Cherry Cough Drops
2f54b9c1-3a37-48fd-b964-6aa2631b8e62
34390-5
HUMAN OTC DRUG LABEL
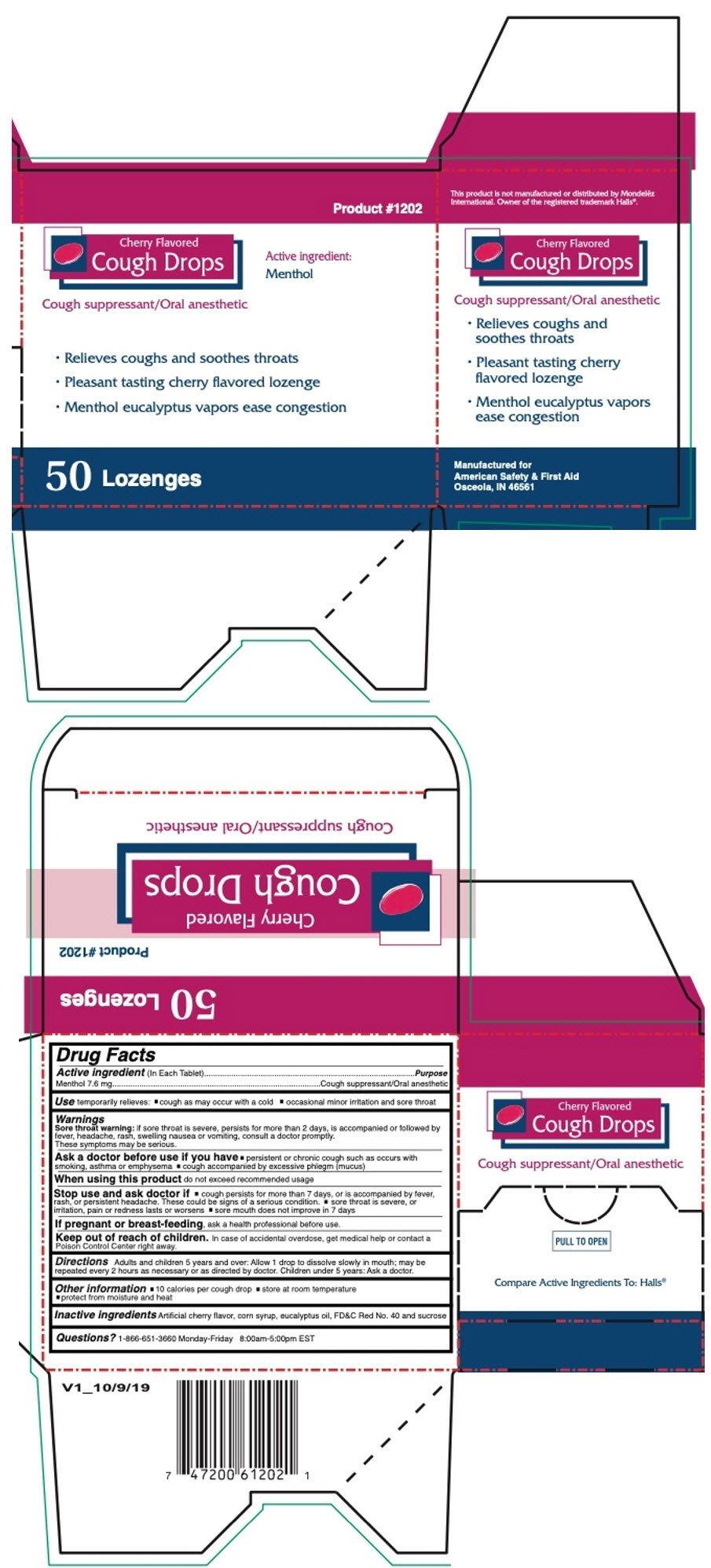
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Cough suppressant/Oral anesthetic
Medication Information
Purpose
Cough suppressant/Oral anesthetic
Description
Drug Facts
Use
temporarily relieves:
- cough as may occur with a cold
- occasional minor irritation and sore throat
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50567-7
When using this product do not exceed recommended usage
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
Adults and children 5 years and over: Allow 1 drop to dissolve slowly in mouth; may be repeated every 2 hours as necessary or as directed by doctor. Children under 5 years: Ask a doctor.
Questions?
1-866-651-3660 Monday-Friday 8:00am-5:00pm EST
Other Information
- 10 calories per cough drop
- store at room temperature
- protect from moisture and heat
Inactive Ingredients
Artificial cherry flavor, corn syrup, eucalyptus oil, FD&C Red No. 40 and sucrose
Stop Use and Ask Doctor If
- cough persists for more than 7 days, or is accompanied by fever, rash, or persistent headache. These could be signs of a serious condition.
- sore throat is severe, or irritation, pain or redness lasts or worsens
- sore mouth does not improve in 7 days
Active Ingredient (in Each Tablet)
Menthol 7.6 mg
Ask A Doctor Before Use If You Have
- persistent or chronic cough such as occurs with smoking, asthma or emphysema
- cough accompanied by excessive phlegm (mucus)
Principal Display Panel 50 Lozenge Box
Product #1202
Cherry Flavored
Cough Drops
Active ingredient:
Menthol
Cough suppressant/Oral anesthetic
- Relieves coughs and soothes throats
- Pleasant tasting cherry flavored lozenge
- Menthol eucalyptus vapors ease congestion
50 Lozenges
Principal Display Panel 100 Lozenge Box
Product #1204
Cherry Flavored
Cough Drops
Active ingredient:
Menthol
Cough suppressant/Oral anesthetic
- Relieves coughs and soothes throats
- Pleasant tasting cherry flavored lozenge
- Menthol eucalyptus vapors ease congestion
100 Lozenges
Structured Label Content
Use
temporarily relieves:
- cough as may occur with a cold
- occasional minor irritation and sore throat
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50567-7 (50567-7)
When using this product do not exceed recommended usage
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Cough suppressant/Oral anesthetic
Directions
Adults and children 5 years and over: Allow 1 drop to dissolve slowly in mouth; may be repeated every 2 hours as necessary or as directed by doctor. Children under 5 years: Ask a doctor.
Questions?
1-866-651-3660 Monday-Friday 8:00am-5:00pm EST
Other Information (Other information)
- 10 calories per cough drop
- store at room temperature
- protect from moisture and heat
Inactive Ingredients (Inactive ingredients)
Artificial cherry flavor, corn syrup, eucalyptus oil, FD&C Red No. 40 and sucrose
Stop Use and Ask Doctor If (Stop use and ask doctor if)
- cough persists for more than 7 days, or is accompanied by fever, rash, or persistent headache. These could be signs of a serious condition.
- sore throat is severe, or irritation, pain or redness lasts or worsens
- sore mouth does not improve in 7 days
Active Ingredient (in Each Tablet) (Active ingredient (In Each Tablet))
Menthol 7.6 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- persistent or chronic cough such as occurs with smoking, asthma or emphysema
- cough accompanied by excessive phlegm (mucus)
Principal Display Panel 50 Lozenge Box (PRINCIPAL DISPLAY PANEL - 50 Lozenge Box)
Product #1202
Cherry Flavored
Cough Drops
Active ingredient:
Menthol
Cough suppressant/Oral anesthetic
- Relieves coughs and soothes throats
- Pleasant tasting cherry flavored lozenge
- Menthol eucalyptus vapors ease congestion
50 Lozenges
Principal Display Panel 100 Lozenge Box (PRINCIPAL DISPLAY PANEL - 100 Lozenge Box)
Product #1204
Cherry Flavored
Cough Drops
Active ingredient:
Menthol
Cough suppressant/Oral anesthetic
- Relieves coughs and soothes throats
- Pleasant tasting cherry flavored lozenge
- Menthol eucalyptus vapors ease congestion
100 Lozenges
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:00.746854 · Updated: 2026-03-14T23:02:50.389879