Pitavastatin Tablets. These Highlights Do Not Include All The Information Needed To Use Pitavastatin Tablets Safely And Effectively. See Full Prescribing Information For Pitavastatin Tablets.
2d5a1b78-4aa6-537b-e063-6394a90a522f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Pitavastatin tablets are indicated as an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in: Adults with primary hyperlipidemia. Adults and pediatric patients aged 8 years and older with heterozygous familial hypercholesterolemia (HeFH).
Indications and Usage
Pitavastatin tablets are indicated as an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in: Adults with primary hyperlipidemia. Adults and pediatric patients aged 8 years and older with heterozygous familial hypercholesterolemia (HeFH).
Dosage and Administration
Take pitavastatin tablets orally once daily with or without food at the same time each day. For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving pitavastatin tablets 4 mg daily, prescribe alternative LDL-C-lowering treatment. Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating pitavastatin tablets, and adjust the dosage if necessary.
Warnings and Precautions
Myopathy and Rhabdomyolysis: Risk factors include age 65 or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher pitavastatin tablets dosage. Discontinue pitavastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue pitavastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the pitavastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever. ( 5.1 , 7 , 8.5 , 8.6 ) Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue pitavastatin if IMNM is suspected. ( 5.2 ) Hepatic Dysfunction : Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue pitavastatin tablets. ( 5.3 )
Contraindications
Pitavastatin tablets is contraindicated in the following conditions: Concomitant use of cyclosporine [see Drug Interactions (7) ] . Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3) ] . Hypersensitivity to pitavastatin or any excipients in pitavastatin tablets. Hypersensitivity reactions including angioedema, rash, pruritus, and urticaria have been reported with pitavastatin tablets [see Adverse Reactions (6) ] .
Adverse Reactions
The following serious adverse reactions are discussed in other sections of the labeling: Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1) ] Immune-Mediated Necrotizing Myopathy [see Warning and Precautions (5.2) ] Hepatic Dysfunction [see Warning and Precautions (5.3) ] Increases in HbA1c and Fasting Serum Glucose Levels [see Warning and Precautions (5.4) ] .
Drug Interactions
Table 2 includes a list of drugs that increase the risk of myopathy and rhabdomyolysis when administered concomitantly with pitavastatin and instructions for preventing or managing drug interactions [see Warnings and Precautions (5.1) , Clinical Pharmacology (12.3) ] . Table 2. Drug Interactions that Increase the Risk of Myopathy and Rhabdomyolysis with Pitavastatin Tablets Cyclosporine Clinical Impact: Cyclosporine significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . Intervention: Concomitant use of cyclosporine with pitavastatin is contraindicated [see Contraindications (4) ] . Gemfibrozil Clinical Impact: Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of gemfibrozil with statins, including pitavastatin. Intervention: Avoid concomitant use of gemfibrozil with pitavastatin. Erythromycin Clinical Impact: Erythromycin significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . Intervention: In patients taking erythromycin, do not exceed pitavastatin tablets 1 mg once daily [see Dosage and Administration (2.4) ] . Rifampin Clinical Impact: Rifampin significantly increases peak pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . Intervention: In patients taking rifampin, do not exceed pitavastatin tablets 2 mg once daily [see Dosage and Administration (2.4) ] . Fibrates Clinical Impact: Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins, including pitavastatin. Intervention: Consider if the benefit of using fibrates concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. Niacin Clinical Impact: The risk of myopathy and rhabdomyolysis may be increased with concomitant use of lipid-modifying doses (≥1 g/day) of niacin with pitavastatin. Intervention: Consider if the benefit of using lipid-modifying doses (>1 g/day) of niacin concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. Colchicine Clinical Impact: Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with statins, including pitavastatin. Intervention: Consider the risk/benefit of concomitant use of colchicine with pitavastatin.
Storage and Handling
Pitavastatin tablets, for oral administration are available as: 1 mg : Round, white to off-white, film-coated tablet, debossed with "P1" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-956-90 2 mg : Round, white to off-white, film-coated tablet, debossed with "P2" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-957-90 4 mg : Round, white to off-white, film-coated tablet, debossed with "P4" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-958-90
How Supplied
Pitavastatin tablets, for oral administration are available as: 1 mg : Round, white to off-white, film-coated tablet, debossed with "P1" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-956-90 2 mg : Round, white to off-white, film-coated tablet, debossed with "P2" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-957-90 4 mg : Round, white to off-white, film-coated tablet, debossed with "P4" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-958-90
Medication Information
Warnings and Precautions
Myopathy and Rhabdomyolysis: Risk factors include age 65 or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher pitavastatin tablets dosage. Discontinue pitavastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue pitavastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the pitavastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever. ( 5.1 , 7 , 8.5 , 8.6 ) Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue pitavastatin if IMNM is suspected. ( 5.2 ) Hepatic Dysfunction : Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue pitavastatin tablets. ( 5.3 )
Indications and Usage
Pitavastatin tablets are indicated as an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in: Adults with primary hyperlipidemia. Adults and pediatric patients aged 8 years and older with heterozygous familial hypercholesterolemia (HeFH).
Dosage and Administration
Take pitavastatin tablets orally once daily with or without food at the same time each day. For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving pitavastatin tablets 4 mg daily, prescribe alternative LDL-C-lowering treatment. Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating pitavastatin tablets, and adjust the dosage if necessary.
Contraindications
Pitavastatin tablets is contraindicated in the following conditions: Concomitant use of cyclosporine [see Drug Interactions (7) ] . Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3) ] . Hypersensitivity to pitavastatin or any excipients in pitavastatin tablets. Hypersensitivity reactions including angioedema, rash, pruritus, and urticaria have been reported with pitavastatin tablets [see Adverse Reactions (6) ] .
Adverse Reactions
The following serious adverse reactions are discussed in other sections of the labeling: Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1) ] Immune-Mediated Necrotizing Myopathy [see Warning and Precautions (5.2) ] Hepatic Dysfunction [see Warning and Precautions (5.3) ] Increases in HbA1c and Fasting Serum Glucose Levels [see Warning and Precautions (5.4) ] .
Drug Interactions
Table 2 includes a list of drugs that increase the risk of myopathy and rhabdomyolysis when administered concomitantly with pitavastatin and instructions for preventing or managing drug interactions [see Warnings and Precautions (5.1) , Clinical Pharmacology (12.3) ] . Table 2. Drug Interactions that Increase the Risk of Myopathy and Rhabdomyolysis with Pitavastatin Tablets Cyclosporine Clinical Impact: Cyclosporine significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . Intervention: Concomitant use of cyclosporine with pitavastatin is contraindicated [see Contraindications (4) ] . Gemfibrozil Clinical Impact: Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of gemfibrozil with statins, including pitavastatin. Intervention: Avoid concomitant use of gemfibrozil with pitavastatin. Erythromycin Clinical Impact: Erythromycin significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . Intervention: In patients taking erythromycin, do not exceed pitavastatin tablets 1 mg once daily [see Dosage and Administration (2.4) ] . Rifampin Clinical Impact: Rifampin significantly increases peak pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . Intervention: In patients taking rifampin, do not exceed pitavastatin tablets 2 mg once daily [see Dosage and Administration (2.4) ] . Fibrates Clinical Impact: Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins, including pitavastatin. Intervention: Consider if the benefit of using fibrates concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. Niacin Clinical Impact: The risk of myopathy and rhabdomyolysis may be increased with concomitant use of lipid-modifying doses (≥1 g/day) of niacin with pitavastatin. Intervention: Consider if the benefit of using lipid-modifying doses (>1 g/day) of niacin concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. Colchicine Clinical Impact: Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with statins, including pitavastatin. Intervention: Consider the risk/benefit of concomitant use of colchicine with pitavastatin.
Storage and Handling
Pitavastatin tablets, for oral administration are available as: 1 mg : Round, white to off-white, film-coated tablet, debossed with "P1" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-956-90 2 mg : Round, white to off-white, film-coated tablet, debossed with "P2" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-957-90 4 mg : Round, white to off-white, film-coated tablet, debossed with "P4" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-958-90
How Supplied
Pitavastatin tablets, for oral administration are available as: 1 mg : Round, white to off-white, film-coated tablet, debossed with "P1" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-956-90 2 mg : Round, white to off-white, film-coated tablet, debossed with "P2" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-957-90 4 mg : Round, white to off-white, film-coated tablet, debossed with "P4" on one side. They are supplied as: Bottles of 90 with a child-resistant closure, NDC 51407-958-90
Description
Pitavastatin tablets are indicated as an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in: Adults with primary hyperlipidemia. Adults and pediatric patients aged 8 years and older with heterozygous familial hypercholesterolemia (HeFH).
Section 42229-5
Risk Factors for Myopathy
Risk factors for myopathy include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use of certain drugs (including other lipid-lowering therapies), and higher pitavastatin tablets dosage [see Dosage and Administration (2.2), Drug Interactions (7) and Use in Specific Populations (8.5, 8.6)] . Dosages of pitavastatin tablets greater than 4 mg once daily were associated with an increased risk for severe myopathy in premarketing clinical studies. The maximum recommended dose of pitavastatin tablets is 4 mg once daily.
Section 44425-7
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light.
Dispense contents in a tight, light-resistant container with a child-resistant closure.
10 Overdosage
No specific treatment for pitavastatin tablets overdose is known. Contact Poison Control (1-800-222-1222) for latest recommendations. Hemodialysis is unlikely to be of benefit due to high protein binding ratio of pitavastatin tablets.
11 Description
Pitavastatin tablets for oral use is an HMG-CoA reductase inhibitor.
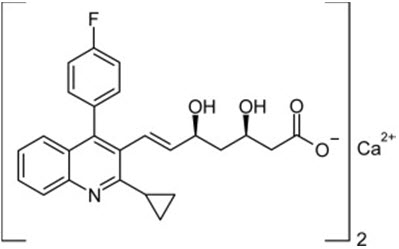
The chemical name for pitavastatin is (+)monocalcium bis{(3R, 5S, 6 E)-7-[2-cyclopropyl-4-(4-fluorophenyl)-3-quinolyl]-3,5- dihydroxy-6-heptenoate}. The structural formula is:
The empirical formula for pitavastatin is C 50H 46CaF 2N 2O 8and the molecular weight is 880.98. Pitavastatin is odorless and occurs as white to pale-yellow powder. It is freely soluble in pyridine, chloroform, dilute hydrochloric acid, and tetrahydrofuran, soluble in ethylene glycol, sparingly soluble in octanol, slightly soluble in methanol, very slightly soluble in water or ethanol, and practically insoluble in acetonitrile or diethyl ether. Pitavastatin is hygroscopic and slightly unstable in light.
Each film-coated pitavastatin tablet contains 1.045 mg, 2.09 mg, or 4.18 mg of pitavastatin calcium, which is equivalent to 1 mg, 2 mg, or 4 mg, respectively of free base and the following inactive ingredients: lactose monohydrate, magnesium carbonate, low-substituted hydroxypropyl cellulose, hypromellose and magnesium stearate. The film coating contains: hypromellose, polyethylene glycol, talc and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of pitavastatin tablets as an adjunctive therapy to diet to reduce elevated LDL-C in pediatric patients aged 8 years and older with HeFH have been established. Use of pitavastatin tablets for this indication is supported by a 12-week, double-blind, placebo-controlled trial in 82 pediatric patients 8 to 16 years of age with HeFH [see Clinical Studies (14)] and a 52-week open-label trial in 85 pediatric patients with HeFH.
The safety and effectiveness of pitavastatin tablets have not been established in pediatric patients younger than 8 years of age with HeFH or in pediatric patients with other types of hyperlipidemia (other than HeFH).
8.5 Geriatric Use
In controlled clinical studies, 1,209 (43%) patients were 65 years and older. No overall differences in safety or effectiveness were observed between these patients and younger patients.
Advanced age (≥65 years) is a risk factor for pitavastatin-associated myopathy and rhabdomyolysis. Dose selection for a geriatric patient should be cautious, recognizing the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy and the higher risk of myopathy. Monitor geriatric patients receiving pitavastatin tablets for the increased risk of myopathy [see Warnings and Precautions (5.1)] .
4 Contraindications
Pitavastatin tablets is contraindicated in the following conditions:
- Concomitant use of cyclosporine [see Drug Interactions (7)] .
- Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3)] .
- Hypersensitivity to pitavastatin or any excipients in pitavastatin tablets. Hypersensitivity reactions including angioedema, rash, pruritus, and urticaria have been reported with pitavastatin tablets [see Adverse Reactions (6)] .
6 Adverse Reactions
The following serious adverse reactions are discussed in other sections of the labeling:
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1)]
- Immune-Mediated Necrotizing Myopathy [see Warning and Precautions (5.2)]
- Hepatic Dysfunction [see Warning and Precautions (5.3)]
- Increases in HbA1c and Fasting Serum Glucose Levels [see Warning and Precautions (5.4)] .
7 Drug Interactions
Table 2 includes a list of drugs that increase the risk of myopathy and rhabdomyolysis when administered concomitantly with pitavastatin and instructions for preventing or managing drug interactions [see Warnings and Precautions (5.1), Clinical Pharmacology (12.3)] .
| Cyclosporine | |
| Clinical Impact: | Cyclosporine significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . |
| Intervention: | Concomitant use of cyclosporine with pitavastatin is contraindicated [see Contraindications (4)] . |
| Gemfibrozil | |
| Clinical Impact: | Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of gemfibrozil with statins, including pitavastatin. |
| Intervention: | Avoid concomitant use of gemfibrozil with pitavastatin. |
| Erythromycin | |
| Clinical Impact: | Erythromycin significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . |
| Intervention: | In patients taking erythromycin, do not exceed pitavastatin tablets 1 mg once daily [see Dosage and Administration (2.4)] . |
| Rifampin | |
| Clinical Impact: | Rifampin significantly increases peak pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . |
| Intervention: | In patients taking rifampin, do not exceed pitavastatin tablets 2 mg once daily [see Dosage and Administration (2.4)] . |
| Fibrates | |
| Clinical Impact: | Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins, including pitavastatin. |
| Intervention: | Consider if the benefit of using fibrates concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. |
| Niacin | |
| Clinical Impact: | The risk of myopathy and rhabdomyolysis may be increased with concomitant use of lipid-modifying doses (≥1 g/day) of niacin with pitavastatin. |
| Intervention: | Consider if the benefit of using lipid-modifying doses (>1 g/day) of niacin concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. |
| Colchicine | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with statins, including pitavastatin. |
| Intervention: | Consider the risk/benefit of concomitant use of colchicine with pitavastatin. |
8.6 Renal Impairment
Renal impairment is a risk factor for myopathy and rhabdomyolysis. Monitor all patients with renal impairment for development of myopathy. Due to the risk of myopathy, a dosage modification of pitavastatin tablets is recommended for patients with moderate and severe renal impairment (estimated glomerular filtration rate 30 to 59 mL/min/1.73 m 2and 15 to 29 mL/min/1.73 m 2, respectively), as well as end-stage renal disease receiving hemodialysis [see Dosage and Administration (2.3), Warnings and Precautions (5.1), Clinical Pharmacology (12.3)] .
8.7 Hepatic Impairment
Pitavastatin tablets are contraindicated in patients with active liver failure or decompensated cirrhosis [see Contraindications (4), Warnings and Precautions (5.3)] .
1 Indications and Usage
Pitavastatin tablets are indicated as an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in:
- Adults with primary hyperlipidemia.
- Adults and pediatric patients aged 8 years and older with heterozygous familial hypercholesterolemia (HeFH).
5.3 Hepatic Dysfunction
Increases in serum transaminases have been reported with pitavastatin tablets [see Adverse Reactions (6)] . In most cases, these changes appeared soon after initiation, were transient, were not accompanied by symptoms, and resolved or improved on continued therapy or after a brief interruption in therapy. There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking statins, including pitavastatin.
Patients who consume substantial quantities of alcohol and/or have a history of liver disease may be at increased risk for hepatic injury.
Consider liver enzyme testing before the initiation of pitavastatin tablets and when clinically indicated thereafter. Pitavastatin tablets is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)] . If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue pitavastatin tablets.
12.1 Mechanism of Action
Pitavastatin is an inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase, the enzyme that catalyzes the conversion of HMG-CoA to mevalonate, a rate-limiting step in the biosynthetic pathway for cholesterol. As a result, the expression of LDL-receptors followed by the uptake of LDL from blood to liver is accelerated and then the plasma TC decreases. Sustained inhibition of cholesterol synthesis in the liver also decreases levels of very low-density lipoproteins.
5 Warnings and Precautions
- Myopathy and Rhabdomyolysis:Risk factors include age 65 or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher pitavastatin tablets dosage. Discontinue pitavastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue pitavastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the pitavastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever. ( 5.1, 7, 8.5, 8.6)
- Immune-Mediated Necrotizing Myopathy (IMNM):Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue pitavastatin if IMNM is suspected. ( 5.2)
- Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue pitavastatin tablets. ( 5.3)
2 Dosage and Administration
- Take orally once daily with or without food at the same time each day. ( 2.1)
- For patients requiring a high-intensity statin or are unable to achieve their LDL-C goal receiving pitavastatin tablets 4 mg daily, prescribe alternative LDL-C-lowering treatment. ( 2.1)
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiation of pitavastatin tablets and adjust the dosage if necessary. ( 2.1)
- Recommended dosage is 2 mg to 4 mg once daily. Maximum recommended dosage is 4 mg once daily ( 2.2)
- Recommended starting dosage for patients with moderate and severe renal impairment and end-stage renal disease on hemodialysis is 1 mg once daily. Maximum recommended dosage is 2 mg once daily. ( 2.3)
- See full prescribing information for pitavastatin tablets dosage modifications due to drug interactions. ( 2.4)
3 Dosage Forms and Strengths
Pitavastatin tablets are supplied as:
1 mg: Round, white to off-white, film-coated tablet, debossed with "P1" on one side.
2 mg: Round, white to off-white, film-coated tablet, debossed with "P2" on one side.
4 mg: Round, white to off-white, film-coated tablet, debossed with "P4" on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of pitavastatin tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders:abdominal discomfort, abdominal pain, dyspepsia, nausea
General disorders:asthenia, fatigue, malaise, dizziness
Hepatobiliary disorders:hepatitis, jaundice, fatal and non-fatal hepatic failure
Immune system disorders:angioedema, immune-mediated necrotizing myopathy associated with statin use
Metabolism and nutrition disorders:increases in HbA1c, fasting serum glucose levels
Musculoskeletal and connective tissue disorders:muscle spasms, myopathy, rhabdomyolysis
Nervous system disorders:hypoesthesia, peripheral neuropathy. There have been rare reports of new onset or exacerbation of myasthenia gravis, including ocular myasthenia, and reports of recurrence when the same or a different statin was administered. Rare reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. Cognitive impairment was generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks).
Psychiatric disorders:insomnia, depression.
Reproductive system and breast disorders:erectile dysfunction
Respiratory, thoracic and mediastinal disorders:interstitial lung disease
Skin and subcutaneous tissue disorders:lichen planus
8 Use in Specific Populations
5.1 Myopathy and Rhabdomyolysis
Pitavastatin tablets may cause myopathy (muscle pain, tenderness, or weakness associated with elevated creatine kinase [CK]) and rhabdomyolysis. Acute kidney injury secondary to myoglobinuria and rare fatalities have occurred as a result of rhabdomyolysis in patients treated with statins, including pitavastatin tablets.
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of one drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
16 How Supplied/storage and Handling
Pitavastatin tablets, for oral administration are available as:
1 mg: Round, white to off-white, film-coated tablet, debossed with "P1" on one side. They are supplied as:
- Bottles of 90 with a child-resistant closure, NDC 51407-956-90
2 mg: Round, white to off-white, film-coated tablet, debossed with "P2" on one side. They are supplied as:
- Bottles of 90 with a child-resistant closure, NDC 51407-957-90
4 mg: Round, white to off-white, film-coated tablet, debossed with "P4" on one side. They are supplied as:
- Bottles of 90 with a child-resistant closure, NDC 51407-958-90
5.2 Immune Mediated Necrotizing Myopathy
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use, including reports of recurrence when the same or a different statin was administered. IMNM is characterized by proximal muscle weakness and elevated serum creatine kinase that persist despite discontinuation of statin treatment; positive anti-HMG CoA reductase antibody; muscle biopsy showing necrotizing myopathy; and improvement with immunosuppressive agents. Additional neuromuscular and serologic testing may be necessary. Treatment with immunosuppressive agents may be required. Discontinue pitavastatin tablets if IMNM is suspected.
Principal Display Panel 1 Mg Tablet Bottle Label
NDC 51407-956-90
Pitavastatin
Tablets
1 mg
90 Tablets
Rx only
Principal Display Panel 2 Mg Tablet Bottle Label
NDC 51407-957-90
Pitavastatin
Tablets
2 mg
90 Tablets
Rx only
Principal Display Panel 4 Mg Tablet Bottle Label
NDC 51407-958-90
Pitavastatin
Tablets
4 mg
90 Tablets
Rx only
2.1 Important Dosage and Administration Information
- Take pitavastatin tablets orally once daily with or without food at the same time each day.
- For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving pitavastatin tablets 4 mg daily, prescribe alternative LDL-C-lowering treatment.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating pitavastatin tablets, and adjust the dosage if necessary.
5.4 Increases in Hba1c and Fasting Serum Glucose Levels
Increases in HbA1c and fasting serum glucose levels have been reported with statins, including pitavastatin tablets. Optimize lifestyle measures, including regular exercise, maintaining a healthy body weight, and making healthy food choices.
2.3 Recommended Dosage in Patients With Renal Impairment
- The recommended starting dosage for patients with moderate and severe renal impairment (estimated glomerular filtration rate 30 to 59 mL/minute/1.73 m 2and 15 to 29 mL/minute/1.73 m 2, respectively) and patients with end-stage renal disease receiving hemodialysis is pitavastatin tablets 1 mg once daily. The maximum recommended dose for these patients is pitavastatin tablets 2 mg once daily [see Use in Specific Populations (8.5)] .
- There are no dosage adjustment recommendations for patients with mild renal impairment.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 92-week carcinogenicity study in mice given pitavastatin, at the maximum tolerated dose of 75 mg/kg/day with systemic maximum exposures (AUC) 26 times the clinical maximum exposure at 4 mg daily, there was an absence of drug-related tumors.
In a 92-week carcinogenicity study in rats given pitavastatin at 1, 5, 25 mg/kg/day by oral gavage there was a significant increase in the incidence of thyroid follicular cell tumors at 25 mg/kg/day, which represents 295 times human systemic exposures based on AUC at the 4 mg daily maximum human dose.
In a 26-week transgenic mouse (Tg rasH2) carcinogenicity study where animals were given pitavastatin at 30, 75, and 150 mg/kg/day by oral gavage, no clinically significant tumors were observed.
Pitavastatin was not mutagenic in the Ames test with Salmonella typhimuriumand Escherichia coliwith and without metabolic activation, the micronucleus test following a single administration in mice and multiple administrations in rats, the unscheduled DNA synthesis test in rats, and a Comet assay in mice. In the chromosomal aberration test, clastogenicity was observed at the highest doses tested, which also elicited high levels of cytotoxicity.
Pitavastatin had no adverse effects on male and female rat fertility at oral doses of 10 and 30 mg/kg/day, respectively, at systemic exposures 56- and 354-times clinical exposure at 4 mg daily based on AUC.
Pitavastatin treatment in rabbits resulted in mortality in males and females given 1 mg/kg/day (30-times clinical systemic exposure at 4 mg daily based on AUC) and higher during a fertility study. Although the cause of death was not determined, rabbits had gross signs of renal toxicity (kidneys whitened) indicative of possible ischemia. Lower doses (15-times human systemic exposure) did not show significant toxicity in adult males and females. However, decreased implantations, increased resorptions, and decreased viability of fetuses were observed.
2.4 Pitavastatin Tablets Dosage Adjustments Due to Drug Interactions
- In patients taking erythromycin, do not exceed pitavastatin tablets 1 mg once daily [see Drug Interactions (7)] .
- In patients taking rifampin, do not exceed pitavastatin tablets 2 mg once daily [see Drug Interactions (7)] .
2.2 Recommended Dosage for Adults and Pediatric Patients Aged 8 Years and Older
- The recommended dosage range of pitavastatin tablets is 2 mg to 4 mg daily.
- The maximum recommended dosage is pitavastatin tablets 4 mg once daily.
Structured Label Content
Section 42229-5 (42229-5)
Risk Factors for Myopathy
Risk factors for myopathy include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use of certain drugs (including other lipid-lowering therapies), and higher pitavastatin tablets dosage [see Dosage and Administration (2.2), Drug Interactions (7) and Use in Specific Populations (8.5, 8.6)] . Dosages of pitavastatin tablets greater than 4 mg once daily were associated with an increased risk for severe myopathy in premarketing clinical studies. The maximum recommended dose of pitavastatin tablets is 4 mg once daily.
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light.
Dispense contents in a tight, light-resistant container with a child-resistant closure.
10 Overdosage (10 OVERDOSAGE)
No specific treatment for pitavastatin tablets overdose is known. Contact Poison Control (1-800-222-1222) for latest recommendations. Hemodialysis is unlikely to be of benefit due to high protein binding ratio of pitavastatin tablets.
11 Description (11 DESCRIPTION)
Pitavastatin tablets for oral use is an HMG-CoA reductase inhibitor.
The chemical name for pitavastatin is (+)monocalcium bis{(3R, 5S, 6 E)-7-[2-cyclopropyl-4-(4-fluorophenyl)-3-quinolyl]-3,5- dihydroxy-6-heptenoate}. The structural formula is:
The empirical formula for pitavastatin is C 50H 46CaF 2N 2O 8and the molecular weight is 880.98. Pitavastatin is odorless and occurs as white to pale-yellow powder. It is freely soluble in pyridine, chloroform, dilute hydrochloric acid, and tetrahydrofuran, soluble in ethylene glycol, sparingly soluble in octanol, slightly soluble in methanol, very slightly soluble in water or ethanol, and practically insoluble in acetonitrile or diethyl ether. Pitavastatin is hygroscopic and slightly unstable in light.
Each film-coated pitavastatin tablet contains 1.045 mg, 2.09 mg, or 4.18 mg of pitavastatin calcium, which is equivalent to 1 mg, 2 mg, or 4 mg, respectively of free base and the following inactive ingredients: lactose monohydrate, magnesium carbonate, low-substituted hydroxypropyl cellulose, hypromellose and magnesium stearate. The film coating contains: hypromellose, polyethylene glycol, talc and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of pitavastatin tablets as an adjunctive therapy to diet to reduce elevated LDL-C in pediatric patients aged 8 years and older with HeFH have been established. Use of pitavastatin tablets for this indication is supported by a 12-week, double-blind, placebo-controlled trial in 82 pediatric patients 8 to 16 years of age with HeFH [see Clinical Studies (14)] and a 52-week open-label trial in 85 pediatric patients with HeFH.
The safety and effectiveness of pitavastatin tablets have not been established in pediatric patients younger than 8 years of age with HeFH or in pediatric patients with other types of hyperlipidemia (other than HeFH).
8.5 Geriatric Use
In controlled clinical studies, 1,209 (43%) patients were 65 years and older. No overall differences in safety or effectiveness were observed between these patients and younger patients.
Advanced age (≥65 years) is a risk factor for pitavastatin-associated myopathy and rhabdomyolysis. Dose selection for a geriatric patient should be cautious, recognizing the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy and the higher risk of myopathy. Monitor geriatric patients receiving pitavastatin tablets for the increased risk of myopathy [see Warnings and Precautions (5.1)] .
4 Contraindications (4 CONTRAINDICATIONS)
Pitavastatin tablets is contraindicated in the following conditions:
- Concomitant use of cyclosporine [see Drug Interactions (7)] .
- Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3)] .
- Hypersensitivity to pitavastatin or any excipients in pitavastatin tablets. Hypersensitivity reactions including angioedema, rash, pruritus, and urticaria have been reported with pitavastatin tablets [see Adverse Reactions (6)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in other sections of the labeling:
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1)]
- Immune-Mediated Necrotizing Myopathy [see Warning and Precautions (5.2)]
- Hepatic Dysfunction [see Warning and Precautions (5.3)]
- Increases in HbA1c and Fasting Serum Glucose Levels [see Warning and Precautions (5.4)] .
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 2 includes a list of drugs that increase the risk of myopathy and rhabdomyolysis when administered concomitantly with pitavastatin and instructions for preventing or managing drug interactions [see Warnings and Precautions (5.1), Clinical Pharmacology (12.3)] .
| Cyclosporine | |
| Clinical Impact: | Cyclosporine significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . |
| Intervention: | Concomitant use of cyclosporine with pitavastatin is contraindicated [see Contraindications (4)] . |
| Gemfibrozil | |
| Clinical Impact: | Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of gemfibrozil with statins, including pitavastatin. |
| Intervention: | Avoid concomitant use of gemfibrozil with pitavastatin. |
| Erythromycin | |
| Clinical Impact: | Erythromycin significantly increases pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . |
| Intervention: | In patients taking erythromycin, do not exceed pitavastatin tablets 1 mg once daily [see Dosage and Administration (2.4)] . |
| Rifampin | |
| Clinical Impact: | Rifampin significantly increases peak pitavastatin exposure and increases the risk of myopathy and rhabdomyolysis . |
| Intervention: | In patients taking rifampin, do not exceed pitavastatin tablets 2 mg once daily [see Dosage and Administration (2.4)] . |
| Fibrates | |
| Clinical Impact: | Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins, including pitavastatin. |
| Intervention: | Consider if the benefit of using fibrates concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. |
| Niacin | |
| Clinical Impact: | The risk of myopathy and rhabdomyolysis may be increased with concomitant use of lipid-modifying doses (≥1 g/day) of niacin with pitavastatin. |
| Intervention: | Consider if the benefit of using lipid-modifying doses (>1 g/day) of niacin concomitantly with pitavastatin outweighs the increased risk of myopathy and rhabdomyolysis. |
| Colchicine | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with statins, including pitavastatin. |
| Intervention: | Consider the risk/benefit of concomitant use of colchicine with pitavastatin. |
8.6 Renal Impairment
Renal impairment is a risk factor for myopathy and rhabdomyolysis. Monitor all patients with renal impairment for development of myopathy. Due to the risk of myopathy, a dosage modification of pitavastatin tablets is recommended for patients with moderate and severe renal impairment (estimated glomerular filtration rate 30 to 59 mL/min/1.73 m 2and 15 to 29 mL/min/1.73 m 2, respectively), as well as end-stage renal disease receiving hemodialysis [see Dosage and Administration (2.3), Warnings and Precautions (5.1), Clinical Pharmacology (12.3)] .
8.7 Hepatic Impairment
Pitavastatin tablets are contraindicated in patients with active liver failure or decompensated cirrhosis [see Contraindications (4), Warnings and Precautions (5.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Pitavastatin tablets are indicated as an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in:
- Adults with primary hyperlipidemia.
- Adults and pediatric patients aged 8 years and older with heterozygous familial hypercholesterolemia (HeFH).
5.3 Hepatic Dysfunction
Increases in serum transaminases have been reported with pitavastatin tablets [see Adverse Reactions (6)] . In most cases, these changes appeared soon after initiation, were transient, were not accompanied by symptoms, and resolved or improved on continued therapy or after a brief interruption in therapy. There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking statins, including pitavastatin.
Patients who consume substantial quantities of alcohol and/or have a history of liver disease may be at increased risk for hepatic injury.
Consider liver enzyme testing before the initiation of pitavastatin tablets and when clinically indicated thereafter. Pitavastatin tablets is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)] . If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue pitavastatin tablets.
12.1 Mechanism of Action
Pitavastatin is an inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase, the enzyme that catalyzes the conversion of HMG-CoA to mevalonate, a rate-limiting step in the biosynthetic pathway for cholesterol. As a result, the expression of LDL-receptors followed by the uptake of LDL from blood to liver is accelerated and then the plasma TC decreases. Sustained inhibition of cholesterol synthesis in the liver also decreases levels of very low-density lipoproteins.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Myopathy and Rhabdomyolysis:Risk factors include age 65 or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher pitavastatin tablets dosage. Discontinue pitavastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue pitavastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the pitavastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever. ( 5.1, 7, 8.5, 8.6)
- Immune-Mediated Necrotizing Myopathy (IMNM):Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue pitavastatin if IMNM is suspected. ( 5.2)
- Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue pitavastatin tablets. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Take orally once daily with or without food at the same time each day. ( 2.1)
- For patients requiring a high-intensity statin or are unable to achieve their LDL-C goal receiving pitavastatin tablets 4 mg daily, prescribe alternative LDL-C-lowering treatment. ( 2.1)
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiation of pitavastatin tablets and adjust the dosage if necessary. ( 2.1)
- Recommended dosage is 2 mg to 4 mg once daily. Maximum recommended dosage is 4 mg once daily ( 2.2)
- Recommended starting dosage for patients with moderate and severe renal impairment and end-stage renal disease on hemodialysis is 1 mg once daily. Maximum recommended dosage is 2 mg once daily. ( 2.3)
- See full prescribing information for pitavastatin tablets dosage modifications due to drug interactions. ( 2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Pitavastatin tablets are supplied as:
1 mg: Round, white to off-white, film-coated tablet, debossed with "P1" on one side.
2 mg: Round, white to off-white, film-coated tablet, debossed with "P2" on one side.
4 mg: Round, white to off-white, film-coated tablet, debossed with "P4" on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of pitavastatin tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders:abdominal discomfort, abdominal pain, dyspepsia, nausea
General disorders:asthenia, fatigue, malaise, dizziness
Hepatobiliary disorders:hepatitis, jaundice, fatal and non-fatal hepatic failure
Immune system disorders:angioedema, immune-mediated necrotizing myopathy associated with statin use
Metabolism and nutrition disorders:increases in HbA1c, fasting serum glucose levels
Musculoskeletal and connective tissue disorders:muscle spasms, myopathy, rhabdomyolysis
Nervous system disorders:hypoesthesia, peripheral neuropathy. There have been rare reports of new onset or exacerbation of myasthenia gravis, including ocular myasthenia, and reports of recurrence when the same or a different statin was administered. Rare reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. Cognitive impairment was generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks).
Psychiatric disorders:insomnia, depression.
Reproductive system and breast disorders:erectile dysfunction
Respiratory, thoracic and mediastinal disorders:interstitial lung disease
Skin and subcutaneous tissue disorders:lichen planus
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.1 Myopathy and Rhabdomyolysis
Pitavastatin tablets may cause myopathy (muscle pain, tenderness, or weakness associated with elevated creatine kinase [CK]) and rhabdomyolysis. Acute kidney injury secondary to myoglobinuria and rare fatalities have occurred as a result of rhabdomyolysis in patients treated with statins, including pitavastatin tablets.
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of one drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Pitavastatin tablets, for oral administration are available as:
1 mg: Round, white to off-white, film-coated tablet, debossed with "P1" on one side. They are supplied as:
- Bottles of 90 with a child-resistant closure, NDC 51407-956-90
2 mg: Round, white to off-white, film-coated tablet, debossed with "P2" on one side. They are supplied as:
- Bottles of 90 with a child-resistant closure, NDC 51407-957-90
4 mg: Round, white to off-white, film-coated tablet, debossed with "P4" on one side. They are supplied as:
- Bottles of 90 with a child-resistant closure, NDC 51407-958-90
5.2 Immune Mediated Necrotizing Myopathy (5.2 Immune-Mediated Necrotizing Myopathy)
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use, including reports of recurrence when the same or a different statin was administered. IMNM is characterized by proximal muscle weakness and elevated serum creatine kinase that persist despite discontinuation of statin treatment; positive anti-HMG CoA reductase antibody; muscle biopsy showing necrotizing myopathy; and improvement with immunosuppressive agents. Additional neuromuscular and serologic testing may be necessary. Treatment with immunosuppressive agents may be required. Discontinue pitavastatin tablets if IMNM is suspected.
Principal Display Panel 1 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label)
NDC 51407-956-90
Pitavastatin
Tablets
1 mg
90 Tablets
Rx only
Principal Display Panel 2 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 2 mg Tablet Bottle Label)
NDC 51407-957-90
Pitavastatin
Tablets
2 mg
90 Tablets
Rx only
Principal Display Panel 4 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 4 mg Tablet Bottle Label)
NDC 51407-958-90
Pitavastatin
Tablets
4 mg
90 Tablets
Rx only
2.1 Important Dosage and Administration Information
- Take pitavastatin tablets orally once daily with or without food at the same time each day.
- For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving pitavastatin tablets 4 mg daily, prescribe alternative LDL-C-lowering treatment.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating pitavastatin tablets, and adjust the dosage if necessary.
5.4 Increases in Hba1c and Fasting Serum Glucose Levels (5.4 Increases in HbA1c and Fasting Serum Glucose Levels)
Increases in HbA1c and fasting serum glucose levels have been reported with statins, including pitavastatin tablets. Optimize lifestyle measures, including regular exercise, maintaining a healthy body weight, and making healthy food choices.
2.3 Recommended Dosage in Patients With Renal Impairment (2.3 Recommended Dosage in Patients with Renal Impairment)
- The recommended starting dosage for patients with moderate and severe renal impairment (estimated glomerular filtration rate 30 to 59 mL/minute/1.73 m 2and 15 to 29 mL/minute/1.73 m 2, respectively) and patients with end-stage renal disease receiving hemodialysis is pitavastatin tablets 1 mg once daily. The maximum recommended dose for these patients is pitavastatin tablets 2 mg once daily [see Use in Specific Populations (8.5)] .
- There are no dosage adjustment recommendations for patients with mild renal impairment.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 92-week carcinogenicity study in mice given pitavastatin, at the maximum tolerated dose of 75 mg/kg/day with systemic maximum exposures (AUC) 26 times the clinical maximum exposure at 4 mg daily, there was an absence of drug-related tumors.
In a 92-week carcinogenicity study in rats given pitavastatin at 1, 5, 25 mg/kg/day by oral gavage there was a significant increase in the incidence of thyroid follicular cell tumors at 25 mg/kg/day, which represents 295 times human systemic exposures based on AUC at the 4 mg daily maximum human dose.
In a 26-week transgenic mouse (Tg rasH2) carcinogenicity study where animals were given pitavastatin at 30, 75, and 150 mg/kg/day by oral gavage, no clinically significant tumors were observed.
Pitavastatin was not mutagenic in the Ames test with Salmonella typhimuriumand Escherichia coliwith and without metabolic activation, the micronucleus test following a single administration in mice and multiple administrations in rats, the unscheduled DNA synthesis test in rats, and a Comet assay in mice. In the chromosomal aberration test, clastogenicity was observed at the highest doses tested, which also elicited high levels of cytotoxicity.
Pitavastatin had no adverse effects on male and female rat fertility at oral doses of 10 and 30 mg/kg/day, respectively, at systemic exposures 56- and 354-times clinical exposure at 4 mg daily based on AUC.
Pitavastatin treatment in rabbits resulted in mortality in males and females given 1 mg/kg/day (30-times clinical systemic exposure at 4 mg daily based on AUC) and higher during a fertility study. Although the cause of death was not determined, rabbits had gross signs of renal toxicity (kidneys whitened) indicative of possible ischemia. Lower doses (15-times human systemic exposure) did not show significant toxicity in adult males and females. However, decreased implantations, increased resorptions, and decreased viability of fetuses were observed.
2.4 Pitavastatin Tablets Dosage Adjustments Due to Drug Interactions
- In patients taking erythromycin, do not exceed pitavastatin tablets 1 mg once daily [see Drug Interactions (7)] .
- In patients taking rifampin, do not exceed pitavastatin tablets 2 mg once daily [see Drug Interactions (7)] .
2.2 Recommended Dosage for Adults and Pediatric Patients Aged 8 Years and Older
- The recommended dosage range of pitavastatin tablets is 2 mg to 4 mg daily.
- The maximum recommended dosage is pitavastatin tablets 4 mg once daily.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:34.044944 · Updated: 2026-03-14T22:22:43.646413