r
326cb5d6-c784-404f-9a13-6d69262dda6e
34390-5
HUMAN OTC DRUG LABEL
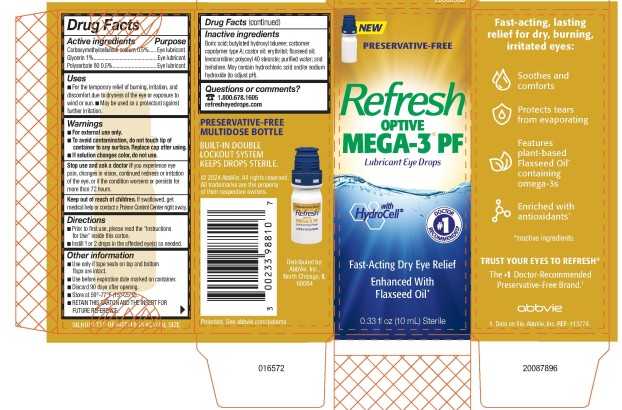
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Eye lubricant Eye lubricant Eye lubricant
Description
Carboxymethylcellulose sodium 0.5% Glycerin 1% Polysorbate 80 0.5%
Medication Information
Warnings
-
For external use only.
-
To avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- If solution changes color, do not use.
Uses
- For the temporary relief of burning, irritation, and discomfort due to dryness of the eye or exposure to wind or sun.
- May be used as a protectant against further irritation.
Purpose
Eye lubricant
Eye lubricant
Eye lubricant
Directions
- Prior to first use, please read the “Instructions for Use” inside this carton.
- Instill 1 or 2 drops in the affected eye(s) as needed.
Other Information
- Use only if tape seals on top and bottom flaps are intact.
- Use before expiration date marked on container.
- Discard 90 days after opening.
- Store at 59°-77°F (15°-25°C).
- RETAIN THIS CARTON FOR FUTURE REFERENCE
Inactive Ingredients
Boric acid; butylated hydroxyl toluene; carbomer copolymer type A; castor oil; erythritol; flaxseed oil; levocarnitine; polyoxyl 40 stearate; purified water; and trehalose. May contain hydrochloric acid and/or sodium hydroxide (to adjust pH).
Description
Carboxymethylcellulose sodium 0.5% Glycerin 1% Polysorbate 80 0.5%
Principal Display Panel
NDC 0023-3988-10
NEW
PRESERVATIVE-FREE
Refresh
OPTIVE
MEGA-3
®
PF
Lubricant Eye Drops
w ith H y d rocell ®
Fast-Acting Dry Eye Relief
Enhanced With
Flaxseed Oil
*
0.33 fl oz (10 mL) Sterile
Active Ingredients
Carboxymethylcellulose sodium 0.5%
Glycerin 1%
Polysorbate 80 0.5%
Questions Or Comments?
1.800.678.160 5
r efresheyedrops.com
PRESERVATIVE-FREE MULTIDOSE BOTTLE
BUILT-IN DOUBLE LOCKOUT SYSTEM KEEPS DROPS STERILE .
©2024 AbbVie. All rights reserved.
All trademarks are the property of their respective owners.
Distributed by: AbbVie, Inc.,
North Chicago, IL 60064
Product of US
Patented. See abbvie.com/patents
Stop Use and Ask Doctor If
you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Warnings
-
For external use only.
-
To avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- If solution changes color, do not use.
Uses
- For the temporary relief of burning, irritation, and discomfort due to dryness of the eye or exposure to wind or sun.
- May be used as a protectant against further irritation.
Purpose
Eye lubricant
Eye lubricant
Eye lubricant
Directions
- Prior to first use, please read the “Instructions for Use” inside this carton.
- Instill 1 or 2 drops in the affected eye(s) as needed.
Other Information (Other information)
- Use only if tape seals on top and bottom flaps are intact.
- Use before expiration date marked on container.
- Discard 90 days after opening.
- Store at 59°-77°F (15°-25°C).
- RETAIN THIS CARTON FOR FUTURE REFERENCE
Inactive Ingredients (Inactive ingredients)
Boric acid; butylated hydroxyl toluene; carbomer copolymer type A; castor oil; erythritol; flaxseed oil; levocarnitine; polyoxyl 40 stearate; purified water; and trehalose. May contain hydrochloric acid and/or sodium hydroxide (to adjust pH).
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 0023-3988-10
NEW
PRESERVATIVE-FREE
Refresh
OPTIVE
MEGA-3
®
PF
Lubricant Eye Drops
w ith H y d rocell ®
Fast-Acting Dry Eye Relief
Enhanced With
Flaxseed Oil
*
0.33 fl oz (10 mL) Sterile
Active Ingredients (Active ingredients)
Carboxymethylcellulose sodium 0.5%
Glycerin 1%
Polysorbate 80 0.5%
Questions Or Comments? (Questions or comments?)
1.800.678.160 5
r efresheyedrops.com
PRESERVATIVE-FREE MULTIDOSE BOTTLE
BUILT-IN DOUBLE LOCKOUT SYSTEM KEEPS DROPS STERILE .
©2024 AbbVie. All rights reserved.
All trademarks are the property of their respective owners.
Distributed by: AbbVie, Inc.,
North Chicago, IL 60064
Product of US
Patented. See abbvie.com/patents
Stop Use and Ask Doctor If (Stop use and ask doctor if)
you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-03-14T20:08:17.505862 · Updated: 2026-03-14T23:14:03.597898