These Highlights Do Not Include All The Information Needed To Use Neffy ®
2d4063e8-f56c-6902-e063-6294a90a0d40
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Neffy is indicated for emergency treatment of type I allergic reactions, including anaphylaxis, in adult and pediatric patients who weigh 30 kg or greater.
Indications and Usage
Neffy is indicated for emergency treatment of type I allergic reactions, including anaphylaxis, in adult and pediatric patients who weigh 30 kg or greater.
Dosage and Administration
Recommended Dosage : one spray of neffy (2 mg of epinephrine) administered into one nostril. In absence of clinical improvement or if symptoms worsen after initial treatment, administer a second dose of neffy in the same nostril with a new nasal spray starting 5 minutes after the first dose. ( 2.1 ) Neffy is for nasal use only. ( 2.2 ) Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required. ( 2.1 ) It is recommended that patients are prescribed and have immediate access to two neffy nasal sprays at all times. See full prescribing information for administration instructions. ( 2.2 )
Warnings and Precautions
Absorption of neffy may be affected by underlying structural and anatomical nasal conditions. ( 5.1 ) Administer with caution in patients with heart disease; may aggravate angina pectoris or produce ventricular arrhythmias. ( 5.2 ) May aggravate certain coexisting conditions. ( 5.2 ) The presence of a sulfite in this product should not deter use. ( 5.3 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Risks Associated with Use of Epinephrine in Certain Coexisting Conditions [see Warnings and Precautions (5.2) ] Allergic Reactions Associated with Sulfite [see Warnings and Precautions (5.3) ]
Drug Interactions
Neffy may alter nasal mucosa for up to 2 weeks after administration and increase systemic absorption of nasal products, including neffy. ( 7.1 ) Cardiac glycosides, diuretics or anti-arrhythmics: observe for development of cardiac arrhythmias. ( 7.2 ) Tricyclic antidepressants, monoamine oxidase inhibitors, levothyroxine sodium, certain antihistamines, and catechol-O-methyl transferase inhibitors may potentiate effects of epinephrine. ( 7.3 ) Beta-adrenergic blocking drugs antagonize cardiostimulating and bronchodilating effects of epinephrine. ( 7.4 ) Alpha-adrenergic blocking drugs antagonize vasoconstricting and hypertensive effects of epinephrine. ( 7.4 ) Ergot alkaloids may reverse the pressor effects of epinephrine. ( 7.4 )
Medication Information
Warnings and Precautions
Absorption of neffy may be affected by underlying structural and anatomical nasal conditions. ( 5.1 ) Administer with caution in patients with heart disease; may aggravate angina pectoris or produce ventricular arrhythmias. ( 5.2 ) May aggravate certain coexisting conditions. ( 5.2 ) The presence of a sulfite in this product should not deter use. ( 5.3 )
Indications and Usage
Neffy is indicated for emergency treatment of type I allergic reactions, including anaphylaxis, in adult and pediatric patients who weigh 30 kg or greater.
Dosage and Administration
Recommended Dosage : one spray of neffy (2 mg of epinephrine) administered into one nostril. In absence of clinical improvement or if symptoms worsen after initial treatment, administer a second dose of neffy in the same nostril with a new nasal spray starting 5 minutes after the first dose. ( 2.1 ) Neffy is for nasal use only. ( 2.2 ) Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required. ( 2.1 ) It is recommended that patients are prescribed and have immediate access to two neffy nasal sprays at all times. See full prescribing information for administration instructions. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Risks Associated with Use of Epinephrine in Certain Coexisting Conditions [see Warnings and Precautions (5.2) ] Allergic Reactions Associated with Sulfite [see Warnings and Precautions (5.3) ]
Drug Interactions
Neffy may alter nasal mucosa for up to 2 weeks after administration and increase systemic absorption of nasal products, including neffy. ( 7.1 ) Cardiac glycosides, diuretics or anti-arrhythmics: observe for development of cardiac arrhythmias. ( 7.2 ) Tricyclic antidepressants, monoamine oxidase inhibitors, levothyroxine sodium, certain antihistamines, and catechol-O-methyl transferase inhibitors may potentiate effects of epinephrine. ( 7.3 ) Beta-adrenergic blocking drugs antagonize cardiostimulating and bronchodilating effects of epinephrine. ( 7.4 ) Alpha-adrenergic blocking drugs antagonize vasoconstricting and hypertensive effects of epinephrine. ( 7.4 ) Ergot alkaloids may reverse the pressor effects of epinephrine. ( 7.4 )
Description
Neffy is indicated for emergency treatment of type I allergic reactions, including anaphylaxis, in adult and pediatric patients who weigh 30 kg or greater.
Section 42229-5
Adverse Reactions in Four Clinical Pharmacology Studies with Neffy for Adult Subjects
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of neffy 2 mg is based on four clinical pharmacology studies in 175 healthy adults and adults with type I allergy without anaphylaxis, who did not have structural or anatomical nasal conditions [see Clinical Pharmacology (12.2, 12.3)] . The four clinical pharmacology studies were designed to compare the pharmacokinetic (PK) and pharmacodynamic (PD) profiles of one dose of neffy 2 mg sprayed into one nostril or two doses of neffy 2 mg sprayed into either the same or opposite nostril, administered 10 minutes apart, with PK and PD profiles of one or two dose(s) of epinephrine injection administered intramuscularly. The common adverse reactions that occurred with neffy 2 mg after one and two dose(s) are listed in Table 1.
| Adverse Reaction
Data include subjects with nasal allergen challenge induced rhinitis
|
Neffy 2 mg
One Dose |
Neffy 2 mg
Two Doses Two nasal doses of neffy 2 mg were administered 10 minutes apart
|
||
|---|---|---|---|---|
| N = 134
The trials used a crossover design and therefore the total number of subjects do not match the number of unique subjects (n = 175)
|
N = 85 | |||
| Throat irritation | 2 | (2%) | 16 | (19%) |
| Headache | 8 | (6%) | 15 | (18%) |
| Nasal discomfort | 13 | (10%) | 11 | (13%) |
| Feeling jittery | 1 | (1%) | 9 | (11%) |
| Tremor | 0 | (0%) | 7 | (8%) |
| Rhinorrhea | 4 | (3%) | 6 | (7%) |
| Nasal pruritus | 0 | (0%) | 3 | (4%) |
| Sneezing | 0 | (0%) | 3 | (4%) |
| Abdominal pain | 1 | (1%) | 3 | (4%) |
| Gingival pain | 0 | (0%) | 3 | (4%) |
| Hypoesthesia oral | 0 | (0%) | 3 | (4%) |
| Nasal Congestion | 0 | (0%) | 2 | (2%) |
| Dizziness | 4 | (3%) | 2 | (2%) |
| Nausea | 4 | (3%) | 2 | (2%) |
| Vomiting | 3 | (2%) | 2 | (2%) |
Section 42230-3
| PATIENT INFORMATION
NEFFY ®(ne'fee) (epinephrine nasal spray) For allergic emergencies (anaphylaxis) |
||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 08/2024 | |||
| Read this Patient Information leaflet carefully before you start using neffy and each time you get a refill. There may be new information. You, your caregiver, or others who may be in a position to administer neffy should know how to use it before you have an allergic emergency. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | ||||
|
||||
|
|
|||
|
||||
What is neffy?
|
||||
Before using neffy, tell your healthcare provider about all your medical conditions, especially if you:
Especially tell your healthcare provider if you take or use:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. Use neffy for the treatment of anaphylaxis as prescribed by your healthcare provider, regardless of your medical conditions or medicine you take. |
||||
How should I use neffy?
|
||||
|
What are the possible side effects of neffy?
Neffy may cause serious side effects. If you have certain medical conditions, or take certain medicines, your condition may get worse or you may have more or longer lasting side effects when you use neffy.Talk to your healthcare provider about all your medical conditions. Common side effects of neffy include: |
||||
|
|
|||
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of neffy .For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store neffy?
|
||||
|
General information about the safe and effective use of neffy.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use neffy for a condition for which it was not prescribed. Do not give neffy to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about neffy that is written for health professionals. |
||||
|
What are the ingredients in neffy?
Active Ingredient: epinephrine Inactive Ingredients: benzalkonium chloride, disodium edetate, n-dodecyl beta-D-maltoside, sodium chloride, sodium metabisulfite, and hydrochloric acid or sodium hydroxide to adjust pH, in water for injection. |
Section 44425-7
Storage and Handling
Store at 68°F to 77°F (20°C to 25°C). Excursions permitted up to 122°F (50°C).
Do not freeze. Neffy freezes below 5°F (-15°C). If neffy freezes, it will not deliver epinephrine.
10 Overdosage
Overdosage of epinephrine has been reported to produce extremely elevated arterial pressure, which may result in cerebrovascular hemorrhage, particularly in elderly patients. Overdosage may also result in pulmonary edema because of peripheral vascular constriction together with cardiac stimulation. Epinephrine overdosage can also cause transient bradycardia followed by tachycardia which may be accompanied by fatal cardiac arrhythmias; premature ventricular contractions followed by multifocal ventricular tachycardia; atrial tachycardia and occasionally by atrioventricular block; extreme pallor and coldness of the skin; metabolic acidosis; kidney failure.
Epinephrine is rapidly inactivated in the body and treatment following overdosage with epinephrine is primarily supportive. Treatment of epinephrine associated pulmonary edema consists of a rapidly acting alpha-adrenergic blocking drug (such as phentolamine mesylate) and respiratory support. Treatment of epinephrine associated arrhythmias consists of administration of a beta-adrenergic blocking drug (such as propranolol). If necessary, pressor effects may be counteracted by rapidly acting vasodilators or α-adrenergic blocking drugs. If prolonged hypotension follows such measures, it may be necessary to administer another pressor drug.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
11 Description
Neffy contains epinephrine, a sympathomimetic catecholamine. Chemically, epinephrine is (-)-3,4-dihydroxy-α-[(methylamino)methyl] benzyl alcohol with molecular weight of 183.21 g/mol and the following structure:
Neffy (epinephrine nasal spray) is supplied as a single-dose nasal spray containing 2 mg of epinephrine in 0.1 mL solution for nasal administration.
Inactive ingredients include benzalkonium chloride, disodium edetate, n-dodecyl beta-D-maltoside, sodium chloride, sodium metabisulfite, and hydrochloric acid or sodium hydroxide to adjust pH, in water for injection. The pH range is approximately 3 to 5.5.
8.4 Pediatric Use
The safety and effectiveness of neffy for emergency treatment of type I allergic reactions, including anaphylaxis, have been established in pediatric patients who weigh 30 kg or greater. Use of neffy for this indication is supported by clinical pharmacology studies in adults that compared the PK/PD profile of neffy to epinephrine injection products with established safety and effectiveness for this indication, and clinical pharmacology data with neffy in pediatric patients who weigh 30 kg or greater [see Adverse Reactions (6)and Clinical Pharmacology (12.2, 12.3)].
The safety and effectiveness of neffy have not been established in pediatric patients who weigh less than 30 kg.
8.5 Geriatric Use
Clinical pharmacology studies of neffy for the emergency treatment of type I allergic reactions, including anaphylaxis, did not include a sufficient number of subjects aged 65 and over to determine whether they respond differently from younger adult subjects. However, other reported clinical experience with use of epinephrine for the treatment of anaphylaxis has identified that geriatric patients may be particularly sensitive to the effects of epinephrine. Neffy should be administered with caution in elderly patients who are at greater risk for developing adverse reactions after epinephrine administration.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks Associated with Use of Epinephrine in Certain Coexisting Conditions [see Warnings and Precautions (5.2)]
- Allergic Reactions Associated with Sulfite [see Warnings and Precautions (5.3)]
7 Drug Interactions
- Neffy may alter nasal mucosa for up to 2 weeks after administration and increase systemic absorption of nasal products, including neffy. ( 7.1)
- Cardiac glycosides, diuretics or anti-arrhythmics: observe for development of cardiac arrhythmias. ( 7.2)
- Tricyclic antidepressants, monoamine oxidase inhibitors, levothyroxine sodium, certain antihistamines, and catechol-O-methyl transferase inhibitors may potentiate effects of epinephrine. ( 7.3)
- Beta-adrenergic blocking drugs antagonize cardiostimulating and bronchodilating effects of epinephrine. ( 7.4)
- Alpha-adrenergic blocking drugs antagonize vasoconstricting and hypertensive effects of epinephrine. ( 7.4)
- Ergot alkaloids may reverse the pressor effects of epinephrine. ( 7.4)
12.2 Pharmacodynamics
Four clinical pharmacology studies of neffy in adults and one clinical pharmacology study in pediatric subjects who weigh 30 kg or greater are described below. All doses were administered by study staff unless otherwise stated.
12.3 Pharmacokinetics
Pharmacokinetics assessments were performed in the clinical pharmacology studies described in the Pharmacodynamicssubsection [see Clinical Pharmacology (12.2)] .
2.1 Recommended Dosage
The recommended dosage of neffy is one spray (2 mg of epinephrine) administered into one nostril. In the absence of clinical improvement or if symptoms worsen after the initial treatment, a second dose of neffy may be administered in the same nostril with a second nasal spray starting 5 minutes after the first dose.
- Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required.
- It is recommended that patients are prescribed and have immediate access to two neffy nasal sprays at all times.
1 Indications and Usage
Neffy is indicated for emergency treatment of type I allergic reactions, including anaphylaxis, in adult and pediatric patients who weigh 30 kg or greater.
12.1 Mechanism of Action
Epinephrine acts on both alpha and beta-adrenergic receptors.
Through its action on alpha-adrenergic receptors, epinephrine lessens the vasodilation and increased vascular permeability that occurs during anaphylaxis, which can lead to loss of intravascular fluid volume and hypotension.
Through its action on beta-adrenergic receptors, epinephrine causes bronchial smooth muscle relaxation and helps alleviate bronchospasm, wheezing and dyspnea that may occur during anaphylaxis.
Epinephrine alleviates pruritus, urticaria, and angioedema. It may also relieve gastrointestinal and genitourinary symptoms associated with anaphylaxis because of its relaxer effects on the smooth muscle of the stomach, intestine, uterus, and urinary bladder.
5 Warnings and Precautions
- Absorption of neffy may be affected by underlying structural and anatomical nasal conditions. ( 5.1)
- Administer with caution in patients with heart disease; may aggravate angina pectoris or produce ventricular arrhythmias. ( 5.2)
- May aggravate certain coexisting conditions. ( 5.2)
- The presence of a sulfite in this product should not deter use. ( 5.3)
2 Dosage and Administration
Recommended Dosage: one spray of neffy (2 mg of epinephrine) administered into one nostril. In absence of clinical improvement or if symptoms worsen after initial treatment, administer a second dose of neffy in the same nostril with a new nasal spray starting 5 minutes after the first dose. ( 2.1)
- Neffy is for nasal use only. ( 2.2)
- Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required. ( 2.1)
- It is recommended that patients are prescribed and have immediate access to two neffy nasal sprays at all times.
- See full prescribing information for administration instructions. ( 2.2)
3 Dosage Forms and Strengths
Nasal spray: 2 mg/0.1 mL of epinephrine per spray in a single-dose nasal spray.
8 Use in Specific Populations
Elderly patients may be at greater risk of developing adverse reactions. ( 8.5)
2.2 Administration Instructions
- Neffy is for nasal use only.
- Each neffy nasal spray is for single use and delivers the entire dose upon activation.
- Do not prime or attempt to reuse neffy for more than one administration.
- Use the right hand to administer neffy to the right nostril and use the left hand to administer neffy to the left nostril.
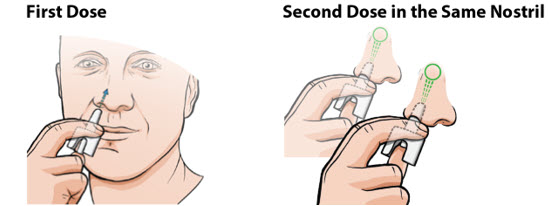
Administer neffy by inserting the nozzle of the nasal spray fully into one nostril until your fingers touch the nose (see Figure 1). Hold the nasal spray straight into the nose - do not angle the nasal spray to the inside septum or outer wall of the nose as some medication may be lost. Press the plunger firmly to activate. Avoid sniffing during and after administration. If a second dose of neffy is needed, administer a new nasal spray into the same nostril starting 5 minutes after the first dose. More than two sequential doses of epinephrine should be administered under direct medical supervision. Refer patients and caregivers to the Instructions for Usefor detailed administration instructions.
Figure 1: Administration of Neffy
If neffy is frozen and is needed in an emergency, do not wait to thaw, seek emergency medical care immediately [see How Supplied/Storage and Handling (16)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Principal Display Panel Outer Package
NDC 71872-7330-1
Rx Only
neffy
®
(epinephrine nasal spray)
2 mg
For Use in the Nose Only (Single-dose)
Read the Instructions for Use for complete
information on how to use neffy.
1 x single-dose nasal spray device
13.2 Animal Toxicology And/or Pharmacology
In a single-dose nasal toxicity study, treatment of neffy in rats induced epinephrine-related histopathology changes in the nose, such as minimal ulceration of the exposed mucosa (at ≥2.3-fold the recommended clinical dose of neffy 2 mg based on local surface area), and nasal passages, such as minimal to mild necrosis in the nasal turbinate and parietal wall in the rostral-most level (at ≥1.2-fold the recommended clinical dose of neffy 2 mg based on local surface area) on day 2. These findings were often associated with minimal to mild neutrophilic inflammation and were reversible after 14 days post-dose.
7.3 Drugs Potentiating Effects of Epinephrine
The effects of epinephrine may be potentiated by tricyclic antidepressants, monoamine oxidase inhibitors, levothyroxine sodium, certain antihistamines, notably chlorpheniramine, tripelennamine, and diphenhydramine, and catechol-O-methyl transferase (COMT) inhibitors such as entacapone.
7.4 Drugs Antagonizing Effects of Epinephrine
The cardiostimulating and bronchodilating effects of epinephrine are antagonized by beta-adrenergic blocking drugs, such as propranolol.
The vasoconstricting and hypertensive effects of epinephrine are antagonized by alpha-adrenergic blocking drugs, such as phentolamine.
Ergot alkaloids may also reverse the pressor effects of epinephrine.
5.3 Allergic Reactions Associated With Sulfite
Epinephrine is the preferred treatment for serious allergic or other emergency situations even though neffy contains sodium metabisulfite, a sulfite that may in other products cause allergic-type reactions including anaphylactic symptoms or life-threatening or less severe asthmatic episodes in certain susceptible persons. The alternatives to using epinephrine in a life-threatening situation may not be satisfactory. The presence of a sulfite(s) in neffy should not deter administration of the drug for treatment of serious allergic or other emergency situations.
7.2 Drugs Increasing Risk of Cardiac Arrhythmias
Patients who receive epinephrine while concomitantly taking cardiac glycosides, diuretics, or anti-arrhythmics should be observed carefully for the development of cardiac arrhythmias [see Warnings and Precautions (5.2)and Adverse Reactions (6)] .
7.1 Potential Increased Exposure of Nasal Spray Drugs
Neffy may alter nasal mucosa for up to 2 weeks after administration, and thus may increase systemic absorption of nasal products, including neffy, potentially increasing the risk of adverse reactions associated with these products.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies to evaluate the carcinogenic potential of epinephrine have not been conducted.
Epinephrine and other catecholamines have been shown to have mutagenic potential in vitro. Epinephrine was positive in the Salmonellabacterial reverse mutation assay, positive in the mouse lymphoma assay, and negative in the in vivomicronucleus assay. Epinephrine is an oxidative mutagen based on the E. coliWP2 Mutoxitest bacterial reverse mutation assay. This should not prevent the use of epinephrine where indicated [see Indications and Usage (1)] .
The potential for epinephrine to impair reproductive performance has not been evaluated, but epinephrine has been shown to decrease implantation in female rabbits dosed subcutaneously with 1.2 mg/kg/day (15-fold the highest human intramuscular or subcutaneous daily dose) during gestation days 3 to 9.
5.2 Risks Associated With Use of Epinephrine in Certain Coexisting Conditions
Some patients may be at greater risk for developing adverse reactions after epinephrine administration. Despite these concerns, it should be recognized that the presence of these conditions is not a contraindication to epinephrine administration in an acute, life-threatening situation. Therefore, patients with these conditions, and/or any other person who might be in a position to administer neffy to a patient experiencing anaphylaxis should be carefully instructed in regard to the circumstances under which epinephrine should be used.
Epinephrine should be administered with caution to patients who have heart disease, including patients with cardiac arrhythmias, coronary artery disease, or hypertension. In such patients, or in patients who are on drugs that may sensitize the heart to arrhythmias, epinephrine may precipitate or aggravate angina pectoris, as well as produce ventricular arrhythmias [see Drug Interactions (7)and Adverse Reactions (6)] .
Epinephrine can temporarily exacerbate the underlying condition or increase symptoms in patients with the following: hyperthyroidism, Parkinson's disease, diabetes, renal impairment. Epinephrine should be administered with caution in patients with these conditions, including elderly patients and pregnant women.
5.1 Potential Altered Absorption of Neffy in Patients With Underlying Structural Or Anatomical Nasal Conditions
Clinical pharmacology studies with neffy included subjects with history of allergic rhinitis, but did not include subjects with underlying structural and anatomical nasal conditions (e.g., polyps, history of nasal fractures or injuries, or history of nasal surgery). Absorption of neffy may be affected by underlying structural and anatomical nasal conditions. Consider use of other epinephrine products given by other routes of administration for patients with underlying structural or anatomical nasal conditions.
Structured Label Content
Section 42229-5 (42229-5)
Adverse Reactions in Four Clinical Pharmacology Studies with Neffy for Adult Subjects
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of neffy 2 mg is based on four clinical pharmacology studies in 175 healthy adults and adults with type I allergy without anaphylaxis, who did not have structural or anatomical nasal conditions [see Clinical Pharmacology (12.2, 12.3)] . The four clinical pharmacology studies were designed to compare the pharmacokinetic (PK) and pharmacodynamic (PD) profiles of one dose of neffy 2 mg sprayed into one nostril or two doses of neffy 2 mg sprayed into either the same or opposite nostril, administered 10 minutes apart, with PK and PD profiles of one or two dose(s) of epinephrine injection administered intramuscularly. The common adverse reactions that occurred with neffy 2 mg after one and two dose(s) are listed in Table 1.
| Adverse Reaction
Data include subjects with nasal allergen challenge induced rhinitis
|
Neffy 2 mg
One Dose |
Neffy 2 mg
Two Doses Two nasal doses of neffy 2 mg were administered 10 minutes apart
|
||
|---|---|---|---|---|
| N = 134
The trials used a crossover design and therefore the total number of subjects do not match the number of unique subjects (n = 175)
|
N = 85 | |||
| Throat irritation | 2 | (2%) | 16 | (19%) |
| Headache | 8 | (6%) | 15 | (18%) |
| Nasal discomfort | 13 | (10%) | 11 | (13%) |
| Feeling jittery | 1 | (1%) | 9 | (11%) |
| Tremor | 0 | (0%) | 7 | (8%) |
| Rhinorrhea | 4 | (3%) | 6 | (7%) |
| Nasal pruritus | 0 | (0%) | 3 | (4%) |
| Sneezing | 0 | (0%) | 3 | (4%) |
| Abdominal pain | 1 | (1%) | 3 | (4%) |
| Gingival pain | 0 | (0%) | 3 | (4%) |
| Hypoesthesia oral | 0 | (0%) | 3 | (4%) |
| Nasal Congestion | 0 | (0%) | 2 | (2%) |
| Dizziness | 4 | (3%) | 2 | (2%) |
| Nausea | 4 | (3%) | 2 | (2%) |
| Vomiting | 3 | (2%) | 2 | (2%) |
Section 42230-3 (42230-3)
| PATIENT INFORMATION
NEFFY ®(ne'fee) (epinephrine nasal spray) For allergic emergencies (anaphylaxis) |
||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 08/2024 | |||
| Read this Patient Information leaflet carefully before you start using neffy and each time you get a refill. There may be new information. You, your caregiver, or others who may be in a position to administer neffy should know how to use it before you have an allergic emergency. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | ||||
|
||||
|
|
|||
|
||||
What is neffy?
|
||||
Before using neffy, tell your healthcare provider about all your medical conditions, especially if you:
Especially tell your healthcare provider if you take or use:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. Use neffy for the treatment of anaphylaxis as prescribed by your healthcare provider, regardless of your medical conditions or medicine you take. |
||||
How should I use neffy?
|
||||
|
What are the possible side effects of neffy?
Neffy may cause serious side effects. If you have certain medical conditions, or take certain medicines, your condition may get worse or you may have more or longer lasting side effects when you use neffy.Talk to your healthcare provider about all your medical conditions. Common side effects of neffy include: |
||||
|
|
|||
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of neffy .For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store neffy?
|
||||
|
General information about the safe and effective use of neffy.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use neffy for a condition for which it was not prescribed. Do not give neffy to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about neffy that is written for health professionals. |
||||
|
What are the ingredients in neffy?
Active Ingredient: epinephrine Inactive Ingredients: benzalkonium chloride, disodium edetate, n-dodecyl beta-D-maltoside, sodium chloride, sodium metabisulfite, and hydrochloric acid or sodium hydroxide to adjust pH, in water for injection. |
Section 44425-7 (44425-7)
Storage and Handling
Store at 68°F to 77°F (20°C to 25°C). Excursions permitted up to 122°F (50°C).
Do not freeze. Neffy freezes below 5°F (-15°C). If neffy freezes, it will not deliver epinephrine.
10 Overdosage (10 OVERDOSAGE)
Overdosage of epinephrine has been reported to produce extremely elevated arterial pressure, which may result in cerebrovascular hemorrhage, particularly in elderly patients. Overdosage may also result in pulmonary edema because of peripheral vascular constriction together with cardiac stimulation. Epinephrine overdosage can also cause transient bradycardia followed by tachycardia which may be accompanied by fatal cardiac arrhythmias; premature ventricular contractions followed by multifocal ventricular tachycardia; atrial tachycardia and occasionally by atrioventricular block; extreme pallor and coldness of the skin; metabolic acidosis; kidney failure.
Epinephrine is rapidly inactivated in the body and treatment following overdosage with epinephrine is primarily supportive. Treatment of epinephrine associated pulmonary edema consists of a rapidly acting alpha-adrenergic blocking drug (such as phentolamine mesylate) and respiratory support. Treatment of epinephrine associated arrhythmias consists of administration of a beta-adrenergic blocking drug (such as propranolol). If necessary, pressor effects may be counteracted by rapidly acting vasodilators or α-adrenergic blocking drugs. If prolonged hypotension follows such measures, it may be necessary to administer another pressor drug.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
11 Description (11 DESCRIPTION)
Neffy contains epinephrine, a sympathomimetic catecholamine. Chemically, epinephrine is (-)-3,4-dihydroxy-α-[(methylamino)methyl] benzyl alcohol with molecular weight of 183.21 g/mol and the following structure:
Neffy (epinephrine nasal spray) is supplied as a single-dose nasal spray containing 2 mg of epinephrine in 0.1 mL solution for nasal administration.
Inactive ingredients include benzalkonium chloride, disodium edetate, n-dodecyl beta-D-maltoside, sodium chloride, sodium metabisulfite, and hydrochloric acid or sodium hydroxide to adjust pH, in water for injection. The pH range is approximately 3 to 5.5.
8.4 Pediatric Use
The safety and effectiveness of neffy for emergency treatment of type I allergic reactions, including anaphylaxis, have been established in pediatric patients who weigh 30 kg or greater. Use of neffy for this indication is supported by clinical pharmacology studies in adults that compared the PK/PD profile of neffy to epinephrine injection products with established safety and effectiveness for this indication, and clinical pharmacology data with neffy in pediatric patients who weigh 30 kg or greater [see Adverse Reactions (6)and Clinical Pharmacology (12.2, 12.3)].
The safety and effectiveness of neffy have not been established in pediatric patients who weigh less than 30 kg.
8.5 Geriatric Use
Clinical pharmacology studies of neffy for the emergency treatment of type I allergic reactions, including anaphylaxis, did not include a sufficient number of subjects aged 65 and over to determine whether they respond differently from younger adult subjects. However, other reported clinical experience with use of epinephrine for the treatment of anaphylaxis has identified that geriatric patients may be particularly sensitive to the effects of epinephrine. Neffy should be administered with caution in elderly patients who are at greater risk for developing adverse reactions after epinephrine administration.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks Associated with Use of Epinephrine in Certain Coexisting Conditions [see Warnings and Precautions (5.2)]
- Allergic Reactions Associated with Sulfite [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Neffy may alter nasal mucosa for up to 2 weeks after administration and increase systemic absorption of nasal products, including neffy. ( 7.1)
- Cardiac glycosides, diuretics or anti-arrhythmics: observe for development of cardiac arrhythmias. ( 7.2)
- Tricyclic antidepressants, monoamine oxidase inhibitors, levothyroxine sodium, certain antihistamines, and catechol-O-methyl transferase inhibitors may potentiate effects of epinephrine. ( 7.3)
- Beta-adrenergic blocking drugs antagonize cardiostimulating and bronchodilating effects of epinephrine. ( 7.4)
- Alpha-adrenergic blocking drugs antagonize vasoconstricting and hypertensive effects of epinephrine. ( 7.4)
- Ergot alkaloids may reverse the pressor effects of epinephrine. ( 7.4)
12.2 Pharmacodynamics
Four clinical pharmacology studies of neffy in adults and one clinical pharmacology study in pediatric subjects who weigh 30 kg or greater are described below. All doses were administered by study staff unless otherwise stated.
12.3 Pharmacokinetics
Pharmacokinetics assessments were performed in the clinical pharmacology studies described in the Pharmacodynamicssubsection [see Clinical Pharmacology (12.2)] .
2.1 Recommended Dosage
The recommended dosage of neffy is one spray (2 mg of epinephrine) administered into one nostril. In the absence of clinical improvement or if symptoms worsen after the initial treatment, a second dose of neffy may be administered in the same nostril with a second nasal spray starting 5 minutes after the first dose.
- Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required.
- It is recommended that patients are prescribed and have immediate access to two neffy nasal sprays at all times.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Neffy is indicated for emergency treatment of type I allergic reactions, including anaphylaxis, in adult and pediatric patients who weigh 30 kg or greater.
12.1 Mechanism of Action
Epinephrine acts on both alpha and beta-adrenergic receptors.
Through its action on alpha-adrenergic receptors, epinephrine lessens the vasodilation and increased vascular permeability that occurs during anaphylaxis, which can lead to loss of intravascular fluid volume and hypotension.
Through its action on beta-adrenergic receptors, epinephrine causes bronchial smooth muscle relaxation and helps alleviate bronchospasm, wheezing and dyspnea that may occur during anaphylaxis.
Epinephrine alleviates pruritus, urticaria, and angioedema. It may also relieve gastrointestinal and genitourinary symptoms associated with anaphylaxis because of its relaxer effects on the smooth muscle of the stomach, intestine, uterus, and urinary bladder.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Absorption of neffy may be affected by underlying structural and anatomical nasal conditions. ( 5.1)
- Administer with caution in patients with heart disease; may aggravate angina pectoris or produce ventricular arrhythmias. ( 5.2)
- May aggravate certain coexisting conditions. ( 5.2)
- The presence of a sulfite in this product should not deter use. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Recommended Dosage: one spray of neffy (2 mg of epinephrine) administered into one nostril. In absence of clinical improvement or if symptoms worsen after initial treatment, administer a second dose of neffy in the same nostril with a new nasal spray starting 5 minutes after the first dose. ( 2.1)
- Neffy is for nasal use only. ( 2.2)
- Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required. ( 2.1)
- It is recommended that patients are prescribed and have immediate access to two neffy nasal sprays at all times.
- See full prescribing information for administration instructions. ( 2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Nasal spray: 2 mg/0.1 mL of epinephrine per spray in a single-dose nasal spray.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Elderly patients may be at greater risk of developing adverse reactions. ( 8.5)
2.2 Administration Instructions
- Neffy is for nasal use only.
- Each neffy nasal spray is for single use and delivers the entire dose upon activation.
- Do not prime or attempt to reuse neffy for more than one administration.
- Use the right hand to administer neffy to the right nostril and use the left hand to administer neffy to the left nostril.
Administer neffy by inserting the nozzle of the nasal spray fully into one nostril until your fingers touch the nose (see Figure 1). Hold the nasal spray straight into the nose - do not angle the nasal spray to the inside septum or outer wall of the nose as some medication may be lost. Press the plunger firmly to activate. Avoid sniffing during and after administration. If a second dose of neffy is needed, administer a new nasal spray into the same nostril starting 5 minutes after the first dose. More than two sequential doses of epinephrine should be administered under direct medical supervision. Refer patients and caregivers to the Instructions for Usefor detailed administration instructions.
Figure 1: Administration of Neffy
If neffy is frozen and is needed in an emergency, do not wait to thaw, seek emergency medical care immediately [see How Supplied/Storage and Handling (16)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Principal Display Panel Outer Package (PRINCIPAL DISPLAY PANEL - OUTER PACKAGE)
NDC 71872-7330-1
Rx Only
neffy
®
(epinephrine nasal spray)
2 mg
For Use in the Nose Only (Single-dose)
Read the Instructions for Use for complete
information on how to use neffy.
1 x single-dose nasal spray device
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In a single-dose nasal toxicity study, treatment of neffy in rats induced epinephrine-related histopathology changes in the nose, such as minimal ulceration of the exposed mucosa (at ≥2.3-fold the recommended clinical dose of neffy 2 mg based on local surface area), and nasal passages, such as minimal to mild necrosis in the nasal turbinate and parietal wall in the rostral-most level (at ≥1.2-fold the recommended clinical dose of neffy 2 mg based on local surface area) on day 2. These findings were often associated with minimal to mild neutrophilic inflammation and were reversible after 14 days post-dose.
7.3 Drugs Potentiating Effects of Epinephrine
The effects of epinephrine may be potentiated by tricyclic antidepressants, monoamine oxidase inhibitors, levothyroxine sodium, certain antihistamines, notably chlorpheniramine, tripelennamine, and diphenhydramine, and catechol-O-methyl transferase (COMT) inhibitors such as entacapone.
7.4 Drugs Antagonizing Effects of Epinephrine
The cardiostimulating and bronchodilating effects of epinephrine are antagonized by beta-adrenergic blocking drugs, such as propranolol.
The vasoconstricting and hypertensive effects of epinephrine are antagonized by alpha-adrenergic blocking drugs, such as phentolamine.
Ergot alkaloids may also reverse the pressor effects of epinephrine.
5.3 Allergic Reactions Associated With Sulfite (5.3 Allergic Reactions Associated with Sulfite)
Epinephrine is the preferred treatment for serious allergic or other emergency situations even though neffy contains sodium metabisulfite, a sulfite that may in other products cause allergic-type reactions including anaphylactic symptoms or life-threatening or less severe asthmatic episodes in certain susceptible persons. The alternatives to using epinephrine in a life-threatening situation may not be satisfactory. The presence of a sulfite(s) in neffy should not deter administration of the drug for treatment of serious allergic or other emergency situations.
7.2 Drugs Increasing Risk of Cardiac Arrhythmias
Patients who receive epinephrine while concomitantly taking cardiac glycosides, diuretics, or anti-arrhythmics should be observed carefully for the development of cardiac arrhythmias [see Warnings and Precautions (5.2)and Adverse Reactions (6)] .
7.1 Potential Increased Exposure of Nasal Spray Drugs
Neffy may alter nasal mucosa for up to 2 weeks after administration, and thus may increase systemic absorption of nasal products, including neffy, potentially increasing the risk of adverse reactions associated with these products.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies to evaluate the carcinogenic potential of epinephrine have not been conducted.
Epinephrine and other catecholamines have been shown to have mutagenic potential in vitro. Epinephrine was positive in the Salmonellabacterial reverse mutation assay, positive in the mouse lymphoma assay, and negative in the in vivomicronucleus assay. Epinephrine is an oxidative mutagen based on the E. coliWP2 Mutoxitest bacterial reverse mutation assay. This should not prevent the use of epinephrine where indicated [see Indications and Usage (1)] .
The potential for epinephrine to impair reproductive performance has not been evaluated, but epinephrine has been shown to decrease implantation in female rabbits dosed subcutaneously with 1.2 mg/kg/day (15-fold the highest human intramuscular or subcutaneous daily dose) during gestation days 3 to 9.
5.2 Risks Associated With Use of Epinephrine in Certain Coexisting Conditions (5.2 Risks Associated with Use of Epinephrine in Certain Coexisting Conditions)
Some patients may be at greater risk for developing adverse reactions after epinephrine administration. Despite these concerns, it should be recognized that the presence of these conditions is not a contraindication to epinephrine administration in an acute, life-threatening situation. Therefore, patients with these conditions, and/or any other person who might be in a position to administer neffy to a patient experiencing anaphylaxis should be carefully instructed in regard to the circumstances under which epinephrine should be used.
Epinephrine should be administered with caution to patients who have heart disease, including patients with cardiac arrhythmias, coronary artery disease, or hypertension. In such patients, or in patients who are on drugs that may sensitize the heart to arrhythmias, epinephrine may precipitate or aggravate angina pectoris, as well as produce ventricular arrhythmias [see Drug Interactions (7)and Adverse Reactions (6)] .
Epinephrine can temporarily exacerbate the underlying condition or increase symptoms in patients with the following: hyperthyroidism, Parkinson's disease, diabetes, renal impairment. Epinephrine should be administered with caution in patients with these conditions, including elderly patients and pregnant women.
5.1 Potential Altered Absorption of Neffy in Patients With Underlying Structural Or Anatomical Nasal Conditions (5.1 Potential Altered Absorption of Neffy in Patients with Underlying Structural or Anatomical Nasal Conditions)
Clinical pharmacology studies with neffy included subjects with history of allergic rhinitis, but did not include subjects with underlying structural and anatomical nasal conditions (e.g., polyps, history of nasal fractures or injuries, or history of nasal surgery). Absorption of neffy may be affected by underlying structural and anatomical nasal conditions. Consider use of other epinephrine products given by other routes of administration for patients with underlying structural or anatomical nasal conditions.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:27.658867 · Updated: 2026-03-14T22:22:33.812720