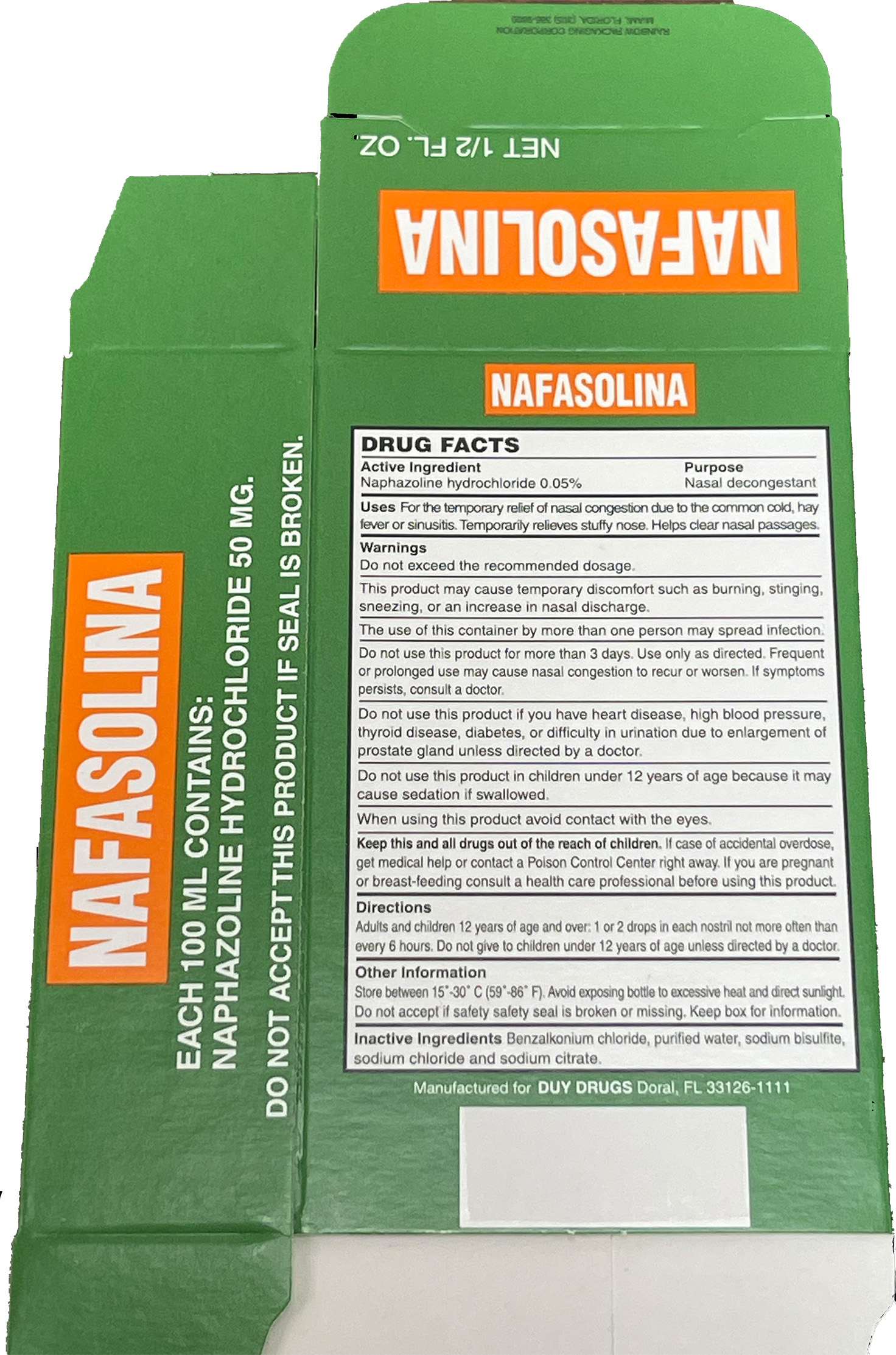
Naphazoline Hydrochloride 0.05%
30cade3d-208f-8594-e063-6394a90ad2a0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Naphazoline hydrochloride 0.05% v/v. Purpose: Nasal decongestant
Medication Information
Warnings and Precautions
Do not exceed recommended dosage.
This product may cause temporary discomfort such as burning, stinging, sneezing, or an increase in nasal discharge.
The use of this container by more than one person may spread infection.
Do not use this product for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen. If symptoms persist, consult a doctor.
Do not use this product if you have heart disease, high blood pressure, thyroid disease, diabetes, or difficulty in urination due to enlargement of the prostate gland unless directed by a doctor.
Do not use this product in children under 12 years of age because it may cause sedation if swallowed.
When using this product avoid contact with the eyes.
Indications and Usage
Naphazoline hydrochloride 0.05% nasal drops are indicated for the temporary relief of nasal congestion due to the common cold, hay fever, or sinusitis. Temporarily relieves a stuffy nose. Helps clear nasal passages.
Dosage and Administration
Adults and children 12 years of age and over: 1 or 2 drops in each nostril not more often than every 6 hours. Do not give to children under 12 years of age unless directed by a doctor.
Description
Naphazoline hydrochloride 0.05% v/v. Purpose: Nasal decongestant
Section 50565-1
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Stop using this product after 3 days.
If symptoms persist, stop, and consult a doctor.
Section 50567-7
Use for the temporary relief of nasal congestion due to the common cold, hay fever, or sinusitis.
Section 50570-1
If you are pregnant or breast-feeding consult a health care professional before using this product.
Do not use this product if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2
weeks after stopping the MAOI drug. If you are uncertain whether your prescription drug contains an MAOI, consult a health professional before taking this product.
Do not use this product for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen. If symptoms persist, consult a doctor.
Do not use this product if you have heart disease, high blood pressure, thyroid disease, diabetes, or difficulty in urination due to enlargement of the prostate gland unless directed by a doctor.
Do not use this product in children under 12 years of age because it may cause sedation if swallowed.
Do not use this product in a child who has heart disease, high blood pressure, thyroid disease, or diabetes unless directed by a doctor.
This product is for nasal use only.
When using this product avoid contact with the eyes. In case of contact with eyes, rinse eyes thoroughly with water.
Section 51727-6
Benzalkonium chloride, purified water, sodium bisulfite, sodium chloride, and sodium citrate.
Section 51945-4
Principal Display Panel
0.5 FL. OZ NDC: 12539-146-02
Section 55105-1
For the temporary relief of nasal congestion due to the common cold, hay fever, or sinusitis.
Temporarily relieves a stuffy nose.
Helps clear nasal passages.
Section 55106-9
Naphazoline hydrochloride 0.05% v/v. Purpose: Nasal decongestant
Structured Label Content
Indications and Usage (34067-9)
Naphazoline hydrochloride 0.05% nasal drops are indicated for the temporary relief of nasal congestion due to the common cold, hay fever, or sinusitis. Temporarily relieves a stuffy nose. Helps clear nasal passages.
Dosage and Administration (34068-7)
Adults and children 12 years of age and over: 1 or 2 drops in each nostril not more often than every 6 hours. Do not give to children under 12 years of age unless directed by a doctor.
Warnings and Precautions (34071-1)
Do not exceed recommended dosage.
This product may cause temporary discomfort such as burning, stinging, sneezing, or an increase in nasal discharge.
The use of this container by more than one person may spread infection.
Do not use this product for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen. If symptoms persist, consult a doctor.
Do not use this product if you have heart disease, high blood pressure, thyroid disease, diabetes, or difficulty in urination due to enlargement of the prostate gland unless directed by a doctor.
Do not use this product in children under 12 years of age because it may cause sedation if swallowed.
When using this product avoid contact with the eyes.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Stop using this product after 3 days.
If symptoms persist, stop, and consult a doctor.
Section 50567-7 (50567-7)
Use for the temporary relief of nasal congestion due to the common cold, hay fever, or sinusitis.
Section 50570-1 (50570-1)
If you are pregnant or breast-feeding consult a health care professional before using this product.
Do not use this product if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2
weeks after stopping the MAOI drug. If you are uncertain whether your prescription drug contains an MAOI, consult a health professional before taking this product.
Do not use this product for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen. If symptoms persist, consult a doctor.
Do not use this product if you have heart disease, high blood pressure, thyroid disease, diabetes, or difficulty in urination due to enlargement of the prostate gland unless directed by a doctor.
Do not use this product in children under 12 years of age because it may cause sedation if swallowed.
Do not use this product in a child who has heart disease, high blood pressure, thyroid disease, or diabetes unless directed by a doctor.
This product is for nasal use only.
When using this product avoid contact with the eyes. In case of contact with eyes, rinse eyes thoroughly with water.
Section 51727-6 (51727-6)
Benzalkonium chloride, purified water, sodium bisulfite, sodium chloride, and sodium citrate.
Section 51945-4 (51945-4)
Principal Display Panel
0.5 FL. OZ NDC: 12539-146-02
Section 55105-1 (55105-1)
For the temporary relief of nasal congestion due to the common cold, hay fever, or sinusitis.
Temporarily relieves a stuffy nose.
Helps clear nasal passages.
Section 55106-9 (55106-9)
Naphazoline hydrochloride 0.05% v/v. Purpose: Nasal decongestant
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:45.141606 · Updated: 2026-03-14T23:11:30.674942