Drx Choice ®
303d7651-9c8f-4a14-9f2c-7181fa0f003e
34390-5
HUMAN OTC DRUG LABEL
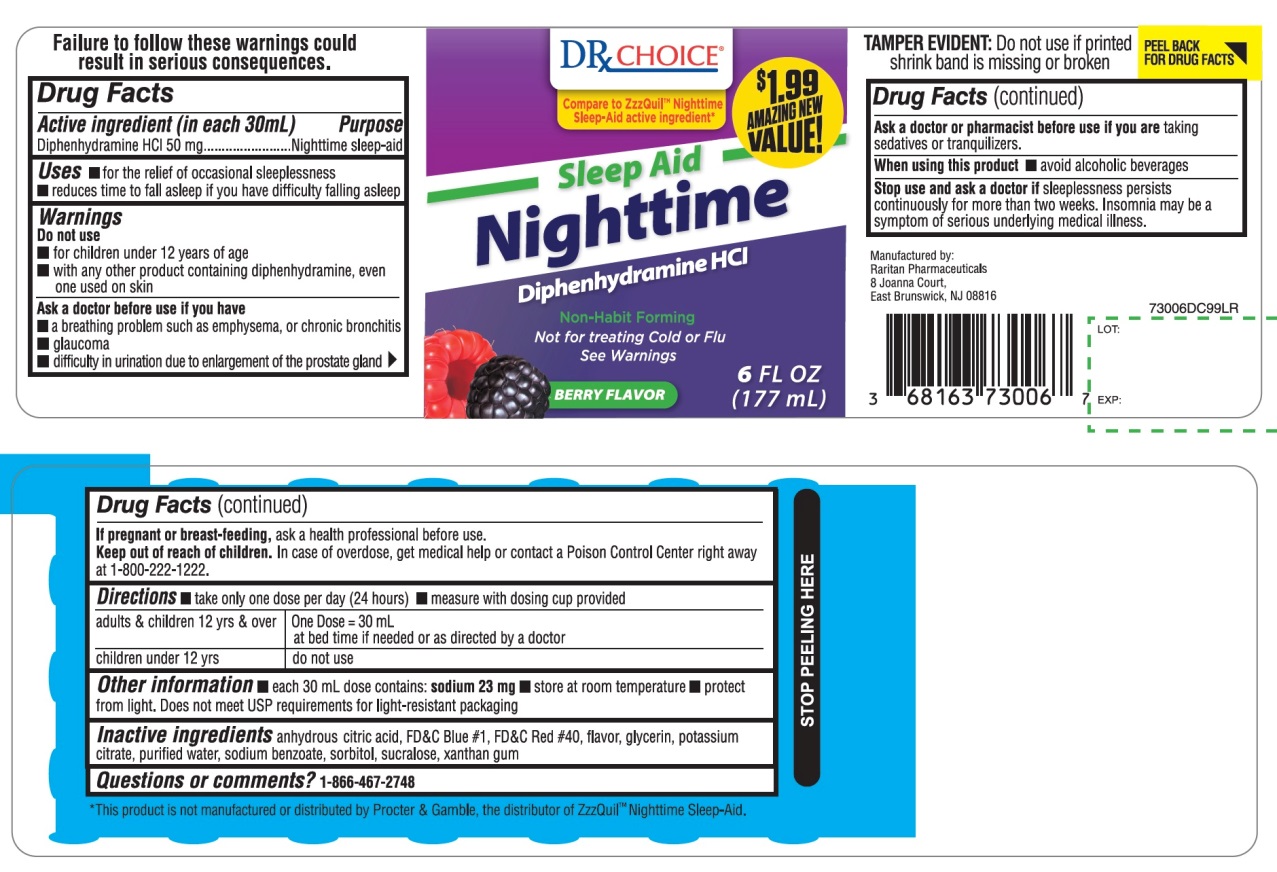
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Nighttime sleep-aid
Medication Information
Purpose
Nighttime sleep-aid
Description
Drug Facts
Uses
- for the relief of occasional sleeplessness
- reduces time to fall asleep if you have difficulty falling asleep
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
Section 50566-9
Stop use and ask a doctor ifsleeplessness persists continuously for more than two weeks. Insomnia may be a symptom of serious underlying medical illness.
Section 50568-5
Ask a doctor or pharmacist before use if you aretaking sedatives or tranquilizers.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
- take only one dose per day (24 hours)
- only use cup provided
|
adults & children 12yrs & over |
One Dose 30 mL at bed time if needed or as directed by a doctor |
|
Children under 12 yrs |
do not use |
Do Not Use
- for children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Other Information
- each 30 mL dose contains:potassium 5 mg, sodium 10 mg
- store at room temperature
Overdose Warning:
In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Inactive Ingredients
anhydrous citric acid, FD&C Blue No. 1, FD&C Red No. 40, flavor, glycerin, potassium citrate, purified water, sodium benzoate, sorbitol, sucralose, xanthan gum.
Principal Display Panel
DRx CHOICE ®
NDC 68163-730-06
Compare to in ZzzQuil® Nighttime Sleep-Aid active ingredient *
Sleep-Aid
Nighttime
Diphenhydramine HCl
Non-Habit Forming
BERRY FLAVOR
Naturally and Artificially Flavored
Not for treating Cold or Flu See Warnings
6 FL OZ (177 mL)
$1.99 AMAZING NEW VALUE!
TAMPER EVIDENT: DO NOT USE IF PRINTED SHRINK BAND IS MISSING OR BROKEN
*This product is not manufactured or distributed by Procter & Gamble, the distributor of ZzzQuil™ Nighttime Sleep-Aid.
Manufactured by:
Raritan Pharmaceuticals
8 Joanna Court,
East Brunswick, NJ 08816
When Using This Product
- avoid alcoholic beverages
Active Ingredient (in Each 30 Ml)
Diphenhydramine HCl 50 mg
Ask A Doctor Before Use If You Have
- a breathing problem such as emphysema, or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
Structured Label Content
Uses
- for the relief of occasional sleeplessness
- reduces time to fall asleep if you have difficulty falling asleep
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50566-9 (50566-9)
Stop use and ask a doctor ifsleeplessness persists continuously for more than two weeks. Insomnia may be a symptom of serious underlying medical illness.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you aretaking sedatives or tranquilizers.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Nighttime sleep-aid
Directions
- take only one dose per day (24 hours)
- only use cup provided
|
adults & children 12yrs & over |
One Dose 30 mL at bed time if needed or as directed by a doctor |
|
Children under 12 yrs |
do not use |
Do Not Use (Do not use)
- for children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Other Information (Other information)
- each 30 mL dose contains:potassium 5 mg, sodium 10 mg
- store at room temperature
Overdose Warning: (Overdose warning:)
In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Inactive Ingredients (Inactive ingredients)
anhydrous citric acid, FD&C Blue No. 1, FD&C Red No. 40, flavor, glycerin, potassium citrate, purified water, sodium benzoate, sorbitol, sucralose, xanthan gum.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
DRx CHOICE ®
NDC 68163-730-06
Compare to in ZzzQuil® Nighttime Sleep-Aid active ingredient *
Sleep-Aid
Nighttime
Diphenhydramine HCl
Non-Habit Forming
BERRY FLAVOR
Naturally and Artificially Flavored
Not for treating Cold or Flu See Warnings
6 FL OZ (177 mL)
$1.99 AMAZING NEW VALUE!
TAMPER EVIDENT: DO NOT USE IF PRINTED SHRINK BAND IS MISSING OR BROKEN
*This product is not manufactured or distributed by Procter & Gamble, the distributor of ZzzQuil™ Nighttime Sleep-Aid.
Manufactured by:
Raritan Pharmaceuticals
8 Joanna Court,
East Brunswick, NJ 08816
When Using This Product (When using this product)
- avoid alcoholic beverages
Active Ingredient (in Each 30 Ml) (Active ingredient (in each 30 mL))
Diphenhydramine HCl 50 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- a breathing problem such as emphysema, or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:06.733646 · Updated: 2026-03-14T23:10:19.800513