2edc4479-fb5a-d8a9-e063-6394a90a0db3
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sodium Hyaluronate
Medication Information
Warnings and Precautions
Precautions for Use
If you experience abnormal symptoms or side effects such as red spots, swelling, or itching on the area of use during or after using the cosmetic product due to direct sunlight, consult with a specialist.
Avoid using on areas with wounds.
Precautions for storage and handling:
Keep out of reach of children.
Store away from direct sunlight.
Use only on the vulva, do not use inside the vagina.
Follow the prescribed usage and dosage carefully.
Do not use on infants and toddlers under 3 years old.
It is advisable not to use during pregnancy, and do not use on the area around the vulva immediately before delivery.
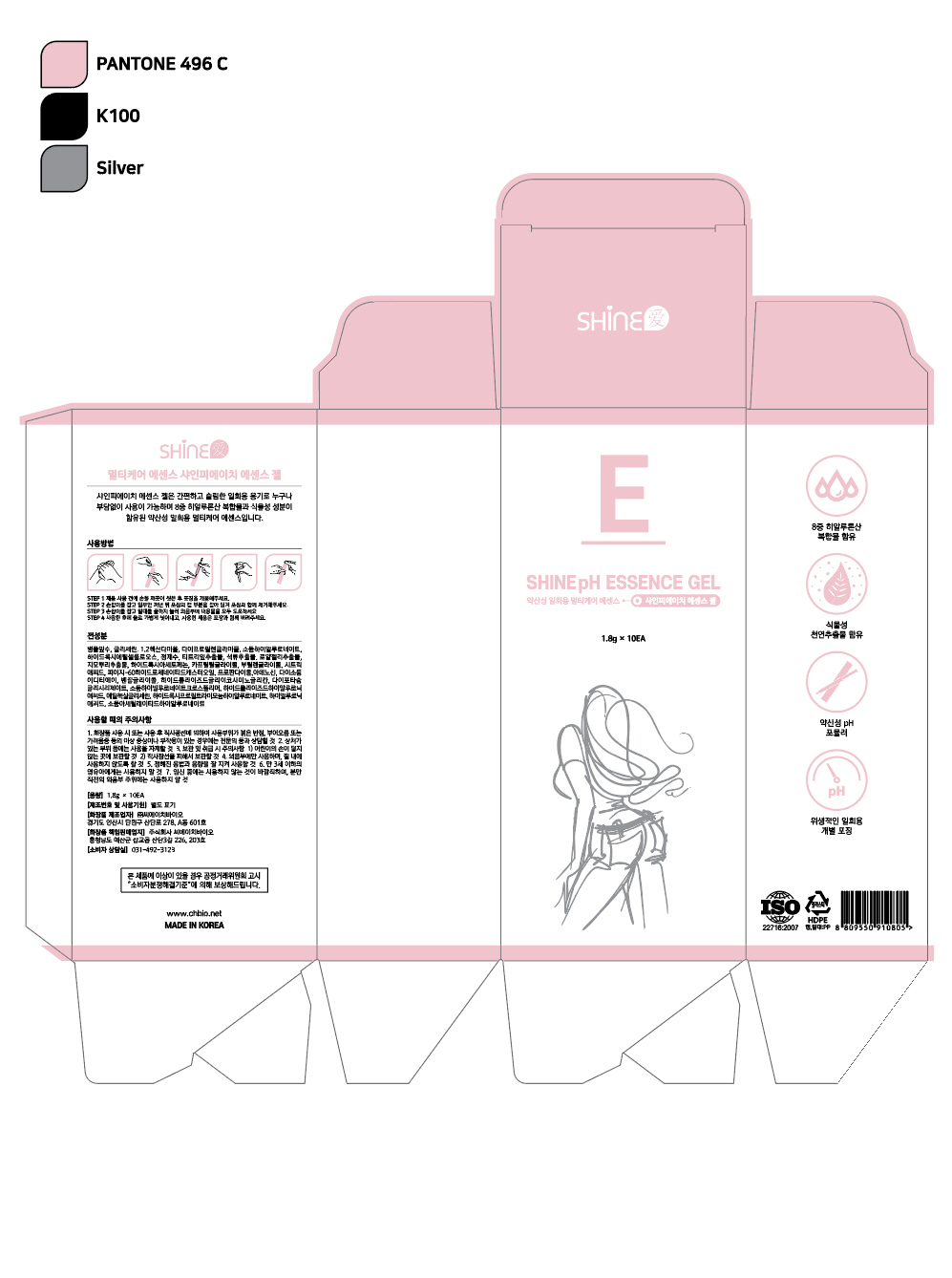
Indications and Usage
Step 1
Wash your hands thoroughly before opening the product packaging.
Step 2
Hold the handle and partially remove the product. Then pull on the cap portion of the packaging to remove it along with the packaging.
Step 3
Holding the handle, press the plunger all the way down to apply the entire contents to the vulva.
Step 4
After use, gently rinse with water. Dispose of the used product along with its packaging.
Dosage and Administration
for external use only
Description
Sodium Hyaluronate
Section 50565-1
keep out or reach of the children
Section 51727-6
Centella Asiatica Leaf Water
Glycerin
1,2-Hexanediol
Dipropylene Glycol
Hydroxyethylcellulose
Water
Melaleuca Alternifolia (Tea Tree) Leaf Extract
Punica Granatum Fruit Extract
Royal Jelly Extract
Anemarrhena Asphodeloides Root Extract
Hydroxyacetophenone
Caprylyl Glycol
Butylene Glycol
Citric Acid
PEG-60 Hydrogenated Castor Oil
Propanediol
Adenosine
Disodium EDTA
Benzyl Glycol
Hydrolyzed Glycosaminoglycans
Dipotassium Glycyrrhizate
Sodium Hyaluronate Crosspolymer
Hydrolyzed Hyaluronic Acid
Ethylhexylglycerin
Hydroxypropyltrimonium Hyaluronate
Hyaluronic Acid
Sodium Acetylated Hyaluronate
Section 51945-4
Section 55105-1
feminine hygiene
Section 55106-9
Sodium Hyaluronate
Structured Label Content
Indications and Usage (34067-9)
Step 1
Wash your hands thoroughly before opening the product packaging.
Step 2
Hold the handle and partially remove the product. Then pull on the cap portion of the packaging to remove it along with the packaging.
Step 3
Holding the handle, press the plunger all the way down to apply the entire contents to the vulva.
Step 4
After use, gently rinse with water. Dispose of the used product along with its packaging.
Dosage and Administration (34068-7)
for external use only
Warnings and Precautions (34071-1)
Precautions for Use
If you experience abnormal symptoms or side effects such as red spots, swelling, or itching on the area of use during or after using the cosmetic product due to direct sunlight, consult with a specialist.
Avoid using on areas with wounds.
Precautions for storage and handling:
Keep out of reach of children.
Store away from direct sunlight.
Use only on the vulva, do not use inside the vagina.
Follow the prescribed usage and dosage carefully.
Do not use on infants and toddlers under 3 years old.
It is advisable not to use during pregnancy, and do not use on the area around the vulva immediately before delivery.
Section 50565-1 (50565-1)
keep out or reach of the children
Section 51727-6 (51727-6)
Centella Asiatica Leaf Water
Glycerin
1,2-Hexanediol
Dipropylene Glycol
Hydroxyethylcellulose
Water
Melaleuca Alternifolia (Tea Tree) Leaf Extract
Punica Granatum Fruit Extract
Royal Jelly Extract
Anemarrhena Asphodeloides Root Extract
Hydroxyacetophenone
Caprylyl Glycol
Butylene Glycol
Citric Acid
PEG-60 Hydrogenated Castor Oil
Propanediol
Adenosine
Disodium EDTA
Benzyl Glycol
Hydrolyzed Glycosaminoglycans
Dipotassium Glycyrrhizate
Sodium Hyaluronate Crosspolymer
Hydrolyzed Hyaluronic Acid
Ethylhexylglycerin
Hydroxypropyltrimonium Hyaluronate
Hyaluronic Acid
Sodium Acetylated Hyaluronate
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
feminine hygiene
Section 55106-9 (55106-9)
Sodium Hyaluronate
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:37.323555 · Updated: 2026-03-14T23:07:42.303933