These Highlights Do Not Include All The Information Needed To Use Vumerity®
2d74414f-6b83-4ea2-9a95-3cecabbce774
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Serious Gastrointestinal Reactions ( 5.7 ) 12/2023
Indications and Usage
VUMERITY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
Dosage and Administration
Blood tests are required prior to initiation of VUMERITY ( 2.1 ) Starting dose: 231 mg twice a day, orally, for 7 days ( 2.2 ) Maintenance dose after 7 days: 462 mg (administered as two 231 mg capsules) twice a day, orally ( 2.2 ) Swallow VUMERITY capsules whole and intact. Do not crush, chew, or sprinkle capsule contents on food ( 2.3 ) Avoid administration of VUMERITY with a high-fat, high-calorie meal/snack ( 2.3 ) Avoid co-administration of VUMERITY with alcohol ( 2.3 )
Warnings and Precautions
Anaphylaxis and Angioedema: Discontinue and do not restart VUMERITY if these occur. ( 5.1 ) Progressive Multifocal Leukoencephalopathy (PML): Withhold VUMERITY at the first sign or symptom suggestive of PML. ( 5.2 ) Herpes Zoster and Other Serious Opportunistic Infections: Consider withholding VUMERITY in cases of serious infection until the infection has resolved. ( 5.3 ) Lymphopenia: Obtain a CBC including lymphocyte count before initiating VUMERITY, after 6 months, and every 6 to 12 months thereafter. Consider interruption of VUMERITY if lymphocyte counts <0.5 × 10 9 /L persist for more than six months. ( 5.4 ) Liver Injury: Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels before initiating VUMERITY and during treatment, as clinically indicated. Discontinue VUMERITY if clinically significant liver injury induced by VUMERITY is suspected. ( 5.6 )
Contraindications
VUMERITY is contraindicated in patients With known hypersensitivity to diroximel fumarate, dimethyl fumarate, or to any of the excipients of VUMERITY. Reactions may include anaphylaxis and angioedema [see Warnings and Precautions ( 5.1 )]. Taking dimethyl fumarate [see Drug Interactions ( 7.1 )] .
Adverse Reactions
The following important adverse reactions are described elsewhere in labeling: Anaphylaxis and Angioedema [see Warnings and Precautions ( 5.1 )] Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions Section ( 5.2 )] Herpes Zoster and Other Serious Opportunistic Infections [see Warnings and Precautions ( 5.3 )] Lymphopenia [see Warnings and Precautions ( 5.4 )] Liver Injury [see Warnings and Precautions ( 5.6 )] Flushing [see Warnings and Precautions ( 5.6 )] Serious Gastrointestinal Reactions [see Warnings and Precautions ( 5.7 )]
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
How Supplied
VUMERITY is available as delayed-release capsules for oral administration, containing 231 mg of diroximel fumarate. The 231 mg capsules have a white cap and a white body, printed with “DRF 231 mg” in black ink on the body. VUMERITY is available as follows: Bottle of 120 capsules, NDC 64406-020-03.
Medication Information
Warnings and Precautions
Anaphylaxis and Angioedema: Discontinue and do not restart VUMERITY if these occur. ( 5.1 ) Progressive Multifocal Leukoencephalopathy (PML): Withhold VUMERITY at the first sign or symptom suggestive of PML. ( 5.2 ) Herpes Zoster and Other Serious Opportunistic Infections: Consider withholding VUMERITY in cases of serious infection until the infection has resolved. ( 5.3 ) Lymphopenia: Obtain a CBC including lymphocyte count before initiating VUMERITY, after 6 months, and every 6 to 12 months thereafter. Consider interruption of VUMERITY if lymphocyte counts <0.5 × 10 9 /L persist for more than six months. ( 5.4 ) Liver Injury: Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels before initiating VUMERITY and during treatment, as clinically indicated. Discontinue VUMERITY if clinically significant liver injury induced by VUMERITY is suspected. ( 5.6 )
Indications and Usage
VUMERITY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
Dosage and Administration
Blood tests are required prior to initiation of VUMERITY ( 2.1 ) Starting dose: 231 mg twice a day, orally, for 7 days ( 2.2 ) Maintenance dose after 7 days: 462 mg (administered as two 231 mg capsules) twice a day, orally ( 2.2 ) Swallow VUMERITY capsules whole and intact. Do not crush, chew, or sprinkle capsule contents on food ( 2.3 ) Avoid administration of VUMERITY with a high-fat, high-calorie meal/snack ( 2.3 ) Avoid co-administration of VUMERITY with alcohol ( 2.3 )
Contraindications
VUMERITY is contraindicated in patients With known hypersensitivity to diroximel fumarate, dimethyl fumarate, or to any of the excipients of VUMERITY. Reactions may include anaphylaxis and angioedema [see Warnings and Precautions ( 5.1 )]. Taking dimethyl fumarate [see Drug Interactions ( 7.1 )] .
Adverse Reactions
The following important adverse reactions are described elsewhere in labeling: Anaphylaxis and Angioedema [see Warnings and Precautions ( 5.1 )] Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions Section ( 5.2 )] Herpes Zoster and Other Serious Opportunistic Infections [see Warnings and Precautions ( 5.3 )] Lymphopenia [see Warnings and Precautions ( 5.4 )] Liver Injury [see Warnings and Precautions ( 5.6 )] Flushing [see Warnings and Precautions ( 5.6 )] Serious Gastrointestinal Reactions [see Warnings and Precautions ( 5.7 )]
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
How Supplied
VUMERITY is available as delayed-release capsules for oral administration, containing 231 mg of diroximel fumarate. The 231 mg capsules have a white cap and a white body, printed with “DRF 231 mg” in black ink on the body. VUMERITY is available as follows: Bottle of 120 capsules, NDC 64406-020-03.
Description
Warnings and Precautions, Serious Gastrointestinal Reactions ( 5.7 ) 12/2023
Section 42229-5
Serious gastrointestinal reactions, including perforation, ulceration, hemorrhage, and obstruction, some with fatal outcomes, have been reported in the postmarketing setting with the use of fumaric acid esters, including VUMERITY, with or without concomitant aspirin use. The majority of these events have occurred within 6 months of fumaric acid ester treatment initiation. In controlled clinical trials, the incidence of serious gastrointestinal adverse reactions was 1% in patients treated with dimethyl fumarate; these events, none of which were fatal, included vomiting (0.3%) and abdominal pain (0.3%) [see Adverse Reactions (6.1)].
Monitor patients, promptly evaluate, and discontinue VUMERITY for new or worsening severe gastrointestinal signs and symptoms.
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 03/2024 |
|
PPI-54499-03 |
|
|
Patient Information
VUMERITY (vue mer' i tee) (diroximel fumarate) delayed-release capsules |
|
What is VUMERITY?
|
|
Do not take VUMERITY if you:
|
|
Before taking and while you take VUMERITY, tell your doctor about all of your medical conditions, including if you:
|
|
| Tell your doctor about all the medicines you take including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |
How should I take VUMERITY?
|
|
What should I avoid while taking VUMERITY?
|
|
|
What are the possible side effects of VUMERITY?
VUMERITY may cause serious side effects including:
|
|
|
|
|
|
The most common side effects of VUMERITY include:
|
|
| These are not all the possible side effects of VUMERITY. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. For more information go to dailymed.nlm.nih.gov |
|
How should I store VUMERITY?
|
|
|
General Information about the safe and effective use of VUMERITY
Medicines are sometimes prescribed for purposes other than those listed in this Patient Information. Do not use VUMERITY for a condition for which it was not prescribed. Do not give VUMERITY to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk to your doctor or pharmacist. You can ask your pharmacist or doctor for information about VUMERITY that is written for healthcare professionals. |
|
|
What are the ingredients in VUMERITY?
|
Section 43683-2
| Warnings and Precautions, Serious Gastrointestinal Reactions (5.7) | 12/2023 |
Section 51945-4
Principal Display Panel – 120 Capsule Carton Label
120
Capsules
RX Only
NDC 64406-020-03
VUMERITY™
(diroximel fumarate)
delayed-release capsules
231 mg
Swallow capsule whole.
Manufactured for:
Biogen Inc.
Cambridge, MA 02142
Product of the
United Kingdom.
Biogen ®
5.6 Flushing
VUMERITY may cause flushing (e.g., warmth, redness, itching, and/or burning sensation). In clinical trials of dimethyl fumarate (which has the same active metabolite as VUMERITY), 40% of dimethyl fumarate-treated patients experienced flushing. Flushing symptoms generally began soon after initiating dimethyl fumarate and usually improved or resolved over time. In the majority of patients who experienced flushing, it was mild or moderate in severity. Three percent (3%) of patients discontinued dimethyl fumarate for flushing and <1% had serious flushing symptoms that were not life-threatening but led to hospitalization.
Administration of VUMERITY with food may reduce the incidence of flushing [see Dosage and Administration (2.3)]. Studies with dimethyl fumarate show that administration of non-enteric coated aspirin (up to a dose of 325 mg) 30 minutes prior to dosing may reduce the incidence or severity of flushing [see Clinical Pharmacology (12.3)].
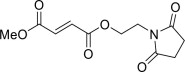
11 Description
VUMERITY contains diroximel fumarate. The chemical name of diroximel fumarate is 2-Butenedioic acid (2E)-, 1-[2-(2,5-dioxo-1-pyrrolidinyl)ethyl] 4-methyl ester, which has a molecular formula of C11H13NO6 and molecular weight of 255.22. Diroximel fumarate has the following structure:
Diroximel fumarate is a white to off-white powder that is slightly soluble in water.
VUMERITY is provided as delayed-release capsules for oral administration. Each capsule contains 231 mg of diroximel fumarate and the following inactive ingredients: crospovidone, colloidal silicon dioxide, magnesium stearate (non-bovine), methacrylic acid and ethyl acrylate copolymer, microcrystalline cellulose, talc, and triethyl citrate.
The capsule shell contains carrageenan, hypromellose, potassium chloride, and titanium dioxide. It is printed with black ink that contains iron oxide, potassium hydroxide, propylene glycol, and shellac.
5.4 Lymphopenia
VUMERITY may decrease lymphocyte counts. In the MS placebo-controlled trials with dimethyl fumarate (which has the same active metabolite as VUMERITY), mean lymphocyte counts decreased by approximately 30% during the first year of treatment with dimethyl fumarate and then remained stable. Four weeks after stopping dimethyl fumarate, mean lymphocyte counts increased but did not return to baseline. Six percent (6%) of dimethyl fumarate patients and <1% of placebo patients experienced lymphocyte counts <0.5 × 109/L (lower limit of normal 0.91 × 109/L). The incidence of infections (60% vs 58%) and serious infections (2% vs 2%) was similar in patients treated with dimethyl fumarate or placebo, respectively. There was no increased incidence of serious infections observed in patients with lymphocyte counts <0.8 × 109/L or ≤0.5 × 109/L in controlled trials, although one patient in an extension study developed PML in the setting of prolonged lymphopenia (lymphocyte counts predominantly <0.5 × 109/L for 3.5 years) [see Warnings and Precautions (5.2)].
In controlled and uncontrolled clinical trials with dimethyl fumarate, 2% of patients experienced prolonged, severe lymphopenia, (defined as lymphocyte counts <0.5 x 109/L for at least six months); in this group of patients, the majority of lymphocyte counts remained <0.5 x 109/L with continued therapy. In these patients with prolonged, severe lymphopenia, the median time for lymphocyte counts to return to normal after discontinuing dimethyl fumarate was 96.0 weeks.
In these controlled and uncontrolled clinical studies, among patients who did not experience prolonged, severe lymphopenia during treatment, the median times for lymphocyte counts to return to normal after discontinuing dimethyl fumarate were as follows:
- 4.3 weeks in patients with mild lymphopenia (lymphocyte count ≥0.8 x 109/L) at discontinuation,
- 10.0 weeks in patients with moderate lymphopenia (lymphocyte count 0.5 to <0.8 x 109/L) at discontinuation, and
- 16.7 weeks in patients with severe lymphopenia (lymphocyte count <0.5 x 109/L) at discontinuation.
Neither VUMERITY nor dimethyl fumarate have been studied in patients with preexisting low lymphocyte counts.
Obtain a complete blood count (CBC), including lymphocyte count, before initiating treatment with VUMERITY, 6 months after starting treatment, and then every 6 to 12 months thereafter, and as clinically indicated. Consider interruption of VUMERITY in patients with lymphocyte counts less than 0.5 × 109/L persisting for more than six months. Given the potential for delayed recovery of lymphocyte counts, continue to obtain lymphocyte counts until their recovery if VUMERITY is discontinued or interrupted because of lymphopenia. Consider withholding treatment from patients with serious infections until resolution. Decisions about whether or not to restart VUMERITY should be individualized based on clinical circumstances.
5.5 Liver Injury
Clinically significant cases of liver injury have been reported in patients treated with dimethyl fumarate (which has the same active metabolite as VUMERITY) in the postmarketing setting. The onset has ranged from a few days to several months after initiation of treatment with dimethyl fumarate. Signs and symptoms of liver injury, including elevation of serum aminotransferases to greater than 5-fold the upper limit of normal and elevation of total bilirubin to greater than 2-fold the upper limit of normal have been observed. These abnormalities resolved upon treatment discontinuation. Some cases required hospitalization. None of the reported cases resulted in liver failure, liver transplant, or death. However, the combination of new serum aminotransferase elevations with increased levels of bilirubin caused by drug-induced hepatocellular injury is an important predictor of serious liver injury that may lead to acute liver failure, liver transplant, or death in some patients.
Elevations of hepatic transaminases (most no greater than 3 times the upper limit of normal) were observed during controlled trials with dimethyl fumarate [see Adverse Reactions (6.1)].
Obtain serum aminotransferase, alkaline phosphatase (ALP), and total bilirubin levels prior to treatment with VUMERITY and during treatment, as clinically indicated. Discontinue VUMERITY if clinically significant liver injury induced by VUMERITY is suspected.
16.1 How Supplied
VUMERITY is available as delayed-release capsules for oral administration, containing 231 mg of diroximel fumarate. The 231 mg capsules have a white cap and a white body, printed with “DRF 231 mg” in black ink on the body. VUMERITY is available as follows:
Bottle of 120 capsules, NDC 64406-020-03.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of dimethyl fumarate and VUMERITY did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently from younger patients.
14 Clinical Studies
The efficacy of VUMERITY is based upon bioavailability studies in patients with relapsing forms of multiple sclerosis and healthy subjects comparing oral dimethyl fumarate delayed-release capsules to VUMERITY delayed-release capsules [see Clinical Pharmacology (12.3)].
The clinical studies described below were conducted using dimethyl fumarate.
The efficacy and safety of dimethyl fumarate were demonstrated in two studies (Studies 1 and 2) that evaluated dimethyl fumarate taken either twice or three times a day in patients with relapsing-remitting multiple sclerosis (RRMS). The starting dose for dimethyl fumarate was 120 mg twice or three times a day for the first 7 days, followed by an increase to 240 mg twice or three times a day. Both studies included patients who had experienced at least 1 relapse over the year preceding the trial or had a brain Magnetic Resonance Imaging (MRI) scan demonstrating at least one gadolinium-enhancing (Gd+) lesion within 6 weeks of randomization. The Expanded Disability Status Scale (EDSS) was also assessed and patients could have scores ranging from 0 to 5. Neurological evaluations were performed at baseline, every 3 months, and at the time of suspected relapse. MRI evaluations were performed at baseline, month 6, and year 1 and 2 in a subset of patients (44% in Study 1 and 48% in Study 2).
4 Contraindications
6 Adverse Reactions
The following important adverse reactions are described elsewhere in labeling:
- Anaphylaxis and Angioedema [see Warnings and Precautions (5.1)]
- Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions Section (5.2)]
- Herpes Zoster and Other Serious Opportunistic Infections [see Warnings and Precautions (5.3)]
- Lymphopenia [see Warnings and Precautions (5.4)]
- Liver Injury [see Warnings and Precautions (5.6)]
- Flushing [see Warnings and Precautions (5.6)]
- Serious Gastrointestinal Reactions [see Warnings and Precautions (5.7)]
8.6 Renal Impairment
No dosage adjustment is necessary in patients with mild renal impairment. Because of an increase in the exposure of a major metabolite [2-hydroxyethyl succinimide (HES)], use of VUMERITY is not recommended in patients with moderate or severe renal impairment [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
After oral administration of VUMERITY, diroximel fumarate undergoes rapid presystemic hydrolysis by esterases and is converted to its active metabolite, monomethyl fumarate (MMF). Diroximel fumarate is not quantifiable in plasma following oral administration of VUMERITY. Therefore, all pharmacokinetic analyses related to VUMERITY were performed with plasma MMF concentrations. Pharmacokinetic data were obtained in subjects with relapsing forms of multiple sclerosis (MS) and healthy volunteers.
2.2 Dosing Information
The starting dosage for VUMERITY is 231 mg twice a day orally. After 7 days, the dosage should be increased to the maintenance dosage of 462 mg (administered as two 231 mg capsules) twice a day orally. Temporary dosage reductions to 231 mg twice a day may be considered for individuals who do not tolerate the maintenance dosage. Within 4 weeks, the recommended dosage of 462 mg twice a day should be resumed. Discontinuation of VUMERITY should be considered for patients unable to tolerate return to the maintenance dosage. Administration of non-enteric coated aspirin (up to a dose of 325 mg) 30 minutes prior to VUMERITY dosing may reduce the incidence or severity of flushing [see Clinical Pharmacology (12.3)].
1 Indications and Usage
VUMERITY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
12.1 Mechanism of Action
The mechanism by which diroximel fumarate exerts its therapeutic effect in multiple sclerosis is unknown. MMF, the active metabolite of diroximel fumarate, has been shown to activate the nuclear factor (erythroid-derived 2)-like 2 (Nrf2) pathway in vitro and in vivo in animals and humans. The Nrf2 pathway is involved in the cellular response to oxidative stress. MMF has been identified as a nicotinic acid receptor agonist in vitro.
16.2 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
5 Warnings and Precautions
- Anaphylaxis and Angioedema: Discontinue and do not restart VUMERITY if these occur. (5.1)
- Progressive Multifocal Leukoencephalopathy (PML): Withhold VUMERITY at the first sign or symptom suggestive of PML. (5.2)
- Herpes Zoster and Other Serious Opportunistic Infections: Consider withholding VUMERITY in cases of serious infection until the infection has resolved. (5.3)
- Lymphopenia: Obtain a CBC including lymphocyte count before initiating VUMERITY, after 6 months, and every 6 to 12 months thereafter. Consider interruption of VUMERITY if lymphocyte counts <0.5 × 109/L persist for more than six months. (5.4)
- Liver Injury: Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels before initiating VUMERITY and during treatment, as clinically indicated. Discontinue VUMERITY if clinically significant liver injury induced by VUMERITY is suspected. (5.6)
2 Dosage and Administration
- Blood tests are required prior to initiation of VUMERITY (2.1)
- Starting dose: 231 mg twice a day, orally, for 7 days (2.2)
- Maintenance dose after 7 days: 462 mg (administered as two 231 mg capsules) twice a day, orally (2.2)
- Swallow VUMERITY capsules whole and intact. Do not crush, chew, or sprinkle capsule contents on food (2.3)
- Avoid administration of VUMERITY with a high-fat, high-calorie meal/snack (2.3)
- Avoid co-administration of VUMERITY with alcohol (2.3)
3 Dosage Forms and Strengths
VUMERITY is available as hard, delayed-release capsules containing 231 mg of diroximel fumarate. The capsules have a white cap and a white body, printed with “DRF 231 mg” in black ink on the body.
6.2 Postmarketing Experience
The following adverse reaction has been identified during post approval use of dimethyl fumarate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Acute pancreatitis; Gastrointestinal perforation, ulceration, obstruction, and hemorrhage [see Warnings and Precautions (5.7)]
Hepatobiliary Disorders: Liver function abnormalities (elevations in transaminases ≥3 times ULN with concomitant elevations in total bilirubin >2 times ULN) [see Warnings and Precautions (5.5)].
Infections and Infestations: Herpes zoster infection and other serious opportunistic infections [See Warnings and Precautions (5.3)].
Respiratory, Thoracic, and Mediastinal Disorders: Rhinorrhea
Skin and Subcutaneous: Alopecia
8 Use in Specific Populations
5.1 Anaphylaxis and Angioedema
VUMERITY can cause anaphylaxis and angioedema after the first dose or at any time during treatment. Signs and symptoms in patients taking dimethyl fumarate (which has the same active metabolite as VUMERITY) have included difficulty breathing, urticaria, and swelling of the throat and tongue. Patients should be instructed to discontinue VUMERITY and seek immediate medical care should they experience signs and symptoms of anaphylaxis or angioedema.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data described in the following sections were obtained using dimethyl fumarate delayed-release capsules, which has the same active metabolite as VUMERITY.
In placebo controlled and uncontrolled clinical studies of dimethyl fumarate (which has the same active metabolite as VUMERITY), a total of 2513 patients have been followed for periods up to 13 years with an overall exposure equivalent to 11,318 person-years. A total of 1169 patients have received at least 5 years of treatment with dimethyl fumarate, and 426 patients have received at least 10 years of treatment with dimethyl fumarate.
2.3 Administration Instructions
Swallow VUMERITY capsules whole and intact. Do not crush or chew, or sprinkle the capsule contents on food.
If taken with food, avoid a high-fat, high-calorie meal/snack; the meal/snack should contain no more than 700 calories and no more than 30 g fat [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
Avoid co-administration of VUMERITY with alcohol [see Clinical Pharmacology (12.3)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
7.1 Concomitant Dimethyl Fumarate
VUMERITY is contraindicated in patients currently taking dimethyl fumarate, which is also metabolized to monomethyl fumarate. VUMERITY may be initiated the day following discontinuation of dimethyl fumarate [see Contraindications (4)].
2.5 Patients With Renal Impairment
13.2 Animal Toxicology And/or Pharmacology
Kidney toxicity, including tubular changes (degeneration, necrosis, regeneration, hypertrophy) and/or interstitial fibrosis, were observed following oral administration of diroximel fumarate in rats and monkeys. In the chronic toxicology study in rats (0, 50, 100, or 300 mg/kg/day), adverse renal findings occurred at all doses tested. Plasma exposures (AUC) at the low dose (50 mg/kg/day) were similar to (MMF) or less than (HES) those in humans at the RHD. In the chronic toxicology study in monkeys (0, 15, 50, or 150 mg/kg/day), adverse renal findings occurred at all but the lowest dose tested (15 mg/kg/day), which was associated with plasma MMF and HES exposures (AUC) less than those in humans at the RHD.
5.2 Progressive Multifocal Leukoencephalopathy
Progressive multifocal leukoencephalopathy (PML) has occurred in patients with MS treated with dimethyl fumarate (which has the same active metabolite as VUMERITY). PML is an opportunistic viral infection of the brain caused by the JC virus (JCV) that typically only occurs in patients who are immunocompromised, and that usually leads to death or severe disability. A fatal case of PML occurred in a patient who received dimethyl fumarate for 4 years while enrolled in a clinical trial. During the clinical trial, the patient experienced prolonged lymphopenia (lymphocyte counts predominantly <0.5 × 109/L for 3.5 years) while taking dimethyl fumarate [see Warnings and Precautions (5.4)]. The patient had no other identified systemic medical conditions resulting in compromised immune system function and had not previously been treated with natalizumab, which has a known association with PML. The patient was also not taking any immunosuppressive or immunomodulatory medications concomitantly.
PML has also occurred in patients taking dimethyl fumarate in the postmarketing setting in the presence of lymphopenia (<0.9 × 109/L). While the role of lymphopenia in these cases is uncertain, the PML cases have occurred predominantly in patients with lymphocyte counts <0.8×109/L persisting for more than 6 months.
At the first sign or symptom suggestive of PML, withhold VUMERITY and perform an appropriate diagnostic evaluation. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes.
Magnetic resonance imaging (MRI) findings may be apparent before clinical signs or symptoms. Cases of PML diagnosed based on MRI findings and the detection of JCV DNA in the cerebrospinal fluid in the absence of clinical signs or symptoms specific to PML, have been reported in patients treated with other MS medications associated with PML. Many of these patients subsequently became symptomatic with PML. Therefore, monitoring with MRI for signs that may be consistent with PML may be useful, and any suspicious findings should lead to further investigation to allow for an early diagnosis of PML, if present. Lower PML-related mortality and morbidity have been reported following discontinuation of another MS medication associated with PML in patients with PML who were initially asymptomatic compared to patients with PML who had characteristic clinical signs and symptoms at diagnosis. It is not known whether these differences are due to early detection and discontinuation of MS treatment or due to differences in disease in these patients.
2.1 Blood Tests Prior to Initiation of Vumerity
5.3 Herpes Zoster and Other Serious Opportunistic Infections
Serious cases of herpes zoster have occurred in patients treated with dimethyl fumarate (which has the same active metabolite as VUMERITY) including disseminated herpes zoster, herpes zoster ophthalmicus, herpes zoster meningoencephalitis, and herpes zoster meningomyelitis. These events may occur at any time during treatment. Monitor patients on VUMERITY for signs and symptoms of herpes zoster. If herpes zoster occurs, appropriate treatment for herpes zoster should be administered.
Other serious opportunistic infections have occurred with dimethyl fumarate, including cases of serious viral (herpes simplex virus, West Nile virus, cytomegalovirus), fungal (Candida and Aspergillus), and bacterial (Nocardia, Listeria monocytogenes, Mycobacterium tuberculosis) infections. These infections have been reported in patients with reduced absolute lymphocyte counts (ALC) as well as in patients with normal ALC. These infections have affected the brain, meninges, spinal cord, gastrointestinal tract, lungs, skin, eye, and ear. Patients with symptoms and signs consistent with any of these infections should undergo prompt diagnostic evaluation and receive appropriate treatment.
Consider withholding VUMERITY treatment in patients with herpes zoster or other serious infections until the infection has resolved [see Adverse Reactions (6.2)].
2.4 Blood Tests to Assess Safety After Initiation of Vumerity
Obtain a complete blood cell count (CBC), including lymphocyte count, 6 months after initiation of VUMERITY and then every 6 to 12 months thereafter, as clinically indicated [see Warnings and Precautions (5.4)].
Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels during treatment with VUMERITY, as clinically indicated [see Warnings and Precautions (5.5)].
Structured Label Content
Section 42229-5 (42229-5)
Serious gastrointestinal reactions, including perforation, ulceration, hemorrhage, and obstruction, some with fatal outcomes, have been reported in the postmarketing setting with the use of fumaric acid esters, including VUMERITY, with or without concomitant aspirin use. The majority of these events have occurred within 6 months of fumaric acid ester treatment initiation. In controlled clinical trials, the incidence of serious gastrointestinal adverse reactions was 1% in patients treated with dimethyl fumarate; these events, none of which were fatal, included vomiting (0.3%) and abdominal pain (0.3%) [see Adverse Reactions (6.1)].
Monitor patients, promptly evaluate, and discontinue VUMERITY for new or worsening severe gastrointestinal signs and symptoms.
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 03/2024 |
|
PPI-54499-03 |
|
|
Patient Information
VUMERITY (vue mer' i tee) (diroximel fumarate) delayed-release capsules |
|
What is VUMERITY?
|
|
Do not take VUMERITY if you:
|
|
Before taking and while you take VUMERITY, tell your doctor about all of your medical conditions, including if you:
|
|
| Tell your doctor about all the medicines you take including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |
How should I take VUMERITY?
|
|
What should I avoid while taking VUMERITY?
|
|
|
What are the possible side effects of VUMERITY?
VUMERITY may cause serious side effects including:
|
|
|
|
|
|
The most common side effects of VUMERITY include:
|
|
| These are not all the possible side effects of VUMERITY. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. For more information go to dailymed.nlm.nih.gov |
|
How should I store VUMERITY?
|
|
|
General Information about the safe and effective use of VUMERITY
Medicines are sometimes prescribed for purposes other than those listed in this Patient Information. Do not use VUMERITY for a condition for which it was not prescribed. Do not give VUMERITY to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk to your doctor or pharmacist. You can ask your pharmacist or doctor for information about VUMERITY that is written for healthcare professionals. |
|
|
What are the ingredients in VUMERITY?
|
Section 43683-2 (43683-2)
| Warnings and Precautions, Serious Gastrointestinal Reactions (5.7) | 12/2023 |
Section 51945-4 (51945-4)
Principal Display Panel – 120 Capsule Carton Label
120
Capsules
RX Only
NDC 64406-020-03
VUMERITY™
(diroximel fumarate)
delayed-release capsules
231 mg
Swallow capsule whole.
Manufactured for:
Biogen Inc.
Cambridge, MA 02142
Product of the
United Kingdom.
Biogen ®
5.6 Flushing
VUMERITY may cause flushing (e.g., warmth, redness, itching, and/or burning sensation). In clinical trials of dimethyl fumarate (which has the same active metabolite as VUMERITY), 40% of dimethyl fumarate-treated patients experienced flushing. Flushing symptoms generally began soon after initiating dimethyl fumarate and usually improved or resolved over time. In the majority of patients who experienced flushing, it was mild or moderate in severity. Three percent (3%) of patients discontinued dimethyl fumarate for flushing and <1% had serious flushing symptoms that were not life-threatening but led to hospitalization.
Administration of VUMERITY with food may reduce the incidence of flushing [see Dosage and Administration (2.3)]. Studies with dimethyl fumarate show that administration of non-enteric coated aspirin (up to a dose of 325 mg) 30 minutes prior to dosing may reduce the incidence or severity of flushing [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
VUMERITY contains diroximel fumarate. The chemical name of diroximel fumarate is 2-Butenedioic acid (2E)-, 1-[2-(2,5-dioxo-1-pyrrolidinyl)ethyl] 4-methyl ester, which has a molecular formula of C11H13NO6 and molecular weight of 255.22. Diroximel fumarate has the following structure:
Diroximel fumarate is a white to off-white powder that is slightly soluble in water.
VUMERITY is provided as delayed-release capsules for oral administration. Each capsule contains 231 mg of diroximel fumarate and the following inactive ingredients: crospovidone, colloidal silicon dioxide, magnesium stearate (non-bovine), methacrylic acid and ethyl acrylate copolymer, microcrystalline cellulose, talc, and triethyl citrate.
The capsule shell contains carrageenan, hypromellose, potassium chloride, and titanium dioxide. It is printed with black ink that contains iron oxide, potassium hydroxide, propylene glycol, and shellac.
5.4 Lymphopenia
VUMERITY may decrease lymphocyte counts. In the MS placebo-controlled trials with dimethyl fumarate (which has the same active metabolite as VUMERITY), mean lymphocyte counts decreased by approximately 30% during the first year of treatment with dimethyl fumarate and then remained stable. Four weeks after stopping dimethyl fumarate, mean lymphocyte counts increased but did not return to baseline. Six percent (6%) of dimethyl fumarate patients and <1% of placebo patients experienced lymphocyte counts <0.5 × 109/L (lower limit of normal 0.91 × 109/L). The incidence of infections (60% vs 58%) and serious infections (2% vs 2%) was similar in patients treated with dimethyl fumarate or placebo, respectively. There was no increased incidence of serious infections observed in patients with lymphocyte counts <0.8 × 109/L or ≤0.5 × 109/L in controlled trials, although one patient in an extension study developed PML in the setting of prolonged lymphopenia (lymphocyte counts predominantly <0.5 × 109/L for 3.5 years) [see Warnings and Precautions (5.2)].
In controlled and uncontrolled clinical trials with dimethyl fumarate, 2% of patients experienced prolonged, severe lymphopenia, (defined as lymphocyte counts <0.5 x 109/L for at least six months); in this group of patients, the majority of lymphocyte counts remained <0.5 x 109/L with continued therapy. In these patients with prolonged, severe lymphopenia, the median time for lymphocyte counts to return to normal after discontinuing dimethyl fumarate was 96.0 weeks.
In these controlled and uncontrolled clinical studies, among patients who did not experience prolonged, severe lymphopenia during treatment, the median times for lymphocyte counts to return to normal after discontinuing dimethyl fumarate were as follows:
- 4.3 weeks in patients with mild lymphopenia (lymphocyte count ≥0.8 x 109/L) at discontinuation,
- 10.0 weeks in patients with moderate lymphopenia (lymphocyte count 0.5 to <0.8 x 109/L) at discontinuation, and
- 16.7 weeks in patients with severe lymphopenia (lymphocyte count <0.5 x 109/L) at discontinuation.
Neither VUMERITY nor dimethyl fumarate have been studied in patients with preexisting low lymphocyte counts.
Obtain a complete blood count (CBC), including lymphocyte count, before initiating treatment with VUMERITY, 6 months after starting treatment, and then every 6 to 12 months thereafter, and as clinically indicated. Consider interruption of VUMERITY in patients with lymphocyte counts less than 0.5 × 109/L persisting for more than six months. Given the potential for delayed recovery of lymphocyte counts, continue to obtain lymphocyte counts until their recovery if VUMERITY is discontinued or interrupted because of lymphopenia. Consider withholding treatment from patients with serious infections until resolution. Decisions about whether or not to restart VUMERITY should be individualized based on clinical circumstances.
5.5 Liver Injury
Clinically significant cases of liver injury have been reported in patients treated with dimethyl fumarate (which has the same active metabolite as VUMERITY) in the postmarketing setting. The onset has ranged from a few days to several months after initiation of treatment with dimethyl fumarate. Signs and symptoms of liver injury, including elevation of serum aminotransferases to greater than 5-fold the upper limit of normal and elevation of total bilirubin to greater than 2-fold the upper limit of normal have been observed. These abnormalities resolved upon treatment discontinuation. Some cases required hospitalization. None of the reported cases resulted in liver failure, liver transplant, or death. However, the combination of new serum aminotransferase elevations with increased levels of bilirubin caused by drug-induced hepatocellular injury is an important predictor of serious liver injury that may lead to acute liver failure, liver transplant, or death in some patients.
Elevations of hepatic transaminases (most no greater than 3 times the upper limit of normal) were observed during controlled trials with dimethyl fumarate [see Adverse Reactions (6.1)].
Obtain serum aminotransferase, alkaline phosphatase (ALP), and total bilirubin levels prior to treatment with VUMERITY and during treatment, as clinically indicated. Discontinue VUMERITY if clinically significant liver injury induced by VUMERITY is suspected.
16.1 How Supplied
VUMERITY is available as delayed-release capsules for oral administration, containing 231 mg of diroximel fumarate. The 231 mg capsules have a white cap and a white body, printed with “DRF 231 mg” in black ink on the body. VUMERITY is available as follows:
Bottle of 120 capsules, NDC 64406-020-03.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of dimethyl fumarate and VUMERITY did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently from younger patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of VUMERITY is based upon bioavailability studies in patients with relapsing forms of multiple sclerosis and healthy subjects comparing oral dimethyl fumarate delayed-release capsules to VUMERITY delayed-release capsules [see Clinical Pharmacology (12.3)].
The clinical studies described below were conducted using dimethyl fumarate.
The efficacy and safety of dimethyl fumarate were demonstrated in two studies (Studies 1 and 2) that evaluated dimethyl fumarate taken either twice or three times a day in patients with relapsing-remitting multiple sclerosis (RRMS). The starting dose for dimethyl fumarate was 120 mg twice or three times a day for the first 7 days, followed by an increase to 240 mg twice or three times a day. Both studies included patients who had experienced at least 1 relapse over the year preceding the trial or had a brain Magnetic Resonance Imaging (MRI) scan demonstrating at least one gadolinium-enhancing (Gd+) lesion within 6 weeks of randomization. The Expanded Disability Status Scale (EDSS) was also assessed and patients could have scores ranging from 0 to 5. Neurological evaluations were performed at baseline, every 3 months, and at the time of suspected relapse. MRI evaluations were performed at baseline, month 6, and year 1 and 2 in a subset of patients (44% in Study 1 and 48% in Study 2).
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described elsewhere in labeling:
- Anaphylaxis and Angioedema [see Warnings and Precautions (5.1)]
- Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions Section (5.2)]
- Herpes Zoster and Other Serious Opportunistic Infections [see Warnings and Precautions (5.3)]
- Lymphopenia [see Warnings and Precautions (5.4)]
- Liver Injury [see Warnings and Precautions (5.6)]
- Flushing [see Warnings and Precautions (5.6)]
- Serious Gastrointestinal Reactions [see Warnings and Precautions (5.7)]
8.6 Renal Impairment
No dosage adjustment is necessary in patients with mild renal impairment. Because of an increase in the exposure of a major metabolite [2-hydroxyethyl succinimide (HES)], use of VUMERITY is not recommended in patients with moderate or severe renal impairment [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
After oral administration of VUMERITY, diroximel fumarate undergoes rapid presystemic hydrolysis by esterases and is converted to its active metabolite, monomethyl fumarate (MMF). Diroximel fumarate is not quantifiable in plasma following oral administration of VUMERITY. Therefore, all pharmacokinetic analyses related to VUMERITY were performed with plasma MMF concentrations. Pharmacokinetic data were obtained in subjects with relapsing forms of multiple sclerosis (MS) and healthy volunteers.
2.2 Dosing Information
The starting dosage for VUMERITY is 231 mg twice a day orally. After 7 days, the dosage should be increased to the maintenance dosage of 462 mg (administered as two 231 mg capsules) twice a day orally. Temporary dosage reductions to 231 mg twice a day may be considered for individuals who do not tolerate the maintenance dosage. Within 4 weeks, the recommended dosage of 462 mg twice a day should be resumed. Discontinuation of VUMERITY should be considered for patients unable to tolerate return to the maintenance dosage. Administration of non-enteric coated aspirin (up to a dose of 325 mg) 30 minutes prior to VUMERITY dosing may reduce the incidence or severity of flushing [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
VUMERITY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
12.1 Mechanism of Action
The mechanism by which diroximel fumarate exerts its therapeutic effect in multiple sclerosis is unknown. MMF, the active metabolite of diroximel fumarate, has been shown to activate the nuclear factor (erythroid-derived 2)-like 2 (Nrf2) pathway in vitro and in vivo in animals and humans. The Nrf2 pathway is involved in the cellular response to oxidative stress. MMF has been identified as a nicotinic acid receptor agonist in vitro.
16.2 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Anaphylaxis and Angioedema: Discontinue and do not restart VUMERITY if these occur. (5.1)
- Progressive Multifocal Leukoencephalopathy (PML): Withhold VUMERITY at the first sign or symptom suggestive of PML. (5.2)
- Herpes Zoster and Other Serious Opportunistic Infections: Consider withholding VUMERITY in cases of serious infection until the infection has resolved. (5.3)
- Lymphopenia: Obtain a CBC including lymphocyte count before initiating VUMERITY, after 6 months, and every 6 to 12 months thereafter. Consider interruption of VUMERITY if lymphocyte counts <0.5 × 109/L persist for more than six months. (5.4)
- Liver Injury: Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels before initiating VUMERITY and during treatment, as clinically indicated. Discontinue VUMERITY if clinically significant liver injury induced by VUMERITY is suspected. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Blood tests are required prior to initiation of VUMERITY (2.1)
- Starting dose: 231 mg twice a day, orally, for 7 days (2.2)
- Maintenance dose after 7 days: 462 mg (administered as two 231 mg capsules) twice a day, orally (2.2)
- Swallow VUMERITY capsules whole and intact. Do not crush, chew, or sprinkle capsule contents on food (2.3)
- Avoid administration of VUMERITY with a high-fat, high-calorie meal/snack (2.3)
- Avoid co-administration of VUMERITY with alcohol (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
VUMERITY is available as hard, delayed-release capsules containing 231 mg of diroximel fumarate. The capsules have a white cap and a white body, printed with “DRF 231 mg” in black ink on the body.
6.2 Postmarketing Experience
The following adverse reaction has been identified during post approval use of dimethyl fumarate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Acute pancreatitis; Gastrointestinal perforation, ulceration, obstruction, and hemorrhage [see Warnings and Precautions (5.7)]
Hepatobiliary Disorders: Liver function abnormalities (elevations in transaminases ≥3 times ULN with concomitant elevations in total bilirubin >2 times ULN) [see Warnings and Precautions (5.5)].
Infections and Infestations: Herpes zoster infection and other serious opportunistic infections [See Warnings and Precautions (5.3)].
Respiratory, Thoracic, and Mediastinal Disorders: Rhinorrhea
Skin and Subcutaneous: Alopecia
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.1 Anaphylaxis and Angioedema
VUMERITY can cause anaphylaxis and angioedema after the first dose or at any time during treatment. Signs and symptoms in patients taking dimethyl fumarate (which has the same active metabolite as VUMERITY) have included difficulty breathing, urticaria, and swelling of the throat and tongue. Patients should be instructed to discontinue VUMERITY and seek immediate medical care should they experience signs and symptoms of anaphylaxis or angioedema.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data described in the following sections were obtained using dimethyl fumarate delayed-release capsules, which has the same active metabolite as VUMERITY.
In placebo controlled and uncontrolled clinical studies of dimethyl fumarate (which has the same active metabolite as VUMERITY), a total of 2513 patients have been followed for periods up to 13 years with an overall exposure equivalent to 11,318 person-years. A total of 1169 patients have received at least 5 years of treatment with dimethyl fumarate, and 426 patients have received at least 10 years of treatment with dimethyl fumarate.
2.3 Administration Instructions
Swallow VUMERITY capsules whole and intact. Do not crush or chew, or sprinkle the capsule contents on food.
If taken with food, avoid a high-fat, high-calorie meal/snack; the meal/snack should contain no more than 700 calories and no more than 30 g fat [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
Avoid co-administration of VUMERITY with alcohol [see Clinical Pharmacology (12.3)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
7.1 Concomitant Dimethyl Fumarate
VUMERITY is contraindicated in patients currently taking dimethyl fumarate, which is also metabolized to monomethyl fumarate. VUMERITY may be initiated the day following discontinuation of dimethyl fumarate [see Contraindications (4)].
2.5 Patients With Renal Impairment
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Kidney toxicity, including tubular changes (degeneration, necrosis, regeneration, hypertrophy) and/or interstitial fibrosis, were observed following oral administration of diroximel fumarate in rats and monkeys. In the chronic toxicology study in rats (0, 50, 100, or 300 mg/kg/day), adverse renal findings occurred at all doses tested. Plasma exposures (AUC) at the low dose (50 mg/kg/day) were similar to (MMF) or less than (HES) those in humans at the RHD. In the chronic toxicology study in monkeys (0, 15, 50, or 150 mg/kg/day), adverse renal findings occurred at all but the lowest dose tested (15 mg/kg/day), which was associated with plasma MMF and HES exposures (AUC) less than those in humans at the RHD.
5.2 Progressive Multifocal Leukoencephalopathy
Progressive multifocal leukoencephalopathy (PML) has occurred in patients with MS treated with dimethyl fumarate (which has the same active metabolite as VUMERITY). PML is an opportunistic viral infection of the brain caused by the JC virus (JCV) that typically only occurs in patients who are immunocompromised, and that usually leads to death or severe disability. A fatal case of PML occurred in a patient who received dimethyl fumarate for 4 years while enrolled in a clinical trial. During the clinical trial, the patient experienced prolonged lymphopenia (lymphocyte counts predominantly <0.5 × 109/L for 3.5 years) while taking dimethyl fumarate [see Warnings and Precautions (5.4)]. The patient had no other identified systemic medical conditions resulting in compromised immune system function and had not previously been treated with natalizumab, which has a known association with PML. The patient was also not taking any immunosuppressive or immunomodulatory medications concomitantly.
PML has also occurred in patients taking dimethyl fumarate in the postmarketing setting in the presence of lymphopenia (<0.9 × 109/L). While the role of lymphopenia in these cases is uncertain, the PML cases have occurred predominantly in patients with lymphocyte counts <0.8×109/L persisting for more than 6 months.
At the first sign or symptom suggestive of PML, withhold VUMERITY and perform an appropriate diagnostic evaluation. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes.
Magnetic resonance imaging (MRI) findings may be apparent before clinical signs or symptoms. Cases of PML diagnosed based on MRI findings and the detection of JCV DNA in the cerebrospinal fluid in the absence of clinical signs or symptoms specific to PML, have been reported in patients treated with other MS medications associated with PML. Many of these patients subsequently became symptomatic with PML. Therefore, monitoring with MRI for signs that may be consistent with PML may be useful, and any suspicious findings should lead to further investigation to allow for an early diagnosis of PML, if present. Lower PML-related mortality and morbidity have been reported following discontinuation of another MS medication associated with PML in patients with PML who were initially asymptomatic compared to patients with PML who had characteristic clinical signs and symptoms at diagnosis. It is not known whether these differences are due to early detection and discontinuation of MS treatment or due to differences in disease in these patients.
2.1 Blood Tests Prior to Initiation of Vumerity (2.1 Blood Tests Prior to Initiation of VUMERITY)
5.3 Herpes Zoster and Other Serious Opportunistic Infections
Serious cases of herpes zoster have occurred in patients treated with dimethyl fumarate (which has the same active metabolite as VUMERITY) including disseminated herpes zoster, herpes zoster ophthalmicus, herpes zoster meningoencephalitis, and herpes zoster meningomyelitis. These events may occur at any time during treatment. Monitor patients on VUMERITY for signs and symptoms of herpes zoster. If herpes zoster occurs, appropriate treatment for herpes zoster should be administered.
Other serious opportunistic infections have occurred with dimethyl fumarate, including cases of serious viral (herpes simplex virus, West Nile virus, cytomegalovirus), fungal (Candida and Aspergillus), and bacterial (Nocardia, Listeria monocytogenes, Mycobacterium tuberculosis) infections. These infections have been reported in patients with reduced absolute lymphocyte counts (ALC) as well as in patients with normal ALC. These infections have affected the brain, meninges, spinal cord, gastrointestinal tract, lungs, skin, eye, and ear. Patients with symptoms and signs consistent with any of these infections should undergo prompt diagnostic evaluation and receive appropriate treatment.
Consider withholding VUMERITY treatment in patients with herpes zoster or other serious infections until the infection has resolved [see Adverse Reactions (6.2)].
2.4 Blood Tests to Assess Safety After Initiation of Vumerity (2.4 Blood Tests to Assess Safety After Initiation of VUMERITY)
Obtain a complete blood cell count (CBC), including lymphocyte count, 6 months after initiation of VUMERITY and then every 6 to 12 months thereafter, as clinically indicated [see Warnings and Precautions (5.4)].
Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels during treatment with VUMERITY, as clinically indicated [see Warnings and Precautions (5.5)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:21.643792 · Updated: 2026-03-14T22:26:55.770037