases atazanavir concentrations. When coadministered with tenofovir disoproxil fumarate, use atazanavir given with ritonavir. ( 7.2)
- Coadministration of tenofovir disoproxil fumarate with certain HIV-1 protease inhibitors or certain drugs to treat HCV increases tenofovir concentrations. Monitor for evidence of tenofovir toxicity. ( 7.2)
- Consult Full Prescribing Information prior to and during treatment for important drug interactions. ( 7.2)
12.3 Pharmacokinetics
The pharmacokinetics of TDF have been evaluated in healthy volunteers and HIV-1 infected individuals. Tenofovir pharmacokinetics are similar between these populations.
8.6 renal Impairment (8.6 Renal Impairment)
The dosing interval for tenofovir disoproxil fumarate should be modified in adult patients with estimated creatinine clearance below 50 mL/min or in patients with end stage renal disease requiring dialysis [see Dosage and Administration (2.4)and Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tenofovir disoproxil fumarate is a nucleotide analog HIV-1 reverse transcriptase inhibitor and an HBV reverse transcriptase inhibitor and is indicated:
1.2 Chronic Hepatitis B
Tenofovir disoproxil fumarate is indicated for the treatment of chronic hepatitis B virus (HBV) in adults and pediatric patients 2 years of age and older weighing at least 35 kg.
Impairment of Fertility
There were no effects on fertility, mating performance or early embryonic development when TDF was administered to male rats at a dose equivalent to 10 times the human dose based on body surface area comparisons for 28 days prior to mating and to female rats for 15 days prior to mating through day seven of gestation. There was, however, an alteration of the estrous cycle in female rats.
12.1 Mechanism of Action
Tenofovir disoproxil fumarate is an antiviral drug [See Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- New onset or worsening renal impairment: Can include acute renal failure and Fanconi syndrome. Avoid administering tenofovir disoproxil fumarate with concurrent or recent use of nephrotoxic drugs.( 5.2)
- HIV testing: HIV antibody testing should be offered to all HBV-infected patients before initiating therapy with tenofovir disoproxil fumarate. Tenofovir disoproxil fumarate should only be used as part of an appropriate antiretroviral combination regimen in HIV-infected patients with or without HBV coinfection. ( 5.3)
- Immune reconstitution syndrome: May necessitate further evaluation and treatment. ( 5.4)
- Decreases in bone mineral density (BMD): Consider assessment of BMD in patients with a history of pathologic fracture or other risk factors for osteoporosis or bone loss. ( 5.5)
- Lactic acidosis/severe hepatomegaly with steatosis: Discontinue treatment in patients who develop symptoms or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity. ( 5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Testing: Prior to or when initiating tenofovir disoproxil fumarate test for hepatitis B virus infection and HIV-1 infection. Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorous. ( 2.1)
- Recommended tablet dosage in adults and pediatric patients weighing at least 35 kg: One tenofovir disoproxil fumarate 300 mg tablet once daily taken orally without regard to food. ( 2.2)
- Recommended dosage in pediatric patients at least 2 years of age and adults:
- For patients weighing at least 35 kg who can swallow an intact tablet, one tenofovir disoproxil fumarate tablet (300 mg based on body weight) once daily taken orally without regard to food. ( 2.2)
- Recommended dosage in renally impaired adult patients:
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tenofovir disoproxil fumarate is available as tablets.
• 300 mg Tablets: 300 mg of TDF (equivalent to 245 mg of tenofovir disoproxil): almond-shaped, blue, film coated, debossed with “32” on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of tenofovir disoproxil fumarate. Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders
allergic reaction, including angioedema
Metabolism and Nutrition Disorders
lactic acidosis, hypokalemia, hypophosphatemia
Respiratory, Thoracic,
and Mediastinal Disorders
dyspnea
Gastrointestinal Disorders
pancreatitis, increased amylase, abdominal pain
Hepatobiliary Disorders
hepatic steatosis, hepatitis, increased liver enzymes (most commonly AST, ALT gamma GT)
Skin and Subcutaneous Tissue Disorders
rash
Musculoskeletal and Connective Tissue Disorders
rhabdomyolysis, osteomalacia (manifested as bone pain and which may contribute to fractures), muscular weakness, myopathy
Renal and Urinary Disorders
acute renal failure, renal failure, acute tubular necrosis, Fanconi syndrome, proximal renal tubulopathy, interstitial nephritis (including acute cases), nephrogenic diabetes insipidus, renal insufficiency, increased creatinine, proteinuria, polyuria
General Disorders and Administration Site Conditions
asthenia
The following adverse reactions, listed under the body system headings above, may occur as a consequence of proximal renal tubulopathy: rhabdomyolysis, osteomalacia, hypokalemia, muscular weakness, myopathy, hypophosphatemia.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Breastfeeding in HIV-1 infected mothers is not recommended due to the potential for HIV-1 transmission. ( 8.2)
6.1 clinical Trials Experience (6.1 Clinical Trials Experience)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
14.1 Overview of Clinical Trials
The efficacy and safety of tenofovir disoproxil fumarate in adults and pediatric subjects were evaluated in the trials summarized in Table 19.
| Trial | Population | Study Arms (N)
Randomized and dosed.
|
Timepoint
(Week) |
|---|---|---|---|
| Trial 903
Randomized, double-blind, active-controlled trial.
( NCT00158821) |
HIV-1 treatment-naïve adults | Tenofovir disoproxil fumarate+lamivudine+efavirenz (299)
stavudine+lamivudine+efavirenz (301) |
144 |
| Trial 934
Randomized, open-label active-controlled trial.
( NCT00112047) |
emtricitabine+tenofovir disoproxil fumarate+efavirenz (257)
zidovudine/lamivudine+efavirenz (254) |
144 | |
| Trial 907
Randomized, double-blind, placebo-controlled trial.
( NCT00002450) |
HIV-1 treatment-experienced adults | Tenofovir disoproxil fumarate (368)
Placebo (182) |
24 |
| Trial 0102
( NCT00117676) |
HBeAg-negative adults with chronic HBV | Tenofovir disoproxil fumarate (250)
HEPSERA (125) |
48 |
| Trial 0103
( NCT00116805) |
HBeAg-positive adults with chronic HBV | Tenofovir disoproxil fumarate (176)
HEPSERA (90) |
48 |
| Trial 121
( NCT00737568) |
Adults with lamivudine-resistant chronic HBV | Tenofovir disoproxil fumarate (141) | 96 |
| Trial 0108
( NCT00298363) |
Adults with chronic HBV and decompensated liver disease | Tenofovir disoproxil fumarate (45) | 48 |
| Trial 352
( NCT00528957) |
HIV-1 treatment experienced pediatric subjects 2 years to <12 years | Tenofovir disoproxil fumarate (44)
stavudine or zidovudine (48) |
48 |
| Trial 321
( NCT00352053) |
HIV-1 treatment-experienced pediatric subjects 12 years to <18 years | Tenofovir disoproxil fumarate (45)
Placebo (42) |
48 |
| Trial 115
( NCT00734162) |
Pediatric subjects 12 years to <18 years with chronic HBV | Tenofovir disoproxil fumarate (52)
Placebo (54) |
72 |
|
Trial 144 § ( NCT01651403) |
Pediatric subjects 2 years to <12 years with chronic HBV | Tenofovir disoproxil fumarate (60)
Placebo (29) |
48 |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Severe Acute Exacerbation of Hepatitis B in Patients Infected with HBV
Inform patients that severe acute exacerbations of hepatitis B have been reported in patients infected with hepatitis B virus (HBV) and have discontinued tenofovir disoproxil fumarate. Advise patients not to discontinue tenofovir disoproxil fumarate without first informing their healthcare provider. All patients should be tested for HBV infection before or when starting tenofovir disoproxil fumarate and those who are infected with HBV need close medical follow-up for several months after stopping tenofovir disoproxil fumarate to monitor for exacerbations of hepatitis [ see Warnings and Precautions (5.1) ].
New Onset or Worsening Renal Impairment
Inform patients that renal impairment, including cases of acute renal failure and Fanconi syndrome, has been reported in association with the use of tenofovir disoproxil fumarate. Advise patients to avoid tenofovir disoproxil fumarate with concurrent or recent use of a nephrotoxic agent (e.g., high-dose or multiple NSAIDs) [ see Warnings and Precautions (5.2) ]. The dosing interval of tenofovir disoproxil fumarate may need adjustment in HIV-1 infected patients with renal impairment.
Immune Reconstitution Syndrome
Inform patients that in some patients with advanced HIV infection (AIDS) signs and symptoms of inflammation from previous infections may occur soon after anti-HIV treatment is started. It is believed that these symptoms are due to an improvement in the body’s immune response, enabling the body to fight infections that may have been present with no obvious symptoms. Advise patients to inform their healthcare provider immediately of any symptoms of infection [ see Warnings and Precautions (5.4) ].
Bone Loss and Mineralization Defects
Inform patients that decreases in bone mineral density have been observed with the use of tenofovir disoproxil fumarate. Consider bone monitoring in patients who have a history of pathologic bone fracture or at risk for osteopenia [ see Warnings and Precautions (5.5) ].
Lactic Acidosis and Severe Hepatomegaly
Inform patients that lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported. Treatment with tenofovir disoproxil fumarate should be suspended in any patient who develops clinical symptoms suggestive of lactic acidosis or pronounced hepatotoxicity [ see Warnings and Precautions (5.6) ].
Drug Interactions
Advise patients that tenofovir disoproxil fumarate may interact with many drugs; therefore, advise patients to report to their healthcare provider the use of any other medication, including other HIV drugs and drugs for treatment of hepatitis C virus [ see Warnings and Precautions (5.7)and Drug Interactions (7) ].
Dosing Recommendations
Inform patients that it is important to take tenofovir disoproxil fumarate on a regular dosing schedule with or without food and to avoid missing doses as it can result in development of resistance [ see Dosage and Administration (2) ].
Pregnancy Registry
Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant women exposed to tenofovir disoproxil fumarate [ see Use in Specific Populations (8.1) ].
Lactation
Instruct mothers not to breastfeed if they are taking tenofovir disoproxil fumarate for the treatment of HIV-1 infection because of the risk of passing the HIV-1 virus to the baby [ see Use in Specific Populations (8.2) ].
Treatment Duration
Advise patients that in the treatment of chronic hepatitis B, the optimal duration of treatment is unknown. The relationship between response and long-term prevention of outcomes such as hepatocellular carcinoma is not known.
Manufactured by:
Qilu Pharmaceutical Co., Ltd.
Jinan, 250101, China
Manufactured for:
Quinn Pharmaceuticals
Boca Raton, FL, USA
Code number: 34040051111I
Revised: March 2023
All brand names listed are the registered trademarks of their respective owners and are not trademarks of Qilu Pharmaceutical Co., Ltd..
7.1 Drugs Affecting Renal Function
Tenofovir is primarily eliminated by the kidneys [ see Clinical Pharmacology (12.3) ]. Coadministration of tenofovir disoproxil fumarate with drugs that are eliminated by active tubular secretion may increase concentrations of tenofovir and/or the coadministered drug. Some examples include, but are not limited to, acyclovir, cidofovir, ganciclovir, valacyclovir, valganciclovir, aminoglycosides (e.g., gentamicin), and high-dose or multiple NSAIDs [ see Warnings and Precautions (5.2) ]. Drugs that decrease renal function may increase concentrations of tenofovir.
In the treatment of chronic hepatitis B, tenofovir disoproxil fumarate should not be administered in combination with HEPSERA (adefovir dipivoxil).
5.4 immune Reconstitution Syndrome (5.4 Immune Reconstitution Syndrome)
Immune reconstitution syndrome has been reported in HIV-1 infected patients treated with combination antiretroviral therapy, including tenofovir disoproxil fumarate. During the initial phase of combination antiretroviral treatment, HIV-1 infected patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium aviuminfection, cytomegalovirus, Pneumocystis jiroveciipneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves’ disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Tenofovir disoproxil fumarate tablets, 300 mg, are almond-shaped, blue, film-coated tablets containing 300 mg of tenofovir DF, which is equivalent to 245 mg of tenofovir disoproxil, and are debossed with “32” on one side.
- NDC: 71335-2364-1: 30 TABLETs in a BOTTLE
- NDC: 71335-2364-2: 15 TABLETs in a BOTTLE
Store tenofovir disoproxil fumarate tablets at 25°C (77°F), excursions permitted to 15-30°C(59-86°F) (see USP Controlled Room Temperature).
• Keep container tightly closed.
• Dispense only in original container.
• Do not use if seal over bottle opening is broken or missing.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
5.5 bone Loss and Mineralization Defects (5.5 Bone Loss and Mineralization Defects)
Bone Mineral Density
In clinical trials in HIV-1 infected adults, tenofovir disoproxil fumarate was associated with slightly greater decreases in bone mineral density (BMD) and increases in biochemical markers of bone metabolism, suggesting increased bone turnover relative to comparators [see Adverse Reactions (6.1)] . Serum parathyroid hormone levels and 1,25 Vitamin D levels were also higher in subjects receiving tenofovir disoproxil fumarate.
Clinical trials evaluating tenofovir disoproxil fumarate in pediatric subjects were conducted. Under normal circumstances, BMD increases rapidly in pediatric patients. In HIV-1 infected subjects 2 years to less than 18 years of age, bone effects were similar to those observed in adult subjects and suggest increased bone turnover. Total body BMD gain was less in the tenofovir disoproxil fumarate-treated HIV-1 infected pediatric subjects as compared to the control groups. Similar trends were observed in chronic HBV-infected pediatric subjects 2 years to less than 18 years of age. In all pediatric trials, normal skeletal growth (height) was not affected for the duration of the clinical trials [see Adverse Reactions (6.1)] .
The effects of tenofovir disoproxil fumarate-associated changes in BMD and biochemical markers on long-term bone health and future fracture risk in adults and pediatric subjects 2 years and older are unknown. The long-term effect of lower spine and total body BMD on skeletal growth in pediatric patients, and in particular, the effects of long-duration exposure in younger children is unknown.
Although the effect of supplementation with calcium and vitamin D was not studied, such supplementation may be beneficial. Assessment of BMD should be considered for adult and pediatric patients who have a history of pathologic bone fracture or other risk factors for osteoporosis or bone loss. If bone abnormalities are suspected, appropriate consultation should be obtained.
Mineralization Defects
Cases of osteomalacia associated with proximal renal tubulopathy, manifested as bone pain or pain in extremities and which may contribute to fractures, have been reported in association with tenofovir disoproxil fumarate use [see Adverse Reactions (6.2)] . Arthralgia and muscle pain or weakness have also been reported in cases of proximal renal tubulopathy. Hypophosphatemia and osteomalacia secondary to proximal renal tubulopathy should be considered in patients at risk of renal dysfunction who present with persistent or worsening bone or muscle symptoms while receiving TDF-containing products [see Warnings and Precautions (5.2)] .
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Tenofovir and TDF administered in toxicology studies to rats, dogs, and monkeys at exposures (based on AUCs) greater than or equal to 6 fold those observed in humans caused bone toxicity. In monkeys the bone toxicity was diagnosed as osteomalacia. Osteomalacia observed in monkeys appeared to be reversible upon dose reduction or discontinuation of tenofovir. In rats and dogs, the bone toxicity manifested as reduced bone mineral density. The mechanism(s) underlying bone toxicity is unknown.
Evidence of renal toxicity was noted in 4 animal species. Increases in serum creatinine, BUN, glycosuria, proteinuria, phosphaturia, and/or calciuria and decreases in serum phosphate were observed to varying degrees in these animals. These toxicities were noted at exposures (based on AUCs) 2–20 times higher than those observed in humans. The relationship of the renal abnormalities, particularly the phosphaturia, to the bone toxicity is not known.
5.2 New Onset Or Worsening Renal Impairment (5.2 New Onset or Worsening Renal Impairment)
Tenofovir is principally eliminated by the kidney. Renal impairment, including cases of acute renal failure and Fanconi syndrome (renal tubular injury with severe hypophosphatemia), has been reported with the use of tenofovir disoproxil fumarate [see Adverse Reactions (6.2)] .
Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorus.
Dosing interval adjustment of tenofovir disoproxil fumarate and close monitoring of renal function are recommended in all patients with creatinine clearance below 50 mL/min [see Dosage and Administration (2.4)] . No safety or efficacy data are available in patients with renal impairment who received tenofovir disoproxil fumarate using these dosing guidelines, so the potential benefit of tenofovir disoproxil fumarate therapy should be assessed against the potential risk of renal toxicity.
Tenofovir disoproxil fumarate should be avoided with concurrent or recent use of a nephrotoxic agent (e.g., high-dose or multiple non-steroidal anti-inflammatory drugs (NSAIDs)) [see Drug Interactions (7.1)] . Cases of acute renal failure after initiation of high-dose or multiple NSAIDs have been reported in HIV-infected patients with risk factors for renal dysfunction who appeared stable on TDF. Some patients required hospitalization and renal replacement therapy. Alternatives to NSAIDs should be considered, if needed, in patients at risk for renal dysfunction.
Persistent or worsening bone pain, pain in extremities, fractures and/or muscular pain or weakness may be manifestations of proximal renal tubulopathy and should prompt an evaluation of renal function in patients at risk of renal dysfunction.
5.3 patients Coinfected With Hiv 1 and Hbv (5.3 Patients Coinfected with HIV-1 and HBV)
Due to the risk of development of HIV-1 resistance, tenofovir disoproxil fumarate should only be used in HIV-1 and HBV coinfected patients as part of an appropriate antiretroviral combination regimen.
HIV-1 antibody testing should be offered to all HBV-infected patients before initiating therapy with tenofovir disoproxil fumarate. It is also recommended that all patients with HIV-1 be tested for the presence of chronic hepatitis B before initiating treatment with tenofovir disoproxil fumarate.
7.2 Established and Significant Interactions
Table 12 provides a listing of established or clinically significant drug interactions. The drug interactions described are based on studies conducted with TDF [see Clinical Pharmacology (12.3)].
| Concomitant Drug Class: Drug Name |
Effect on Concentration
=Increase, ↓=Decrease
|
Clinical Comment |
|
NRTI:
didanosine |
↑ didanosine |
Patients receiving tenofovir disoproxil fumarate and didanosine should be monitored closely for didanosine-associated adverse reactions. Discontinue didanosine in patients who develop didanosine-associated adverse reactions. Higher didanosine concentrations could potentiate didanosine-associated adverse reactions, including pancreatitis, and neuropathy. Suppression of CD4+ cell counts has been observed in patients receiving tenofovir disoproxil fumarate with didanosine 400 mg daily. In patients weighing greater than 60 kg, reduce the didanosine dose to 250 mg when it is coadministered with tenofovir disoproxil fumarate. In patients weighing less than 60 kg, reduce the didanosine dose to 200 mg when it is coadministered with tenofovir disoproxil fumarate. When coadministered, tenofovir disoproxil fumarate and Videx EC may be taken under fasted conditions or with a light meal (less than 400 kcal, 20% fat). |
|
HIV-1 Protease
Inhibitors:
lopinavir/ritonavir atazanavir/ritonavir darunavir/ritonavir |
↓ atazanavir ↑ tenofovir |
When coadministered with tenofovir disoproxil fumarate, atazanavir 300 mg should be given with ritonavir 100 mg. Monitor patients receiving tenofovir disoproxil fumarate concomitantly with lopinavir/ritonavir, ritonavir-boosted atazanavir, or ritonavir boosted darunavir for TDF-associated adverse reactions. Discontinue tenofovir disoproxil fumarate in patients who develop TDF-associated adverse reactions. |
|
Hepatitis C Antiviral Agents: sofosbuvir/velpatasvir sofosbuvir/velpatasvir/ voxilaprevir
|
↑ tenofovir |
Monitor patients receiving tenofovir disoproxil fumarate concomitantly with EPCLUSA ®(sofosbuvir/velpatasvir) for adverse reactions associated with TDF. Monitor patients receiving tenofovir disoproxil fumarate concomitantly with HARVONI ®(ledipasvir/sofosbuvir) without an HIV-1 protease inhibitor/ritonavir or an HIV-1 protease inhibitor/cobicistat combination, for adverse reactions associated with TDF. In patients receiving tenofovir disoproxil fumarate concomitantly with HARVONI and an HIV-1 protease inhibitor/ritonavir or an HIV-1 protease inhibitor/cobicistat combination, consider an alternative HCV or antiretroviral therapy, as the safety of increased tenofovir concentrations in this setting has not been established. If coadministration is necessary, monitor for adverse reactions associated with TDF. |
5.7 risk of Adverse Reactions Due to Drug Interactions (5.7 Risk of Adverse Reactions Due to Drug Interactions)
The concomitant use of tenofovir disoproxil fumarate and other drugs may result in known or potentially significant drug interactions, some of which may lead to possible clinically significant adverse reactions from greater exposures of concomitant drugs [see Drug Interactions (7.2)] .
See Table 12 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during therapy with tenofovir disoproxil fumarate; review concomitant medications during therapy with tenofovir disoproxil fumarate; and monitor for adverse reactions associated with the concomitant drugs.
2.4 dosage Adjustment in Patients With Renal Impairment (2.4 Dosage Adjustment in Patients with Renal Impairment)
Significant increase in drug exposures occurred when tenofovir disoproxil fumarate was administered to subjects with moderate to severe renal impairment (creatinine clearance below 50 mL/min). Table 3 provides dosage interval adjustment for patients with renal impairment. No dosage adjustment of tenofovir disoproxil fumarate tablets 300 mg is necessary for patients with mild renal impairment (creatinine clearance 50–80 mL/min) [see Warnings and Precautions (5.3), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)] .
| Creatinine Clearance
(mL/min) Calculated using ideal (lean) body weight.
|
||||
|---|---|---|---|---|
| 50 or greater | 30-49 | 10-29 | Hemodialysis Patients | |
| Recommended 300 mg Dosing Interval | Every 24 hours | Every 48 hours | Every 72 to 96 hours | Every 7 days or after a total of approximately 12 hours of dialysis
Generally once weekly assuming 3 hemodialysis sessions a week of approximately 4 hours' duration. Tenofovir disoproxil fumarate should be administered following completion of dialysis.
|
No data are available to make dosage recommendations in patients with creatinine clearance below 10 mL/min who are not on hemodialysis.
No data are available to make dosage recommendations in pediatric patients with renal impairment.
Warning: Posttreatment Acute Exacerbation of Hepatitis B (WARNING: POSTTREATMENT ACUTE EXACERBATION OF HEPATITIS B)
Severe acute exacerbations of hepatitis B virus (HBV) have been reported in HBV-infected patients who have discontinued anti-hepatitis B therapy, including tenofovir disoproxil fumarate. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in HBV-infected patients who discontinue anti-hepatitis B therapy, including tenofovir disoproxil fumarate. If appropriate, resumption of anti-hepatitis B therapy may be warranted [See Warnings and Precautions (5.1)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14.3 Clinical Trial Results in Pediatric Subjects With Hiv 1 Infection (14.3 Clinical Trial Results in Pediatric Subjects with HIV-1 Infection)
In Trial 352, 92 treatment-experienced subjects 2 years to less than 12 years of age with stable, virologic suppression on a stavudine (d4T)- or zidovudine (AZT)-containing regimen were randomized to either replace d4T or AZT with tenofovir disoproxil fumarate (N=44) or continue their original regimen (N=48) for 48 weeks. Five additional subjects over the age of 12 years were enrolled and randomized (tenofovir disoproxil fumarate N=4, original regimen N=1) but are not included in the efficacy analysis. After 48 weeks, all eligible subjects were allowed to continue in the trial receiving open-label tenofovir disoproxil fumarate. At Week 48, 89% of subjects in the tenofovir disoproxil fumarate treatment group and 90% of subjects in the d4T or AZT treatment group had HIV-1 RNA concentrations <400 copies/mL. During the 48-week randomized phase of the trial, 1 subject in the tenofovir disoproxil fumarate group discontinued the trial prematurely because of virologic failure/lack of efficacy and 3 subjects (2 subjects in the tenofovir disoproxil fumarate group and 1 subject in the d4T or AZT group) discontinued for other reasons.
In Trial 321, 87 treatment-experienced subjects 12 years to less than 18 years of age were treated with tenofovir disoproxil fumarate (N=45) or placebo (N=42) in combination with an optimized background regimen (OBR) for 48 weeks. The mean baseline CD4 cell count was 374 cells/mm 3and the mean baseline plasma HIV-1 RNA was 4.6 log 10copies/mL. At baseline, 90% of subjects harbored NRTI resistance-associated substitutions in their HIV-1 isolates. Overall, the trial failed to show a difference in virologic response between the tenofovir disoproxil fumarate and placebo groups. Subgroup analyses suggest the lack of difference in virologic response may be attributable to imbalances between treatment arms in baseline viral susceptibility to tenofovir disoproxil fumarate and OBR.
Although changes in HIV-1 RNA in these highly treatment-experienced subjects were less than anticipated, the comparability of the pharmacokinetic and safety data to that observed in adults supports the use of tenofovir disoproxil fumarate in pediatric patients 12 years and older who weigh at least 35 kg and whose HIV-1 isolate is expected to be sensitive to tenofovir disoproxil fumarate [see Warnings and Precautions (5.5), Adverse Reactions (6.1), and Clinical Pharmacology (12.3)].
5.1 severe Acute Exacerbation of Hepatitis B in Patients With Hbv Infection (5.1 Severe Acute Exacerbation of Hepatitis B in Patients with HBV Infection)
All patients should be tested for the presence of chronic hepatitis B virus (HBV) before or when initiating tenofovir disoproxil fumarate [see Dosage and Administration (2.1)] .
Discontinuation of anti-HBV therapy, including tenofovir disoproxil fumarate, may be associated with severe acute exacerbations of hepatitis B. Patients infected with HBV who discontinue tenofovir disoproxil fumarate should be closely monitored with both clinical and laboratory follow-up for at least several months after stopping treatment. If appropriate, resumption of anti-hepatitis B therapy may be warranted, especially in patients with advanced liver disease or cirrhosis, since posttreatment exacerbation of hepatitis may lead to hepatic decompensation and liver failure.
2.1 testing Prior to Initiation of Tenofovir Disoproxil Fumarate for Treatment of Hiv 1 Infection Or Chronic Hepatitis B (2.1 Testing Prior to Initiation of Tenofovir Disoproxil Fumarate for Treatment of HIV-1 Infection or Chronic Hepatitis B)
Prior to or when initiating tenofovir disoproxil fumarate, test patients for HBV infection and HIV-1 infection. Tenofovir disoproxil fumarate alone should not be used in patients with HIV-1 infection [see Warnings and Precautions (5.3)] .
Prior to initiation and during use of tenofovir disoproxil fumarate, on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorus [see Warnings and Precautions (5.2)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:55.846805 · Updated: 2026-03-14T22:35:08.076257