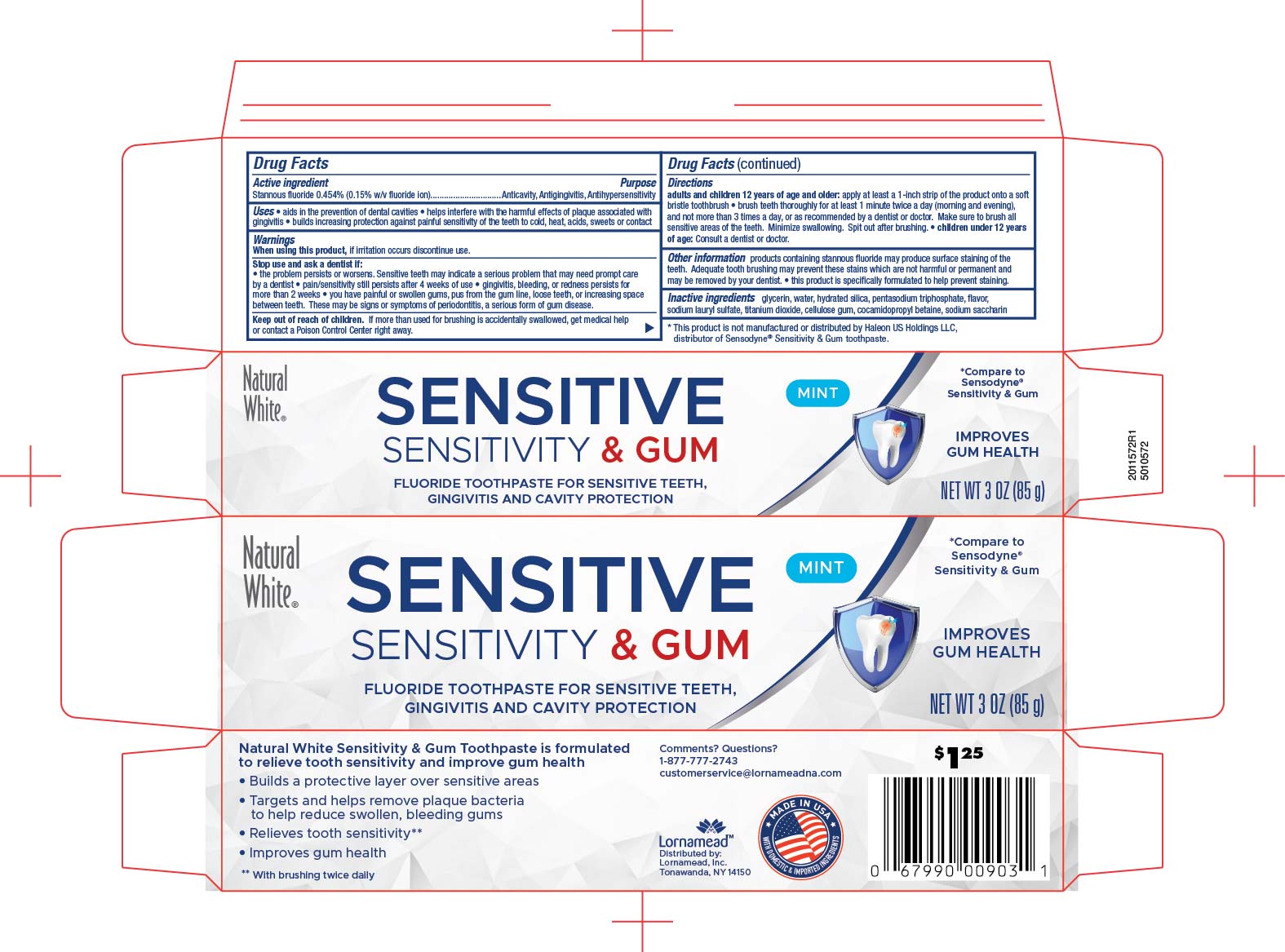
5010572 Dollar Tree Nw Sensitive & Gum Tp (8015053)
2e83bda8-4d4b-640f-e063-6294a90abc60
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Stannous fluoride 0.454% (0.15% w/v fluoride ion)
Medication Information
Indications and Usage
Directions
adults and children 12 years of age and older: apply at least 1-inch strip of the product onto a soft bristle toothbrush
brush teeth thoroughly for at least 1 minute twice a day (morning and evening), and not more than 3 times a day, or as recommended by a dentist or doctor. make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing.
- children under 12 years of age: Consult a dentist or doctor.
Dosage and Administration
Directions
adults and children 12 years of age and older: apply at least 1-inch strip of the product onto a soft bristle toothbrush
- brush teeth thoroughly for at least 1 minute twice a day (morning and evening), and not more than 3 times a day, or as recommended by a dentist or doctor. make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing.
- children under 12 years of age: Consult a dentist or doctor.
Description
Stannous fluoride 0.454% (0.15% w/v fluoride ion)
Section 50565-1
keep out of reach of children. If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a dentist if:
- the problem persists or worsens. Sensitive teeths may indicate a serious problem that may need prompt care by a dentist
- pain/sensitivity still persists after 4 weeks of use
- gingivitis, bleeding, or redness persists formore that 2 weeks
- you have painful or swollen gums,pus from the gum line, loose teeth, or increasinf space between teeth. These may be signs or symptoms of periodontitis, a serious form of gum disease.
Section 50567-7
When using this product, if irritation occurs discontinue use.
Section 51727-6
Inactive ingredients glycerin, water, hydrated silica pentasodium triphosphate, flavor, sodium lauryl sulfate, titanium dioxide, cellulose gum, cocamidopropyl betaine, sodium saccharin
Section 51945-4
Section 55105-1
Anticavity, Antigingivitis, Antihypersensitivity
Section 55106-9
Stannous fluoride 0.454% (0.15% w/v fluoride ion)
Section 60561-8
Other information products containing stannous fluoride may produce surface staining of the teeth. Adequate tooth brushing may prevent these stains which are not harmful or permanent and may be removed by your dentist.
- this product is specifically formulated to help prevent staining.
Structured Label Content
Indications and Usage (34067-9)
Directions
adults and children 12 years of age and older: apply at least 1-inch strip of the product onto a soft bristle toothbrush
brush teeth thoroughly for at least 1 minute twice a day (morning and evening), and not more than 3 times a day, or as recommended by a dentist or doctor. make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing.
- children under 12 years of age: Consult a dentist or doctor.
Dosage and Administration (34068-7)
Directions
adults and children 12 years of age and older: apply at least 1-inch strip of the product onto a soft bristle toothbrush
- brush teeth thoroughly for at least 1 minute twice a day (morning and evening), and not more than 3 times a day, or as recommended by a dentist or doctor. make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing.
- children under 12 years of age: Consult a dentist or doctor.
Section 50565-1 (50565-1)
keep out of reach of children. If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a dentist if:
- the problem persists or worsens. Sensitive teeths may indicate a serious problem that may need prompt care by a dentist
- pain/sensitivity still persists after 4 weeks of use
- gingivitis, bleeding, or redness persists formore that 2 weeks
- you have painful or swollen gums,pus from the gum line, loose teeth, or increasinf space between teeth. These may be signs or symptoms of periodontitis, a serious form of gum disease.
Section 50567-7 (50567-7)
When using this product, if irritation occurs discontinue use.
Section 51727-6 (51727-6)
Inactive ingredients glycerin, water, hydrated silica pentasodium triphosphate, flavor, sodium lauryl sulfate, titanium dioxide, cellulose gum, cocamidopropyl betaine, sodium saccharin
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Anticavity, Antigingivitis, Antihypersensitivity
Section 55106-9 (55106-9)
Stannous fluoride 0.454% (0.15% w/v fluoride ion)
Section 60561-8 (60561-8)
Other information products containing stannous fluoride may produce surface staining of the teeth. Adequate tooth brushing may prevent these stains which are not harmful or permanent and may be removed by your dentist.
- this product is specifically formulated to help prevent staining.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:12.512444 · Updated: 2026-03-14T23:10:26.709421