Valacyclovir Tablets
3146e367-8438-4141-9368-95ce6f59c724
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Valacyclovir tablets are a deoxynucleoside analogue DNA polymerase inhibitor indicated for: Adult Patients ( ) • Cold Sores (Herpes Labialis) • Genital Herpes o Treatment in immunocompetent patients (initial or recurrent episode) o Suppression in immunocompetent or HIV-1-infected patients o Reduction of transmission • Herpes Zoster Pediatric Patients ( ) • Cold Sores (Herpes Labialis) • Chickenpox Limitations of Use ( 1.3 ) • The efficacy and safety of valacyclovir tablets have not been established in immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients.
Dosage and Administration
• Valacyclovir tablets may be given without regard to meals. • Valacyclovir oral suspension (25 mg/mL or 50 mg/mL) may be prepared extemporaneously from the 500-mg valacyclovir tablets for use in pediatric patients for whom a solid dosage form is not appropriate [see ].
Contraindications
Valacyclovir tablets are contraindicated in patients who have had a demonstrated clinically significant hypersensitivity reaction (e.g., anaphylaxis) to valacyclovir, acyclovir, or any component of the formulation [see ].
Warnings and Precautions
• Thrombotic thrombocytopenic purpura/hemolytic uremic syndrome (TTP/HUS): Has occurred in patients with advanced HIV-1 disease and in allogenic bone marrow transplant and renal transplant patients receiving 8 grams per day of valacyclovir tablets in clinical trials. Discontinue treatment if clinical symptoms and laboratory findings consistent with TTP/HUS occur. () • Acute renal failure: May occur in elderly patients (with or without reduced renal function), patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function, patients who receive concomitant nephrotoxic drugs, or inadequately hydrated patients. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (, 5.2 ) • Central nervous system adverse reactions (e.g., agitation, hallucinations, confusion, and encephalopathy): May occur in both adult and pediatric patients (with or without reduced renal function) and in patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function. Elderly patients are more likely to have central nervous system adverse reactions. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (,)
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: • Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome [see ]. • Acute Renal Failure [see Warnings and Precautions (5.2) ]. • Central Nervous System Effects [see ]. The most common adverse reactions reported in at least 1 indication by greater than 10% of adult subjects treated with valacyclovir tablets and observed more frequently with valacyclovir tablets compared with placebo are headache, nausea, and abdominal pain. The only adverse reaction reported in greater than 10% of pediatric subjects aged less than 18 years was headache.
Drug Interactions
No clinically significant drug-drug or drug-food interactions with valacyclovir tablets are known [see ].
How Supplied
Valacyclovir Tablets, USP 1 gram (blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side) containing 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram valacyclovir. Bottle of 0 NDC 68788-8705-4 Bottle of 0 NDC 68788-8705-0 Bottle of 0 NDC 68788-8705-1 Bottle of 0 NDC 68788-8705-5 Bottle of 0 NDC 68788-8705-2 Bottle of 0 NDC 68788-8705-7 Bottle of 0 NDC 68788-8705-3
Storage and Handling
Valacyclovir Tablets, USP 1 gram (blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side) containing 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram valacyclovir. Bottle of 0 NDC 68788-8705-4 Bottle of 0 NDC 68788-8705-0 Bottle of 0 NDC 68788-8705-1 Bottle of 0 NDC 68788-8705-5 Bottle of 0 NDC 68788-8705-2 Bottle of 0 NDC 68788-8705-7 Bottle of 0 NDC 68788-8705-3
Description
Valacyclovir tablets are a deoxynucleoside analogue DNA polymerase inhibitor indicated for: Adult Patients ( ) • Cold Sores (Herpes Labialis) • Genital Herpes o Treatment in immunocompetent patients (initial or recurrent episode) o Suppression in immunocompetent or HIV-1-infected patients o Reduction of transmission • Herpes Zoster Pediatric Patients ( ) • Cold Sores (Herpes Labialis) • Chickenpox Limitations of Use ( 1.3 ) • The efficacy and safety of valacyclovir tablets have not been established in immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients.
Medication Information
Warnings and Precautions
• Thrombotic thrombocytopenic purpura/hemolytic uremic syndrome (TTP/HUS): Has occurred in patients with advanced HIV-1 disease and in allogenic bone marrow transplant and renal transplant patients receiving 8 grams per day of valacyclovir tablets in clinical trials. Discontinue treatment if clinical symptoms and laboratory findings consistent with TTP/HUS occur. () • Acute renal failure: May occur in elderly patients (with or without reduced renal function), patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function, patients who receive concomitant nephrotoxic drugs, or inadequately hydrated patients. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (, 5.2 ) • Central nervous system adverse reactions (e.g., agitation, hallucinations, confusion, and encephalopathy): May occur in both adult and pediatric patients (with or without reduced renal function) and in patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function. Elderly patients are more likely to have central nervous system adverse reactions. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (,)
Indications and Usage
Valacyclovir tablets are a deoxynucleoside analogue DNA polymerase inhibitor indicated for: Adult Patients ( ) • Cold Sores (Herpes Labialis) • Genital Herpes o Treatment in immunocompetent patients (initial or recurrent episode) o Suppression in immunocompetent or HIV-1-infected patients o Reduction of transmission • Herpes Zoster Pediatric Patients ( ) • Cold Sores (Herpes Labialis) • Chickenpox Limitations of Use ( 1.3 ) • The efficacy and safety of valacyclovir tablets have not been established in immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients.
Dosage and Administration
• Valacyclovir tablets may be given without regard to meals. • Valacyclovir oral suspension (25 mg/mL or 50 mg/mL) may be prepared extemporaneously from the 500-mg valacyclovir tablets for use in pediatric patients for whom a solid dosage form is not appropriate [see ].
Contraindications
Valacyclovir tablets are contraindicated in patients who have had a demonstrated clinically significant hypersensitivity reaction (e.g., anaphylaxis) to valacyclovir, acyclovir, or any component of the formulation [see ].
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: • Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome [see ]. • Acute Renal Failure [see Warnings and Precautions (5.2) ]. • Central Nervous System Effects [see ]. The most common adverse reactions reported in at least 1 indication by greater than 10% of adult subjects treated with valacyclovir tablets and observed more frequently with valacyclovir tablets compared with placebo are headache, nausea, and abdominal pain. The only adverse reaction reported in greater than 10% of pediatric subjects aged less than 18 years was headache.
Drug Interactions
No clinically significant drug-drug or drug-food interactions with valacyclovir tablets are known [see ].
Storage and Handling
Valacyclovir Tablets, USP 1 gram (blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side) containing 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram valacyclovir. Bottle of 0 NDC 68788-8705-4 Bottle of 0 NDC 68788-8705-0 Bottle of 0 NDC 68788-8705-1 Bottle of 0 NDC 68788-8705-5 Bottle of 0 NDC 68788-8705-2 Bottle of 0 NDC 68788-8705-7 Bottle of 0 NDC 68788-8705-3
How Supplied
Valacyclovir Tablets, USP 1 gram (blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side) containing 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram valacyclovir. Bottle of 0 NDC 68788-8705-4 Bottle of 0 NDC 68788-8705-0 Bottle of 0 NDC 68788-8705-1 Bottle of 0 NDC 68788-8705-5 Bottle of 0 NDC 68788-8705-2 Bottle of 0 NDC 68788-8705-7 Bottle of 0 NDC 68788-8705-3
Description
Valacyclovir tablets are a deoxynucleoside analogue DNA polymerase inhibitor indicated for: Adult Patients ( ) • Cold Sores (Herpes Labialis) • Genital Herpes o Treatment in immunocompetent patients (initial or recurrent episode) o Suppression in immunocompetent or HIV-1-infected patients o Reduction of transmission • Herpes Zoster Pediatric Patients ( ) • Cold Sores (Herpes Labialis) • Chickenpox Limitations of Use ( 1.3 ) • The efficacy and safety of valacyclovir tablets have not been established in immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients.
Section 42229-5
Cold Sores (Herpes Labialis)
Valacyclovir tablets are indicated for treatment of cold sores (herpes labialis). The efficacy of valacyclovir tablets initiated after the development of clinical signs of a cold sore (e.g., papule, vesicle, or ulcer) has not been established.
Section 44425-7
Storage:
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
10 Overdosage
Caution should be exercised to prevent inadvertent overdose [see , ]. Precipitation of acyclovir in renal tubules may occur when the solubility (2.5 mg/mL) is exceeded in the intratubular fluid. In the event of acute renal failure and anuria, the patient may benefit from hemodialysis until renal function is restored [see ].
11 Description
Valacyclovir Tablets, USP is the hydrochloride salt of the L-valyl ester of the antiviral drug acyclovir.
Valacyclovir Tablets, USP are for oral administration. Each tablet contains 556.2 mg or 1.112 grams of valacyclovir hydrochloride equivalent to 500 mg or 1 gram of valacyclovir, respectively, and the inactive ingredients anhydrous dibasic calcium phosphate, colloidal silicon dioxide, croscarmellose sodium, FD&C blue #2, hypromellose, magnesium stearate, polyethylene glycol 400, polysorbate 80, povidone K90, and titanium dioxide.
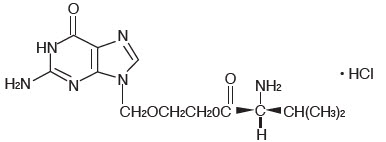
The chemical name of valacyclovir hydrochloride is L-valine, 2-[(2-amino-1,6-dihydro-6-oxo-9H-purin-9-yl)methoxy]ethyl ester, monohydrochloride. It has the following structural formula:
Valacyclovir hydrochloride is a white to off-white powder with the molecular formula C13H20N6O4∙HCl and a molecular weight of 360.80. The maximum solubility in water at 25°C is 174 mg/mL. The pkas for valacyclovir hydrochloride are 1.90, 7.47, and 9.43.
14.4 Chickenpox
The use of valacyclovir tablets for treatment of chickenpox in pediatric subjects aged 2 to less than 18 years is based on single-dose pharmacokinetic and multiple-dose safety data from an open-label trial with valacyclovir and supported by safety and extrapolated efficacy data from 3 randomized, double-blind, placebo-controlled trials evaluating oral acyclovir in pediatric subjects.
The single-dose pharmacokinetic and multiple-dose safety trial enrolled 27 pediatric subjects aged 1 to less than 12 years with clinically suspected VZV infection. Each subject was dosed with valacyclovir oral suspension, 20 mg/kg 3 times daily for 5 days. Acyclovir systemic exposures in pediatric subjects following valacyclovir oral suspension were compared with historical acyclovir systemic exposures in immunocompetent adults receiving the solid oral dosage form of valacyclovir or acyclovir for the treatment of herpes zoster. The mean projected daily acyclovir exposures in pediatric subjects across all age-groups (1 to less than 12 years) were lower (Cmax: ↓13%, AUC: ↓30%)than the mean daily historical exposures in adults receiving valacyclovir 1 gram 3 times daily, but were higher (daily AUC: ↑50%) than the mean daily historical exposures in adults receiving acyclovir 800 mg 5 times daily. The projected daily exposures in pediatric subjects were greater (daily AUC approximately 100% greater) than the exposures seen in immunocompetent pediatric subjects receiving acyclovir 20 mg/kg 4 times daily for the treatment of chickenpox. Based on the pharmacokinetic and safety data from this trial and the safety and extrapolated efficacy data from the acyclovir trials, oral valacyclovir 20 mg/kg 3 times a day for 5 days (not to exceed 1 gram 3 times daily) is recommended for the treatment of chickenpox in pediatric patients aged 2 to less than 18 years. Because the efficacy and safety of acyclovir for the treatment of chickenpox in children aged less than 2 years have not been established, efficacy data cannot be extrapolated to support valacyclovir treatment in children aged less than 2 years with chickenpox.
Valacyclovir is also not recommended for the treatment of herpes zoster in children because safety data up to 7 days' duration are not available [see Use in Specific Populations (8.4)].
8.4 Pediatric Use
Valacyclovir tablets are indicated for treatment of cold sores in pediatric patients aged greater than or equal to 12 years and for treatment of chickenpox in pediatric patients aged 2 to less than 18 years [see , ].
The use of valacyclovir tablets for treatment of cold sores is based on 2 double-blind, placebo-controlled clinical trials in healthy adults and adolescents (aged greater than or equal to 12 years) with a history of recurrent cold sores [see ].
The use of valacyclovir tablets for treatment of chickenpox in pediatric patients aged 2 to less than 18 years is based on single-dose pharmacokinetic and multiple-dose safety data from an open-label trial with valacyclovir and supported by efficacy and safety data from 3 randomized, double-blind, placebo-controlled trials evaluating oral acyclovir in pediatric subjects with chickenpox [see , , , ].
The efficacy and safety of valacyclovir have not been established in pediatric patients:
-
•aged less than 12 years with cold sores
-
•aged less than 18 years with genital herpes
-
•aged less than 18 years with herpes zoster
-
•aged less than 2 years with chickenpox
-
•for suppressive therapy following neonatal HSV infection.
The pharmacokinetic profile and safety of valacyclovir oral suspension in children aged less than 12 years were studied in 3 open label trials. No efficacy evaluations were conducted in any of the 3 trials.
Trial 1 was a single dose pharmacokinetic, multiple dose safety trial in 27 pediatric subjects aged 1 to less than 12 years with clinically suspected varicella-zoster virus (VZV) infection [see Error! Hyperlink reference not valid. , , , ].
Trial 2 was a single-dose pharmacokinetic and safety trial in pediatric subjects aged 1 month to less than 6 years who had an active herpes virus infection or who were at risk for herpes virus infection. Fifty-seven subjects were enrolled and received a single dose of 25 mg/kg valacyclovir oral suspension. In infants and children aged 3 months to less than 6 years, this dose provided comparable systemic acyclovir exposures to that from a 1-gram dose of valacyclovir in adults (historical data). In infants aged 1 month to less than 3 months, mean acyclovir exposures resulting from a 25-mg/kg dose were higher (Cmax: ↑30%, AUC: ↑60%) than acyclovir exposures following a 1-gram dose of valacyclovir in adults. Acyclovir is not approved for suppressive therapy in infants and children following neonatal HSV infections; therefore valacyclovir is not recommended for this indication because efficacy cannot be extrapolated from acyclovir.
Trial 3 was a single-dose pharmacokinetic, multiple-dose safety trial in 28 pediatric subjects aged 1 to less than 12 years with clinically suspected HSV infection. None of the subjects enrolled in this trial had genital herpes. Each subject was dosed with valacyclovir oral suspension, 10 mg/kg twice daily for 3 to 5 days. Acyclovir systemic exposures in pediatric subjects following valacyclovir oral suspension were compared with historical acyclovir systemic exposures in immunocompetent adults receiving the solid oral dosage form of valacyclovir or acyclovir for the treatment of recurrent genital herpes. The mean projected daily acyclovir systemic exposures in pediatric subjects across all age groups (1 to less than 12 years) were lower (Cmax : ↓20%, AUC: ↓33%) compared with the acyclovir systemic exposures in adults receiving valacyclovir 500 mg twice daily, but were higher (daily AUC: ↑16%) than systemic exposures in adults receiving acyclovir 200 mg 5 times daily. Insufficient data are available to support valacyclovir for the treatment of recurrent genital herpes in this age-group because clinical information on recurrent genital herpes in young children is limited; therefore, extrapolating efficacy data from adults to this population is not possible. Moreover, valacyclovir has not been studied in children aged 1 to less than 12 years with recurrent genital herpes.
8.5 Geriatric Use
Of the total number of subjects in clinical trials of valacyclovir tablets, 906 were 65 and over, and 352 were 75 and over. In a clinical trial of herpes zoster, the duration of pain after healing (post-herpetic neuralgia) was longer in subjects 65 and older compared with younger adults. Elderly patients are more likely to have reduced renal function and require dose reduction. Elderly patients are also more likely to have renal or CNS adverse events [see , Warnings and Precautions (5.2, , ].
14.3 Herpes Zoster
Two randomized double-blind clinical trials in immunocompetent adults with localized herpes zoster were conducted. Valacyclovir tablets was compared with placebo in subjects aged less than 50 years, and with oral acyclovir in subjects aged greater than 50 years. All subjects were treated within 72 hours of appearance of zoster rash. In subjects aged less than 50 years, the median time to cessation of new lesion formation was 2 days for those treated with valacyclovir tablets compared with 3 days for those treated with placebo. In subjects aged greater than 50 years, the median time to cessation of new lesions was 3 days in subjects treated with either valacyclovir tablets or oral acyclovir. In subjects aged less than 50 years, no difference was found with respect to the duration of pain after healing (post-herpetic neuralgia) between the recipients of valacyclovir tablets and placebo. In subjects aged greater than 50 years, among the 83% who reported pain after healing (post-herpetic neuralgia), the median duration of pain after healing (95% CI) in days was: 40 [31, 51], 43 [36, 55], and 59 [41, 77] for 7-day valacyclovir tablets, 14-day valacyclovir tablets, and 7-day oral acyclovir, respectively.
4 Contraindications
Valacyclovir tablets are contraindicated in patients who have had a demonstrated clinically significant hypersensitivity reaction (e.g., anaphylaxis) to valacyclovir, acyclovir, or any component of the formulation [see ].
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome [see ].
-
•Acute Renal Failure [see Warnings and Precautions (5.2)].
-
•Central Nervous System Effects [see ].
The most common adverse reactions reported in at least 1 indication by greater than 10% of adult subjects treated with valacyclovir tablets and observed more frequently with valacyclovir tablets compared with placebo are headache, nausea, and abdominal pain. The only adverse reaction reported in greater than 10% of pediatric subjects aged less than 18 years was headache.
7 Drug Interactions
No clinically significant drug-drug or drug-food interactions with valacyclovir tablets are known [see ].
8.6 Renal Impairment
Dosage reduction is recommended when administering valacyclovir tablets to patients with renal impairment [see , Warnings and Precautions (5.2, ].
12.3 Pharmacokinetics
The pharmacokinetics of valacyclovir and acyclovir after oral administration of valacyclovir tablets have been investigated in 14 volunteer trials involving 283 adults and in 3 trials involving 112 pediatric subjects aged 1 month to less than 12 years.
1.3 Limitations of Use
The efficacy and safety of valacyclovir tablets have not been established in:
-
•Immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients with a CD4+ cell count greater than or equal to 100 cells/mm3.
-
•Patients aged less than 12 years with cold sores (herpes labialis).
-
•Patients aged less than 2 years or greater than or equal to 18 years with chickenpox.
-
•Patients aged less than 18 years with genital herpes.
-
•Patients aged less than 18 years with herpes zoster.
-
•Neonates and infants as suppressive therapy following neonatal herpes simplex virus (HSV) infection.
1 Indications and Usage
Valacyclovir tablets are a deoxynucleoside analogue DNA polymerase inhibitor indicated for:
Adult Patients ( )
-
•Cold Sores (Herpes Labialis)
-
•Genital Herpes
-
oTreatment in immunocompetent patients (initial or recurrent episode)
-
oSuppression in immunocompetent or HIV-1-infected patients
-
oReduction of transmission
-
-
•Herpes Zoster
Pediatric Patients ( )
-
•Cold Sores (Herpes Labialis)
-
•Chickenpox
Limitations of Use ( 1.3 )
-
•The efficacy and safety of valacyclovir tablets have not been established in immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients.
5.2 Acute Renal Failure
Cases of acute renal failure have been reported in:
-
•Elderly patients with or without reduced renal function. Caution should be exercised when administering valacyclovir tablets to geriatric patients, and dosage reduction is recommended for those with impaired renal function [see , ].
-
•Patients with underlying renal disease who received higher-than-recommended doses of valacyclovir tablets for their level of renal function. Dosage reduction is recommended when administering valacyclovir tablets to patients with renal impairment [see , ].
-
•Patients receiving other nephrotoxic drugs. Caution should be exercised when administering valacyclovir tablets to patients receiving potentially nephrotoxic drugs.
-
•Patients without adequate hydration. Precipitation of acyclovir in renal tubules may occur when the solubility (2.5 mg/mL) is exceeded in the intratubular fluid. Adequate hydration should be maintained for all patients.
In the event of acute renal failure and anuria, the patient may benefit from hemodialysis until renal function is restored [see , ].
12.1 Mechanism of Action
Valacyclovir is an antiviral drug active against α-herpes viruses [see ].
5 Warnings and Precautions
-
•Thrombotic thrombocytopenic purpura/hemolytic uremic syndrome (TTP/HUS): Has occurred in patients with advanced HIV-1 disease and in allogenic bone marrow transplant and renal transplant patients receiving 8 grams per day of valacyclovir tablets in clinical trials. Discontinue treatment if clinical symptoms and laboratory findings consistent with TTP/HUS occur. ()
-
•Acute renal failure: May occur in elderly patients (with or without reduced renal function), patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function, patients who receive concomitant nephrotoxic drugs, or inadequately hydrated patients. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (, 5.2)
-
•Central nervous system adverse reactions (e.g., agitation, hallucinations, confusion, and encephalopathy): May occur in both adult and pediatric patients (with or without reduced renal function) and in patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function. Elderly patients are more likely to have central nervous system adverse reactions. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (,)
2 Dosage and Administration
-
•Valacyclovir tablets may be given without regard to meals.
-
•Valacyclovir oral suspension (25 mg/mL or 50 mg/mL) may be prepared extemporaneously from the 500-mg valacyclovir tablets for use in pediatric patients for whom a solid dosage form is not appropriate [see ].
3 Dosage Forms and Strengths
Tablets:
-
•Valacyclovir Tablets, USP 500-mg: Blue, film-coated, capsule-shaped tablets, debossed with "Y131" on one side contains 556.2 mg of valacyclovir hydrochloride equivalent to 500 mg of the free base.
-
•Valacyclovir Tablets, USP 1-gram: Blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side contains 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram of the free base.
6.3 Postmarketing Experience
In addition to adverse events reported from clinical trials, the following events have been identified during postmarketing use of valacyclovir tablets. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to valacyclovir tablets.
17 Patient Counseling Information
Advise the patient to read the FDA-Approved Patient Labeling (Medication Guide,available at www.wprx- .com/prescription-catalog).
14.1 Cold Sores (herpes Labialis )
Two double-blind, placebo-controlled clinical trials were conducted in 1,856 healthy adults and adolescents (aged greater than or equal to 12 years) with a history of recurrent cold sores. Subjects self-initiated therapy at the earliest symptoms and prior to any signs of a cold sore. The majority of subjects initiated treatment within 2 hours of onset of symptoms. Subjects were randomized to valacyclovir tablets 2 grams twice daily on Day 1 followed by placebo on Day 2, valacyclovir tablets 2 grams twice daily on Day 1 followed by 1 gram twice daily on Day 2, or placebo on Days 1 and 2.
The mean duration of cold sore episodes was about 1 day shorter in treated subjects as compared with placebo. The 2-day regimen did not offer additional benefit over the 1-day regimen.
No significant difference was observed between subjects receiving valacyclovir tablets or placebo in the prevention of progression of cold sore lesions beyond the papular stage.
2.4 Patients With Renal Impairment
Dosage recommendations for adult patients with reduced renal function are provided in Table 1 [see , , ]. Data are not available for the use of valacyclovir tablets in pediatric patients with a creatinine clearance less than 50 mL/min/1.73 m2.
| Indications |
Normal Dosage Regimen
(Creatinine Clearance ≥50 mL/min) |
Creatinine Clearance (mL/min) | ||
|---|---|---|---|---|
| 30-49 | 10-29 | <10 | ||
|
Cold sores (Herpes labialis )
|
Two 2-gram doses taken 12 hours apart |
Two 1-gram doses taken 12 hours apart |
Two 500-mg doses taken 12 hours apart |
500-mg single dose |
|
Genital herpes :
|
1 gram every 12 hours |
no reduction |
1 gram every 24 hours |
500 mg every 24 hours |
|
Genital herpes :
|
500 mg every 12 hours |
no reduction |
500 mg every 24 hours |
500 mg every 24 hours |
|
Genital herpes:
|
||||
|
Immunocompetent patients |
1 gram every 24 hours |
no reduction |
500 mg every 24 hours |
500 mg every 24 hours |
|
Alternate dose for immunocompetent patients with less than or equal to 9 recurrences/year |
500 mg every 24 hours |
no reduction |
500 mg every 48 hours |
500 mg every 48 hours |
|
HIV-1-infected patients |
500 mg every 12 hours |
no reduction |
500 mg every 24 hours |
500 mg every 24 hours |
|
Herpes zoster |
1 gram every 8 hours |
1 gram every 12 hours |
1 gram every 24 hours |
500 mg every 24 hours |
5.3 Central Nervous System Effects
Central nervous system adverse reactions, including agitation, hallucinations, confusion, delirium, seizures, and encephalopathy, have been reported in both adult and pediatric patients with or without reduced renal function and in patients with underlying renal disease who received higher-than-recommended doses of valacyclovir tablets for their level of renal function. Elderly patients are more likely to have central nervous system adverse reactions. Valacyclovir tablets should be discontinued if central nervous system adverse reactions occur [see , , ].
16 How Supplied/storage and Handling
Valacyclovir Tablets, USP 1 gram (blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side) containing 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram valacyclovir.
|
Bottle of 0 NDC 68788-8705-4 Bottle of 0 NDC 68788-8705-0 |
|
Bottle of 0 NDC 68788-8705-1 |
Bottle of 0 NDC 68788-8705-5
|
Bottle of 0 NDC 68788-8705-2 Bottle of 0 NDC 68788-8705-7 |
|
Bottle of 0 NDC 68788-8705-3 |
6.1 Clinical Trials Experience in Adult Subjects
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
6.2 Clinical Trials Experience in Pediatric Subjects
The safety profile of valacyclovir tablets has been studied in 177 pediatric subjects aged 1 month to less than 18 years. Sixty-five of these pediatric subjects, aged 12 to less than 18 years, received oral tablets for 1 to 2 days for treatment of cold sores. The remaining 112 pediatric subjects, aged 1 month to less than 12 years, participated in 3 pharmacokinetic and safety trials and received valacyclovir oral suspension. Fifty-one of these 112 pediatric subjects received oral suspension for 3 to 6 days. The frequency, intensity, and nature of clinical adverse reactions and laboratory abnormalities were similar to those seen in adults.
Principal Display Panel 1 Gram Tablet Bottle Label
NDC 68788-8705
Rx Only
Valacyclovir
Tablets, USP
1 gram
Dispense the Medication Guide provided
separately to each patient.
Westminster
Pharmaceuticals
Repackaged By: Preferred Pharmaceuticals Inc.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The data presented below include references to the steady-state acyclovir AUC observed in humans treated with 1-gram of valacyclovir tablets given orally 3 times a day to treat herpes zoster. Plasma drug concentrations in animal studies are expressed as multiples of human exposure to acyclovir [see ].
5.1 Thrombotic Thrombocytopenic Purpura/hemolytic Uremic Syndrome (ttp/hus)
TTP/HUS, in some cases resulting in death, has occurred in patients with advanced HIV-1 disease and also in allogeneic bone marrow transplant and renal transplant recipients participating in clinical trials of valacyclovir tablets at doses of 8 grams per day. Treatment with valacyclovir tablets should be stopped immediately if clinical signs, symptoms, and laboratory abnormalities consistent with TTP/HUS occur.
Structured Label Content
Section 42229-5 (42229-5)
Cold Sores (Herpes Labialis)
Valacyclovir tablets are indicated for treatment of cold sores (herpes labialis). The efficacy of valacyclovir tablets initiated after the development of clinical signs of a cold sore (e.g., papule, vesicle, or ulcer) has not been established.
Section 44425-7 (44425-7)
Storage:
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Caution should be exercised to prevent inadvertent overdose [see , ]. Precipitation of acyclovir in renal tubules may occur when the solubility (2.5 mg/mL) is exceeded in the intratubular fluid. In the event of acute renal failure and anuria, the patient may benefit from hemodialysis until renal function is restored [see ].
11 Description (11 DESCRIPTION)
Valacyclovir Tablets, USP is the hydrochloride salt of the L-valyl ester of the antiviral drug acyclovir.
Valacyclovir Tablets, USP are for oral administration. Each tablet contains 556.2 mg or 1.112 grams of valacyclovir hydrochloride equivalent to 500 mg or 1 gram of valacyclovir, respectively, and the inactive ingredients anhydrous dibasic calcium phosphate, colloidal silicon dioxide, croscarmellose sodium, FD&C blue #2, hypromellose, magnesium stearate, polyethylene glycol 400, polysorbate 80, povidone K90, and titanium dioxide.
The chemical name of valacyclovir hydrochloride is L-valine, 2-[(2-amino-1,6-dihydro-6-oxo-9H-purin-9-yl)methoxy]ethyl ester, monohydrochloride. It has the following structural formula:
Valacyclovir hydrochloride is a white to off-white powder with the molecular formula C13H20N6O4∙HCl and a molecular weight of 360.80. The maximum solubility in water at 25°C is 174 mg/mL. The pkas for valacyclovir hydrochloride are 1.90, 7.47, and 9.43.
14.4 Chickenpox
The use of valacyclovir tablets for treatment of chickenpox in pediatric subjects aged 2 to less than 18 years is based on single-dose pharmacokinetic and multiple-dose safety data from an open-label trial with valacyclovir and supported by safety and extrapolated efficacy data from 3 randomized, double-blind, placebo-controlled trials evaluating oral acyclovir in pediatric subjects.
The single-dose pharmacokinetic and multiple-dose safety trial enrolled 27 pediatric subjects aged 1 to less than 12 years with clinically suspected VZV infection. Each subject was dosed with valacyclovir oral suspension, 20 mg/kg 3 times daily for 5 days. Acyclovir systemic exposures in pediatric subjects following valacyclovir oral suspension were compared with historical acyclovir systemic exposures in immunocompetent adults receiving the solid oral dosage form of valacyclovir or acyclovir for the treatment of herpes zoster. The mean projected daily acyclovir exposures in pediatric subjects across all age-groups (1 to less than 12 years) were lower (Cmax: ↓13%, AUC: ↓30%)than the mean daily historical exposures in adults receiving valacyclovir 1 gram 3 times daily, but were higher (daily AUC: ↑50%) than the mean daily historical exposures in adults receiving acyclovir 800 mg 5 times daily. The projected daily exposures in pediatric subjects were greater (daily AUC approximately 100% greater) than the exposures seen in immunocompetent pediatric subjects receiving acyclovir 20 mg/kg 4 times daily for the treatment of chickenpox. Based on the pharmacokinetic and safety data from this trial and the safety and extrapolated efficacy data from the acyclovir trials, oral valacyclovir 20 mg/kg 3 times a day for 5 days (not to exceed 1 gram 3 times daily) is recommended for the treatment of chickenpox in pediatric patients aged 2 to less than 18 years. Because the efficacy and safety of acyclovir for the treatment of chickenpox in children aged less than 2 years have not been established, efficacy data cannot be extrapolated to support valacyclovir treatment in children aged less than 2 years with chickenpox.
Valacyclovir is also not recommended for the treatment of herpes zoster in children because safety data up to 7 days' duration are not available [see Use in Specific Populations (8.4)].
8.4 Pediatric Use
Valacyclovir tablets are indicated for treatment of cold sores in pediatric patients aged greater than or equal to 12 years and for treatment of chickenpox in pediatric patients aged 2 to less than 18 years [see , ].
The use of valacyclovir tablets for treatment of cold sores is based on 2 double-blind, placebo-controlled clinical trials in healthy adults and adolescents (aged greater than or equal to 12 years) with a history of recurrent cold sores [see ].
The use of valacyclovir tablets for treatment of chickenpox in pediatric patients aged 2 to less than 18 years is based on single-dose pharmacokinetic and multiple-dose safety data from an open-label trial with valacyclovir and supported by efficacy and safety data from 3 randomized, double-blind, placebo-controlled trials evaluating oral acyclovir in pediatric subjects with chickenpox [see , , , ].
The efficacy and safety of valacyclovir have not been established in pediatric patients:
-
•aged less than 12 years with cold sores
-
•aged less than 18 years with genital herpes
-
•aged less than 18 years with herpes zoster
-
•aged less than 2 years with chickenpox
-
•for suppressive therapy following neonatal HSV infection.
The pharmacokinetic profile and safety of valacyclovir oral suspension in children aged less than 12 years were studied in 3 open label trials. No efficacy evaluations were conducted in any of the 3 trials.
Trial 1 was a single dose pharmacokinetic, multiple dose safety trial in 27 pediatric subjects aged 1 to less than 12 years with clinically suspected varicella-zoster virus (VZV) infection [see Error! Hyperlink reference not valid. , , , ].
Trial 2 was a single-dose pharmacokinetic and safety trial in pediatric subjects aged 1 month to less than 6 years who had an active herpes virus infection or who were at risk for herpes virus infection. Fifty-seven subjects were enrolled and received a single dose of 25 mg/kg valacyclovir oral suspension. In infants and children aged 3 months to less than 6 years, this dose provided comparable systemic acyclovir exposures to that from a 1-gram dose of valacyclovir in adults (historical data). In infants aged 1 month to less than 3 months, mean acyclovir exposures resulting from a 25-mg/kg dose were higher (Cmax: ↑30%, AUC: ↑60%) than acyclovir exposures following a 1-gram dose of valacyclovir in adults. Acyclovir is not approved for suppressive therapy in infants and children following neonatal HSV infections; therefore valacyclovir is not recommended for this indication because efficacy cannot be extrapolated from acyclovir.
Trial 3 was a single-dose pharmacokinetic, multiple-dose safety trial in 28 pediatric subjects aged 1 to less than 12 years with clinically suspected HSV infection. None of the subjects enrolled in this trial had genital herpes. Each subject was dosed with valacyclovir oral suspension, 10 mg/kg twice daily for 3 to 5 days. Acyclovir systemic exposures in pediatric subjects following valacyclovir oral suspension were compared with historical acyclovir systemic exposures in immunocompetent adults receiving the solid oral dosage form of valacyclovir or acyclovir for the treatment of recurrent genital herpes. The mean projected daily acyclovir systemic exposures in pediatric subjects across all age groups (1 to less than 12 years) were lower (Cmax : ↓20%, AUC: ↓33%) compared with the acyclovir systemic exposures in adults receiving valacyclovir 500 mg twice daily, but were higher (daily AUC: ↑16%) than systemic exposures in adults receiving acyclovir 200 mg 5 times daily. Insufficient data are available to support valacyclovir for the treatment of recurrent genital herpes in this age-group because clinical information on recurrent genital herpes in young children is limited; therefore, extrapolating efficacy data from adults to this population is not possible. Moreover, valacyclovir has not been studied in children aged 1 to less than 12 years with recurrent genital herpes.
8.5 Geriatric Use
Of the total number of subjects in clinical trials of valacyclovir tablets, 906 were 65 and over, and 352 were 75 and over. In a clinical trial of herpes zoster, the duration of pain after healing (post-herpetic neuralgia) was longer in subjects 65 and older compared with younger adults. Elderly patients are more likely to have reduced renal function and require dose reduction. Elderly patients are also more likely to have renal or CNS adverse events [see , Warnings and Precautions (5.2, , ].
14.3 Herpes Zoster
Two randomized double-blind clinical trials in immunocompetent adults with localized herpes zoster were conducted. Valacyclovir tablets was compared with placebo in subjects aged less than 50 years, and with oral acyclovir in subjects aged greater than 50 years. All subjects were treated within 72 hours of appearance of zoster rash. In subjects aged less than 50 years, the median time to cessation of new lesion formation was 2 days for those treated with valacyclovir tablets compared with 3 days for those treated with placebo. In subjects aged greater than 50 years, the median time to cessation of new lesions was 3 days in subjects treated with either valacyclovir tablets or oral acyclovir. In subjects aged less than 50 years, no difference was found with respect to the duration of pain after healing (post-herpetic neuralgia) between the recipients of valacyclovir tablets and placebo. In subjects aged greater than 50 years, among the 83% who reported pain after healing (post-herpetic neuralgia), the median duration of pain after healing (95% CI) in days was: 40 [31, 51], 43 [36, 55], and 59 [41, 77] for 7-day valacyclovir tablets, 14-day valacyclovir tablets, and 7-day oral acyclovir, respectively.
4 Contraindications (4 CONTRAINDICATIONS)
Valacyclovir tablets are contraindicated in patients who have had a demonstrated clinically significant hypersensitivity reaction (e.g., anaphylaxis) to valacyclovir, acyclovir, or any component of the formulation [see ].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome [see ].
-
•Acute Renal Failure [see Warnings and Precautions (5.2)].
-
•Central Nervous System Effects [see ].
The most common adverse reactions reported in at least 1 indication by greater than 10% of adult subjects treated with valacyclovir tablets and observed more frequently with valacyclovir tablets compared with placebo are headache, nausea, and abdominal pain. The only adverse reaction reported in greater than 10% of pediatric subjects aged less than 18 years was headache.
7 Drug Interactions (7 DRUG INTERACTIONS)
No clinically significant drug-drug or drug-food interactions with valacyclovir tablets are known [see ].
8.6 Renal Impairment
Dosage reduction is recommended when administering valacyclovir tablets to patients with renal impairment [see , Warnings and Precautions (5.2, ].
12.3 Pharmacokinetics
The pharmacokinetics of valacyclovir and acyclovir after oral administration of valacyclovir tablets have been investigated in 14 volunteer trials involving 283 adults and in 3 trials involving 112 pediatric subjects aged 1 month to less than 12 years.
1.3 Limitations of Use
The efficacy and safety of valacyclovir tablets have not been established in:
-
•Immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients with a CD4+ cell count greater than or equal to 100 cells/mm3.
-
•Patients aged less than 12 years with cold sores (herpes labialis).
-
•Patients aged less than 2 years or greater than or equal to 18 years with chickenpox.
-
•Patients aged less than 18 years with genital herpes.
-
•Patients aged less than 18 years with herpes zoster.
-
•Neonates and infants as suppressive therapy following neonatal herpes simplex virus (HSV) infection.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Valacyclovir tablets are a deoxynucleoside analogue DNA polymerase inhibitor indicated for:
Adult Patients ( )
-
•Cold Sores (Herpes Labialis)
-
•Genital Herpes
-
oTreatment in immunocompetent patients (initial or recurrent episode)
-
oSuppression in immunocompetent or HIV-1-infected patients
-
oReduction of transmission
-
-
•Herpes Zoster
Pediatric Patients ( )
-
•Cold Sores (Herpes Labialis)
-
•Chickenpox
Limitations of Use ( 1.3 )
-
•The efficacy and safety of valacyclovir tablets have not been established in immunocompromised patients other than for the suppression of genital herpes in HIV-1-infected patients.
5.2 Acute Renal Failure
Cases of acute renal failure have been reported in:
-
•Elderly patients with or without reduced renal function. Caution should be exercised when administering valacyclovir tablets to geriatric patients, and dosage reduction is recommended for those with impaired renal function [see , ].
-
•Patients with underlying renal disease who received higher-than-recommended doses of valacyclovir tablets for their level of renal function. Dosage reduction is recommended when administering valacyclovir tablets to patients with renal impairment [see , ].
-
•Patients receiving other nephrotoxic drugs. Caution should be exercised when administering valacyclovir tablets to patients receiving potentially nephrotoxic drugs.
-
•Patients without adequate hydration. Precipitation of acyclovir in renal tubules may occur when the solubility (2.5 mg/mL) is exceeded in the intratubular fluid. Adequate hydration should be maintained for all patients.
In the event of acute renal failure and anuria, the patient may benefit from hemodialysis until renal function is restored [see , ].
12.1 Mechanism of Action
Valacyclovir is an antiviral drug active against α-herpes viruses [see ].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Thrombotic thrombocytopenic purpura/hemolytic uremic syndrome (TTP/HUS): Has occurred in patients with advanced HIV-1 disease and in allogenic bone marrow transplant and renal transplant patients receiving 8 grams per day of valacyclovir tablets in clinical trials. Discontinue treatment if clinical symptoms and laboratory findings consistent with TTP/HUS occur. ()
-
•Acute renal failure: May occur in elderly patients (with or without reduced renal function), patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function, patients who receive concomitant nephrotoxic drugs, or inadequately hydrated patients. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (, 5.2)
-
•Central nervous system adverse reactions (e.g., agitation, hallucinations, confusion, and encephalopathy): May occur in both adult and pediatric patients (with or without reduced renal function) and in patients with underlying renal disease who receive higher-than-recommended doses of valacyclovir tablets for their level of renal function. Elderly patients are more likely to have central nervous system adverse reactions. Use with caution in elderly patients and reduce dosage in patients with renal impairment. (,)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Valacyclovir tablets may be given without regard to meals.
-
•Valacyclovir oral suspension (25 mg/mL or 50 mg/mL) may be prepared extemporaneously from the 500-mg valacyclovir tablets for use in pediatric patients for whom a solid dosage form is not appropriate [see ].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets:
-
•Valacyclovir Tablets, USP 500-mg: Blue, film-coated, capsule-shaped tablets, debossed with "Y131" on one side contains 556.2 mg of valacyclovir hydrochloride equivalent to 500 mg of the free base.
-
•Valacyclovir Tablets, USP 1-gram: Blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side contains 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram of the free base.
6.3 Postmarketing Experience
In addition to adverse events reported from clinical trials, the following events have been identified during postmarketing use of valacyclovir tablets. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to valacyclovir tablets.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-Approved Patient Labeling (Medication Guide,available at www.wprx- .com/prescription-catalog).
14.1 Cold Sores (herpes Labialis ) (14.1 Cold Sores (Herpes Labialis ))
Two double-blind, placebo-controlled clinical trials were conducted in 1,856 healthy adults and adolescents (aged greater than or equal to 12 years) with a history of recurrent cold sores. Subjects self-initiated therapy at the earliest symptoms and prior to any signs of a cold sore. The majority of subjects initiated treatment within 2 hours of onset of symptoms. Subjects were randomized to valacyclovir tablets 2 grams twice daily on Day 1 followed by placebo on Day 2, valacyclovir tablets 2 grams twice daily on Day 1 followed by 1 gram twice daily on Day 2, or placebo on Days 1 and 2.
The mean duration of cold sore episodes was about 1 day shorter in treated subjects as compared with placebo. The 2-day regimen did not offer additional benefit over the 1-day regimen.
No significant difference was observed between subjects receiving valacyclovir tablets or placebo in the prevention of progression of cold sore lesions beyond the papular stage.
2.4 Patients With Renal Impairment (2.4 Patients with Renal Impairment)
Dosage recommendations for adult patients with reduced renal function are provided in Table 1 [see , , ]. Data are not available for the use of valacyclovir tablets in pediatric patients with a creatinine clearance less than 50 mL/min/1.73 m2.
| Indications |
Normal Dosage Regimen
(Creatinine Clearance ≥50 mL/min) |
Creatinine Clearance (mL/min) | ||
|---|---|---|---|---|
| 30-49 | 10-29 | <10 | ||
|
Cold sores (Herpes labialis )
|
Two 2-gram doses taken 12 hours apart |
Two 1-gram doses taken 12 hours apart |
Two 500-mg doses taken 12 hours apart |
500-mg single dose |
|
Genital herpes :
|
1 gram every 12 hours |
no reduction |
1 gram every 24 hours |
500 mg every 24 hours |
|
Genital herpes :
|
500 mg every 12 hours |
no reduction |
500 mg every 24 hours |
500 mg every 24 hours |
|
Genital herpes:
|
||||
|
Immunocompetent patients |
1 gram every 24 hours |
no reduction |
500 mg every 24 hours |
500 mg every 24 hours |
|
Alternate dose for immunocompetent patients with less than or equal to 9 recurrences/year |
500 mg every 24 hours |
no reduction |
500 mg every 48 hours |
500 mg every 48 hours |
|
HIV-1-infected patients |
500 mg every 12 hours |
no reduction |
500 mg every 24 hours |
500 mg every 24 hours |
|
Herpes zoster |
1 gram every 8 hours |
1 gram every 12 hours |
1 gram every 24 hours |
500 mg every 24 hours |
5.3 Central Nervous System Effects
Central nervous system adverse reactions, including agitation, hallucinations, confusion, delirium, seizures, and encephalopathy, have been reported in both adult and pediatric patients with or without reduced renal function and in patients with underlying renal disease who received higher-than-recommended doses of valacyclovir tablets for their level of renal function. Elderly patients are more likely to have central nervous system adverse reactions. Valacyclovir tablets should be discontinued if central nervous system adverse reactions occur [see , , ].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Valacyclovir Tablets, USP 1 gram (blue, film-coated, capsule-shaped tablets, with a partial scorebar on both sides, debossed with "Y132" on one side) containing 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram valacyclovir.
|
Bottle of 0 NDC 68788-8705-4 Bottle of 0 NDC 68788-8705-0 |
|
Bottle of 0 NDC 68788-8705-1 |
Bottle of 0 NDC 68788-8705-5
|
Bottle of 0 NDC 68788-8705-2 Bottle of 0 NDC 68788-8705-7 |
|
Bottle of 0 NDC 68788-8705-3 |
6.1 Clinical Trials Experience in Adult Subjects
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
6.2 Clinical Trials Experience in Pediatric Subjects
The safety profile of valacyclovir tablets has been studied in 177 pediatric subjects aged 1 month to less than 18 years. Sixty-five of these pediatric subjects, aged 12 to less than 18 years, received oral tablets for 1 to 2 days for treatment of cold sores. The remaining 112 pediatric subjects, aged 1 month to less than 12 years, participated in 3 pharmacokinetic and safety trials and received valacyclovir oral suspension. Fifty-one of these 112 pediatric subjects received oral suspension for 3 to 6 days. The frequency, intensity, and nature of clinical adverse reactions and laboratory abnormalities were similar to those seen in adults.
Principal Display Panel 1 Gram Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 1 gram Tablet Bottle Label)
NDC 68788-8705
Rx Only
Valacyclovir
Tablets, USP
1 gram
Dispense the Medication Guide provided
separately to each patient.
Westminster
Pharmaceuticals
Repackaged By: Preferred Pharmaceuticals Inc.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The data presented below include references to the steady-state acyclovir AUC observed in humans treated with 1-gram of valacyclovir tablets given orally 3 times a day to treat herpes zoster. Plasma drug concentrations in animal studies are expressed as multiples of human exposure to acyclovir [see ].
5.1 Thrombotic Thrombocytopenic Purpura/hemolytic Uremic Syndrome (ttp/hus) (5.1 Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome (TTP/HUS))
TTP/HUS, in some cases resulting in death, has occurred in patients with advanced HIV-1 disease and also in allogeneic bone marrow transplant and renal transplant recipients participating in clinical trials of valacyclovir tablets at doses of 8 grams per day. Treatment with valacyclovir tablets should be stopped immediately if clinical signs, symptoms, and laboratory abnormalities consistent with TTP/HUS occur.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:06.628290 · Updated: 2026-03-14T22:36:03.395056