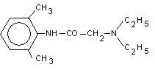lidocaine patch 5%
323e0ec2-c2e5-47d1-a39f-69ff5c99e324
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Dosage and Administration
Apply LIDOCAINE PATCH 5% to intact skin to cover the most painful area. Apply the prescribed number of patches (maximum of 3), only once for up to 12 hours within a 24 hour period. Patches may be cut into smaller sizes with scissors prior to removal of the release liner. (See HANDLING AND DISPOSAL ) Clothing may be worn over the area of application. Smaller areas of treatment are recommended in a debilitated patient, or a patient with impaired elimination. If irritation or a burning sensation occurs during application, remove the patch(es) and do not reapply until the irritation subsides. When LIDOCAINE PATCH 5% is used concomitantly with other products containing local anesthetic agents, the amount absorbed from all formulations must be considered. LIDOCAINE PATCH 5% may not stick if it gets wet. Avoid contact with water, such as bathing, swimming or showering.
Contraindications
LIDOCAINE PATCH 5% is contraindicated in patients with a known history of sensitivity to local anesthetics of the amide type, or to any other component of the product.
Drug Interactions
Antiarrhythmic Drugs LIDOCAINE PATCH 5% should be used with caution in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) since the toxic effects are additive and potentially synergistic. Local Anesthetics When LIDOCAINE PATCH 5% is used concomitantly with other products containing local anesthetic agents, the amount absorbed from all formulations must be considered. Drugs That May Cause Methemoglobinemia When Used with LIDOCAINE PATCH 5% Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics: Examples of Drugs Associated with Methemoglobinemia: Class Examples Nitrates/Nitrites nitric oxide, nitroglycerin, nitroprusside, nitrous oxide Local anesthetics articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine Antineoplastic agents cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase Antibiotics dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides Antimalarials chloroquine, primaquine Anticonvulsants Phenobarbital, phenytoin, sodium valproate Other drugs acetaminophen, metoclopramide, quinine, sulfasalazine
How Supplied
LIDOCAINE PATCH 5% is available as the following: Carton of 30 patches, packaged into individual child-resistant envelopes NDC 68788-7159-3 Store at 25 o C (77 o F); excursions permitted to 15 o -30 o C (59 o -86 o F). [See USP Controlled Room Temperature]. For more information, call Par Pharmacutical at 1-800-828-9393. Manufactured for: Par Pharmaceutical Chestnut Ridge, NY 10977 Printed in U.S.A. Revised: April 2024 Relabeled By: Preferred Pharmaceuticals Inc.
Description
R x only
Medication Information
Dosage and Administration
Apply LIDOCAINE PATCH 5% to intact skin to cover the most painful area. Apply the prescribed number of patches (maximum of 3), only once for up to 12 hours within a 24 hour period. Patches may be cut into smaller sizes with scissors prior to removal of the release liner. (See HANDLING AND DISPOSAL) Clothing may be worn over the area of application. Smaller areas of treatment are recommended in a debilitated patient, or a patient with impaired elimination.
If irritation or a burning sensation occurs during application, remove the patch(es) and do not reapply until the irritation subsides.
When LIDOCAINE PATCH 5% is used concomitantly with other products containing local anesthetic agents, the amount absorbed from all formulations must be considered.
LIDOCAINE PATCH 5% may not stick if it gets wet. Avoid contact with water, such as bathing, swimming or showering.
Contraindications
LIDOCAINE PATCH 5% is contraindicated in patients with a known history of sensitivity to local anesthetics of the amide type, or to any other component of the product.
Drug Interactions
Antiarrhythmic Drugs
LIDOCAINE PATCH 5% should be used with caution in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) since the toxic effects are additive and potentially synergistic.
Local Anesthetics
When LIDOCAINE PATCH 5% is used concomitantly with other products containing local anesthetic agents, the amount absorbed from all formulations must be considered.
Drugs That May Cause Methemoglobinemia When Used with LIDOCAINE PATCH 5%
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
|
Class |
Examples |
|
Nitrates/Nitrites |
nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
|
Local anesthetics |
articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
|
Antineoplastic agents |
cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
|
Antibiotics |
dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
|
Antimalarials |
chloroquine, primaquine |
|
Anticonvulsants |
Phenobarbital, phenytoin, sodium valproate |
|
Other drugs |
acetaminophen, metoclopramide, quinine, sulfasalazine |
How Supplied
LIDOCAINE PATCH 5% is available as the following:
Carton of 30 patches, packaged into individual child-resistant envelopes
NDC 68788-7159-3
Store at 25oC (77oF); excursions permitted to 15o-30oC (59o-86oF). [See USP Controlled Room Temperature].
For more information, call Par Pharmacutical at 1-800-828-9393.
Manufactured for:
Par Pharmaceutical
Chestnut Ridge, NY 10977
Printed in U.S.A.
Revised: April 2024
Relabeled By: Preferred Pharmaceuticals Inc.
Description
LIDOCAINE PATCH 5% is comprised of an adhesive material containing 5% lidocaine, which is applied to a non‑woven polyester felt backing and covered with a polyethylene terephthalate (PET) film release liner. The release liner is removed prior to application to the skin. The size of the patch is 10 cm x 14 cm.
Lidocaine is chemically designated as acetamide, 2‑(diethylamino)‑N‑(2,6‑ dimethylphenyl), has an octanol: water partition ratio of 43 at pH 7.4, and has the following structure:
Each adhesive patch contains 700 mg of lidocaine (50 mg per gram adhesive) in an aqueous base. It also contains the following inactive ingredients: dihydroxyaluminum aminoacetate, disodium edetate, gelatin, glycerin, kaolin, methylparaben, polyacrylic acid, polyvinyl alcohol, propylene glycol, propylparaben, sodium carboxymethylcellulose, sodium polyacrylate, D-sorbitol, tartaric acid, and urea.
Section 42229-5
Rx only
Section 51945-4
Package Label - Principal Display Panel – 30 Count Carton, Lidocaine Patch
General
Hepatic Disease
Patients with severe hepatic disease are at greater risk of developing toxic blood concentrations of lidocaine, because of their inability to metabolize lidocaine normally.
Allergic Reactions
Patients allergic to para‑aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine. However, LIDOCAINE PATCH 5% should be used with caution in patients with a history of drug sensitivities, especially if the etiologic agent is uncertain.
Non-intact Skin
Application to broken or inflamed skin, although not tested, may result in higher blood concentrations of lidocaine from increased absorption. LIDOCAINE PATCH 5% is only recommended for use on intact skin.
External Heat Sources
Placement of external heat sources, such as heating pads or electric blankets, over LIDOCAINE PATCH 5% patches is not recommended as this has not been evaluated and may increase plasma lidocaine levels.
Eye Exposure
The contact of LIDOCAINE PATCH 5% with eyes, although not studied, should be avoided based on the findings of severe eye irritation with the use of similar products in animals. If eye contact occurs, immediately wash out the eye with water or saline and protect the eye until sensation returns.
Pregnancy
Teratogenic Effects
Pregnancy Category B.
LIDOCAINE PATCH 5% has not been studied in pregnancy. Reproduction studies with lidocaine have been performed in rats at doses up to 30 mg/kg subcutaneously and have revealed no evidence of harm to the fetus due to lidocaine. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, LIDOCAINE PATCH 5% should be used during pregnancy only if clearly needed.
Overdosage
Lidocaine overdose from cutaneous absorption is rare, but could occur. If there is any suspicion of lidocaine overdose (see ADVERSE REACTIONS, Systemic Reactions), drug blood concentration should be checked. The management of overdose includes close monitoring, supportive care, and symptomatic treatment. Dialysis is of negligible value in the treatment of acute overdose with lidocaine.
In the absence of massive topical overdose or oral ingestion, evaluation of symptoms of toxicity should include consideration of other etiologies for the clinical effects, or overdosage from other sources of lidocaine or other local anesthetics.
The oral LD50 of lidocaine HCl is 459 (346-773) mg/kg (as the salt) in non-fasted female rats and 214 (159-324) mg/kg (as the salt) in fasted female rats, which are equivalent to roughly 4000 mg and 2000 mg, respectively, in a 60 to 70 kg man based on the equivalent surface area dosage conversion factors between species.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Nursing Mothers
LIDOCAINE PATCH 5% has not been studied in nursing mothers. Lidocaine is excreted in human milk, and the milk: plasma ratio of lidocaine is 0.4. Caution should be exercised when LIDOCAINE PATCH 5% is administered to a nursing woman.
Clinical Studies
Single-dose treatment with LIDOCAINE PATCH 5% was compared to treatment with vehicle patch (without lidocaine), and to no treatment (observation only) in a double‑blind, crossover clinical trial with 35 post‑herpetic neuralgia patients. Pain intensity and pain relief scores were evaluated periodically for 12 hours. LIDOCAINE PATCH 5% performed statistically better than vehicle patch in terms of pain intensity from 4 to 12 hours.
Multiple-dose, two-week treatment with LIDOCAINE PATCH 5% was compared to vehicle patch (without lidocaine) in a double-blind, crossover clinical trial of withdrawal-type design conducted in 32 patients, who were considered as responders to the open-label use of LIDOCAINE PATCH 5% prior to the study. The constant type of pain was evaluated but not the pain induced by sensory stimuli (dysesthesia). Statistically significant differences favoring LIDOCAINE PATCH 5% were observed in terms of time to exit from the trial (14 versus 3.8 days at p-value <0.001), daily average pain relief, and patient’s preference of treatment. About half of the patients also took oral medication commonly used in the treatment of post-herpetic neuralgia. The extent of use of concomitant medication was similar in the two treatment groups.
Excessive Dosing
Excessive dosing by applying LIDOCAINE PATCH 5% to larger areas or for longer than the recommended wearing time could result in increased absorption of lidocaine and high blood concentrations, leading to serious adverse effects (see ADVERSE REACTIONS, Systemic Reactions). Lidocaine toxicity could be expected at lidocaine blood concentrations above 5 mcg/mL. The blood concentration of lidocaine is determined by the rate of systemic absorption and elimination. Longer duration of application, application of more than the recommended number of patches, smaller patients, or impaired elimination may all contribute to increasing the blood concentration of lidocaine. With recommended dosing of LIDOCAINE PATCH 5%, the average peak blood concentration is about 0.13 mcg/mL, but concentrations higher than 0.25 mcg/mL have been observed in some individuals.
Pharmacodynamics
Lidocaine is an amide-type local anesthetic agent and is suggested to stabilize neuronal membranes by inhibiting the ionic fluxes required for the initiation and conduction of impulses.
The penetration of lidocaine into intact skin after application of LIDOCAINE PATCH 5% is sufficient to produce an analgesic effect, but less than the amount necessary to produce a complete sensory block.
Allergic Reactions
Allergic and anaphylactoid reactions associated with lidocaine, although rare, can occur. They are characterized by angioedema, bronchospasm, dermatitis, dyspnea, hypersensitivity, laryngospasm, pruritus, shock, and urticaria. If they occur, they should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
Labor and Delivery
LIDOCAINE PATCH 5% has not been studied in labor and delivery. Lidocaine is not contraindicated in labor and delivery. Should LIDOCAINE PATCH 5% be used concomitantly with other products containing lidocaine, total doses contributed by all formulations must be considered.
Indication and Usage
LIDOCAINE PATCH 5% is indicated for relief of pain associated with post-herpetic neuralgia. It should be applied only to intact skin.
Other Adverse Events
Due to the nature and limitation of spontaneous reports in postmarketing surveillance, causality has not been established for additional reported adverse events including:
Asthenia, confusion, disorientation, dizziness, headache, hyperesthesia, hypoesthesia, lightheadedness, metallic taste, nausea, nervousness, pain exacerbated, paresthesia, somnolence, taste alteration, vomiting, visual disturbances such as blurred vision, flushing, tinnitus, and tremor.
Handling and Disposal
Hands should be washed after the handling of LIDOCAINE PATCH 5%, and eye contact with LIDOCAINE PATCH 5% should be avoided. Do not store patch outside the sealed envelope. Apply immediately after removal from the protective envelope. Fold used patches so that the adhesive side sticks to itself and safely discard used patches or pieces of cut patches where children and pets cannot get to them. LIDOCAINE PATCH 5% should be kept out of the reach of children.
Information for Patients
Methemoglobinemia
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to stop use and seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.
Risk of Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue LIDOCAINE PATCH 5% and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
Application Site Reactions
During or immediately after treatment with LIDOCAINE PATCH 5%, the skin at the site of application may develop blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erythema, exfoliation, irritation, papules, petechia, pruritus, vesicles, or may be the locus of abnormal sensation. These reactions are generally mild and transient, resolving spontaneously within a few minutes to hours.
Accidental Exposure in Children
Even a used LIDOCAINE PATCH 5% patch contains a large amount of lidocaine (at least 665 mg). The potential exists for a small child or a pet to suffer serious adverse effects from chewing or ingesting a new or used LIDOCAINE PATCH 5% patch, although the risk with this formulation has not been evaluated. It is important for patients to store and dispose of LIDOCAINE PATCH 5% out of the reach of children, pets and others. (See HANDLING AND DISPOSAL)
Systemic (dose Related) Reactions
Systemic adverse reactions following appropriate use of LIDOCAINE PATCH 5% are unlikely, due to the small dose absorbed (see CLINICAL PHARMACOLOGY, Pharmacokinetics). Systemic adverse effects of lidocaine are similar in nature to those observed with other amide local anesthetic agents, including CNS excitation and/or depression (light headedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest). Excitatory CNS reactions may be brief or not occur at all, in which case the first manifestation may be drowsiness merging into unconsciousness. Cardiovascular manifestations may include bradycardia, hypotension and cardiovascular collapse leading to arrest.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
A minor metabolite, 2, 6-xylidine, has been found to be carcinogenic in rats. The blood concentration of this metabolite is negligible following application of LIDOCAINE PATCH 5%.
Mutagenesis
Lidocaine HCl is not mutagenic in Salmonella/mammalian microsome test nor clastogenic in chromosome aberration assay with human lymphocytes and mouse micronucleus test.
Impairment of Fertility
The effect of LIDOCAINE PATCH 5% on fertility has not been studied.
Structured Label Content
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Apply LIDOCAINE PATCH 5% to intact skin to cover the most painful area. Apply the prescribed number of patches (maximum of 3), only once for up to 12 hours within a 24 hour period. Patches may be cut into smaller sizes with scissors prior to removal of the release liner. (See HANDLING AND DISPOSAL) Clothing may be worn over the area of application. Smaller areas of treatment are recommended in a debilitated patient, or a patient with impaired elimination.
If irritation or a burning sensation occurs during application, remove the patch(es) and do not reapply until the irritation subsides.
When LIDOCAINE PATCH 5% is used concomitantly with other products containing local anesthetic agents, the amount absorbed from all formulations must be considered.
LIDOCAINE PATCH 5% may not stick if it gets wet. Avoid contact with water, such as bathing, swimming or showering.
Contraindications (CONTRAINDICATIONS)
LIDOCAINE PATCH 5% is contraindicated in patients with a known history of sensitivity to local anesthetics of the amide type, or to any other component of the product.
Drug Interactions
Antiarrhythmic Drugs
LIDOCAINE PATCH 5% should be used with caution in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) since the toxic effects are additive and potentially synergistic.
Local Anesthetics
When LIDOCAINE PATCH 5% is used concomitantly with other products containing local anesthetic agents, the amount absorbed from all formulations must be considered.
Drugs That May Cause Methemoglobinemia When Used with LIDOCAINE PATCH 5%
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
|
Class |
Examples |
|
Nitrates/Nitrites |
nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
|
Local anesthetics |
articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
|
Antineoplastic agents |
cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
|
Antibiotics |
dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
|
Antimalarials |
chloroquine, primaquine |
|
Anticonvulsants |
Phenobarbital, phenytoin, sodium valproate |
|
Other drugs |
acetaminophen, metoclopramide, quinine, sulfasalazine |
How Supplied (HOW SUPPLIED)
LIDOCAINE PATCH 5% is available as the following:
Carton of 30 patches, packaged into individual child-resistant envelopes
NDC 68788-7159-3
Store at 25oC (77oF); excursions permitted to 15o-30oC (59o-86oF). [See USP Controlled Room Temperature].
For more information, call Par Pharmacutical at 1-800-828-9393.
Manufactured for:
Par Pharmaceutical
Chestnut Ridge, NY 10977
Printed in U.S.A.
Revised: April 2024
Relabeled By: Preferred Pharmaceuticals Inc.
Description (DESCRIPTION)
LIDOCAINE PATCH 5% is comprised of an adhesive material containing 5% lidocaine, which is applied to a non‑woven polyester felt backing and covered with a polyethylene terephthalate (PET) film release liner. The release liner is removed prior to application to the skin. The size of the patch is 10 cm x 14 cm.
Lidocaine is chemically designated as acetamide, 2‑(diethylamino)‑N‑(2,6‑ dimethylphenyl), has an octanol: water partition ratio of 43 at pH 7.4, and has the following structure:
Each adhesive patch contains 700 mg of lidocaine (50 mg per gram adhesive) in an aqueous base. It also contains the following inactive ingredients: dihydroxyaluminum aminoacetate, disodium edetate, gelatin, glycerin, kaolin, methylparaben, polyacrylic acid, polyvinyl alcohol, propylene glycol, propylparaben, sodium carboxymethylcellulose, sodium polyacrylate, D-sorbitol, tartaric acid, and urea.
Section 42229-5 (42229-5)
Rx only
Section 51945-4 (51945-4)
Package Label - Principal Display Panel – 30 Count Carton, Lidocaine Patch
General
Hepatic Disease
Patients with severe hepatic disease are at greater risk of developing toxic blood concentrations of lidocaine, because of their inability to metabolize lidocaine normally.
Allergic Reactions
Patients allergic to para‑aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine. However, LIDOCAINE PATCH 5% should be used with caution in patients with a history of drug sensitivities, especially if the etiologic agent is uncertain.
Non-intact Skin
Application to broken or inflamed skin, although not tested, may result in higher blood concentrations of lidocaine from increased absorption. LIDOCAINE PATCH 5% is only recommended for use on intact skin.
External Heat Sources
Placement of external heat sources, such as heating pads or electric blankets, over LIDOCAINE PATCH 5% patches is not recommended as this has not been evaluated and may increase plasma lidocaine levels.
Eye Exposure
The contact of LIDOCAINE PATCH 5% with eyes, although not studied, should be avoided based on the findings of severe eye irritation with the use of similar products in animals. If eye contact occurs, immediately wash out the eye with water or saline and protect the eye until sensation returns.
Pregnancy
Teratogenic Effects
Pregnancy Category B.
LIDOCAINE PATCH 5% has not been studied in pregnancy. Reproduction studies with lidocaine have been performed in rats at doses up to 30 mg/kg subcutaneously and have revealed no evidence of harm to the fetus due to lidocaine. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, LIDOCAINE PATCH 5% should be used during pregnancy only if clearly needed.
Overdosage (OVERDOSAGE)
Lidocaine overdose from cutaneous absorption is rare, but could occur. If there is any suspicion of lidocaine overdose (see ADVERSE REACTIONS, Systemic Reactions), drug blood concentration should be checked. The management of overdose includes close monitoring, supportive care, and symptomatic treatment. Dialysis is of negligible value in the treatment of acute overdose with lidocaine.
In the absence of massive topical overdose or oral ingestion, evaluation of symptoms of toxicity should include consideration of other etiologies for the clinical effects, or overdosage from other sources of lidocaine or other local anesthetics.
The oral LD50 of lidocaine HCl is 459 (346-773) mg/kg (as the salt) in non-fasted female rats and 214 (159-324) mg/kg (as the salt) in fasted female rats, which are equivalent to roughly 4000 mg and 2000 mg, respectively, in a 60 to 70 kg man based on the equivalent surface area dosage conversion factors between species.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Nursing Mothers
LIDOCAINE PATCH 5% has not been studied in nursing mothers. Lidocaine is excreted in human milk, and the milk: plasma ratio of lidocaine is 0.4. Caution should be exercised when LIDOCAINE PATCH 5% is administered to a nursing woman.
Clinical Studies (CLINICAL STUDIES)
Single-dose treatment with LIDOCAINE PATCH 5% was compared to treatment with vehicle patch (without lidocaine), and to no treatment (observation only) in a double‑blind, crossover clinical trial with 35 post‑herpetic neuralgia patients. Pain intensity and pain relief scores were evaluated periodically for 12 hours. LIDOCAINE PATCH 5% performed statistically better than vehicle patch in terms of pain intensity from 4 to 12 hours.
Multiple-dose, two-week treatment with LIDOCAINE PATCH 5% was compared to vehicle patch (without lidocaine) in a double-blind, crossover clinical trial of withdrawal-type design conducted in 32 patients, who were considered as responders to the open-label use of LIDOCAINE PATCH 5% prior to the study. The constant type of pain was evaluated but not the pain induced by sensory stimuli (dysesthesia). Statistically significant differences favoring LIDOCAINE PATCH 5% were observed in terms of time to exit from the trial (14 versus 3.8 days at p-value <0.001), daily average pain relief, and patient’s preference of treatment. About half of the patients also took oral medication commonly used in the treatment of post-herpetic neuralgia. The extent of use of concomitant medication was similar in the two treatment groups.
Excessive Dosing
Excessive dosing by applying LIDOCAINE PATCH 5% to larger areas or for longer than the recommended wearing time could result in increased absorption of lidocaine and high blood concentrations, leading to serious adverse effects (see ADVERSE REACTIONS, Systemic Reactions). Lidocaine toxicity could be expected at lidocaine blood concentrations above 5 mcg/mL. The blood concentration of lidocaine is determined by the rate of systemic absorption and elimination. Longer duration of application, application of more than the recommended number of patches, smaller patients, or impaired elimination may all contribute to increasing the blood concentration of lidocaine. With recommended dosing of LIDOCAINE PATCH 5%, the average peak blood concentration is about 0.13 mcg/mL, but concentrations higher than 0.25 mcg/mL have been observed in some individuals.
Pharmacodynamics
Lidocaine is an amide-type local anesthetic agent and is suggested to stabilize neuronal membranes by inhibiting the ionic fluxes required for the initiation and conduction of impulses.
The penetration of lidocaine into intact skin after application of LIDOCAINE PATCH 5% is sufficient to produce an analgesic effect, but less than the amount necessary to produce a complete sensory block.
Allergic Reactions
Allergic and anaphylactoid reactions associated with lidocaine, although rare, can occur. They are characterized by angioedema, bronchospasm, dermatitis, dyspnea, hypersensitivity, laryngospasm, pruritus, shock, and urticaria. If they occur, they should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
Labor and Delivery
LIDOCAINE PATCH 5% has not been studied in labor and delivery. Lidocaine is not contraindicated in labor and delivery. Should LIDOCAINE PATCH 5% be used concomitantly with other products containing lidocaine, total doses contributed by all formulations must be considered.
Indication and Usage (INDICATION AND USAGE)
LIDOCAINE PATCH 5% is indicated for relief of pain associated with post-herpetic neuralgia. It should be applied only to intact skin.
Other Adverse Events
Due to the nature and limitation of spontaneous reports in postmarketing surveillance, causality has not been established for additional reported adverse events including:
Asthenia, confusion, disorientation, dizziness, headache, hyperesthesia, hypoesthesia, lightheadedness, metallic taste, nausea, nervousness, pain exacerbated, paresthesia, somnolence, taste alteration, vomiting, visual disturbances such as blurred vision, flushing, tinnitus, and tremor.
Handling and Disposal (HANDLING AND DISPOSAL)
Hands should be washed after the handling of LIDOCAINE PATCH 5%, and eye contact with LIDOCAINE PATCH 5% should be avoided. Do not store patch outside the sealed envelope. Apply immediately after removal from the protective envelope. Fold used patches so that the adhesive side sticks to itself and safely discard used patches or pieces of cut patches where children and pets cannot get to them. LIDOCAINE PATCH 5% should be kept out of the reach of children.
Information for Patients
Methemoglobinemia
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to stop use and seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.
Risk of Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue LIDOCAINE PATCH 5% and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
Application Site Reactions
During or immediately after treatment with LIDOCAINE PATCH 5%, the skin at the site of application may develop blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erythema, exfoliation, irritation, papules, petechia, pruritus, vesicles, or may be the locus of abnormal sensation. These reactions are generally mild and transient, resolving spontaneously within a few minutes to hours.
Accidental Exposure in Children
Even a used LIDOCAINE PATCH 5% patch contains a large amount of lidocaine (at least 665 mg). The potential exists for a small child or a pet to suffer serious adverse effects from chewing or ingesting a new or used LIDOCAINE PATCH 5% patch, although the risk with this formulation has not been evaluated. It is important for patients to store and dispose of LIDOCAINE PATCH 5% out of the reach of children, pets and others. (See HANDLING AND DISPOSAL)
Systemic (dose Related) Reactions (Systemic (Dose-Related) Reactions)
Systemic adverse reactions following appropriate use of LIDOCAINE PATCH 5% are unlikely, due to the small dose absorbed (see CLINICAL PHARMACOLOGY, Pharmacokinetics). Systemic adverse effects of lidocaine are similar in nature to those observed with other amide local anesthetic agents, including CNS excitation and/or depression (light headedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest). Excitatory CNS reactions may be brief or not occur at all, in which case the first manifestation may be drowsiness merging into unconsciousness. Cardiovascular manifestations may include bradycardia, hypotension and cardiovascular collapse leading to arrest.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
A minor metabolite, 2, 6-xylidine, has been found to be carcinogenic in rats. The blood concentration of this metabolite is negligible following application of LIDOCAINE PATCH 5%.
Mutagenesis
Lidocaine HCl is not mutagenic in Salmonella/mammalian microsome test nor clastogenic in chromosome aberration assay with human lymphocytes and mouse micronucleus test.
Impairment of Fertility
The effect of LIDOCAINE PATCH 5% on fertility has not been studied.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:16.047594 · Updated: 2026-03-14T22:29:27.892649