2f2030c2-f72e-4142-9cff-540bc8fbf8a1
2f2030c2-f72e-4142-9cff-540bc8fbf8a1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
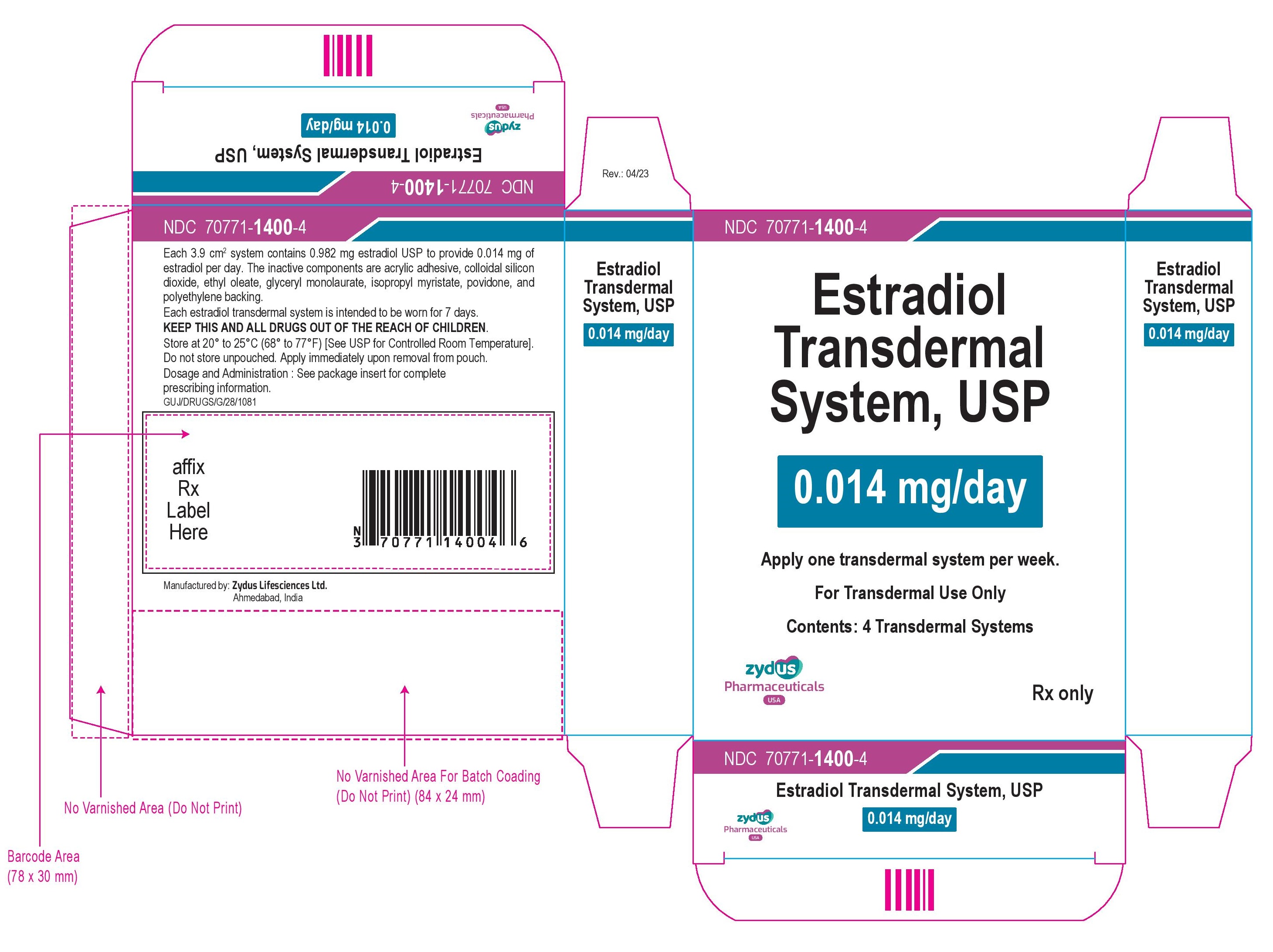
NDC 70771- 1400 -4 Estradiol Transdermal System, USP 0.014 mg/day Apply one transdermal system per week. For Transdermal Use Only Contents: 4 Transdermal Systems Rx Only zydus Pharmaceuticals USA
Medication Information
Description
NDC 70771- 1400 -4 Estradiol Transdermal System, USP 0.014 mg/day Apply one transdermal system per week. For Transdermal Use Only Contents: 4 Transdermal Systems Rx Only zydus Pharmaceuticals USA
Package Label.principal Display Panel
NDC 70771-1400-4
Estradiol Transdermal System, USP
0.014 mg/day
Apply one transdermal system per week.
For Transdermal Use Only
Contents: 4 Transdermal Systems
Rx Only
zydus Pharmaceuticals USA
Structured Label Content
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 70771-1400-4
Estradiol Transdermal System, USP
0.014 mg/day
Apply one transdermal system per week.
For Transdermal Use Only
Contents: 4 Transdermal Systems
Rx Only
zydus Pharmaceuticals USA
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:12.718926 · Updated: 2026-03-14T21:56:58.197015