These Highlights Do Not Include All The Information Needed To Use Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate And Amphetamine Sulfate Extended-release Capsules Safely And Effectively. See Full Prescribing Information For Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, And Amphetamine Sulfate Extended-release Capsules.
302a2f67-e6d0-4831-9032-2d30e8692af2
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2 , 9.3) ].
Indications and Usage
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, a CNS stimulant, are indicated for the treatment of attention deficit hyperactivity disorder (ADHD). ( 1 ) Children (ages 6 to 12): Efficacy was established in one 3-week outpatient, controlled trial and one analogue classroom, controlled trial in children with ADHD. ( 14 ) Adolescents (ages 13 to 17): Efficacy was established in one 4-week controlled trial in adolescents with ADHD. ( 14 ) Adults: Efficacy was established in one 4-week controlled trial in adults with ADHD. ( 14 )
Dosage and Administration
Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 is 30 mg once daily. ( 2.2 , 2.3 , 2.4 ) Adults: 20 mg once daily in the morning. ( 2.5 ) Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. ( 2.6 , 8.6 ) Adults with severe renal impairment: 15 mg once daily in the morning. ( 2.6 , 8.6 ) Patients with ESRD: Not recommended. ( 2.6 , 8.6 )
Warnings and Precautions
Serious Cardiovascular Events: Sudden death has been reported with usual doses of CNS stimulants in children and adolescents with structural cardiac abnormalities or other serious heart problems; sudden death, stroke, and myocardial infarction have been reported in adults taking CNS stimulants at usual doses. Stimulant drugs should not be used in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, or other serious heart problems. ( 5.1 ) Increase in Blood Pressure: Monitor blood pressure and pulse at appropriate intervals. Use with caution in patients for whom blood pressure increases may be problematic. ( 5.2 ) Psychiatric Adverse Events: Stimulants may cause treatment-emergent psychotic or manic symptoms in patients with no prior history, or exacerbation of symptoms in patients with pre-existing psychosis. Evaluate for bipolar disorder prior to stimulant use. Monitor for aggressive behavior. ( 5.3 ) Long-term Suppression of Growth: Monitor height and weight at appropriate intervals. ( 5.4 ) Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. ( 5.5 ) Peripheral Vasculopathy, including Raynaud's phenomenon: Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants. ( 5.6 ) Serotonin Syndrome: Increased risk when co-administered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and initiate supportive treatment ( 4 , 5.7 , 10 ). Visual Disturbance: Difficulties with accommodation and blurring of vision have been reported with stimulant treatment. ( 5.8 ) Tics: May exacerbate tics. Evaluate for tics and Tourette's syndrome prior to stimulant administration. ( 5.9 )
Contraindications
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules administration is contraindicated in patients with the following conditions: Advanced arteriosclerosis Symptomatic cardiovascular disease Moderate to severe hypertension Hyperthyroidism In patients known to be hypersensitive to amphetamine, or other components of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions (6.2) ]. Glaucoma Agitated states History of drug abuse Patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions (5.6) and Drug Interactions (7.1) ].
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
MAOI antidepressants are contraindicated; MAOIs potentiate the effects of amphetamine. Do not administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules during or within 14 days after use of MAOI. ( 4 , 7.1 ) Alkalinizing agents (GI antacids and urinary): These agents increase blood levels of amphetamine. ( 7.1 ) Acidifying agents (GI and urinary): These agents reduce blood levels of amphetamine. ( 7.1 ) Adrenergic blockers, antihistamines, antihypertensives, phenobarbital, phenytoin, veratrum alkaloids, and ethosuximide: Effects may be reduced by amphetamines. ( 7.1 ) Tricyclic antidepressants, norepinephrine, and meperidine: Effects may be potentiated by amphetamines. ( 7.1 )
Storage and Handling
30 mg capsules: Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads. Bottles of 100 capsules: 63629-9481-01 Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required. Store at 20ºC to 25°C (68°F to 77ºF) [see USP Controlled Room Temperature]. Disposal Comply with local laws and regulations on drug disposal of CNS stimulants. Dispose of remaining, unused, or expired dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with an undesirable, nontoxic substance to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and discard dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the household trash. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
How Supplied
30 mg capsules: Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads. Bottles of 100 capsules: 63629-9481-01 Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required. Store at 20ºC to 25°C (68°F to 77ºF) [see USP Controlled Room Temperature]. Disposal Comply with local laws and regulations on drug disposal of CNS stimulants. Dispose of remaining, unused, or expired dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with an undesirable, nontoxic substance to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and discard dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the household trash. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Medication Information
Warnings and Precautions
Serious Cardiovascular Events: Sudden death has been reported with usual doses of CNS stimulants in children and adolescents with structural cardiac abnormalities or other serious heart problems; sudden death, stroke, and myocardial infarction have been reported in adults taking CNS stimulants at usual doses. Stimulant drugs should not be used in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, or other serious heart problems. ( 5.1 ) Increase in Blood Pressure: Monitor blood pressure and pulse at appropriate intervals. Use with caution in patients for whom blood pressure increases may be problematic. ( 5.2 ) Psychiatric Adverse Events: Stimulants may cause treatment-emergent psychotic or manic symptoms in patients with no prior history, or exacerbation of symptoms in patients with pre-existing psychosis. Evaluate for bipolar disorder prior to stimulant use. Monitor for aggressive behavior. ( 5.3 ) Long-term Suppression of Growth: Monitor height and weight at appropriate intervals. ( 5.4 ) Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. ( 5.5 ) Peripheral Vasculopathy, including Raynaud's phenomenon: Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants. ( 5.6 ) Serotonin Syndrome: Increased risk when co-administered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and initiate supportive treatment ( 4 , 5.7 , 10 ). Visual Disturbance: Difficulties with accommodation and blurring of vision have been reported with stimulant treatment. ( 5.8 ) Tics: May exacerbate tics. Evaluate for tics and Tourette's syndrome prior to stimulant administration. ( 5.9 )
Indications and Usage
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, a CNS stimulant, are indicated for the treatment of attention deficit hyperactivity disorder (ADHD). ( 1 ) Children (ages 6 to 12): Efficacy was established in one 3-week outpatient, controlled trial and one analogue classroom, controlled trial in children with ADHD. ( 14 ) Adolescents (ages 13 to 17): Efficacy was established in one 4-week controlled trial in adolescents with ADHD. ( 14 ) Adults: Efficacy was established in one 4-week controlled trial in adults with ADHD. ( 14 )
Dosage and Administration
Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 is 30 mg once daily. ( 2.2 , 2.3 , 2.4 ) Adults: 20 mg once daily in the morning. ( 2.5 ) Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. ( 2.6 , 8.6 ) Adults with severe renal impairment: 15 mg once daily in the morning. ( 2.6 , 8.6 ) Patients with ESRD: Not recommended. ( 2.6 , 8.6 )
Contraindications
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules administration is contraindicated in patients with the following conditions: Advanced arteriosclerosis Symptomatic cardiovascular disease Moderate to severe hypertension Hyperthyroidism In patients known to be hypersensitive to amphetamine, or other components of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions (6.2) ]. Glaucoma Agitated states History of drug abuse Patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions (5.6) and Drug Interactions (7.1) ].
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
MAOI antidepressants are contraindicated; MAOIs potentiate the effects of amphetamine. Do not administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules during or within 14 days after use of MAOI. ( 4 , 7.1 ) Alkalinizing agents (GI antacids and urinary): These agents increase blood levels of amphetamine. ( 7.1 ) Acidifying agents (GI and urinary): These agents reduce blood levels of amphetamine. ( 7.1 ) Adrenergic blockers, antihistamines, antihypertensives, phenobarbital, phenytoin, veratrum alkaloids, and ethosuximide: Effects may be reduced by amphetamines. ( 7.1 ) Tricyclic antidepressants, norepinephrine, and meperidine: Effects may be potentiated by amphetamines. ( 7.1 )
Storage and Handling
30 mg capsules: Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads. Bottles of 100 capsules: 63629-9481-01 Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required. Store at 20ºC to 25°C (68°F to 77ºF) [see USP Controlled Room Temperature]. Disposal Comply with local laws and regulations on drug disposal of CNS stimulants. Dispose of remaining, unused, or expired dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with an undesirable, nontoxic substance to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and discard dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the household trash. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
How Supplied
30 mg capsules: Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads. Bottles of 100 capsules: 63629-9481-01 Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required. Store at 20ºC to 25°C (68°F to 77ºF) [see USP Controlled Room Temperature]. Disposal Comply with local laws and regulations on drug disposal of CNS stimulants. Dispose of remaining, unused, or expired dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with an undesirable, nontoxic substance to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and discard dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the household trash. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Description
CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2 , 9.3) ].
Section 42229-5
Special Diagnostic Considerations
Specific etiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use not only of medical but of special psychological, educational, and social resources. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the patient and not solely on the presence of the required number of DSM-IV® characteristics.
Section 51945-4
Mixed Amphe Salts ER 30mg Caps (CII) #100
5.9 Tics
Amphetamines have been reported to exacerbate motor and phonic tics and Tourette's syndrome. Therefore, clinical evaluation for tics and Tourette's syndrome in patients and their families should precede use of stimulant medications.
9.2 Abuse
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are a CNS stimulant that contains amphetamine, which has a high potential for abuse. Abuse is characterized by impaired control of drug use, compulsive use despite harm, and craving.
Signs and symptoms of amphetamine abuse may include increased heart rate, respiratory rate, blood pressure, and/or sweating, dilated pupils, hyperactivity, restlessness, insomnia, decreased appetite, loss of coordination, tremors, flushed skin, vomiting, and/or abdominal pain. Anxiety, psychosis, hostility, aggression, suicidal or homicidal ideation have also been observed. Abusers of amphetamines may use other unapproved routes of administration which can result in overdose and death [see Overdosage (10)].
To reduce the abuse of CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, assess the risk of abuse prior to prescribing. After prescribing, keep careful prescription records, educate patients and their families about abuse and proper storage and disposal of CNS stimulants. Monitor for signs of abuse while on therapy and re-evaluate the need for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules use.
2.5 Adults
In adults with ADHD who are either starting treatment for the first time or switching from another medication, the recommended dose is 20 mg/day.
2.3 Children
In children with ADHD who are 6 to 12 years of age and are either starting treatment for the first time or switching from another medication, start with 10 mg once daily in the morning; daily dosage may be adjusted in increments of 5 mg or 10 mg at weekly intervals. When in the judgment of the clinician a lower initial dose is appropriate, patients may begin treatment with 5 mg once daily in the morning. The maximum recommended dose for children 6 to 12 years of age is 30 mg/day; doses greater than 30 mg/day have not been studied in children. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have not been studied in children under 6 years of age.
5.5 Seizures
There is some clinical evidence that stimulants may lower the convulsive threshold in patients with prior history of seizures, in patients with prior EEG abnormalities in the absence of seizures, and very rarely, in patients without a history of seizures and no prior EEG evidence of seizures. In the presence of seizures, dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be discontinued.
10 Overdosage
Manifestations of amphetamine overdose include restlessness, tremor, hyperreflexia, rapid respiration, confusion, assaultiveness, hallucinations, panic states, hyperpyrexia, and rhabdomyolysis. Fatigue and depression usually follow the central nervous system stimulation. Serotonin syndrome has been reported with amphetamine use, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Cardiovascular effects include arrhythmias, hypertension or hypotension and circulatory collapse. Gastrointestinal symptoms include nausea, vomiting, diarrhea and abdominal cramps. Fatal poisoning is usually preceded by convulsions and coma.
11 Description
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are a once daily extended-release, single-entity amphetamine product. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules combine the neutral sulfate salts of dextroamphetamine and amphetamine, with the dextro isomer of amphetamine saccharate and d,l-amphetamine aspartate monohydrate. The dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended release capsules are a dose proportional composition using a multi-layer fluid bed bead coating process, followed by encapsulation using hard gelatin capsules. The dosage utilizes a bead that is coated with an extended-release drug layer, that is then coated with an enteric polymer to control drug release, and finally coated with an immediate-release drug layer.
| Each capsule contains: Capsule Strength |
5 mg | 10 mg | 15 mg | 20 mg | 25 mg | 30 mg |
|---|---|---|---|---|---|---|
| Dextroamphetamine Saccharate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine (D,L)-Aspartate Monohydrate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Dextroamphetamine Sulfate, USP | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine Sulfate, USP | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Total amphetamine base equivalence mg | 3.1 mg | 6.3 mg | 9.4 mg | 12.5 mg | 15.6 mg | 18.8 mg |
| d-amphetamine base equivalence mg | 2.4 mg | 4.7 mg | 7.1 mg | 9.5 mg | 11.9 mg | 14.2 mg |
| l-amphetamine base equivalence | 0.75 mg | 1.5 mg | 2.3 mg | 3.0 mg | 3.8 mg | 4.5 mg |
9.3 Dependence
Tolerance (a state of adaptation in which exposure to a specific dose of a drug results in a reduction of the drug's desired and/or undesired effects over time, in such a way that a higher dose of the drug is required to produce the same effect that was once obtained at a lower dose) may occur during chronic therapy of CNS stimulants including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Physical Dependence (which is manifested by a withdrawal syndrome produced by abrupt cessation, rapid dose reduction, or administration of an antagonist) may occur in patients treated with CNS stimulants including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Withdrawal symptoms after abrupt cessation of CNS stimulants include dysphoric mood; fatigue; vivid, unpleasant dreams; insomnia or hypersomnia; increased appetite; and psychomotor retardation or agitation.
2.4 Adolescents
The recommended starting dose for adolescents with ADHD who are 13 to17 years of age and are either starting treatment for the first time or switching from another medication is 10 mg/day. The dose may be increased to 20 mg/day after one week if ADHD symptoms are not adequately controlled.
Medication Guide
Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate extended-release capsules, CII
(DEX-troe am FET-uh-meen Sack-uh-rate/ am-FET-uh-meen as-PAR-tate MAH-no-HIGH-drate/ DEX-troe-am-FET-uh-meen SULL-fate/am-FET-uh-meen-SULL-fate)
Read the Medication Guide that comes with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules before you or your child starts taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your doctor about you or your child's treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
What is the most important information I should know about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is a stimulant medicine. The following have been reported with use of stimulant medicines.
1. Heart-related problems:
- sudden death in patients who have heart problems or heart defects
- stroke and heart attack in adults
- increased blood pressure and heart rate
Tell your doctor if you or your child have any heart problems, heart defects, high blood pressure, or a family history of these problems.
Your doctor should check you or your child carefully for heart problems before starting dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Your doctor should check you or your child's blood pressure and heart rate regularly during treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Call your doctor right away if you or your child has any signs of heart problems such as chest pain, shortness of breath, or fainting while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
2. Mental (Psychiatric) problems:
All Patients
- new or worse behavior and thought problems
- new or worse bipolar illness
- new or worse aggressive behavior or hostility
Children and Teenagers
- new psychotic symptoms (such as hearing voices, believing things that are not true, are suspicious) or new manic symptoms
Tell your doctor about any mental problems you or your child have, or about a family history of suicide, bipolar illness, or depression.
Call your doctor right away if you or your child have any new or worsening mental symptoms or problems while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules especially seeing or hearing things that are not real, believing things that are not real, or are suspicious.
3. Circulation problems in fingers and toes [Peripheral vasculopathy, including Raynaud's phenomenon]:
- Fingers or toes may feel numb, cool, painful
- Fingers or toes may change from pale, to blue, to red
Tell your doctor if you have or your child has numbness, pain, skin color change, or sensitivity to temperature in your fingers or toes.
Call your doctor right away if you have or your child has any unexplained wounds appearing on fingers or toes while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
What is dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is a once daily central nervous system stimulant prescription medicine. It is used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD). Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may help increase attention and decrease impulsiveness and hyperactivity in patients with ADHD.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be used as a part of a total treatment program for ADHD that may include counseling or other therapies.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are a federally controlled substance (CII) because it can be abused or lead to dependence. Keep dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in a safe place to prevent misuse and abuse. Selling or giving away dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may harm others, and is against the law.
Tell your doctor if you or your child have (or have a family history of) ever abused or been dependent on alcohol, prescription medicines or street drugs.
Who should not take dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should not be taken if you or your child:
- have heart disease or hardening of the arteries
- have moderate to severe high blood pressure
- have hyperthyroidism
- have an eye problem called glaucoma
- are very anxious, tense, or agitated
- have a history of drug abuse
- are taking or have taken within the past 14 days an anti-depression medicine called a monoamine oxidase inhibitor or MAOI.
- is sensitive to, allergic to, or had a reaction to other stimulant medicines
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have not been studied in children less than 6 years old.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may not be right for you or your child. Before starting Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules tell you or your child's doctor about all health conditions (or a family history of) including if you or your child:
- have heart problems, heart defects, or high blood pressure
- have mental problems including psychosis, mania, bipolar illness, or depression
- have tics or Tourette's syndrome
- have liver problems
- have kidney problems
- have end stage renal disease (ESRD)
- have thyroid problems
- have seizures or have had an abnormal brain wave test (EEG)
- have circulation problems in fingers and toes
- are pregnant or plan to become pregnant. It is not known if dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules will harm your unborn baby.
- There is a pregnancy registry for females who are exposed to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules during pregnancy. The purpose of the registry is to collect information about the health of females exposed to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and their baby. If you or your child becomes pregnant during treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, talk to your healthcare provider about registering with the National Pregnancy Registry of Psychostimulants at 1-866-961-2388 or visit online at https://womensmentalhealth.org/clinical-and-research-programs/pregnancyregistry/othermedications/.
- are breastfeeding or plan to breastfeed. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules passes into breast milk. You or your child should not breastfeed during treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Can dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules be taken with other medicines?
Tell your doctor about all of the medicines that you or your child takes including prescription and non-prescription medicines, vitamins, and herbal supplements. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be adjusted while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules
Your doctor will decide whether dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules can be taken with other medicines.
Especially tell your doctor if you or your child takes:
- anti-depression medicines including MAOIs
- anti-psychotic medicines
- lithium
- narcotic pain medicines
- seizure medicines
- blood thinner medicines
- blood pressure medicines
- stomach acid medicines
- cold or allergy medicines that contain decongestants
Know the medicines that you or your child takes. Keep a list of your medicines with you to show your doctor and pharmacist.
Do not start any new medicine while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules without talking to your doctor first.
How should dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules be taken?
- Take dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules exactly as prescribed. Your doctor may adjust the dose until it is right for you or your child.
- Take dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules once a day in the morning when you first wake up. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is an extended release capsule. It releases medicine into your body throughout the day.
- Swallow dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules whole with water or other liquids. If you or your child cannot swallow the capsule, open it and sprinkle the medicine over a spoonful of applesauce. Swallow all of the applesauce and medicine mixture without chewing immediately. Follow with a drink of water or other liquid. Never chew or crush the capsule or the medicine inside the capsule.
- Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules can be taken with or without food.
- From time to time, your doctor may stop dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules treatment for a while to check ADHD symptoms.
- Your doctor may do regular checks of the blood, heart, and blood pressure while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Children should have their height and weight checked often while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules treatment may be stopped if a problem is found during these check-ups.
- If you or your child takes too much dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules or overdoses, call your doctor or poison control center right away, or get emergency treatment.
What are possible side effects of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
See "What is the most important information I should know about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?" for information on reported heart and mental problems.
Other serious side effects include:
- slowing of growth (height and weight) in children
- seizures, mainly in patients with a history of seizures
- eyesight changes or blurred vision
Common side effects include:
- headache
- decreased appetite
- stomach ache
- nervousness
- trouble sleeping
- mood swings
- weight loss
- dizziness
- dry mouth
- fast heart beat
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may affect you or your child's ability to drive or do other dangerous activities.
Talk to your doctor if you or your child has side effects that are bothersome or do not go away.
This is not a complete list of possible side effects. Ask your doctor or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
- Store dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in a safe place at room temperature, 68°F to 77ºF (20ºC to 25°C) [see USP Controlled Room Temperature].
- Keep dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and all medicines out of the reach of children.
General information about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules for a condition for which it was not prescribed. Do not give dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules to other people, even if they have the same condition. It may harm them and it is against the law.
This Medication Guide summarizes the most important information about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules that was written for healthcare professionals. For more information, you may also contact Rhodes Pharmaceuticals L.P. (the maker of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules) at 1-888-827-0616 or visit the website at http://www.rhodespharma.com
What are the ingredients in dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Active Ingredients: Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, USP, and amphetamine sulfate USP
Inactive Ingredients: The inactive ingredients in dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules include: sugar spheres (sucrose, sugar syrup and corn starch), hypromellose, talc, triethyl citrate, methacrylic acid copolymer dispersion and gelatin capsules. Gelatin capsules contain: titanium dioxide, and FD&C Blue# 1. The 5 mg, 20 mg, 25 mg, and 30 mg capsules also contain FD&C Yellow#6. The 5 mg, 10 mg, and 15 mg also contain D&C Yellow#10 and D&C Red#33. Additionally, the imprint ink contains: shellac, black iron oxide, and potassium hydroxide.
Marketed by:
Rhodes Pharmaceuticals L.P., Coventry, RI 02816
Rev: 03/2022
This Medication Guide has been approved by the U.S. Food and Drug Administration.
8.4 Pediatric Use
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are indicated for use in children 6 years of age and older.
The safety and efficacy of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in children under 6 years of age have not been studied. Long-term effects of amphetamines in children have not been well established.
8.5 Geriatric Use
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have not been studied in the geriatric population.
4 Contraindications
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules administration is contraindicated in patients with the following conditions:
- Advanced arteriosclerosis
- Symptomatic cardiovascular disease
- Moderate to severe hypertension
- Hyperthyroidism
- In patients known to be hypersensitive to amphetamine, or other components of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions (6.2)].
- Glaucoma
- Agitated states
- History of drug abuse
- Patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions (5.6) and Drug Interactions (7.1)].
6 Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions
- MAOI antidepressants are contraindicated; MAOIs potentiate the effects of amphetamine. Do not administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules during or within 14 days after use of MAOI. (4, 7.1)
- Alkalinizing agents (GI antacids and urinary): These agents increase blood levels of amphetamine. (7.1)
- Acidifying agents (GI and urinary): These agents reduce blood levels of amphetamine. (7.1)
- Adrenergic blockers, antihistamines, antihypertensives, phenobarbital, phenytoin, veratrum alkaloids, and ethosuximide: Effects may be reduced by amphetamines. (7.1)
- Tricyclic antidepressants, norepinephrine, and meperidine: Effects may be potentiated by amphetamines. (7.1)
8.6 Renal Impairment
Due to reduced clearance of amphetamines in patients with severe renal impairment (GFR 15 to <30 mL/min/1.73m2), the recommended dose should be reduced. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are not recommended in patients with ESRD (GFR < 15 ml/min/1.73m2) [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
d-Amphetamine is not dialyzable.
12.2 Pharmacodynamics
Amphetamines block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
Pharmacokinetic studies of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have been conducted in healthy adult and pediatric (children aged 6 to 12 yrs) subjects, and adolescent (13 to 17 yrs) and children with ADHD. Both dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release) and dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules contain d-amphetamine and l-amphetamine salts in the ratio of 3:1. Following administration of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release), the peak plasma concentrations occurred in about 3 hours for both d-amphetamine and l-amphetamine.
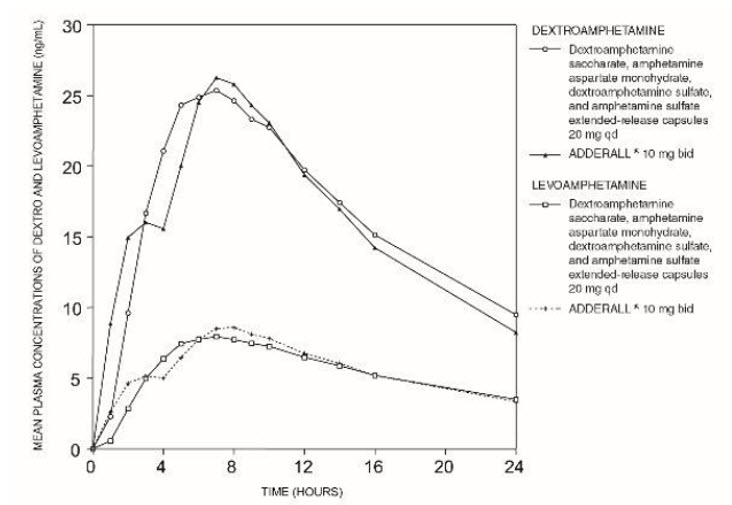
The time to reach maximum plasma concentration (Tmax) for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is about 7 hours, which is about 4 hours longer compared to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release). This is consistent with the extended-release nature of the product.
Figure 1: Mean d-amphetamine and l-amphetamine Plasma Concentrations Following Administration of Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Extended-Release Capsules 20 mg (8 am) and Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Tablets (immediate-release) 10 mg Twice Daily (8 am and 12 noon) in the Fed State.
A single dose of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended release 20 mg capsules provided comparable plasma concentration profiles of both d-amphetamine and l-amphetamine to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release) 10 mg twice daily administered 4 hours apart.
The mean elimination half-life for d-amphetamine is 10 hours in adults; 11 hours in adolescents aged 13 to 17 years and weighing less than or equal to 75 kg/165 lbs; and 9 hours in children aged 6 to 12 years. For the l-amphetamine, the mean elimination half-life in adults is 13 hours; 13 to 14 hours in adolescents; and 11 hours in children aged 6 to 12 years. On a mg/kg body weight basis, children have a higher clearance than adolescents or adults (see Special Populations below).
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules demonstrates linear pharmacokinetics over the dose range of 20 to 60 mg in adults and adolescents weighing greater than 75 kg/165 lbs, over the dose range of 10 to 40 mg in adolescents weighing less than or equal to 75 kg/165 lbs, and 5 to 30 mg in children aged 6 to 12 years. There is no unexpected accumulation at steady state in children.
Food does not affect the extent of absorption of d-amphetamine and l-amphetamine, but prolongs Tmax by 2.5 hours (from 5.2 hrs. at fasted state to 7.7 hrs. after a high-fat meal) for d-amphetamine and 2.7 hours (from 5.6 hrs. at fasted state to 8.3 hrs. after a high fat meal) for l-amphetamine after administration of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release, 30 mg capsule. Opening the capsule and sprinkling the contents on applesauce results in comparable absorption to the intact capsule taken in the fasted state. Equal doses of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules strengths are bioequivalent.
5.7 Serotonin Syndrome
Serotonin syndrome, a potentially life-threatening reaction, may occur when amphetamines are used in combination with other drugs that affect the serotonergic neurotransmitter systems such as MAOIs, selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John's Wort [see Drug Interactions (7.1)]. Amphetamines and amphetamine derivatives are known to be metabolized, to some degree, by cytochrome P450 2D6 (CYP2D6) and display minor inhibition of CYP2D6 metabolism [see Clinical Pharmacology (12.3)]. The potential for a pharmacokinetic interaction exists with the co-administration of CYP2D6 inhibitors which may increase the risk with increased exposure to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. In these situations, consider an alternative non-serotonergic drug or an alternative drug that does not inhibit CYP2D6 [see Drug Interactions (7.1)]. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
Concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with MAOI drugs is contraindicated [see Contraindications (4)].
Discontinue treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and any concomitant serotonergic agents immediately if symptoms of serotonin syndrome occur, and initiate supportive symptomatic treatment. Concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with other serotonergic drugs or CYP2D6 inhibitors should be used only if the potential benefit justifies the potential risk. If clinically warranted, consider initiating dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with lower doses, monitoring patients for the emergence of serotonin syndrome during drug initiation or titration, and informing patients of the increased risk for serotonin syndrome.
5.8 Visual Disturbance
Difficulties with accommodation and blurring of vision have been reported with stimulant treatment.
1 Indications and Usage
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, a CNS stimulant, are indicated for the treatment of attention deficit hyperactivity disorder (ADHD). (1)
- Children (ages 6 to 12): Efficacy was established in one 3-week outpatient, controlled trial and one analogue classroom, controlled trial in children with ADHD. (14)
- Adolescents (ages 13 to 17): Efficacy was established in one 4-week controlled trial in adolescents with ADHD. (14)
- Adults: Efficacy was established in one 4-week controlled trial in adults with ADHD. (14)
12.1 Mechanism of Action
Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules contain amphetamine, a Schedule II controlled substance.
5 Warnings and Precautions
- Serious Cardiovascular Events: Sudden death has been reported with usual doses of CNS stimulants in children and adolescents with structural cardiac abnormalities or other serious heart problems; sudden death, stroke, and myocardial infarction have been reported in adults taking CNS stimulants at usual doses. Stimulant drugs should not be used in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, or other serious heart problems. (5.1)
- Increase in Blood Pressure: Monitor blood pressure and pulse at appropriate intervals. Use with caution in patients for whom blood pressure increases may be problematic. (5.2)
- Psychiatric Adverse Events: Stimulants may cause treatment-emergent psychotic or manic symptoms in patients with no prior history, or exacerbation of symptoms in patients with pre-existing psychosis. Evaluate for bipolar disorder prior to stimulant use. Monitor for aggressive behavior. (5.3)
- Long-term Suppression of Growth: Monitor height and weight at appropriate intervals. (5.4)
- Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. (5.5)
- Peripheral Vasculopathy, including Raynaud's phenomenon: Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants. (5.6)
- Serotonin Syndrome: Increased risk when co-administered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and initiate supportive treatment (4, 5.7, 10).
- Visual Disturbance: Difficulties with accommodation and blurring of vision have been reported with stimulant treatment. (5.8)
- Tics: May exacerbate tics. Evaluate for tics and Tourette's syndrome prior to stimulant administration. (5.9)
2 Dosage and Administration
- Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 is 30 mg once daily. (2.2, 2.3, 2.4)
- Adults: 20 mg once daily in the morning. (2.5)
- Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. (2.6, 8.6)
- Adults with severe renal impairment: 15 mg once daily in the morning. (2.6, 8.6)
- Patients with ESRD: Not recommended. (2.6, 8.6)
3 Dosage Forms and Strengths
| 5 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Blue Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "705" on the body in black ink containing white to off-white beads. | |
| 10 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Blue Opaque Cap/Blue Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "710" on the body in black ink containing white to off-white beads. | |
| 15 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| White Opaque Cap/Blue Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "715" on the body in black ink containing white to off-white beads. | |
| 20 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Orange Opaque Cap/ Orange Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "720" on the body in black ink containing white to off-white beads. | |
| 25 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| White Opaque Cap/Orange Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "725" on the body in black ink containing white to off-white beads. | |
| 30 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads. |
8 Use in Specific Populations
Warning: Abuse and Dependence
CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2, 9.3)].
6.1 Clinical Trials Experience
The premarketing development program for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules included exposures in a total of 1315 participants in clinical trials (635 pediatric patients, 350 adolescent patients, 248 adult patients, and 82 healthy adult subjects). Of these, 635 patients (ages 6 to 12) were evaluated in two controlled clinical studies, one open-label clinical study, and two single-dose clinical pharmacology studies (N= 40). Safety data on all patients are included in the discussion that follows. Adverse reactions were assessed by collecting adverse reactions, results of physical examinations, vital signs, weights, laboratory analyses, and ECGs.
Adverse reactions during exposure were obtained primarily by general inquiry and recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse reactions without first grouping similar types of reactions into a smaller number of standardized event categories. In the tables and listings that follow, COSTART terminology has been used to classify reported adverse reactions.
The stated frequencies of adverse reactions represent the proportion of individuals who experienced, at least once, a treatment-emergent adverse event of the type listed.
5.10 Prescribing and Dispensing
The least amount of amphetamine feasible should be prescribed or dispensed at one time in order to minimize the possibility of overdosage. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be used with caution in patients who use other sympathomimetic drugs.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.4 Long Term Suppression of Growth
Monitor growth in children during treatment with stimulants. Patients who are not growing or gaining weight as expected may need to have their treatment interrupted.
Careful follow-up of weight and height in children ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated children over 36 months (to the ages of 10 to 13 years), suggests that consistently medicated children (i.e., treatment for 7 days per week throughout the year) have a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this period of development.
In a controlled trial of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in adolescents, mean weight change from baseline within the initial 4 weeks of therapy was -1.1 lbs. and -2.8 lbs., respectively, for patients receiving 10 mg and 20 mg dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Higher doses were associated with greater weight loss within the initial 4 weeks of treatment. Chronic use of amphetamines can be expected to cause a similar suppression of growth.
16 How Supplied/storage and Handling
30 mg capsules: Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads.
Bottles of 100 capsules: 63629-9481-01
Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required.
Store at 20ºC to 25°C (68°F to 77ºF) [see USP Controlled Room Temperature].
Disposal
Comply with local laws and regulations on drug disposal of CNS stimulants. Dispose of remaining, unused, or expired dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with an undesirable, nontoxic substance to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and discard dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the household trash.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
5.1 Potential for Abuse and Dependence
CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Boxed Warning, Drug Abuse and Dependence (9.2, 9.3)].
13.2 Animal Toxicology And/or Pharmacology
Acute administration of high doses of amphetamine (d- or d,l-) has been shown to produce long-lasting neurotoxic effects, including irreversible nerve fiber damage, in rodents. The significance of these findings to humans is unknown.
2.2 Dosing Considerations for All Patients
Individualize the dosage according to the therapeutic needs and response of the patient. Administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at the lowest effective dosage.
Based on bioequivalence data, patients taking divided doses of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release), (for example, twice daily), may be switched to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at the same total daily dose taken once daily. Titrate at weekly intervals to appropriate efficacy and tolerability as indicated.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may be taken whole, or the capsule may be opened and the entire contents sprinkled on applesauce. If the patient is using the sprinkle administration method, the sprinkled applesauce should be consumed immediately; it should not be stored. Patients should take the applesauce with sprinkled beads in its entirety without chewing. The dose of a single capsule should not be divided. The contents of the entire capsule should be taken, and patients should not take anything less than one capsule per day.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may be taken with or without food.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be given upon awakening. Afternoon doses should be avoided because of the potential for insomnia.
Where possible, dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules therapy should be interrupted occasionally to determine if there is a recurrence of behavioral symptoms sufficient to require continued therapy.
1.1 Attention Deficit Hyperactivity Disorder
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are indicated for the treatment of attention deficit hyperactivity disorder (ADHD).
The efficacy of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the treatment of ADHD was established on the basis of two controlled trials in children aged 6 to 12, one controlled trial in adolescents aged 13 to 17, and one controlled trial in adults who met DSM-IV® criteria for ADHD [see Clinical Studies (14)].
A diagnosis of ADHD (DSM-IV®) implies the presence of hyperactive-impulsive or inattentive symptoms that caused impairment and were present before age 7 years. The symptoms must cause clinically significant impairment, e.g., in social, academic, or occupational functioning, and be present in two or more settings, e.g., school (or work) and at home. The symptoms must not be better accounted for by another mental disorder. For the Inattentive Type, at least six of the following symptoms must have persisted for at least 6 months: lack of attention to details/careless mistakes; lack of sustained attention; poor listener; failure to follow through on tasks; poor organization; avoids tasks requiring sustained mental effort; loses things; easily distracted; forgetful. For the Hyperactive-Impulsive Type, at least six of the following symptoms must have persisted for at least 6 months: fidgeting/squirming; leaving seat; inappropriate running/climbing; difficulty with quiet activities; "on the go;" excessive talking; blurting answers; can't wait turn; intrusive. The Combined Type requires both inattentive and hyperactive-impulsive criteria to be met.
2.6 Dosage in Patients With Renal Impairment
In adult patients with severe renal impairment (GFR 15 to < 30 mL/min/1.73m2), the recommended dose is 15 mg once daily in the morning. In pediatric patients (6 to 17 years of age) with severe renal impairment, the recommended dose is 5 mg once daily. The maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are not recommended in patients with end stage renal disease (ESRD) (GFR < 15 mL/min/1.73m2 [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.1 Important Information Prior to Initiating Treatment
Prior to initiating treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, assess for the presence of cardiac disease (e.g., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2)].
Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy. Maintain careful prescription records, educate patients about abuse, monitor for signs for abuse and overdose, and periodically re-evaluate the need for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules use [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9)].
7.1 Clinically Important Interactions With Amphetamines
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact | Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure. |
| Intervention | Do not administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules concomitantly or within 14 days after discontinuing MAOI [see Contraindications (4) . |
| Examples | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Serotonergic Drugs | |
| Clinical Impact | The concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and serotonergic drugs increases the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules initiation or dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and the concomitant serotonergic drug(s) [see Warnings and Precautions (5.6)]. |
| Examples | selective serotonin reuptake inhibitors (SSRI), serotonin norepinephrine reuptake inhibitors (SNRI), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John's Wort |
| CYP2D6 Inhibitors | |
| Clinical Impact | The concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and CYP2D6 inhibitors may increase the exposure of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules compared to the use of the drug alone and increase the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules initiation and after a dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and the CYP2D6 inhibitor [see Warnings and Precautions (5.6) and Overdosage (10)]. |
| Examples | paroxetine and fluoxetine (also serotonergic drugs), quinidine, ritonavir |
| Alkalinizing Agents | |
| Clinical Impact | Increase blood levels and potentiate the action of amphetamine. |
| Intervention | Co-administration of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and gastrointestinal or urinary alkalinizing agents should be avoided. |
| Examples | Gastrointestinal alkalinizing agents (e.g., sodium bicarbonate). Urinary alkalinizing agents (e.g. acetazolamide, some thiazides). |
| Acidifying Agents | |
| Clinical Impact | Lower blood levels and efficacy of amphetamines. |
| Intervention | Increase dose based on clinical response. |
| Examples | Gastrointestinal acidifying agents (e.g., guanethidine, reserpine, glutamic acid HCl, ascorbic acid). Urinary acidifying agents (e.g., ammonium chloride, sodium acid phosphate, methenamine salts). |
| Tricyclic Antidepressants | |
| Clinical Impact | May enhance the activity of tricyclic or sympathomimetic agents causing striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated. |
| Intervention | Monitor frequently and adjust or use alternative therapy based on clinical response. |
| Examples | desipramine, protriptyline |
| Proton Pump Inhibitors | |
| Clinical Impact | Time to maximum concentration (Tmax) of amphetamine is decreased compared to when administered alone. |
| Intervention | Monitor patients for changes in clinical effect and adjust therapy based on clinical response. |
| Examples | Omeprazole |
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon
Stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.
6.2 Adverse Reactions Associated With the Use of Amphetamine; Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Extended Release Capsules; Or Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Tablets (immediate Release)
The following adverse reactions have been identified during post-approval use of amphetamine; dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules; or dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Structured Label Content
Section 42229-5 (42229-5)
Special Diagnostic Considerations
Specific etiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use not only of medical but of special psychological, educational, and social resources. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the patient and not solely on the presence of the required number of DSM-IV® characteristics.
Section 51945-4 (51945-4)
Mixed Amphe Salts ER 30mg Caps (CII) #100
5.9 Tics
Amphetamines have been reported to exacerbate motor and phonic tics and Tourette's syndrome. Therefore, clinical evaluation for tics and Tourette's syndrome in patients and their families should precede use of stimulant medications.
9.2 Abuse
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are a CNS stimulant that contains amphetamine, which has a high potential for abuse. Abuse is characterized by impaired control of drug use, compulsive use despite harm, and craving.
Signs and symptoms of amphetamine abuse may include increased heart rate, respiratory rate, blood pressure, and/or sweating, dilated pupils, hyperactivity, restlessness, insomnia, decreased appetite, loss of coordination, tremors, flushed skin, vomiting, and/or abdominal pain. Anxiety, psychosis, hostility, aggression, suicidal or homicidal ideation have also been observed. Abusers of amphetamines may use other unapproved routes of administration which can result in overdose and death [see Overdosage (10)].
To reduce the abuse of CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, assess the risk of abuse prior to prescribing. After prescribing, keep careful prescription records, educate patients and their families about abuse and proper storage and disposal of CNS stimulants. Monitor for signs of abuse while on therapy and re-evaluate the need for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules use.
2.5 Adults
In adults with ADHD who are either starting treatment for the first time or switching from another medication, the recommended dose is 20 mg/day.
2.3 Children
In children with ADHD who are 6 to 12 years of age and are either starting treatment for the first time or switching from another medication, start with 10 mg once daily in the morning; daily dosage may be adjusted in increments of 5 mg or 10 mg at weekly intervals. When in the judgment of the clinician a lower initial dose is appropriate, patients may begin treatment with 5 mg once daily in the morning. The maximum recommended dose for children 6 to 12 years of age is 30 mg/day; doses greater than 30 mg/day have not been studied in children. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have not been studied in children under 6 years of age.
5.5 Seizures
There is some clinical evidence that stimulants may lower the convulsive threshold in patients with prior history of seizures, in patients with prior EEG abnormalities in the absence of seizures, and very rarely, in patients without a history of seizures and no prior EEG evidence of seizures. In the presence of seizures, dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be discontinued.
10 Overdosage (10 OVERDOSAGE)
Manifestations of amphetamine overdose include restlessness, tremor, hyperreflexia, rapid respiration, confusion, assaultiveness, hallucinations, panic states, hyperpyrexia, and rhabdomyolysis. Fatigue and depression usually follow the central nervous system stimulation. Serotonin syndrome has been reported with amphetamine use, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Cardiovascular effects include arrhythmias, hypertension or hypotension and circulatory collapse. Gastrointestinal symptoms include nausea, vomiting, diarrhea and abdominal cramps. Fatal poisoning is usually preceded by convulsions and coma.
11 Description (11 DESCRIPTION)
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are a once daily extended-release, single-entity amphetamine product. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules combine the neutral sulfate salts of dextroamphetamine and amphetamine, with the dextro isomer of amphetamine saccharate and d,l-amphetamine aspartate monohydrate. The dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended release capsules are a dose proportional composition using a multi-layer fluid bed bead coating process, followed by encapsulation using hard gelatin capsules. The dosage utilizes a bead that is coated with an extended-release drug layer, that is then coated with an enteric polymer to control drug release, and finally coated with an immediate-release drug layer.
| Each capsule contains: Capsule Strength |
5 mg | 10 mg | 15 mg | 20 mg | 25 mg | 30 mg |
|---|---|---|---|---|---|---|
| Dextroamphetamine Saccharate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine (D,L)-Aspartate Monohydrate | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Dextroamphetamine Sulfate, USP | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Amphetamine Sulfate, USP | 1.25 mg | 2.5 mg | 3.75 mg | 5.0 mg | 6.25 mg | 7.5 mg |
| Total amphetamine base equivalence mg | 3.1 mg | 6.3 mg | 9.4 mg | 12.5 mg | 15.6 mg | 18.8 mg |
| d-amphetamine base equivalence mg | 2.4 mg | 4.7 mg | 7.1 mg | 9.5 mg | 11.9 mg | 14.2 mg |
| l-amphetamine base equivalence | 0.75 mg | 1.5 mg | 2.3 mg | 3.0 mg | 3.8 mg | 4.5 mg |
9.3 Dependence
Tolerance (a state of adaptation in which exposure to a specific dose of a drug results in a reduction of the drug's desired and/or undesired effects over time, in such a way that a higher dose of the drug is required to produce the same effect that was once obtained at a lower dose) may occur during chronic therapy of CNS stimulants including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Physical Dependence (which is manifested by a withdrawal syndrome produced by abrupt cessation, rapid dose reduction, or administration of an antagonist) may occur in patients treated with CNS stimulants including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Withdrawal symptoms after abrupt cessation of CNS stimulants include dysphoric mood; fatigue; vivid, unpleasant dreams; insomnia or hypersomnia; increased appetite; and psychomotor retardation or agitation.
2.4 Adolescents
The recommended starting dose for adolescents with ADHD who are 13 to17 years of age and are either starting treatment for the first time or switching from another medication is 10 mg/day. The dose may be increased to 20 mg/day after one week if ADHD symptoms are not adequately controlled.
Medication Guide (MEDICATION GUIDE)
Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate extended-release capsules, CII
(DEX-troe am FET-uh-meen Sack-uh-rate/ am-FET-uh-meen as-PAR-tate MAH-no-HIGH-drate/ DEX-troe-am-FET-uh-meen SULL-fate/am-FET-uh-meen-SULL-fate)
Read the Medication Guide that comes with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules before you or your child starts taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your doctor about you or your child's treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
What is the most important information I should know about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is a stimulant medicine. The following have been reported with use of stimulant medicines.
1. Heart-related problems:
- sudden death in patients who have heart problems or heart defects
- stroke and heart attack in adults
- increased blood pressure and heart rate
Tell your doctor if you or your child have any heart problems, heart defects, high blood pressure, or a family history of these problems.
Your doctor should check you or your child carefully for heart problems before starting dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Your doctor should check you or your child's blood pressure and heart rate regularly during treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Call your doctor right away if you or your child has any signs of heart problems such as chest pain, shortness of breath, or fainting while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
2. Mental (Psychiatric) problems:
All Patients
- new or worse behavior and thought problems
- new or worse bipolar illness
- new or worse aggressive behavior or hostility
Children and Teenagers
- new psychotic symptoms (such as hearing voices, believing things that are not true, are suspicious) or new manic symptoms
Tell your doctor about any mental problems you or your child have, or about a family history of suicide, bipolar illness, or depression.
Call your doctor right away if you or your child have any new or worsening mental symptoms or problems while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules especially seeing or hearing things that are not real, believing things that are not real, or are suspicious.
3. Circulation problems in fingers and toes [Peripheral vasculopathy, including Raynaud's phenomenon]:
- Fingers or toes may feel numb, cool, painful
- Fingers or toes may change from pale, to blue, to red
Tell your doctor if you have or your child has numbness, pain, skin color change, or sensitivity to temperature in your fingers or toes.
Call your doctor right away if you have or your child has any unexplained wounds appearing on fingers or toes while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
What is dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is a once daily central nervous system stimulant prescription medicine. It is used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD). Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may help increase attention and decrease impulsiveness and hyperactivity in patients with ADHD.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be used as a part of a total treatment program for ADHD that may include counseling or other therapies.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are a federally controlled substance (CII) because it can be abused or lead to dependence. Keep dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in a safe place to prevent misuse and abuse. Selling or giving away dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may harm others, and is against the law.
Tell your doctor if you or your child have (or have a family history of) ever abused or been dependent on alcohol, prescription medicines or street drugs.
Who should not take dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should not be taken if you or your child:
- have heart disease or hardening of the arteries
- have moderate to severe high blood pressure
- have hyperthyroidism
- have an eye problem called glaucoma
- are very anxious, tense, or agitated
- have a history of drug abuse
- are taking or have taken within the past 14 days an anti-depression medicine called a monoamine oxidase inhibitor or MAOI.
- is sensitive to, allergic to, or had a reaction to other stimulant medicines
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have not been studied in children less than 6 years old.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may not be right for you or your child. Before starting Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules tell you or your child's doctor about all health conditions (or a family history of) including if you or your child:
- have heart problems, heart defects, or high blood pressure
- have mental problems including psychosis, mania, bipolar illness, or depression
- have tics or Tourette's syndrome
- have liver problems
- have kidney problems
- have end stage renal disease (ESRD)
- have thyroid problems
- have seizures or have had an abnormal brain wave test (EEG)
- have circulation problems in fingers and toes
- are pregnant or plan to become pregnant. It is not known if dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules will harm your unborn baby.
- There is a pregnancy registry for females who are exposed to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules during pregnancy. The purpose of the registry is to collect information about the health of females exposed to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and their baby. If you or your child becomes pregnant during treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, talk to your healthcare provider about registering with the National Pregnancy Registry of Psychostimulants at 1-866-961-2388 or visit online at https://womensmentalhealth.org/clinical-and-research-programs/pregnancyregistry/othermedications/.
- are breastfeeding or plan to breastfeed. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules passes into breast milk. You or your child should not breastfeed during treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules.
Can dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules be taken with other medicines?
Tell your doctor about all of the medicines that you or your child takes including prescription and non-prescription medicines, vitamins, and herbal supplements. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be adjusted while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules
Your doctor will decide whether dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules can be taken with other medicines.
Especially tell your doctor if you or your child takes:
- anti-depression medicines including MAOIs
- anti-psychotic medicines
- lithium
- narcotic pain medicines
- seizure medicines
- blood thinner medicines
- blood pressure medicines
- stomach acid medicines
- cold or allergy medicines that contain decongestants
Know the medicines that you or your child takes. Keep a list of your medicines with you to show your doctor and pharmacist.
Do not start any new medicine while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules without talking to your doctor first.
How should dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules be taken?
- Take dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules exactly as prescribed. Your doctor may adjust the dose until it is right for you or your child.
- Take dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules once a day in the morning when you first wake up. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is an extended release capsule. It releases medicine into your body throughout the day.
- Swallow dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules whole with water or other liquids. If you or your child cannot swallow the capsule, open it and sprinkle the medicine over a spoonful of applesauce. Swallow all of the applesauce and medicine mixture without chewing immediately. Follow with a drink of water or other liquid. Never chew or crush the capsule or the medicine inside the capsule.
- Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules can be taken with or without food.
- From time to time, your doctor may stop dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules treatment for a while to check ADHD symptoms.
- Your doctor may do regular checks of the blood, heart, and blood pressure while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Children should have their height and weight checked often while taking dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules treatment may be stopped if a problem is found during these check-ups.
- If you or your child takes too much dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules or overdoses, call your doctor or poison control center right away, or get emergency treatment.
What are possible side effects of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
See "What is the most important information I should know about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?" for information on reported heart and mental problems.
Other serious side effects include:
- slowing of growth (height and weight) in children
- seizures, mainly in patients with a history of seizures
- eyesight changes or blurred vision
Common side effects include:
- headache
- decreased appetite
- stomach ache
- nervousness
- trouble sleeping
- mood swings
- weight loss
- dizziness
- dry mouth
- fast heart beat
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may affect you or your child's ability to drive or do other dangerous activities.
Talk to your doctor if you or your child has side effects that are bothersome or do not go away.
This is not a complete list of possible side effects. Ask your doctor or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
- Store dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in a safe place at room temperature, 68°F to 77ºF (20ºC to 25°C) [see USP Controlled Room Temperature].
- Keep dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and all medicines out of the reach of children.
General information about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules for a condition for which it was not prescribed. Do not give dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules to other people, even if they have the same condition. It may harm them and it is against the law.
This Medication Guide summarizes the most important information about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules that was written for healthcare professionals. For more information, you may also contact Rhodes Pharmaceuticals L.P. (the maker of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules) at 1-888-827-0616 or visit the website at http://www.rhodespharma.com
What are the ingredients in dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules?
Active Ingredients: Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, USP, and amphetamine sulfate USP
Inactive Ingredients: The inactive ingredients in dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules include: sugar spheres (sucrose, sugar syrup and corn starch), hypromellose, talc, triethyl citrate, methacrylic acid copolymer dispersion and gelatin capsules. Gelatin capsules contain: titanium dioxide, and FD&C Blue# 1. The 5 mg, 20 mg, 25 mg, and 30 mg capsules also contain FD&C Yellow#6. The 5 mg, 10 mg, and 15 mg also contain D&C Yellow#10 and D&C Red#33. Additionally, the imprint ink contains: shellac, black iron oxide, and potassium hydroxide.
Marketed by:
Rhodes Pharmaceuticals L.P., Coventry, RI 02816
Rev: 03/2022
This Medication Guide has been approved by the U.S. Food and Drug Administration.
8.4 Pediatric Use
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are indicated for use in children 6 years of age and older.
The safety and efficacy of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in children under 6 years of age have not been studied. Long-term effects of amphetamines in children have not been well established.
8.5 Geriatric Use
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have not been studied in the geriatric population.
4 Contraindications (4 CONTRAINDICATIONS)
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules administration is contraindicated in patients with the following conditions:
- Advanced arteriosclerosis
- Symptomatic cardiovascular disease
- Moderate to severe hypertension
- Hyperthyroidism
- In patients known to be hypersensitive to amphetamine, or other components of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with other amphetamine products [see Adverse Reactions (6.2)].
- Glaucoma
- Agitated states
- History of drug abuse
- Patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions (5.6) and Drug Interactions (7.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions (7 DRUG INTERACTIONS)
- MAOI antidepressants are contraindicated; MAOIs potentiate the effects of amphetamine. Do not administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules during or within 14 days after use of MAOI. (4, 7.1)
- Alkalinizing agents (GI antacids and urinary): These agents increase blood levels of amphetamine. (7.1)
- Acidifying agents (GI and urinary): These agents reduce blood levels of amphetamine. (7.1)
- Adrenergic blockers, antihistamines, antihypertensives, phenobarbital, phenytoin, veratrum alkaloids, and ethosuximide: Effects may be reduced by amphetamines. (7.1)
- Tricyclic antidepressants, norepinephrine, and meperidine: Effects may be potentiated by amphetamines. (7.1)
8.6 Renal Impairment
Due to reduced clearance of amphetamines in patients with severe renal impairment (GFR 15 to <30 mL/min/1.73m2), the recommended dose should be reduced. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are not recommended in patients with ESRD (GFR < 15 ml/min/1.73m2) [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
d-Amphetamine is not dialyzable.
12.2 Pharmacodynamics
Amphetamines block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
Pharmacokinetic studies of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules have been conducted in healthy adult and pediatric (children aged 6 to 12 yrs) subjects, and adolescent (13 to 17 yrs) and children with ADHD. Both dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release) and dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules contain d-amphetamine and l-amphetamine salts in the ratio of 3:1. Following administration of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release), the peak plasma concentrations occurred in about 3 hours for both d-amphetamine and l-amphetamine.
The time to reach maximum plasma concentration (Tmax) for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules is about 7 hours, which is about 4 hours longer compared to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release). This is consistent with the extended-release nature of the product.
Figure 1: Mean d-amphetamine and l-amphetamine Plasma Concentrations Following Administration of Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Extended-Release Capsules 20 mg (8 am) and Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Tablets (immediate-release) 10 mg Twice Daily (8 am and 12 noon) in the Fed State.
A single dose of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended release 20 mg capsules provided comparable plasma concentration profiles of both d-amphetamine and l-amphetamine to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release) 10 mg twice daily administered 4 hours apart.
The mean elimination half-life for d-amphetamine is 10 hours in adults; 11 hours in adolescents aged 13 to 17 years and weighing less than or equal to 75 kg/165 lbs; and 9 hours in children aged 6 to 12 years. For the l-amphetamine, the mean elimination half-life in adults is 13 hours; 13 to 14 hours in adolescents; and 11 hours in children aged 6 to 12 years. On a mg/kg body weight basis, children have a higher clearance than adolescents or adults (see Special Populations below).
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules demonstrates linear pharmacokinetics over the dose range of 20 to 60 mg in adults and adolescents weighing greater than 75 kg/165 lbs, over the dose range of 10 to 40 mg in adolescents weighing less than or equal to 75 kg/165 lbs, and 5 to 30 mg in children aged 6 to 12 years. There is no unexpected accumulation at steady state in children.
Food does not affect the extent of absorption of d-amphetamine and l-amphetamine, but prolongs Tmax by 2.5 hours (from 5.2 hrs. at fasted state to 7.7 hrs. after a high-fat meal) for d-amphetamine and 2.7 hours (from 5.6 hrs. at fasted state to 8.3 hrs. after a high fat meal) for l-amphetamine after administration of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release, 30 mg capsule. Opening the capsule and sprinkling the contents on applesauce results in comparable absorption to the intact capsule taken in the fasted state. Equal doses of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules strengths are bioequivalent.
5.7 Serotonin Syndrome
Serotonin syndrome, a potentially life-threatening reaction, may occur when amphetamines are used in combination with other drugs that affect the serotonergic neurotransmitter systems such as MAOIs, selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John's Wort [see Drug Interactions (7.1)]. Amphetamines and amphetamine derivatives are known to be metabolized, to some degree, by cytochrome P450 2D6 (CYP2D6) and display minor inhibition of CYP2D6 metabolism [see Clinical Pharmacology (12.3)]. The potential for a pharmacokinetic interaction exists with the co-administration of CYP2D6 inhibitors which may increase the risk with increased exposure to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. In these situations, consider an alternative non-serotonergic drug or an alternative drug that does not inhibit CYP2D6 [see Drug Interactions (7.1)]. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
Concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with MAOI drugs is contraindicated [see Contraindications (4)].
Discontinue treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and any concomitant serotonergic agents immediately if symptoms of serotonin syndrome occur, and initiate supportive symptomatic treatment. Concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with other serotonergic drugs or CYP2D6 inhibitors should be used only if the potential benefit justifies the potential risk. If clinically warranted, consider initiating dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with lower doses, monitoring patients for the emergence of serotonin syndrome during drug initiation or titration, and informing patients of the increased risk for serotonin syndrome.
5.8 Visual Disturbance
Difficulties with accommodation and blurring of vision have been reported with stimulant treatment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, a CNS stimulant, are indicated for the treatment of attention deficit hyperactivity disorder (ADHD). (1)
- Children (ages 6 to 12): Efficacy was established in one 3-week outpatient, controlled trial and one analogue classroom, controlled trial in children with ADHD. (14)
- Adolescents (ages 13 to 17): Efficacy was established in one 4-week controlled trial in adolescents with ADHD. (14)
- Adults: Efficacy was established in one 4-week controlled trial in adults with ADHD. (14)
12.1 Mechanism of Action
Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules contain amphetamine, a Schedule II controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serious Cardiovascular Events: Sudden death has been reported with usual doses of CNS stimulants in children and adolescents with structural cardiac abnormalities or other serious heart problems; sudden death, stroke, and myocardial infarction have been reported in adults taking CNS stimulants at usual doses. Stimulant drugs should not be used in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, or other serious heart problems. (5.1)
- Increase in Blood Pressure: Monitor blood pressure and pulse at appropriate intervals. Use with caution in patients for whom blood pressure increases may be problematic. (5.2)
- Psychiatric Adverse Events: Stimulants may cause treatment-emergent psychotic or manic symptoms in patients with no prior history, or exacerbation of symptoms in patients with pre-existing psychosis. Evaluate for bipolar disorder prior to stimulant use. Monitor for aggressive behavior. (5.3)
- Long-term Suppression of Growth: Monitor height and weight at appropriate intervals. (5.4)
- Seizures: May lower the convulsive threshold. Discontinue in the presence of seizures. (5.5)
- Peripheral Vasculopathy, including Raynaud's phenomenon: Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants. (5.6)
- Serotonin Syndrome: Increased risk when co-administered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and initiate supportive treatment (4, 5.7, 10).
- Visual Disturbance: Difficulties with accommodation and blurring of vision have been reported with stimulant treatment. (5.8)
- Tics: May exacerbate tics. Evaluate for tics and Tourette's syndrome prior to stimulant administration. (5.9)
2 Dosage and Administration (2 DOSAGE and ADMINISTRATION)
- Pediatric patients (ages 6 to 17): 10 mg once daily in the morning. Maximum dose for children 6 to 12 is 30 mg once daily. (2.2, 2.3, 2.4)
- Adults: 20 mg once daily in the morning. (2.5)
- Pediatric patients (ages 6 to 17) with severe renal impairment: 5 mg once daily in the morning. Maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. (2.6, 8.6)
- Adults with severe renal impairment: 15 mg once daily in the morning. (2.6, 8.6)
- Patients with ESRD: Not recommended. (2.6, 8.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
| 5 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Blue Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "705" on the body in black ink containing white to off-white beads. | |
| 10 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Blue Opaque Cap/Blue Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "710" on the body in black ink containing white to off-white beads. | |
| 15 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| White Opaque Cap/Blue Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "715" on the body in black ink containing white to off-white beads. | |
| 20 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Orange Opaque Cap/ Orange Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "720" on the body in black ink containing white to off-white beads. | |
| 25 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| White Opaque Cap/Orange Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "725" on the body in black ink containing white to off-white beads. | |
| 30 mg | Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules: |
| Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads. |
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Warning: Abuse and Dependence (WARNING: ABUSE AND DEPENDENCE)
CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2, 9.3)].
6.1 Clinical Trials Experience
The premarketing development program for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules included exposures in a total of 1315 participants in clinical trials (635 pediatric patients, 350 adolescent patients, 248 adult patients, and 82 healthy adult subjects). Of these, 635 patients (ages 6 to 12) were evaluated in two controlled clinical studies, one open-label clinical study, and two single-dose clinical pharmacology studies (N= 40). Safety data on all patients are included in the discussion that follows. Adverse reactions were assessed by collecting adverse reactions, results of physical examinations, vital signs, weights, laboratory analyses, and ECGs.
Adverse reactions during exposure were obtained primarily by general inquiry and recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse reactions without first grouping similar types of reactions into a smaller number of standardized event categories. In the tables and listings that follow, COSTART terminology has been used to classify reported adverse reactions.
The stated frequencies of adverse reactions represent the proportion of individuals who experienced, at least once, a treatment-emergent adverse event of the type listed.
5.10 Prescribing and Dispensing
The least amount of amphetamine feasible should be prescribed or dispensed at one time in order to minimize the possibility of overdosage. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be used with caution in patients who use other sympathomimetic drugs.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.4 Long Term Suppression of Growth (5.4 Long-Term Suppression of Growth)
Monitor growth in children during treatment with stimulants. Patients who are not growing or gaining weight as expected may need to have their treatment interrupted.
Careful follow-up of weight and height in children ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated children over 36 months (to the ages of 10 to 13 years), suggests that consistently medicated children (i.e., treatment for 7 days per week throughout the year) have a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this period of development.
In a controlled trial of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in adolescents, mean weight change from baseline within the initial 4 weeks of therapy was -1.1 lbs. and -2.8 lbs., respectively, for patients receiving 10 mg and 20 mg dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules. Higher doses were associated with greater weight loss within the initial 4 weeks of treatment. Chronic use of amphetamines can be expected to cause a similar suppression of growth.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
30 mg capsules: Orange Opaque Cap/Beige Opaque Body Hard Gelatin Capsule. Printed Laterally with "RP" on the cap and "730" on the body in black ink containing white to off-white beads.
Bottles of 100 capsules: 63629-9481-01
Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required.
Store at 20ºC to 25°C (68°F to 77ºF) [see USP Controlled Room Temperature].
Disposal
Comply with local laws and regulations on drug disposal of CNS stimulants. Dispose of remaining, unused, or expired dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules with an undesirable, nontoxic substance to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and discard dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the household trash.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
5.1 Potential for Abuse and Dependence
CNS stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Boxed Warning, Drug Abuse and Dependence (9.2, 9.3)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Acute administration of high doses of amphetamine (d- or d,l-) has been shown to produce long-lasting neurotoxic effects, including irreversible nerve fiber damage, in rodents. The significance of these findings to humans is unknown.
2.2 Dosing Considerations for All Patients
Individualize the dosage according to the therapeutic needs and response of the patient. Administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at the lowest effective dosage.
Based on bioequivalence data, patients taking divided doses of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release), (for example, twice daily), may be switched to dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules at the same total daily dose taken once daily. Titrate at weekly intervals to appropriate efficacy and tolerability as indicated.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may be taken whole, or the capsule may be opened and the entire contents sprinkled on applesauce. If the patient is using the sprinkle administration method, the sprinkled applesauce should be consumed immediately; it should not be stored. Patients should take the applesauce with sprinkled beads in its entirety without chewing. The dose of a single capsule should not be divided. The contents of the entire capsule should be taken, and patients should not take anything less than one capsule per day.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules may be taken with or without food.
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules should be given upon awakening. Afternoon doses should be avoided because of the potential for insomnia.
Where possible, dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules therapy should be interrupted occasionally to determine if there is a recurrence of behavioral symptoms sufficient to require continued therapy.
1.1 Attention Deficit Hyperactivity Disorder
Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are indicated for the treatment of attention deficit hyperactivity disorder (ADHD).
The efficacy of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules in the treatment of ADHD was established on the basis of two controlled trials in children aged 6 to 12, one controlled trial in adolescents aged 13 to 17, and one controlled trial in adults who met DSM-IV® criteria for ADHD [see Clinical Studies (14)].
A diagnosis of ADHD (DSM-IV®) implies the presence of hyperactive-impulsive or inattentive symptoms that caused impairment and were present before age 7 years. The symptoms must cause clinically significant impairment, e.g., in social, academic, or occupational functioning, and be present in two or more settings, e.g., school (or work) and at home. The symptoms must not be better accounted for by another mental disorder. For the Inattentive Type, at least six of the following symptoms must have persisted for at least 6 months: lack of attention to details/careless mistakes; lack of sustained attention; poor listener; failure to follow through on tasks; poor organization; avoids tasks requiring sustained mental effort; loses things; easily distracted; forgetful. For the Hyperactive-Impulsive Type, at least six of the following symptoms must have persisted for at least 6 months: fidgeting/squirming; leaving seat; inappropriate running/climbing; difficulty with quiet activities; "on the go;" excessive talking; blurting answers; can't wait turn; intrusive. The Combined Type requires both inattentive and hyperactive-impulsive criteria to be met.
2.6 Dosage in Patients With Renal Impairment (2.6 Dosage in Patients with Renal Impairment)
In adult patients with severe renal impairment (GFR 15 to < 30 mL/min/1.73m2), the recommended dose is 15 mg once daily in the morning. In pediatric patients (6 to 17 years of age) with severe renal impairment, the recommended dose is 5 mg once daily. The maximum dose for children 6 to 12 years of age with severe renal impairment is 20 mg once daily. Dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules are not recommended in patients with end stage renal disease (ESRD) (GFR < 15 mL/min/1.73m2 [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.1 Important Information Prior to Initiating Treatment
Prior to initiating treatment with dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, assess for the presence of cardiac disease (e.g., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2)].
Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy. Maintain careful prescription records, educate patients about abuse, monitor for signs for abuse and overdose, and periodically re-evaluate the need for dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules use [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9)].
7.1 Clinically Important Interactions With Amphetamines (7.1 Clinically Important Interactions with Amphetamines)
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact | Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure. |
| Intervention | Do not administer dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules concomitantly or within 14 days after discontinuing MAOI [see Contraindications (4) . |
| Examples | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Serotonergic Drugs | |
| Clinical Impact | The concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and serotonergic drugs increases the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules initiation or dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and the concomitant serotonergic drug(s) [see Warnings and Precautions (5.6)]. |
| Examples | selective serotonin reuptake inhibitors (SSRI), serotonin norepinephrine reuptake inhibitors (SNRI), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John's Wort |
| CYP2D6 Inhibitors | |
| Clinical Impact | The concomitant use of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and CYP2D6 inhibitors may increase the exposure of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules compared to the use of the drug alone and increase the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules initiation and after a dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and the CYP2D6 inhibitor [see Warnings and Precautions (5.6) and Overdosage (10)]. |
| Examples | paroxetine and fluoxetine (also serotonergic drugs), quinidine, ritonavir |
| Alkalinizing Agents | |
| Clinical Impact | Increase blood levels and potentiate the action of amphetamine. |
| Intervention | Co-administration of dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules and gastrointestinal or urinary alkalinizing agents should be avoided. |
| Examples | Gastrointestinal alkalinizing agents (e.g., sodium bicarbonate). Urinary alkalinizing agents (e.g. acetazolamide, some thiazides). |
| Acidifying Agents | |
| Clinical Impact | Lower blood levels and efficacy of amphetamines. |
| Intervention | Increase dose based on clinical response. |
| Examples | Gastrointestinal acidifying agents (e.g., guanethidine, reserpine, glutamic acid HCl, ascorbic acid). Urinary acidifying agents (e.g., ammonium chloride, sodium acid phosphate, methenamine salts). |
| Tricyclic Antidepressants | |
| Clinical Impact | May enhance the activity of tricyclic or sympathomimetic agents causing striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated. |
| Intervention | Monitor frequently and adjust or use alternative therapy based on clinical response. |
| Examples | desipramine, protriptyline |
| Proton Pump Inhibitors | |
| Clinical Impact | Time to maximum concentration (Tmax) of amphetamine is decreased compared to when administered alone. |
| Intervention | Monitor patients for changes in clinical effect and adjust therapy based on clinical response. |
| Examples | Omeprazole |
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon (5.6 Peripheral Vasculopathy, including Raynaud's Phenomenon)
Stimulants, including dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.
6.2 Adverse Reactions Associated With the Use of Amphetamine; Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Extended Release Capsules; Or Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Tablets (immediate Release) (6.2 Adverse Reactions Associated with the Use of Amphetamine; Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Extended-Release Capsules; or Dextroamphetamine Saccharate, Amphetamine Aspartate Monohydrate, Dextroamphetamine Sulfate, and Amphetamine Sulfate Tablets (immediate-release))
The following adverse reactions have been identified during post-approval use of amphetamine; dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate extended-release capsules; or dextroamphetamine saccharate, amphetamine aspartate monohydrate, dextroamphetamine sulfate, and amphetamine sulfate tablets (immediate-release). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:54.371539 · Updated: 2026-03-14T22:00:00.633808