2f80abc0-49d8-968d-e063-6294a90a555b
34390-5
HUMAN OTC DRUG LABEL
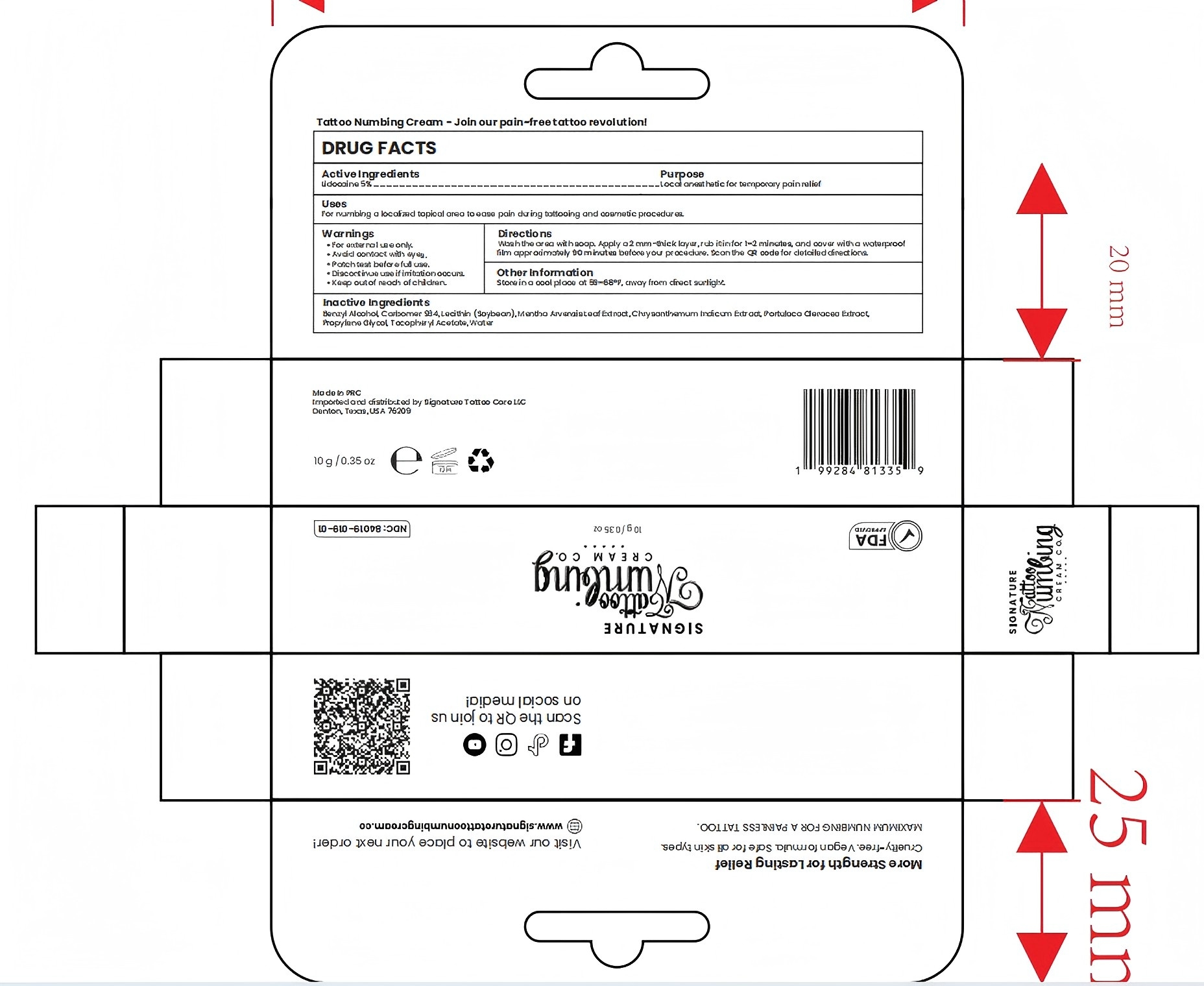
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Stop use and consulta doctor if an allergicreaction occurs. Keep out of reach of children.
Medication Information
Warnings and Precautions
Stop use and consulta doctor if an allergicreaction occurs.
Keep out of reach of children.
Indications and Usage
Apply a 2mm thick layer of cream, rub it in, and cover it with a waterproof film.Leave on for approximately 90 minutes before your procedure for maximum numbing effect.
Dosage and Administration
Apply 1-3 times a week
Description
Stop use and consulta doctor if an allergicreaction occurs. Keep out of reach of children.
Section 50566-9
Discontinueuse if signs of irritation or rash occur
Section 50567-7
Wash the area with soap and water.
Section 51727-6
Benzyl Alcohol, Carbomer 934, Lecithin(Soybean), Propylene Glycol, Tocopheryl Acetate, Water
Section 51945-4
Section 55105-1
Used for numbing local area prior to tattooing
Section 55106-9
5% Lidocaine
Structured Label Content
Indications and Usage (34067-9)
Apply a 2mm thick layer of cream, rub it in, and cover it with a waterproof film.Leave on for approximately 90 minutes before your procedure for maximum numbing effect.
Dosage and Administration (34068-7)
Apply 1-3 times a week
Warnings and Precautions (34071-1)
Stop use and consulta doctor if an allergicreaction occurs.
Keep out of reach of children.
Section 50566-9 (50566-9)
Discontinueuse if signs of irritation or rash occur
Section 50567-7 (50567-7)
Wash the area with soap and water.
Section 51727-6 (51727-6)
Benzyl Alcohol, Carbomer 934, Lecithin(Soybean), Propylene Glycol, Tocopheryl Acetate, Water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Used for numbing local area prior to tattooing
Section 55106-9 (55106-9)
5% Lidocaine
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:40.038693 · Updated: 2026-03-14T23:07:49.806657