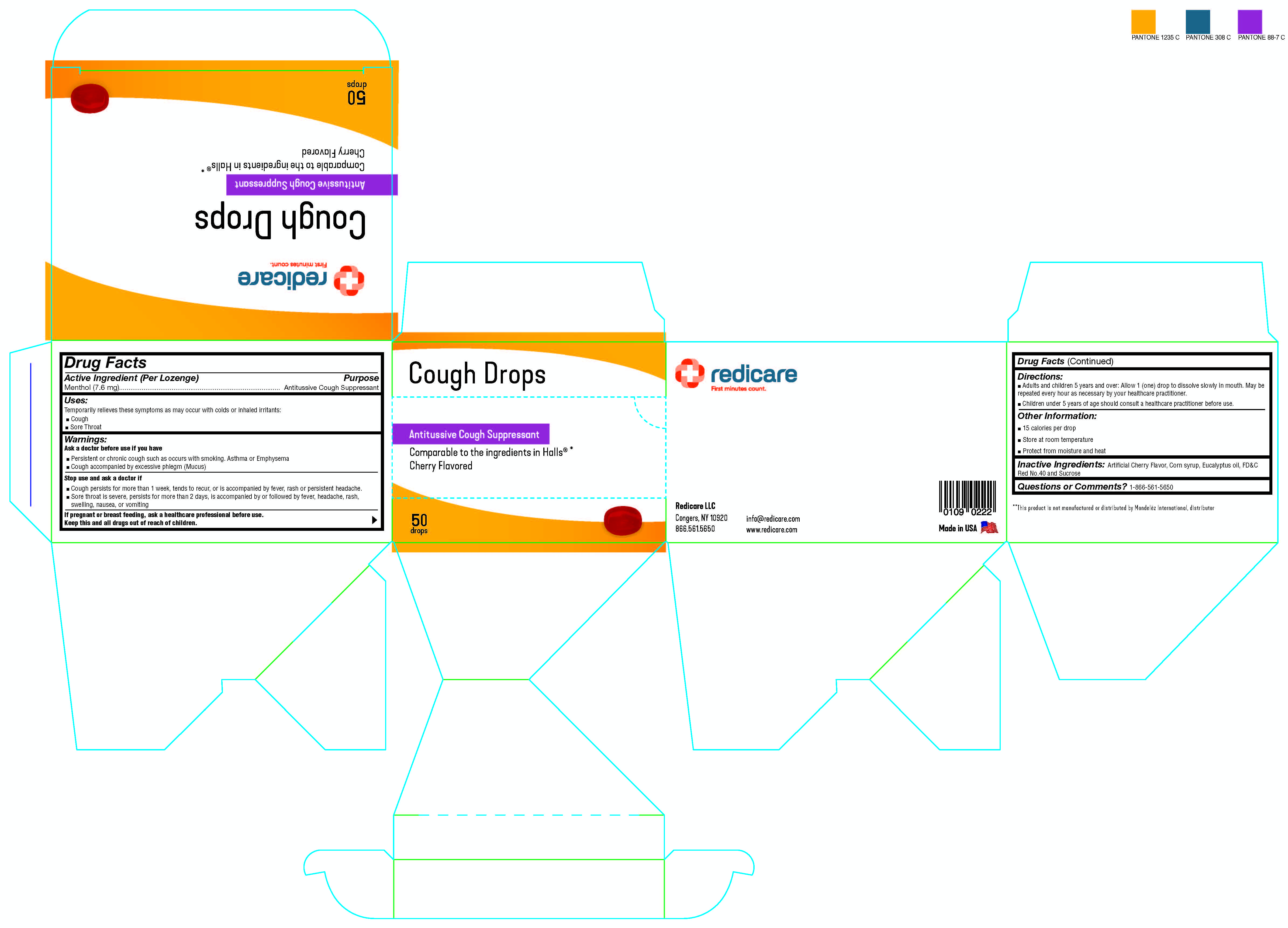
Redicare Cherry Cough Drops
2efeaafa-ee96-37ee-e063-6394a90a9a4b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Menthol (7.6 mg)
Purpose
Antitussive Cough Suppressent
Medication Information
Purpose
Antitussive Cough Suppressent
Description
Menthol (7.6 mg)
Uses
Temporarily relieves these symptoms as may occur with colds or inhaled irritants:
- Cough
- Sore Throat
Section 51945-4
Directions
Adults and children 5 years and over:
- Allow 1 (one) drop to dissolve slowly in mouth. May be repeated every hour as necessary or as directed by your healthcare practitioner.
- Children under 5 years of age should consult a healthcare practitioner before use.
Other Information
- 15 Calories Per Drop
- Store at room temperature
- Protecture from moisture and heat
Inactive Ingredients
Artificial Cherry Flavor, Corn Syrup, Eucalyptus Oil, FD&C Red No. 40, and Sucrose
Questions Or Comments?
1-866-561-5650
Stop Use and Ask A Doctor If
- Cough persists for more than one week, tends to recur, or is accompanied by fever, rash, or persistent headache.
- Sore throat is severe, persists for more than two days, is accompanied by or followed by fever, headache, rash, swelling, nausea, or vomiting
Active Ingredient (per Lozenge)
Menthol (7.6 mg)
Ask A Doctor Before Use If You Have
- Persistent or chronic cough such as occurs with smoking, Asthma, or Emphysema
- Cough accompanied by excessive phlegm (Mucus)
Structured Label Content
Uses
Temporarily relieves these symptoms as may occur with colds or inhaled irritants:
- Cough
- Sore Throat
Section 51945-4 (51945-4)
Purpose
Antitussive Cough Suppressent
Directions
Adults and children 5 years and over:
- Allow 1 (one) drop to dissolve slowly in mouth. May be repeated every hour as necessary or as directed by your healthcare practitioner.
- Children under 5 years of age should consult a healthcare practitioner before use.
Other Information
- 15 Calories Per Drop
- Store at room temperature
- Protecture from moisture and heat
Inactive Ingredients (Inactive ingredients)
Artificial Cherry Flavor, Corn Syrup, Eucalyptus Oil, FD&C Red No. 40, and Sucrose
Questions Or Comments? (Questions or Comments?)
1-866-561-5650
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- Cough persists for more than one week, tends to recur, or is accompanied by fever, rash, or persistent headache.
- Sore throat is severe, persists for more than two days, is accompanied by or followed by fever, headache, rash, swelling, nausea, or vomiting
Active Ingredient (per Lozenge) (Active Ingredient (per lozenge))
Menthol (7.6 mg)
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- Persistent or chronic cough such as occurs with smoking, Asthma, or Emphysema
- Cough accompanied by excessive phlegm (Mucus)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:40.026048 · Updated: 2026-03-14T23:07:49.806657