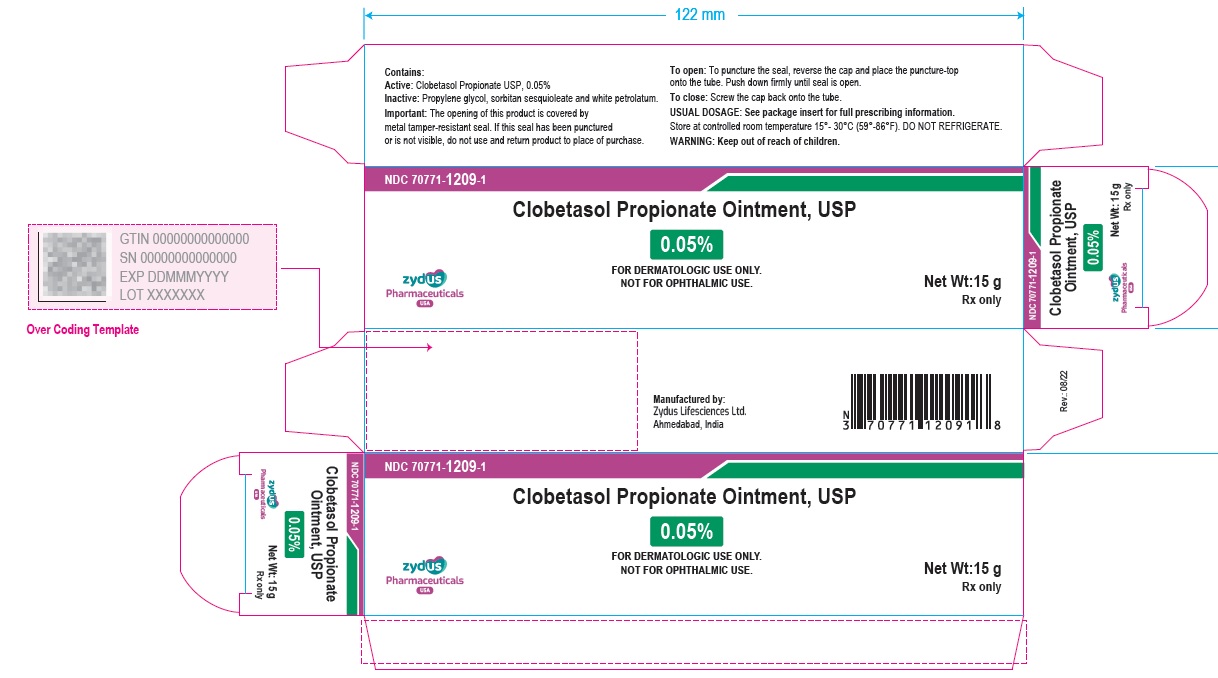
Clobetasol Propionate Ointment Usp, 0.05%
30f4c173-189d-4b29-8d7d-e82e5c0743cf
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
NDC 70771-1209-1 Clobetasol Propionate Ointment USP, 0.05% Rx only 15 g
Medication Information
Description
NDC 70771-1209-1 Clobetasol Propionate Ointment USP, 0.05% Rx only 15 g
Package Label.principal Display Panel
NDC 70771-1209-1
Clobetasol Propionate Ointment USP, 0.05%
Rx only
15 g
Structured Label Content
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 70771-1209-1
Clobetasol Propionate Ointment USP, 0.05%
Rx only
15 g
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:58.023981 · Updated: 2026-03-14T22:18:18.443233