49483-234 Pseudo Time Pe
3116f273-0517-4755-95cd-be3a7c0dd76b
34390-5
HUMAN OTC DRUG LABEL
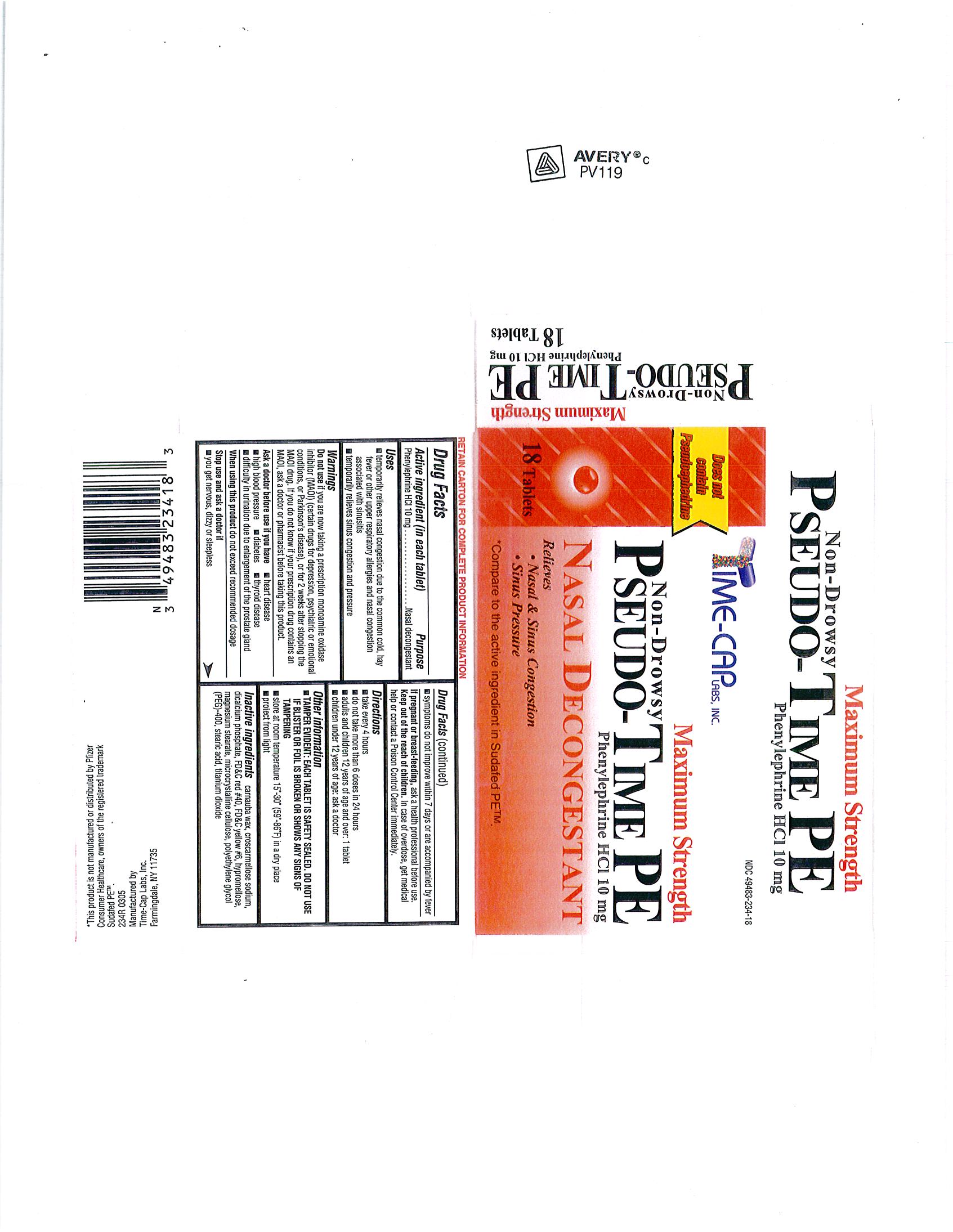
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Each tablet contains: Phenylephrine HCl 10 mg
Medication Information
Warnings and Precautions
|
WARNINGS; Do not use if you are taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease) or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product. |
Indications and Usage
|
Uses:
Temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies and nasal congestion associated with sinusitis Temporarily relieves sinus congestion and pressure |
Dosage and Administration
|
DIRECTIONS:
Take every 4 hours; Do not take more than 6 doses in 24 hours; Adults and children 12 years of age and over; 1 tablet Children under 12 years of age; ask a doctor |
Description
Each tablet contains: Phenylephrine HCl 10 mg
Section 50565-1
| keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center immediately. |
Section 51727-6
| INACTIVE INGREDIENTS: CARNAUBA WAX, CROSCARMELLOSE SODIUM, DIBASIC CALCIUM PHOSPHATE DIHYDRATE, FD-C RED#40 ALUMINUM LAKE, FD-C YELLOW#6 ALUMINUM LAKE, HYPROMELLOSE, MAGNESIUM STEARATE, MICROCRYSTALLINE CELLULOSE, POLYETHYLENE GLYCOL (PEG) 400, STEARIC ACID, TITANIUM DIOXIDE |
Section 51945-4
Section 55105-1
| PURPOSE: nasal decongestant |
Section 55106-9
| Each tablet contains: Phenylephrine HCl 10 mg |
Structured Label Content
Indications and Usage (34067-9)
|
Uses:
Temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies and nasal congestion associated with sinusitis Temporarily relieves sinus congestion and pressure |
Dosage and Administration (34068-7)
|
DIRECTIONS:
Take every 4 hours; Do not take more than 6 doses in 24 hours; Adults and children 12 years of age and over; 1 tablet Children under 12 years of age; ask a doctor |
Warnings and Precautions (34071-1)
|
WARNINGS; Do not use if you are taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease) or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product. |
Section 50565-1 (50565-1)
| keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center immediately. |
Section 51727-6 (51727-6)
| INACTIVE INGREDIENTS: CARNAUBA WAX, CROSCARMELLOSE SODIUM, DIBASIC CALCIUM PHOSPHATE DIHYDRATE, FD-C RED#40 ALUMINUM LAKE, FD-C YELLOW#6 ALUMINUM LAKE, HYPROMELLOSE, MAGNESIUM STEARATE, MICROCRYSTALLINE CELLULOSE, POLYETHYLENE GLYCOL (PEG) 400, STEARIC ACID, TITANIUM DIOXIDE |
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
| PURPOSE: nasal decongestant |
Section 55106-9 (55106-9)
| Each tablet contains: Phenylephrine HCl 10 mg |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:42.570254 · Updated: 2026-03-14T22:55:22.018235