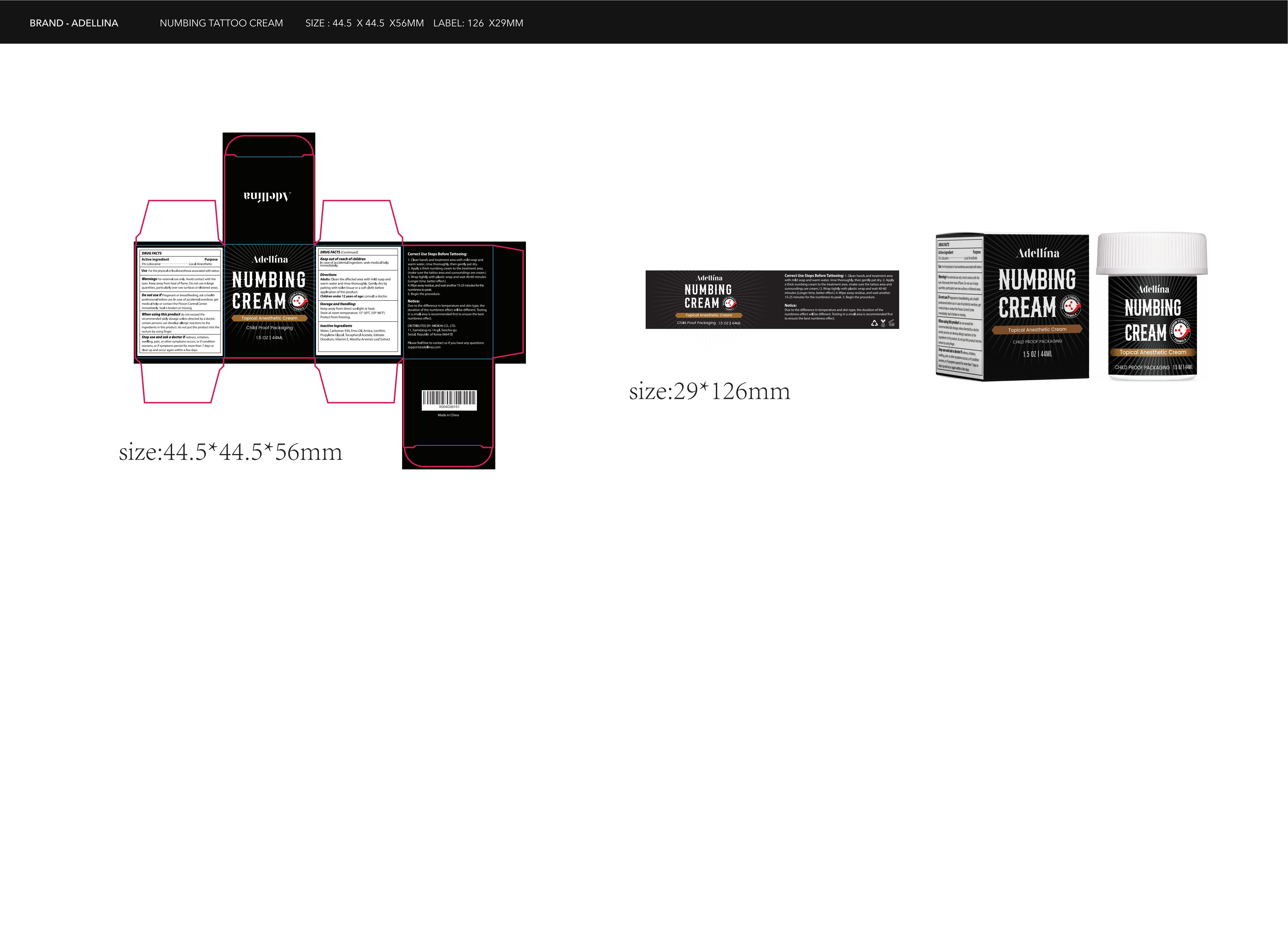
Adellina Numbing Cream
314a83fe-8bdb-766f-e063-6294a90a61c6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 5%
Purpose
Local Anesthetic
Medication Information
Purpose
Local Anesthetic
Description
Lidocaine 5%
Uses
For the physical or local anesthesia associated with tattoo
Dosage
Squeeze out an appropriate amount of product and spread evenly on skin.
Section 50566-9
Stop use and ask a doctor if redness, irritation, swelling, pain, or other symptoms occurs,or If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days.
Section 50567-7
Do not exceed the recommended daily dosage unless directed by a doctor. certain persons can develop allergic reactions to ingredients in this product. do not put this product into the rectum by using finger.
Section 50570-1
Do not use this product if Pregnant or breastfeeding, ask a health professional before use. In case of accidental overdose, get medical help or contact the Poison Control Center immediately. Seal is broken or missing.
Section 51945-4
Active Ingredients
Lidocaine 5%
Inactive Ingredients
AQUA 71.3%
PROPYLENE 5%
CARBOMER 0.25%
GLYCERIN 5%
DISODIUM EDTA 0.05%
HYDROXYACETOPHENONE 0.4%
1,2-HEXANEDIOL 5%
MENTHA ARVENSIS LEAF EXTRACT 2%
CHRYSANTHELLUM INDICUM EXTRACT 2%
PORTULACA OLERACEA EXTRACT 2%
PEG-40 HYDROGENATED CASTOR OIL 2%
For External Use Only.
Avoid contact with the eyes Keep away from heat or flame. Do not use in large quantities, particularly over raw surfaces or blistered areas.
Keep Out of Reach of Children
In case of accidental ingestion, seek medical help immediately.
Structured Label Content
Uses
For the physical or local anesthesia associated with tattoo
Dosage
Squeeze out an appropriate amount of product and spread evenly on skin.
Section 50566-9 (50566-9)
Stop use and ask a doctor if redness, irritation, swelling, pain, or other symptoms occurs,or If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days.
Section 50567-7 (50567-7)
Do not exceed the recommended daily dosage unless directed by a doctor. certain persons can develop allergic reactions to ingredients in this product. do not put this product into the rectum by using finger.
Section 50570-1 (50570-1)
Do not use this product if Pregnant or breastfeeding, ask a health professional before use. In case of accidental overdose, get medical help or contact the Poison Control Center immediately. Seal is broken or missing.
Section 51945-4 (51945-4)
Purpose
Local Anesthetic
Active Ingredients (Active ingredients)
Lidocaine 5%
Inactive Ingredients (Inactive ingredients)
AQUA 71.3%
PROPYLENE 5%
CARBOMER 0.25%
GLYCERIN 5%
DISODIUM EDTA 0.05%
HYDROXYACETOPHENONE 0.4%
1,2-HEXANEDIOL 5%
MENTHA ARVENSIS LEAF EXTRACT 2%
CHRYSANTHELLUM INDICUM EXTRACT 2%
PORTULACA OLERACEA EXTRACT 2%
PEG-40 HYDROGENATED CASTOR OIL 2%
For External Use Only. (For external use only.)
Avoid contact with the eyes Keep away from heat or flame. Do not use in large quantities, particularly over raw surfaces or blistered areas.
Keep Out of Reach of Children (Keep out of reach of children)
In case of accidental ingestion, seek medical help immediately.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:47.660200 · Updated: 2026-03-14T23:08:06.304944