Quality Plus 44-533
2fea3533-8218-4875-affe-5b33a22c553a
34390-5
HUMAN OTC DRUG LABEL
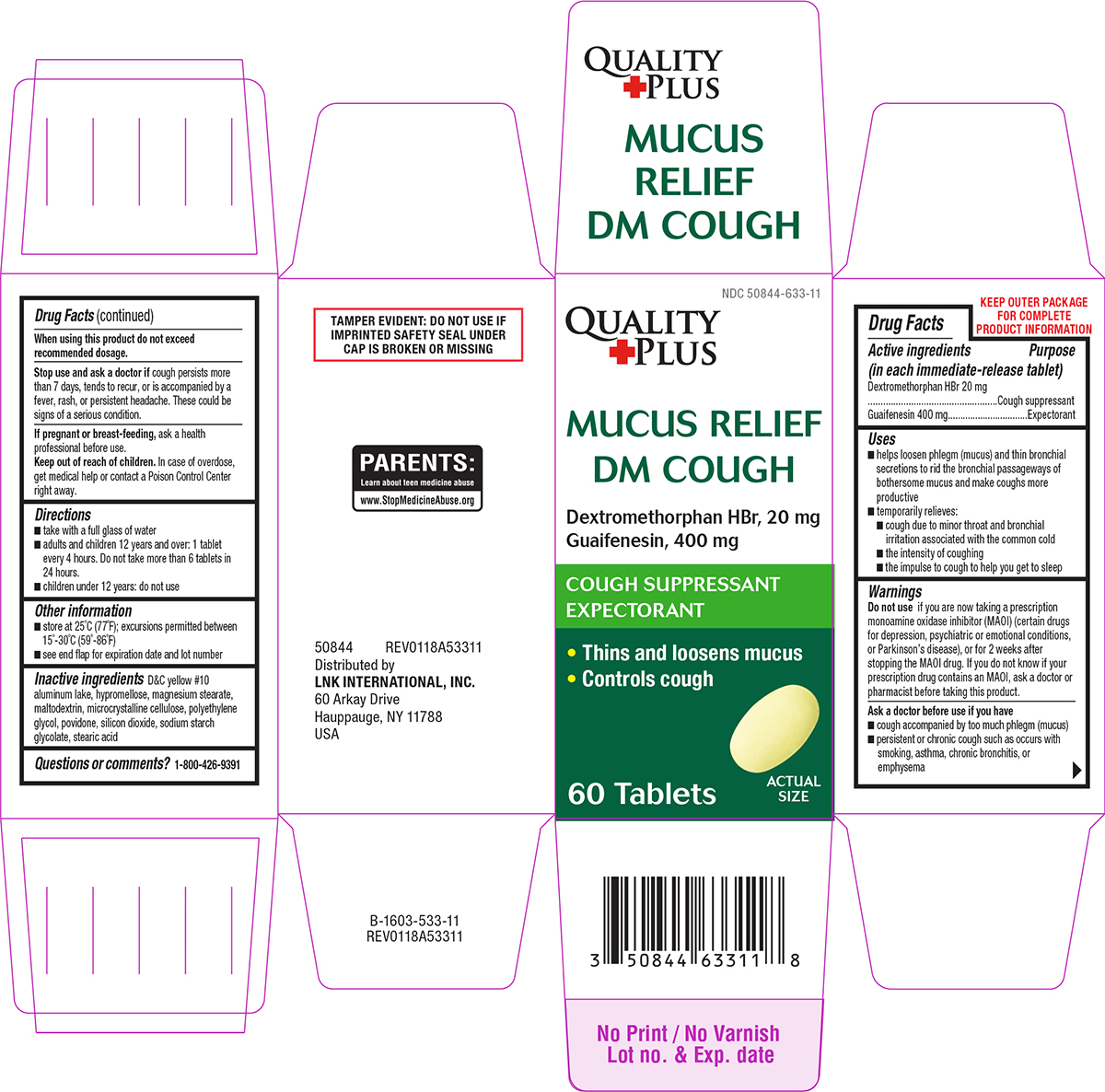
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dextromethorphan HBr 20 mg Guaifenesin 400 mg
Purpose
Cough suppressant Expectorant
Medication Information
Purpose
Cough suppressant
Expectorant
Description
Dextromethorphan HBr 20 mg Guaifenesin 400 mg
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation associated with the common cold
- the intensity of coughing
- the impulse to cough to help you get to sleep
Directions
- take with a full glass of water
- adults and children 12 years and over: 1 tablet every 4 hours. Do not take more than 6 tablets in 24 hours.
- children under 12 years: do not use
Do Not Use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Other Information
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive Ingredients
D&C yellow #10 aluminum lake, hypromellose, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, povidone, silicon dioxide, sodium starch glycolate, stearic acid
Questions Or Comments?
1-800-426-9391
Principal Display Panel
Quality
+Plus
NDC 50844-633-11
MUCUS RELIEF
DM COUGH
Dextromethorphan HBr, 20 mg
Guaifenesin, 400 mg
COUGH SUPPRESSANT
EXPECTORANT
• Thins and loosens mucus
• Controls cough
60 Tablets
ACTUAL
SIZE
50844 REV0118A53311
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive
Hauppauge, NY 11788
USA
TAMPER EVIDENT: DO NOT USE IF
IMPRINTED SAFETY SEAL UNDER
CAP IS BROKEN OR MISSING
PARENTS:
Learn about teen medicine abuse
www.StopMedicineAbuse.org
When Using This Product
do not exceed recommended dosage.
Stop Use and Ask A Doctor If
cough persists more than 7 days, tends to recur, or is accompanied by a fever, rash, or persistent headache. These could be signs of a serious condition.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have
- cough accompanied by too much phlegm (mucus)
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
Active Ingredients (in Each Immediate Release Tablet)
Dextromethorphan HBr 20 mg
Guaifenesin 400 mg
Structured Label Content
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation associated with the common cold
- the intensity of coughing
- the impulse to cough to help you get to sleep
Purpose
Cough suppressant
Expectorant
Directions
- take with a full glass of water
- adults and children 12 years and over: 1 tablet every 4 hours. Do not take more than 6 tablets in 24 hours.
- children under 12 years: do not use
Do Not Use (Do not use)
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Other Information (Other information)
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive Ingredients (Inactive ingredients)
D&C yellow #10 aluminum lake, hypromellose, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, povidone, silicon dioxide, sodium starch glycolate, stearic acid
Questions Or Comments? (Questions or comments?)
1-800-426-9391
Principal Display Panel (Principal display panel)
Quality
+Plus
NDC 50844-633-11
MUCUS RELIEF
DM COUGH
Dextromethorphan HBr, 20 mg
Guaifenesin, 400 mg
COUGH SUPPRESSANT
EXPECTORANT
• Thins and loosens mucus
• Controls cough
60 Tablets
ACTUAL
SIZE
50844 REV0118A53311
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive
Hauppauge, NY 11788
USA
TAMPER EVIDENT: DO NOT USE IF
IMPRINTED SAFETY SEAL UNDER
CAP IS BROKEN OR MISSING
PARENTS:
Learn about teen medicine abuse
www.StopMedicineAbuse.org
When Using This Product (When using this product)
do not exceed recommended dosage.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
cough persists more than 7 days, tends to recur, or is accompanied by a fever, rash, or persistent headache. These could be signs of a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- cough accompanied by too much phlegm (mucus)
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
Active Ingredients (in Each Immediate Release Tablet) (Active ingredients (in each immediate-release tablet))
Dextromethorphan HBr 20 mg
Guaifenesin 400 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:55.007343 · Updated: 2026-03-14T23:08:21.075335