85244-001
2e67e417-4dba-73a2-e063-6294a90aa4b2
34390-5
HUMAN OTC DRUG LABEL
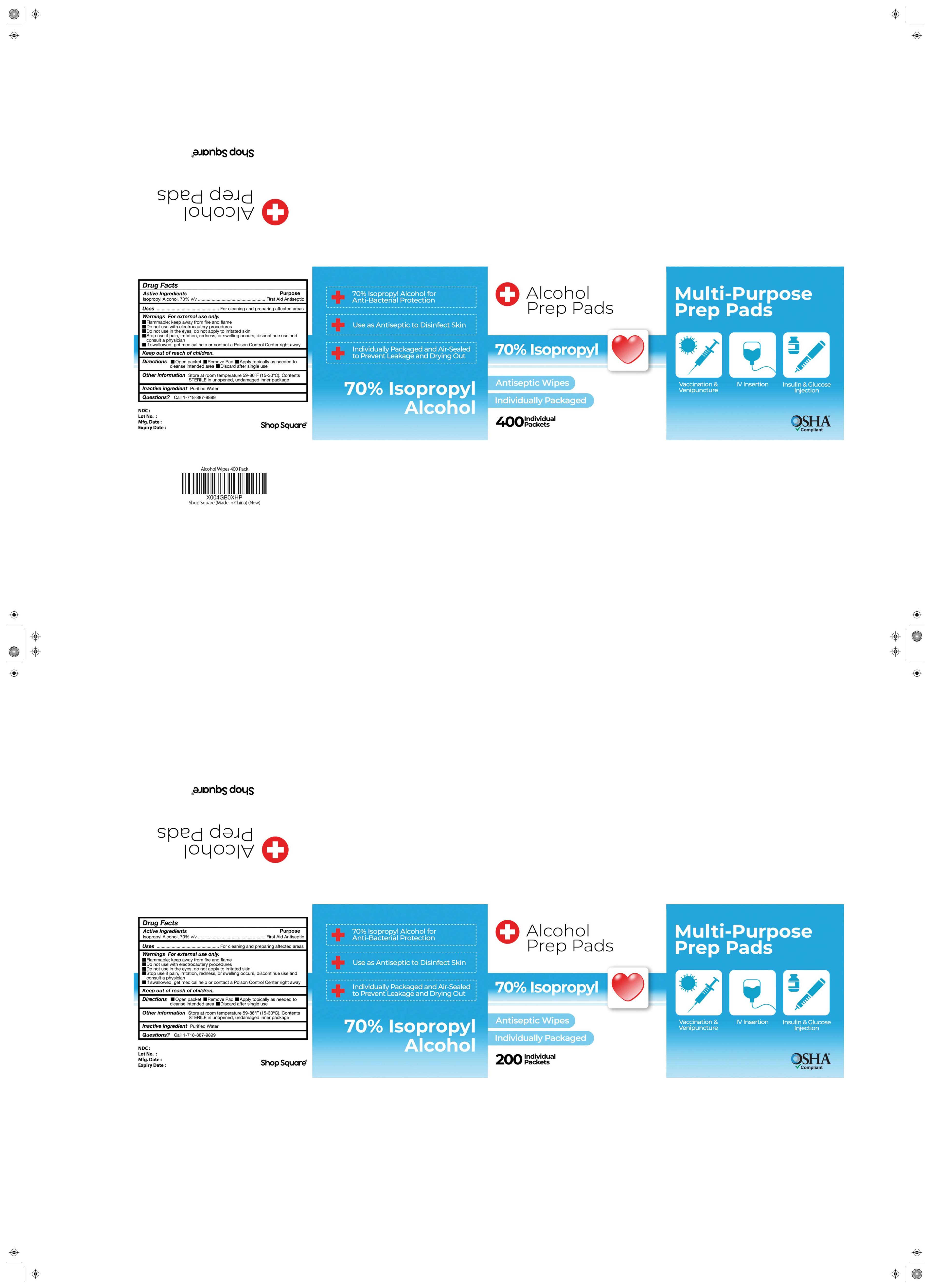
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Isopropyl Aicohol, 70% v/v
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Isopropyl Aicohol, 70% v/v
Use
External use
Stop Use
Stop use if redness or irritationdevelops.discontinue use and consulta physician
Warnings
Flammable.keep away from fire or flame
Questions
call-1-718-887-9899
Directions
Open packet /Remove pad/ Apply topically as needed to cleanse intende area/ Discard after single use
Do Not Use
Do not use with electrocautery proceduresor in the eyes
Active Ingredient
Isopropyl Aicohol, 70% v/v
Other Information
Store at room temperature 59-86℉(15-30°C).ContentsSTERlLE in unopened, undamaged package
Inactive Ingredients
Water
Principal Display Panel
Keep Out of Reach of Children
Keep out of reach of children.
Structured Label Content
Use
External use
Purpose
Antiseptic
Stop Use
Stop use if redness or irritationdevelops.discontinue use and consulta physician
Warnings
Flammable.keep away from fire or flame
Questions
call-1-718-887-9899
Directions
Open packet /Remove pad/ Apply topically as needed to cleanse intende area/ Discard after single use
Do Not Use (Do not use)
Do not use with electrocautery proceduresor in the eyes
Active Ingredient
Isopropyl Aicohol, 70% v/v
Other Information (Other information)
Store at room temperature 59-86℉(15-30°C).ContentsSTERlLE in unopened, undamaged package
Inactive Ingredients (Inactive ingredients)
Water
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Keep Out of Reach of Children (Keep Out Of Reach Of Children)
Keep out of reach of children.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:53.002651 · Updated: 2026-03-14T23:08:17.487612