These Highlights Do Not Include All The Information Needed To Use Rexulti Safely And Effectively. See Full Prescribing Information For Rexulti.
2d301358-6291-4ec1-bd87-37b4ad9bd850
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS See full prescribing information for complete boxed warning. Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at increased risk of death. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease. ( 5.1 ) Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Safety and effectiveness of REXULTI have not been established in pediatric patients with MDD. ( 5.2 , 8.4 )
Indications and Usage
REXULTI is indicated for: Adjunctive treatment to antidepressants for major depressive disorder (MDD) in adults Treatment of schizophrenia in adults and pediatric patients ages 13 years and older Treatment of agitation associated with dementia due to Alzheimer's disease Limitations of Use: REXULTI is not indicated as an as needed ("prn") treatment for agitation associated with dementia due to Alzheimer's disease [see Clinical Studies (14.3) ] .
Dosage and Administration
Administer REXULTI orally once daily with or without food. ( 2 , 12.3 ) Indication Starting Dosage Recommended Target Dosage Maximum Dosage MDD Adults ( 2.2 ) 0.5 mg/day or 1 mg/day 2 mg/day 3 mg/day Schizophrenia Adults ( 2.3 ) 1 mg/day 2 to 4 mg/day 4 mg/day Schizophrenia Pediatric (13 - 17 years) ( 2.3 ) 0.5 mg/day 2 to 4 mg/day 4 mg/day Agitation associated with dementia due to Alzheimer's disease ( 2.4 ) 0.5 mg/day 2 mg/day 3 mg/day Moderate to Severe Hepatic Impairment: Maximum recommended dosage is 2 mg once daily for patients with MDD or agitation associated with dementia due to Alzheimer's disease and 3 mg once daily for patients with schizophrenia. ( 2.5 ) CrCl<60 mL/minute: Maximum recommended dosage is 2 mg once daily for patients with MDD or agitation associated with dementia due to Alzheimer's disease and 3 mg once daily for patients with schizophrenia. ( 2.6 ) See Full Prescribing Information for dosage modifications for CYP2D6 poor metabolizers and for concomitant use with CYP inhibitors or inducers. ( 2.7 )
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack) ( 5.3 ) Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring. ( 5.4 ) Tardive Dyskinesia: Discontinue if clinically appropriate. ( 5.5 ) Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.6 ) Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation. ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or history of leukopenia or neutropenia. Consider discontinuing REXULTI if a clinically significant decline in WBC occurs in absence of other causative factors. ( 5.8 ) Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. ( 5.9 ) Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.11 ) Potential for Cognitive and Motor Impairment: Use caution when operating machinery. ( 5.14 )
Contraindications
REXULTI is contraindicated in patients with a known hypersensitivity to brexpiprazole or any of its components. Reactions have included rash, facial swelling, urticaria, and anaphylaxis.
Adverse Reactions
In placebo-controlled trials in elderly patients with dementia, patients randomized to risperidone, aripiprazole, and olanzapine had a higher incidence of stroke and transient ischemic attack, including fatal stroke. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Boxed Warning , Warnings and Precautions (5.1) ].
Drug Interactions
Factors Dosage Adjustments for REXULTI ( 2.7 ) Strong CYP2D6 REXULTI may be administered without dosage adjustment in patients with MDD when administered with strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). or CYP3A4 inhibitors Administer half of recommended dosage. Strong/moderate CYP2D6 with Strong/moderate CYP3A4 inhibitors Administer a quarter of the recommended dosage. Known CYP2D6 poor metabolizers taking strong/moderate CYP3A4 inhibitors Administer a quarter of the recommended dosage. Strong CYP3A4 inducers Double the recommended dosage and further adjust based on clinical response.
Medication Information
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack) ( 5.3 ) Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring. ( 5.4 ) Tardive Dyskinesia: Discontinue if clinically appropriate. ( 5.5 ) Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.6 ) Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation. ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or history of leukopenia or neutropenia. Consider discontinuing REXULTI if a clinically significant decline in WBC occurs in absence of other causative factors. ( 5.8 ) Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. ( 5.9 ) Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.11 ) Potential for Cognitive and Motor Impairment: Use caution when operating machinery. ( 5.14 )
Indications and Usage
REXULTI is indicated for: Adjunctive treatment to antidepressants for major depressive disorder (MDD) in adults Treatment of schizophrenia in adults and pediatric patients ages 13 years and older Treatment of agitation associated with dementia due to Alzheimer's disease Limitations of Use: REXULTI is not indicated as an as needed ("prn") treatment for agitation associated with dementia due to Alzheimer's disease [see Clinical Studies (14.3) ] .
Dosage and Administration
Dosage and Administration
Advise patients that REXULTI can be taken with or without food. Advise patients regarding importance of following dosage escalation instructions [see Dosage and Administration (2)].
Contraindications
REXULTI is contraindicated in patients with a known hypersensitivity to brexpiprazole or any of its components. Reactions have included rash, facial swelling, urticaria, and anaphylaxis.
Adverse Reactions
In placebo-controlled trials in elderly patients with dementia, patients randomized to risperidone, aripiprazole, and olanzapine had a higher incidence of stroke and transient ischemic attack, including fatal stroke. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Boxed Warning , Warnings and Precautions (5.1) ].
Drug Interactions
Factors Dosage Adjustments for REXULTI ( 2.7 ) Strong CYP2D6 REXULTI may be administered without dosage adjustment in patients with MDD when administered with strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). or CYP3A4 inhibitors Administer half of recommended dosage. Strong/moderate CYP2D6 with Strong/moderate CYP3A4 inhibitors Administer a quarter of the recommended dosage. Known CYP2D6 poor metabolizers taking strong/moderate CYP3A4 inhibitors Administer a quarter of the recommended dosage. Strong CYP3A4 inducers Double the recommended dosage and further adjust based on clinical response.
Description
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS See full prescribing information for complete boxed warning. Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at increased risk of death. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease. ( 5.1 ) Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Safety and effectiveness of REXULTI have not been established in pediatric patients with MDD. ( 5.2 , 8.4 )
Section 42229-5
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Warnings and Precautions (5.1)].
Section 42231-1
| MEDICATION GUIDE REXULTI ® (REX-ul-TE) (brexpiprazole) tablets, for oral use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 5/2025 | ||
|
What is the most important information I should know about REXULTI? REXULTI may cause serious side effects, including:
|
|||
|
|
||
| See "What are the possible side effects of REXULTI?" for more information about side effects. | |||
|
What is REXULTI?
REXULTI is a prescription medicine used:
It is not known if REXULTI is safe and effective for the treatment of MDD in children. It is not known if REXULTI is safe and effective for the treatment of schizophrenia in children under 13 years of age. |
|||
|
Who should not take REXULTI? Do not take REXULTI if you are allergic to brexpiprazole or any of the ingredients in REXULTI. See the end of this Medication Guide for a complete list of ingredients in REXULTI. |
|||
Before taking REXULTI, tell your healthcare provider about all of your medical conditions, including if you:
REXULTI and other medicines may affect each other causing possible serious side effects. REXULTI may affect the way other medicines work, and other medicines may affect how REXULTI works. Your healthcare provider can tell you if it is safe to take REXULTI with your other medicines. Do not start or stop any medicines during treatment with REXULTI without first talking to your healthcare provider. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I take REXULTI?
|
|||
What should I avoid while taking REXULTI?
|
|||
|
What are the possible side effects of REXULTI? REXULTI may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
The most common side effects of REXULTI in children 13 to 17 years of age include difficulty moving or slow movement, tremors (shaking), abnormal eye movements, and muscle stiffness. These are not all the possible side effects of REXULTI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store REXULTI?
|
|||
|
General information about the safe and effective use of REXULTI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use REXULTI for a condition for which it was not prescribed. Do not give REXULTI to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about REXULTI that is written for health professionals. |
|||
|
What are the ingredients in REXULTI?
Active ingredient: brexpiprazole Inactive ingredients: lactose monohydrate, corn starch, microcrystalline cellulose, hydroxypropyl cellulose, low-substituted hydroxypropyl cellulose, magnesium stearate, hypromellose, and talc For color: titanium dioxide, iron oxide, and ferrosoferric oxide Manufactured by Otsuka Pharmaceutical Co., Ltd., Tokyo 101-8535, Japan Distributed and Marketed by Otsuka America Pharmaceutical, Inc., Rockville, MD 20850 USA Marketed by Lundbeck, Deerfield, IL 60015 USA ©2025 For more information about REXULTI, go to www.REXULTI.com or call 1-800-441-6763. |
Section 43683-2
| Warnings and Precautions (5.6) | 5/2025 |
Section 44425-7
Storage
Store REXULTI tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
9.2 Abuse
Animals given access to REXULTI did not self-administer the drug, suggesting that REXULTI does not have rewarding properties.
5.10 Falls
Antipsychotics, including REXULTI, may cause somnolence, postural hypotension, motor, and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic treatment.
10 Overdosage
There is limited clinical trial experience regarding human overdosage with REXULTI.
Management of a REXULTI overdose should concentrate on supportive therapy, maintaining an adequate airway, oxygenation and ventilation, and management of symptoms. Close medical supervision and monitoring should continue until the patient recovers. Consider contacting the Poison Help Line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
Oral activated charcoal and sorbitol (50 g/240 mL), administered one hour after ingesting oral REXULTI, decreased brexpiprazole Cmax and area under the curve (AUC) by approximately 5% to 23% and 31% to 39% respectively; however, there is insufficient information available on the therapeutic potential of activated charcoal in treating an overdose with REXULTI.
There is no information on the effect of hemodialysis in treating an overdose with REXULTI; hemodialysis is unlikely to be useful because brexpiprazole is highly bound to plasma proteins.
5.11 Seizures
Like other antipsychotic drugs, REXULTI may cause seizures. This risk is greatest in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in older patients.
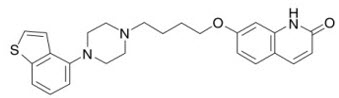
11 Description
Brexpiprazole, an atypical antipsychotic, is available as REXULTI® (brexpiprazole) tablets. Brexpiprazole is 7-{4-[4-(1-Benzothiophen-4-yl)piperazin-1-yl]butoxy}quinolin-2(1H)-one. The empirical formula is C25H27N3O2S, and its molecular weight is 433.57. The chemical structure is:
REXULTI tablets are for oral administration and are available in 0.25 mg, 0.5 mg, 1 mg, 2 mg, 3 mg, and 4 mg strengths. Inactive ingredients include lactose monohydrate, corn starch, microcrystalline cellulose, hydroxypropyl cellulose, low-substituted hydroxypropyl cellulose, magnesium stearate, hypromellose, and talc. Colorants include titanium dioxide, iron oxide, and ferrosoferric oxide.
5.13 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Antipsychotic drugs, including REXULTI, should be used cautiously in patients at risk for aspiration.
9.3 Dependence
Humans and animals that received chronic REXULTI administration did not demonstrate any withdrawal signs upon drug discontinuation. This suggests that REXULTI does not produce physical dependence.
8.5 Geriatric Use
Antipsychotic drugs increase the risk of death in elderly patients with dementia-related psychosis. REXULTI is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions (5.1)].
4 Contraindications
REXULTI is contraindicated in patients with a known hypersensitivity to brexpiprazole or any of its components. Reactions have included rash, facial swelling, urticaria, and anaphylaxis.
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning, Warnings and Precautions (5.1)]
- Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Boxed Warning, Warnings and Precautions (5.2)]
- Cerebrovascular Adverse Reactions Including Stroke in Elderly Patients with Dementia-Related Psychosis [see Warnings and Precautions (5.3)]
- Neuroleptic Malignant Syndrome (NMS) [see Warnings and Precautions (5.4)]
- Tardive Dyskinesia [see Warnings and Precautions (5.5)]
- Metabolic Changes [see Warnings and Precautions (5.6)]
- Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions (5.7)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.8)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.9)]
- Falls [see Warnings and Precautions (5.10)]
- Seizures [see Warnings and Precautions (5.11)]
- Body Temperature Dysregulation [see Warnings and Precautions (5.12)]
- Dysphagia [see Warnings and Precautions (5.13)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.14)]
7 Drug Interactions
| Factors | Dosage Adjustments for REXULTI ( 2.7 ) |
| Strong CYP2D6 REXULTI may be administered without dosage adjustment in patients with MDD when administered with strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). or CYP3A4 inhibitors |
Administer half of recommended dosage. |
| Strong/moderate CYP2D6 with Strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Known CYP2D6 poor metabolizers taking strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Strong CYP3A4 inducers | Double the recommended dosage and further adjust based on clinical response. |
8.8 Renal Impairment
The maximum recommended dosage in patients with CrCl<60 mL/minute is lower than those with mild renal impairment and those with normal renal function [see Dosage and Administration (2.6)]. Patients with renal impairment had higher exposure to brexpiprazole than patients with normal renal function [see Clinical Pharmacology (12.3)]. Greater exposure may increase the risk of REXULTI-associated adverse reactions.
12.2 Pharmacodynamics
Brexpiprazole has affinity (expressed as Ki) for multiple monoaminergic receptors including serotonin 5-HT1A (0.12 nM), 5-HT2A (0.47 nM), 5-HT2B (1.9 nM), 5-HT7 (3.7 nM), dopamine D2 (0.30 nM), D3 (1.1 nM), and noradrenergic α1A (3.8 nM), α1B (0.17 nM), α1D (2.6 nM), and α2C (0.59 nM) receptors. Brexpiprazole acts as a partial agonist at the 5-HT1A, D2, and D3 receptors and as an antagonist at 5-HT2A, 5-HT2B, 5-HT7, α1A, α1B, α1D, and α2C receptors. Brexpiprazole also exhibits affinity for histamine H1 receptor (19 nM) and for muscarinic M1 receptor (67% inhibition at 10 µM).
5.6 Metabolic Changes
Atypical antipsychotic drugs, including REXULTI, have caused metabolic changes including hyperglycemia, diabetes mellitus, dyslipidemia, and body weight gain. Although all of the drugs in the class to date have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.5 Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. The risk appears to be highest among the elderly, especially elderly women, but it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drugs differ in their potential to cause tardive dyskinesia is unknown.
The risk of tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the cumulative dose increases. The syndrome can develop after relatively brief treatment periods, at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect that symptomatic suppression has upon the long-term course of tardive dyskinesia is unknown.
Given these considerations, REXULTI should be prescribed in a manner most likely to reduce the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that 1) is known to respond to antipsychotic drugs and 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, use the lowest dose and the shortest duration of treatment needed to produce a satisfactory clinical response. Periodically reassess the need for continued treatment.
If signs and symptoms of tardive dyskinesia appear in a patient treated with REXULTI, drug discontinuation should be considered. However, some patients may require treatment with REXULTI despite the presence of the syndrome.
8.7 Hepatic Impairment
The maximum recommended dosage in patients with moderate to severe hepatic impairment (Child-Pugh score ≥7) is lower than those with mild hepatic impairment and those with normal hepatic function [see Dosage and Administration (2.4)]. Patients with moderate to severe hepatic impairment generally had higher exposure to brexpiprazole than patients with normal hepatic function [see Clinical Pharmacology (12.3)]. Greater exposure may increase the risk of REXULTI-associated adverse reactions .
1 Indications and Usage
REXULTI is indicated for:
- Adjunctive treatment to antidepressants for major depressive disorder (MDD) in adults
- Treatment of schizophrenia in adults and pediatric patients ages 13 years and older
- Treatment of agitation associated with dementia due to Alzheimer's disease
Limitations of Use:
REXULTI is not indicated as an as needed ("prn") treatment for agitation associated with dementia due to Alzheimer's disease [see Clinical Studies (14.3)].
12.1 Mechanism of Action
The mechanism of action of REXULTI in the adjunctive treatment of major depressive disorder, treatment of agitation associated with dementia due to Alzheimer's disease, or treatment of schizophrenia is unknown. However, the efficacy of REXULTI may be mediated through a combination of partial agonist activity at serotonin 5-HT1A and dopamine D2 receptors, and antagonist activity at serotonin 5-HT2A receptors.
9.1 Controlled Substance
REXULTI contains brexpiprazole, which is not a controlled substance.
5 Warnings and Precautions
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack) (5.3)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring. (5.4)
- Tardive Dyskinesia: Discontinue if clinically appropriate. (5.5)
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. (5.6)
- Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation. (5.7)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or history of leukopenia or neutropenia. Consider discontinuing REXULTI if a clinically significant decline in WBC occurs in absence of other causative factors. (5.8)
- Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. (5.9)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. (5.11)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery. (5.14)
2 Dosage and Administration
Administer REXULTI orally once daily with or without food. (2, 12.3)
| Indication | Starting Dosage | Recommended Target Dosage | Maximum Dosage |
|---|---|---|---|
| MDD Adults (2.2) |
0.5 mg/day or 1 mg/day |
2 mg/day | 3 mg/day |
| Schizophrenia Adults (2.3) |
1 mg/day | 2 to 4 mg/day | 4 mg/day |
| Schizophrenia Pediatric (13 - 17 years) (2.3) |
0.5 mg/day | 2 to 4 mg/day | 4 mg/day |
| Agitation associated with dementia due to Alzheimer's disease (2.4) |
0.5 mg/day | 2 mg/day | 3 mg/day |
- Moderate to Severe Hepatic Impairment: Maximum recommended dosage is 2 mg once daily for patients with MDD or agitation associated with dementia due to Alzheimer's disease and 3 mg once daily for patients with schizophrenia. (2.5)
- CrCl<60 mL/minute: Maximum recommended dosage is 2 mg once daily for patients with MDD or agitation associated with dementia due to Alzheimer's disease and 3 mg once daily for patients with schizophrenia. (2.6)
- See Full Prescribing Information for dosage modifications for CYP2D6 poor metabolizers and for concomitant use with CYP inhibitors or inducers. (2.7)
3 Dosage Forms and Strengths
REXULTI tablets are available in 6 strengths:
- 0.25 mg tablets are light brown, round, shallow convex, bevel-edged body with "BRX" and "0.25" imprinted on one side.
- 0.5 mg tablets: are light orange, round, shallow convex, bevel-edged body with "BRX" and "0.5" imprinted on one side.
- 1 mg tablets are light yellow, round, shallow convex, bevel-edged body with "BRX" and "1" imprinted on one side.
- 2 mg tablets are light green, round, shallow convex, bevel-edged body with "BRX" and "2" imprinted on one side.
- 3 mg tablets are light purple, round, shallow convex, bevel-edged body with "BRX" and "3" imprinted on one side.
- 4 mg tablets are white, round, shallow convex, bevel-edged body with "BRX" and "4" imprinted on one side.
6.2 Postmarketing Experience
The following adverse reaction has been identified during post-approval use of REXULTI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Nervous System disorders: Neuroleptic Malignant Syndrome
8.6 Cyp2d6 Poor Metabolizers
Dosage adjustment is recommended in known CYP2D6 poor metabolizers because these patients have higher brexpiprazole concentrations than normal metabolizers of CYP2D6. Approximately 8% of Caucasians and 3 to 8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers [see Dosage and Administration (2.7), Clinical Pharmacology (12.3)].
8 Use in Specific Populations
Pregnancy: May cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most common adverse reactions in adult patients in clinical trials (≥5%) were weight increased, akathisia, headache, somnolence, and insomnia.
The most common adverse reactions in pediatric patients in clinical trials (≥5%) were weight increased, somnolence, headache, akathisia, and nasopharyngitis.
Brexpiprazole has been evaluated for safety in 12,550 adult patients who participated in multiple-dose clinical trials for major depressive disorder, schizophrenia, agitation associated with dementia due to Alzheimer's disease, attention deficit hyperactivity disorder (ADHD), post-traumatic stress disorder (PTSD), bipolar mania, and borderline personality disorder (BPD). Among them, 3,870 patients were treated with brexpiprazole for at least 180 days, and 1,910 patients were treated for at least one year of exposure.
Additionally, brexpiprazole has been evaluated for safety in 119 pediatric patients who participated in short-term trials, and 314 patients in long-term multiple-dose clinical trials for pediatric schizophrenia and autism spectrum disorders (ASD).
8.9 Other Specific Populations
The recommended dosage for REXULTI is the same in males and females, in different racial groups, and in smokers and nonsmokers [see Clinical Pharmacology (12.3)].
17 Patient Counseling Information
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide).
5.12 Body Temperature Dysregulation
Atypical antipsychotics may disrupt the body's ability to reduce core body temperature. Strenuous exercise, exposure to extreme heat, dehydration, and anticholinergic medications may contribute to an elevation in core body temperature; use REXULTI with caution in patients who may experience these conditions.
5.9 Orthostatic Hypotension and Syncope
Atypical antipsychotics cause orthostatic hypotension and syncope. Generally, the risk is greatest during initial dose titration and when increasing the dose. In the short-term, placebo-controlled clinical studies of REXULTI plus ADT in adult patients with MDD, the incidence of orthostatic hypotension-related adverse reactions in REXULTI plus ADT-treated patients compared to placebo plus ADT-treated patients included: dizziness (2% versus 2%) and orthostatic hypotension (0.1% versus 0%). In the short-term, placebo-controlled clinical studies of REXULTI in adult patients with schizophrenia, the incidence of orthostatic hypotension-related adverse reactions in REXULTI-treated patients compared to placebo patients included: dizziness (2% versus 2%), orthostatic hypotension (0.4% versus 0.2%), and syncope (0.1% versus 0%). In 12-week, placebo-controlled clinical studies of REXULTI in patients with agitation associated with dementia due to Alzheimer's disease, the incidence of orthostatic hypotension-related adverse reactions in patients treated with REXULTI compared to patients treated with placebo included: dizziness (3% versus 3%), orthostatic hypotension (1% versus 1%), and syncope (0.2% versus 0.8%).
Orthostatic vital signs should be monitored in patients who are vulnerable to hypotension (e.g., elderly patients, patients with dehydration, hypovolemia, concomitant treatment with antihypertensive medication), patients with known cardiovascular disease (history of myocardial infarction, ischemic heart disease, heart failure, or conduction abnormalities), and patients with cerebrovascular disease. REXULTI has not been evaluated in patients with a recent history of myocardial infarction or unstable cardiovascular disease. Such patients were excluded from the premarketing clinical studies.
5.4 Neuroleptic Malignant Syndrome (nms)
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with administration of antipsychotic drugs, including REXULTI.
Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmia). Additional signs may include elevated creatinine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue REXULTI and provide intensive symptomatic treatment and monitoring.
5.8 Leukopenia, Neutropenia, and Agranulocytosis
Leukopenia and neutropenia have been reported during treatment with antipsychotic agents. Agranulocytosis (including fatal cases) has been reported with other agents in this class.
Possible risk factors for leukopenia and neutropenia include pre-existing low white blood cell count (WBC) or absolute neutrophil count (ANC) and history of drug-induced leukopenia or neutropenia. In patients with a pre-existing low WBC or ANC or a history of drug-induced leukopenia or neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of REXULTI at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue REXULTI in patients with absolute neutrophil count <1000/mm3 and follow their WBC until recovery.
5.14 Potential for Cognitive and Motor Impairment
REXULTI, like other antipsychotics, may cause somnolence and has the potential to impair judgment, thinking, or motor skills. In the 6-week placebo-controlled clinical studies in patients with MDD, somnolence (including sedation and hypersomnia) was reported in 4% of REXULTI plus ADT-treated patients compared to 1% of placebo plus ADT-treated patients.
In the 6-week placebo-controlled clinical studies in adult patients with schizophrenia, somnolence (including sedation and hypersomnia) was reported in 5% of REXULTI-treated patients compared to 3% of placebo-treated patients.
In the 12-week placebo-controlled, fixed-dose clinical studies in patients (51 to 90 years of age) with agitation associated with dementia due to Alzheimer's disease, somnolence (including sedation) was reported in 3% of patients treated with REXULTI compared to 1% of patients treated with placebo.
Patients should be cautioned about operating hazardous machinery, including motor vehicles, until they are reasonably certain that REXULTI therapy does not affect them adversely.
Principal Display Panel 1 Mg Tablet Bottle Label
1 mg
30 Tablets
NDC 59148-037-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 2 Mg Tablet Bottle Label
2 mg
30 Tablets
NDC 59148-038-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 3 Mg Tablet Bottle Label
3 mg
30 Tablets
NDC 59148-039-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 4 Mg Tablet Bottle Label
4 mg
30 Tablets
NDC 59148-040-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 1 Mg Tablet Bottle Carton
1 mg
30 Tablets
NDC 59148-037-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 2 Mg Tablet Bottle Carton
2 mg
30 Tablets
NDC 59148-038-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 3 Mg Tablet Bottle Carton
3 mg
30 Tablets
NDC 59148-039-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 4 Mg Tablet Bottle Carton
4 mg
30 Tablets
NDC 59148-040-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 0.5 Mg Tablet Bottle Label
0.5 mg
30 Tablets
NDC 59148-036-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 0.25 Mg Tablet Bottle Label
0.25 mg
30 Tablets
NDC 59148-035-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 0.5 Mg Tablet Bottle Carton
0.5 mg
30 Tablets
NDC 59148-036-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
14.1 Adjunctive Treatment of Major Depressive Disorder
The efficacy of REXULTI in the adjunctive treatment of major depressive disorder (MDD) was evaluated in two 6-week double-blind, placebo-controlled, fixed-dose studies of adult patients meeting DSM-IV-TR criteria for MDD, with or without symptoms of anxiety, who had an inadequate response to prior antidepressant therapy (1 to 3 courses) in the current episode and who had also demonstrated an inadequate response throughout the 8 weeks of prospective antidepressant treatment (with escitalopram, fluoxetine, paroxetine controlled-release, sertraline, duloxetine delayed release, or venlafaxine extended release). Inadequate response during the prospective antidepressant treatment phase was defined as having persistent symptoms without substantial improvement throughout the course of treatment.
Patients in Study 1 (NCT01360645) were randomized to REXULTI 2 mg once a day or placebo. Patients in Study 2 (NCT01360632) were randomized to REXULTI 1 or 3 mg once a day or placebo. For patients randomized to REXULTI, all patients initiated treatment at 0.5 mg once daily during Week 1. At Week 2, the REXULTI dosage was increased to 1 mg in all treatment groups, and either maintained at 1 mg or increased to 2 mg or 3 mg once daily, based on treatment assignment, from Week 3 onwards. The dosages were then maintained for the 4 remaining weeks.
The primary endpoint was change from baseline to Week 6 in the Montgomery-Asberg Depression Rating Scale (MADRS), a 10-item clinician-related scale used to assess the degree of depressive symptomatology, with 0 representing no symptoms and 60 representing worst symptoms.
At randomization, the mean MADRS total score was 27. In Studies 1 and 2, REXULTI (plus ADT) 2 mg once daily and 3 mg once daily were superior to placebo plus ADT in reducing mean MADRS total scores. Results from the primary efficacy parameters for both fixed dose studies are shown below in Table 13. Figure 4 below shows the time course of response based on the primary efficacy measure (MADRS) in Study 1.
| Study | Treatment Group | N | Mean Baseline Score (SD) | LS Mean Change from Baseline (SE) | Placebo-subtracted Difference Difference (drug minus placebo) in least-squares mean change from baseline
(95% CI) |
|---|---|---|---|---|---|
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval | |||||
| 1 | REXULTI (2 mg/day) + ADT Dosages statistically significantly superior to placebo
|
175 | 26.9 (5.7) | -8.4 (0.6) | -3.2 (-4.9, -1.5) |
| Placebo + ADT | 178 | 27.3 (5.6) | -5.2 (0.6) | -- | |
| 2 | REXULTI (1 mg/day) + ADT | 211 | 26.5 (5.6) | -7.6 (0.5) | -1.3 (-2.7, 0.1) |
| REXULTI (3 mg/day) + ADT | 213 | 26.5 (5.3) | -8.3 (0.5) | -2.0 (-3.4, -0.5) | |
| Placebo + ADT | 203 | 26.5 (5.2) | -6.3 (0.5) | -- |
An examination of population subgroups did not suggest differential response based on age, gender, race, or choice of prospective antidepressant.
Figure 4 Change from Baseline in MADRS Total Score by Study Visit (Week) in Patients with MDD in Adults (Study 1)
Principal Display Panel 0.25 Mg Tablet Bottle Carton
0.25 mg
30 Tablets
NDC 59148-035-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
2.6 Recommended Dosage in Patients With Renal Impairment
The maximum recommended dosage in patients with creatinine clearance CrCl<60 mL/minute is [see Use in Specific Populations (8.8), Clinical Pharmacology (12.3)].
2 mg orally once daily in patients with MDD or agitation associated with dementia due to Alzheimer's disease and
3 mg orally once daily in patients with schizophrenia
5.7 Pathological Gambling and Other Compulsive Behaviors
Post-marketing case reports suggest that patients can experience intense urges, particularly for gambling, and the inability to control these urges while taking REXULTI. Other compulsive urges, reported less frequently, include sexual urges, shopping, eating, or binge eating, and other impulsive or compulsive behaviors. Because patients may not recognize these behaviors as abnormal, it is important for prescribers to ask patients or their caregivers specifically about the development of new or intense gambling urges, compulsive sexual urges, compulsive shopping, binge or compulsive eating, or other urges while being treated with REXULTI. In some cases, although not all, urges were reported to have stopped when the dose was reduced, or the medication was discontinued. Compulsive behaviors may result in harm to the patient and others if not recognized. Consider dose reduction or stopping the medication if a patient develops such urges.
2.5 Recommended Dosage in Patients With Hepatic Impairment
The maximum recommended dosage in patients with moderate to severe hepatic impairment (Child-Pugh score ≥7) is [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2 mg orally once daily in patients with MDD or agitation associated with dementia due to Alzheimer's disease, and
3 mg orally once daily in patients with schizophrenia
7.1 Drugs Having Clinically Important Interactions With Rexulti
See Table 12 for clinically important drug interactions with REXULTI.
| Strong CYP3A4 Inhibitors | |
| Clinical Impact: | Concomitant use of REXULTI with strong CYP3A4 inhibitors increased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with a strong CYP3A4 inhibitor, reduce the REXULTI dosage [see Dosage and Administration (2.7)]. |
|
Strong CYP2D6 Inhibitors In the clinical studies examining the adjunctive use of REXULTI in the treatment of MDD, dosage was not adjusted for strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). Thus, CYP considerations are already factored into general dosing recommendations, and REXULTI may be administered without dosage adjustment in patients with MDD.
|
|
| Clinical Impact: | Concomitant use of REXULTI with strong CYP2D6 inhibitors increased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with a strong CYP2D6 inhibitor, reduce the REXULTI dosage [see Dosage and Administration (2.7)]. |
| Both CYP3A4 Inhibitors and CYP2D6 Inhibitors | |
| Clinical Impact: | Concomitant use of REXULTI with 1) a strong CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 2) a moderate CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 3) a strong CYP3A4 inhibitor and a moderate CYP2D6 inhibitor; or 4) a moderate CYP3A4 inhibitor and a moderate CYP2D6 inhibitor increased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with 1) a strong CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 2) a moderate CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 3) a strong CYP3A4 inhibitor and a moderate CYP2D6 inhibitor; or 4) a moderate CYP3A4 inhibitor and a moderate CYP2D6 inhibitor, decrease the REXULTI dosage [see Dosage and Administration (2.7)]. |
| Strong CYP3A4 Inducers | |
| Clinical Impact: | Concomitant use of REXULTI and a strong CYP3A4 inducer decreased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with a strong CYP3A4 inducer, increase the REXULTI dosage [see Dosage and Administration (2.7)]. |
14.3 Agitation Associated With Dementia Due to Alzheimer's Disease
The efficacy of REXULTI in the treatment of agitation associated with dementia due to Alzheimer's disease was demonstrated in two 12-week, randomized, double-blind, placebo-controlled, fixed-dose studies (Study 6, NCT01862640 and Study 7, NCT03548584). In these studies, patients were required to:
- Have a diagnosis of probable Alzheimer's disease according to NINCDS-ADRDA criteria,
- Have a Mini-Mental State Examination (MMSE) score of ≥5 and ≤22 and have a total score of ≥4 by the agitation/aggression item of the NPI/NPI-NH, and
- Exhibit sufficient agitation behaviors at time of entry to warrant use of pharmacotherapy, after excluding other factors.
Patients in:
- Study 6 were randomized to an oral dosage of either REXULTI 1 mg once a day, REXULTI 2 mg once a day, or placebo. Patients in both REXULTI groups started on 0.25 mg once daily for approximately three days, then received 0.5 mg once daily for approximately 12 days. Subsequently, patients in the 1 mg group received 1 mg once daily for the remainder of the 12-week study, and patients in the 2 mg group received 1 mg once daily for approximately two weeks and then received 2 mg for the remainder of the study.
- Study 7 were randomized to an oral dose of either REXULTI 2 mg or 3 mg once a day (combined treatment arm) or placebo. Patients in both REXULTI groups started on 0.5 mg once daily for 7 days, then received 1 mg once daily for 7 days and then 2 mg once daily for 14 days. Subsequently, patients in the 2 mg group received 2 mg once daily for the remainder of the 12-week study, and patients in the 3 mg group received 3 mg once daily for the remainder of the study.
Study 6 included 433 patients with a mean age of 74 years old, and a range of 51 and 90 years old; 45% were male; 96%, 3%, and 1%, were White, Black or African American, and Asian, respectively; and 16% and 83% were Latino/Hispanic and not Latino/Hispanic, respectively. Study 7 included 345 patients with a mean age of 74 years old, and a range of 56 and 90 years old; 44% were male; 95%, 4%, and 1% were White, Black or African American, and Asian, respectively; and 31% and 69% were Latino/Hispanic and not Latino/Hispanic, respectively.
The primary efficacy endpoint in these two studies was the change from baseline in the Cohen-Mansfield Agitation Inventory total (CMAI) score at Week 12. The CMAI is a clinician rated questionnaire consisting of 29 items, which assess the frequency of manifestations of agitated behaviors in elderly patients, based on caregiver input. Three specific factors can be derived from the CMAI scale: 1) Aggressive Behavior (e.g., screaming, throwing things, cursing/verbal aggression, kicking, pushing scratching, hurting self or others); 2) Physically Non-Aggressive Behavior (e.g., repetitive mannerisms, general restlessness, pacing); and 3) Verbally Agitated Behavior (e.g., complaining, repetitive questions, constant requests for attention). Each CMAI behavior was rated on a scale of 1 (never) to 7 (very frequent agitated behaviors); the total CMAI scores range from 29 (best) to 203 (worst). A negative change indicates improvement.
In Trial 6, patients in the REXULTI 2 mg group showed improved total CMAI scores compared to patients in the placebo group at Week 12. In Trial 7, patients in the REXULTI 2 mg/3 mg group showed improved total CMAI scores compared to patients in the placebo group at Week 12.
As shown in Table 16 and Figure 8, the mean change from baseline in the total CMAI score after 12 weeks in the 2 mg/or 3 mg REXULTI group was statistically significantly superior to the placebo group. The 1 mg REXULTI group did not demonstrate significantly greater mean changes at baseline from the placebo group in the total CMAI score in this patient population. The 1 mg once day REXULTI dosage is not approved and is not recommended for the treatment of agitation associated with dementia due to Alzheimer's disease [see Dosage and Administration (2.4)].
| Study | Treatment Group | N | Mean Baseline Score (SD) |
LS Mean Change from Baseline (SE) |
Placebo-subtracted Difference Difference (drug minus placebo) in least-squares mean change from baseline
(95% CI) |
|---|---|---|---|---|---|
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval | |||||
| 6 | REXULTI 1 mg/day | 134 | 70.5 (16.0) | -17.6 (1.3) | 0.2 (-3.4, 3.9) |
| REXULTI 2 mg/day Dosages statistically significantly superior to placebo.
|
138 | 71.0 (16.6) | -21.6 (1.3) | -3.8 (-7.4, -0.2) | |
| Placebo | 131 | 72.2 (17.9) | -17.8 (1.3) | — | |
| 7 | REXULTI 2 mg/day or 3 mg/day | 225 | 80.6 (16.6) | -22.6 (1.1) | -5.3 (-8.8, -1.9) |
| Placebo | 116 | 79.2 (17.5) | -17.3 (1.4) | — |
Figure 8: Change from Baseline in Total CMAI Score by Study Week in Patients with Agitation Associated with Dementia Due to Alzheimer's Disease (Study 7)
7.2 Drugs Having No Clinically Important Interactions With Rexulti
Based on pharmacokinetic studies, no dosage adjustment of REXULTI is required when administered concomitantly with CYP2B6 inhibitors (e.g., ticlopidine) or gastric pH modifiers (e.g., omeprazole). Additionally, no dosage adjustment for substrates of CYP2D6 (e.g., dextromethorphan), CYP3A4 (e.g., lovastatin), CYP2B6 (e.g., bupropion), BCRP (e.g., rosuvastatin), or P-gp (e.g., fexofenadine) is required when administered concomitantly with REXULTI.
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in the drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group.
Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Boxed Warning, Warnings and Precautions (5.3)].
5.2 Suicidal Thoughts and Behaviors in Children, Adolescents, and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and over 4,400 pediatric patients, the incidence of suicidal thoughts and behaviors in patients 24 years of age and younger was greater in antidepressant-treated patients than in placebo-treated patients. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1,000 patients treated are provided in Table 2.
No suicides occurred in any of the pediatric studies. There were suicides in the adult studies, but the number was not sufficient to reach any conclusion about antidepressant drug effect on suicide.
| Age Range (years) | Drug-Placebo Difference in Number of Patients with Suicidal Thoughts or Behaviors per 1,000 Patients Treated |
|---|---|
| Increases Compared to Placebo | |
| <18 | 14 additional patients |
| 18 to 24 | 5 additional patients |
| Decreases Compared to Placebo | |
| 25 to 64 | 1 fewer patient |
| ≥65 | 6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance studies in adults with MDD that antidepressants delay the recurrence of depression.
Monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing REXULTI, in patients whose depression is persistently worse or who are experiencing emergent suicidal thoughts or behaviors.
2.2 Recommended Dosage for Adjunctive Treatment of Major Depressive Disorder (adults)
The recommended starting REXULTI dosage for the adjunctive treatment of MDD in adults is 0.5 mg or 1 mg orally once daily. Titrate to 1 mg once daily, then titrate to the target dosage of 2 mg once daily (based on the patient's clinical response and tolerability, increase the dosage at weekly intervals). The maximum recommended daily dosage is 3 mg. Periodically reassess to determine the continued need and appropriate dosage for treatment.
2.4 Recommended Dosage for Agitation Associated With Dementia Due to Alzheimer's Disease
The recommended starting REXULTI dosage for the treatment of agitation associated with dementia due to Alzheimer's disease is 0.5 mg orally once daily on Days 1 to 7. Increase the dosage on Days 8 through 14 to 1 mg once daily, and on Day 15 to 2 mg once daily. The recommended target dose is 2 mg once daily. The dosage can be increased to the maximum recommended daily dosage of 3 mg once daily after at least 14 days, based on clinical response and tolerability.
5.3 Cerebrovascular Adverse Reactions Including Stroke in Elderly Patients With Dementia Related Psychosis
In placebo-controlled trials in elderly patients with dementia, patients randomized to risperidone, aripiprazole, and olanzapine had a higher incidence of stroke and transient ischemic attack, including fatal stroke. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Boxed Warning, Warnings and Precautions (5.1)].
2.7 Dosage Modifications for Cyp2d6 Poor Metabolizers and for Concomitant Use With Cyp Inhibitors Or Inducers
Dosage modifications are recommended in patients who are known cytochrome P450 (CYP) 2D6 poor metabolizers and in patients taking concomitant CYP3A4 inhibitors, CYP2D6 inhibitors, or strong CYP3A4 inducers (see Table 1). If the concomitant drug is discontinued, adjust the REXULTI dosage to its original level. If the concomitant CYP3A4 inducer is discontinued, reduce the REXULTI dosage to the original level over 1 to 2 weeks [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
| Factors | Adjusted REXULTI Dosage |
|---|---|
| CYP2D6 Poor Metabolizers | |
| CYP2D6 poor metabolizers | Administer half of the recommended dosage. |
| Known CYP2D6 poor metabolizers taking strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Patients Taking CYP2D6 Inhibitors and/or CYP3A4 Inhibitors | |
| Strong CYP2D6 inhibitors In the clinical studies examining the use of REXULTI for the adjunctive treatment of MDD, dosage was not adjusted for strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). Thus, CYP considerations are already factored into general dosing recommendations, and REXULTI may be administered without dosage adjustment in patients with MDD.
|
Administer half of the recommended dosage. |
| Strong CYP3A4 inhibitors | Administer half of the recommended dosage. |
| Strong/moderate CYP2D6 inhibitors with strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Patients Taking CYP3A4 Inducers | |
| Strong CYP3A4 inducers | Double the recommended dosage over 1 to 2 weeks. |
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis and Suicidal Thoughts and Behaviors
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS
See full prescribing information for complete boxed warning.
- Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at increased risk of death. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease. (5.1)
- Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Safety and effectiveness of REXULTI have not been established in pediatric patients with MDD. (5.2, 8.4)
Structured Label Content
Dosage and Administration (34068-7)
Dosage and Administration
Advise patients that REXULTI can be taken with or without food. Advise patients regarding importance of following dosage escalation instructions [see Dosage and Administration (2)].
Section 42229-5 (42229-5)
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
| MEDICATION GUIDE REXULTI ® (REX-ul-TE) (brexpiprazole) tablets, for oral use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 5/2025 | ||
|
What is the most important information I should know about REXULTI? REXULTI may cause serious side effects, including:
|
|||
|
|
||
| See "What are the possible side effects of REXULTI?" for more information about side effects. | |||
|
What is REXULTI?
REXULTI is a prescription medicine used:
It is not known if REXULTI is safe and effective for the treatment of MDD in children. It is not known if REXULTI is safe and effective for the treatment of schizophrenia in children under 13 years of age. |
|||
|
Who should not take REXULTI? Do not take REXULTI if you are allergic to brexpiprazole or any of the ingredients in REXULTI. See the end of this Medication Guide for a complete list of ingredients in REXULTI. |
|||
Before taking REXULTI, tell your healthcare provider about all of your medical conditions, including if you:
REXULTI and other medicines may affect each other causing possible serious side effects. REXULTI may affect the way other medicines work, and other medicines may affect how REXULTI works. Your healthcare provider can tell you if it is safe to take REXULTI with your other medicines. Do not start or stop any medicines during treatment with REXULTI without first talking to your healthcare provider. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I take REXULTI?
|
|||
What should I avoid while taking REXULTI?
|
|||
|
What are the possible side effects of REXULTI? REXULTI may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
The most common side effects of REXULTI in children 13 to 17 years of age include difficulty moving or slow movement, tremors (shaking), abnormal eye movements, and muscle stiffness. These are not all the possible side effects of REXULTI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store REXULTI?
|
|||
|
General information about the safe and effective use of REXULTI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use REXULTI for a condition for which it was not prescribed. Do not give REXULTI to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about REXULTI that is written for health professionals. |
|||
|
What are the ingredients in REXULTI?
Active ingredient: brexpiprazole Inactive ingredients: lactose monohydrate, corn starch, microcrystalline cellulose, hydroxypropyl cellulose, low-substituted hydroxypropyl cellulose, magnesium stearate, hypromellose, and talc For color: titanium dioxide, iron oxide, and ferrosoferric oxide Manufactured by Otsuka Pharmaceutical Co., Ltd., Tokyo 101-8535, Japan Distributed and Marketed by Otsuka America Pharmaceutical, Inc., Rockville, MD 20850 USA Marketed by Lundbeck, Deerfield, IL 60015 USA ©2025 For more information about REXULTI, go to www.REXULTI.com or call 1-800-441-6763. |
Section 43683-2 (43683-2)
| Warnings and Precautions (5.6) | 5/2025 |
Section 44425-7 (44425-7)
Storage
Store REXULTI tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
9.2 Abuse
Animals given access to REXULTI did not self-administer the drug, suggesting that REXULTI does not have rewarding properties.
5.10 Falls
Antipsychotics, including REXULTI, may cause somnolence, postural hypotension, motor, and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic treatment.
10 Overdosage (10 OVERDOSAGE)
There is limited clinical trial experience regarding human overdosage with REXULTI.
Management of a REXULTI overdose should concentrate on supportive therapy, maintaining an adequate airway, oxygenation and ventilation, and management of symptoms. Close medical supervision and monitoring should continue until the patient recovers. Consider contacting the Poison Help Line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
Oral activated charcoal and sorbitol (50 g/240 mL), administered one hour after ingesting oral REXULTI, decreased brexpiprazole Cmax and area under the curve (AUC) by approximately 5% to 23% and 31% to 39% respectively; however, there is insufficient information available on the therapeutic potential of activated charcoal in treating an overdose with REXULTI.
There is no information on the effect of hemodialysis in treating an overdose with REXULTI; hemodialysis is unlikely to be useful because brexpiprazole is highly bound to plasma proteins.
5.11 Seizures
Like other antipsychotic drugs, REXULTI may cause seizures. This risk is greatest in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in older patients.
11 Description (11 DESCRIPTION)
Brexpiprazole, an atypical antipsychotic, is available as REXULTI® (brexpiprazole) tablets. Brexpiprazole is 7-{4-[4-(1-Benzothiophen-4-yl)piperazin-1-yl]butoxy}quinolin-2(1H)-one. The empirical formula is C25H27N3O2S, and its molecular weight is 433.57. The chemical structure is:
REXULTI tablets are for oral administration and are available in 0.25 mg, 0.5 mg, 1 mg, 2 mg, 3 mg, and 4 mg strengths. Inactive ingredients include lactose monohydrate, corn starch, microcrystalline cellulose, hydroxypropyl cellulose, low-substituted hydroxypropyl cellulose, magnesium stearate, hypromellose, and talc. Colorants include titanium dioxide, iron oxide, and ferrosoferric oxide.
5.13 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Antipsychotic drugs, including REXULTI, should be used cautiously in patients at risk for aspiration.
9.3 Dependence
Humans and animals that received chronic REXULTI administration did not demonstrate any withdrawal signs upon drug discontinuation. This suggests that REXULTI does not produce physical dependence.
8.5 Geriatric Use
Antipsychotic drugs increase the risk of death in elderly patients with dementia-related psychosis. REXULTI is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions (5.1)].
4 Contraindications (4 CONTRAINDICATIONS)
REXULTI is contraindicated in patients with a known hypersensitivity to brexpiprazole or any of its components. Reactions have included rash, facial swelling, urticaria, and anaphylaxis.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning, Warnings and Precautions (5.1)]
- Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Boxed Warning, Warnings and Precautions (5.2)]
- Cerebrovascular Adverse Reactions Including Stroke in Elderly Patients with Dementia-Related Psychosis [see Warnings and Precautions (5.3)]
- Neuroleptic Malignant Syndrome (NMS) [see Warnings and Precautions (5.4)]
- Tardive Dyskinesia [see Warnings and Precautions (5.5)]
- Metabolic Changes [see Warnings and Precautions (5.6)]
- Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions (5.7)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.8)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.9)]
- Falls [see Warnings and Precautions (5.10)]
- Seizures [see Warnings and Precautions (5.11)]
- Body Temperature Dysregulation [see Warnings and Precautions (5.12)]
- Dysphagia [see Warnings and Precautions (5.13)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.14)]
7 Drug Interactions (7 DRUG INTERACTIONS)
| Factors | Dosage Adjustments for REXULTI ( 2.7 ) |
| Strong CYP2D6 REXULTI may be administered without dosage adjustment in patients with MDD when administered with strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). or CYP3A4 inhibitors |
Administer half of recommended dosage. |
| Strong/moderate CYP2D6 with Strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Known CYP2D6 poor metabolizers taking strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Strong CYP3A4 inducers | Double the recommended dosage and further adjust based on clinical response. |
8.8 Renal Impairment
The maximum recommended dosage in patients with CrCl<60 mL/minute is lower than those with mild renal impairment and those with normal renal function [see Dosage and Administration (2.6)]. Patients with renal impairment had higher exposure to brexpiprazole than patients with normal renal function [see Clinical Pharmacology (12.3)]. Greater exposure may increase the risk of REXULTI-associated adverse reactions.
12.2 Pharmacodynamics
Brexpiprazole has affinity (expressed as Ki) for multiple monoaminergic receptors including serotonin 5-HT1A (0.12 nM), 5-HT2A (0.47 nM), 5-HT2B (1.9 nM), 5-HT7 (3.7 nM), dopamine D2 (0.30 nM), D3 (1.1 nM), and noradrenergic α1A (3.8 nM), α1B (0.17 nM), α1D (2.6 nM), and α2C (0.59 nM) receptors. Brexpiprazole acts as a partial agonist at the 5-HT1A, D2, and D3 receptors and as an antagonist at 5-HT2A, 5-HT2B, 5-HT7, α1A, α1B, α1D, and α2C receptors. Brexpiprazole also exhibits affinity for histamine H1 receptor (19 nM) and for muscarinic M1 receptor (67% inhibition at 10 µM).
5.6 Metabolic Changes
Atypical antipsychotic drugs, including REXULTI, have caused metabolic changes including hyperglycemia, diabetes mellitus, dyslipidemia, and body weight gain. Although all of the drugs in the class to date have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.5 Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. The risk appears to be highest among the elderly, especially elderly women, but it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drugs differ in their potential to cause tardive dyskinesia is unknown.
The risk of tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the cumulative dose increases. The syndrome can develop after relatively brief treatment periods, at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect that symptomatic suppression has upon the long-term course of tardive dyskinesia is unknown.
Given these considerations, REXULTI should be prescribed in a manner most likely to reduce the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that 1) is known to respond to antipsychotic drugs and 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, use the lowest dose and the shortest duration of treatment needed to produce a satisfactory clinical response. Periodically reassess the need for continued treatment.
If signs and symptoms of tardive dyskinesia appear in a patient treated with REXULTI, drug discontinuation should be considered. However, some patients may require treatment with REXULTI despite the presence of the syndrome.
8.7 Hepatic Impairment
The maximum recommended dosage in patients with moderate to severe hepatic impairment (Child-Pugh score ≥7) is lower than those with mild hepatic impairment and those with normal hepatic function [see Dosage and Administration (2.4)]. Patients with moderate to severe hepatic impairment generally had higher exposure to brexpiprazole than patients with normal hepatic function [see Clinical Pharmacology (12.3)]. Greater exposure may increase the risk of REXULTI-associated adverse reactions .
1 Indications and Usage (1 INDICATIONS AND USAGE)
REXULTI is indicated for:
- Adjunctive treatment to antidepressants for major depressive disorder (MDD) in adults
- Treatment of schizophrenia in adults and pediatric patients ages 13 years and older
- Treatment of agitation associated with dementia due to Alzheimer's disease
Limitations of Use:
REXULTI is not indicated as an as needed ("prn") treatment for agitation associated with dementia due to Alzheimer's disease [see Clinical Studies (14.3)].
12.1 Mechanism of Action
The mechanism of action of REXULTI in the adjunctive treatment of major depressive disorder, treatment of agitation associated with dementia due to Alzheimer's disease, or treatment of schizophrenia is unknown. However, the efficacy of REXULTI may be mediated through a combination of partial agonist activity at serotonin 5-HT1A and dopamine D2 receptors, and antagonist activity at serotonin 5-HT2A receptors.
9.1 Controlled Substance
REXULTI contains brexpiprazole, which is not a controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack) (5.3)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring. (5.4)
- Tardive Dyskinesia: Discontinue if clinically appropriate. (5.5)
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. (5.6)
- Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation. (5.7)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or history of leukopenia or neutropenia. Consider discontinuing REXULTI if a clinically significant decline in WBC occurs in absence of other causative factors. (5.8)
- Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope. (5.9)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. (5.11)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery. (5.14)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Administer REXULTI orally once daily with or without food. (2, 12.3)
| Indication | Starting Dosage | Recommended Target Dosage | Maximum Dosage |
|---|---|---|---|
| MDD Adults (2.2) |
0.5 mg/day or 1 mg/day |
2 mg/day | 3 mg/day |
| Schizophrenia Adults (2.3) |
1 mg/day | 2 to 4 mg/day | 4 mg/day |
| Schizophrenia Pediatric (13 - 17 years) (2.3) |
0.5 mg/day | 2 to 4 mg/day | 4 mg/day |
| Agitation associated with dementia due to Alzheimer's disease (2.4) |
0.5 mg/day | 2 mg/day | 3 mg/day |
- Moderate to Severe Hepatic Impairment: Maximum recommended dosage is 2 mg once daily for patients with MDD or agitation associated with dementia due to Alzheimer's disease and 3 mg once daily for patients with schizophrenia. (2.5)
- CrCl<60 mL/minute: Maximum recommended dosage is 2 mg once daily for patients with MDD or agitation associated with dementia due to Alzheimer's disease and 3 mg once daily for patients with schizophrenia. (2.6)
- See Full Prescribing Information for dosage modifications for CYP2D6 poor metabolizers and for concomitant use with CYP inhibitors or inducers. (2.7)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
REXULTI tablets are available in 6 strengths:
- 0.25 mg tablets are light brown, round, shallow convex, bevel-edged body with "BRX" and "0.25" imprinted on one side.
- 0.5 mg tablets: are light orange, round, shallow convex, bevel-edged body with "BRX" and "0.5" imprinted on one side.
- 1 mg tablets are light yellow, round, shallow convex, bevel-edged body with "BRX" and "1" imprinted on one side.
- 2 mg tablets are light green, round, shallow convex, bevel-edged body with "BRX" and "2" imprinted on one side.
- 3 mg tablets are light purple, round, shallow convex, bevel-edged body with "BRX" and "3" imprinted on one side.
- 4 mg tablets are white, round, shallow convex, bevel-edged body with "BRX" and "4" imprinted on one side.
6.2 Postmarketing Experience
The following adverse reaction has been identified during post-approval use of REXULTI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Nervous System disorders: Neuroleptic Malignant Syndrome
8.6 Cyp2d6 Poor Metabolizers (8.6 CYP2D6 Poor Metabolizers)
Dosage adjustment is recommended in known CYP2D6 poor metabolizers because these patients have higher brexpiprazole concentrations than normal metabolizers of CYP2D6. Approximately 8% of Caucasians and 3 to 8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers [see Dosage and Administration (2.7), Clinical Pharmacology (12.3)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: May cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most common adverse reactions in adult patients in clinical trials (≥5%) were weight increased, akathisia, headache, somnolence, and insomnia.
The most common adverse reactions in pediatric patients in clinical trials (≥5%) were weight increased, somnolence, headache, akathisia, and nasopharyngitis.
Brexpiprazole has been evaluated for safety in 12,550 adult patients who participated in multiple-dose clinical trials for major depressive disorder, schizophrenia, agitation associated with dementia due to Alzheimer's disease, attention deficit hyperactivity disorder (ADHD), post-traumatic stress disorder (PTSD), bipolar mania, and borderline personality disorder (BPD). Among them, 3,870 patients were treated with brexpiprazole for at least 180 days, and 1,910 patients were treated for at least one year of exposure.
Additionally, brexpiprazole has been evaluated for safety in 119 pediatric patients who participated in short-term trials, and 314 patients in long-term multiple-dose clinical trials for pediatric schizophrenia and autism spectrum disorders (ASD).
8.9 Other Specific Populations
The recommended dosage for REXULTI is the same in males and females, in different racial groups, and in smokers and nonsmokers [see Clinical Pharmacology (12.3)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide).
5.12 Body Temperature Dysregulation
Atypical antipsychotics may disrupt the body's ability to reduce core body temperature. Strenuous exercise, exposure to extreme heat, dehydration, and anticholinergic medications may contribute to an elevation in core body temperature; use REXULTI with caution in patients who may experience these conditions.
5.9 Orthostatic Hypotension and Syncope
Atypical antipsychotics cause orthostatic hypotension and syncope. Generally, the risk is greatest during initial dose titration and when increasing the dose. In the short-term, placebo-controlled clinical studies of REXULTI plus ADT in adult patients with MDD, the incidence of orthostatic hypotension-related adverse reactions in REXULTI plus ADT-treated patients compared to placebo plus ADT-treated patients included: dizziness (2% versus 2%) and orthostatic hypotension (0.1% versus 0%). In the short-term, placebo-controlled clinical studies of REXULTI in adult patients with schizophrenia, the incidence of orthostatic hypotension-related adverse reactions in REXULTI-treated patients compared to placebo patients included: dizziness (2% versus 2%), orthostatic hypotension (0.4% versus 0.2%), and syncope (0.1% versus 0%). In 12-week, placebo-controlled clinical studies of REXULTI in patients with agitation associated with dementia due to Alzheimer's disease, the incidence of orthostatic hypotension-related adverse reactions in patients treated with REXULTI compared to patients treated with placebo included: dizziness (3% versus 3%), orthostatic hypotension (1% versus 1%), and syncope (0.2% versus 0.8%).
Orthostatic vital signs should be monitored in patients who are vulnerable to hypotension (e.g., elderly patients, patients with dehydration, hypovolemia, concomitant treatment with antihypertensive medication), patients with known cardiovascular disease (history of myocardial infarction, ischemic heart disease, heart failure, or conduction abnormalities), and patients with cerebrovascular disease. REXULTI has not been evaluated in patients with a recent history of myocardial infarction or unstable cardiovascular disease. Such patients were excluded from the premarketing clinical studies.
5.4 Neuroleptic Malignant Syndrome (nms) (5.4 Neuroleptic Malignant Syndrome (NMS))
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with administration of antipsychotic drugs, including REXULTI.
Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmia). Additional signs may include elevated creatinine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue REXULTI and provide intensive symptomatic treatment and monitoring.
5.8 Leukopenia, Neutropenia, and Agranulocytosis
Leukopenia and neutropenia have been reported during treatment with antipsychotic agents. Agranulocytosis (including fatal cases) has been reported with other agents in this class.
Possible risk factors for leukopenia and neutropenia include pre-existing low white blood cell count (WBC) or absolute neutrophil count (ANC) and history of drug-induced leukopenia or neutropenia. In patients with a pre-existing low WBC or ANC or a history of drug-induced leukopenia or neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of REXULTI at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue REXULTI in patients with absolute neutrophil count <1000/mm3 and follow their WBC until recovery.
5.14 Potential for Cognitive and Motor Impairment
REXULTI, like other antipsychotics, may cause somnolence and has the potential to impair judgment, thinking, or motor skills. In the 6-week placebo-controlled clinical studies in patients with MDD, somnolence (including sedation and hypersomnia) was reported in 4% of REXULTI plus ADT-treated patients compared to 1% of placebo plus ADT-treated patients.
In the 6-week placebo-controlled clinical studies in adult patients with schizophrenia, somnolence (including sedation and hypersomnia) was reported in 5% of REXULTI-treated patients compared to 3% of placebo-treated patients.
In the 12-week placebo-controlled, fixed-dose clinical studies in patients (51 to 90 years of age) with agitation associated with dementia due to Alzheimer's disease, somnolence (including sedation) was reported in 3% of patients treated with REXULTI compared to 1% of patients treated with placebo.
Patients should be cautioned about operating hazardous machinery, including motor vehicles, until they are reasonably certain that REXULTI therapy does not affect them adversely.
Principal Display Panel 1 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label)
1 mg
30 Tablets
NDC 59148-037-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 2 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 2 mg Tablet Bottle Label)
2 mg
30 Tablets
NDC 59148-038-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 3 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 3 mg Tablet Bottle Label)
3 mg
30 Tablets
NDC 59148-039-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 4 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 4 mg Tablet Bottle Label)
4 mg
30 Tablets
NDC 59148-040-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 1 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Carton)
1 mg
30 Tablets
NDC 59148-037-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 2 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 2 mg Tablet Bottle Carton)
2 mg
30 Tablets
NDC 59148-038-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 3 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 3 mg Tablet Bottle Carton)
3 mg
30 Tablets
NDC 59148-039-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 4 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 4 mg Tablet Bottle Carton)
4 mg
30 Tablets
NDC 59148-040-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Principal Display Panel 0.5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Bottle Label)
0.5 mg
30 Tablets
NDC 59148-036-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 0.25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Bottle Label)
0.25 mg
30 Tablets
NDC 59148-035-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
Otsuka
Lundbeck
Principal Display Panel 0.5 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Bottle Carton)
0.5 mg
30 Tablets
NDC 59148-036-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
14.1 Adjunctive Treatment of Major Depressive Disorder
The efficacy of REXULTI in the adjunctive treatment of major depressive disorder (MDD) was evaluated in two 6-week double-blind, placebo-controlled, fixed-dose studies of adult patients meeting DSM-IV-TR criteria for MDD, with or without symptoms of anxiety, who had an inadequate response to prior antidepressant therapy (1 to 3 courses) in the current episode and who had also demonstrated an inadequate response throughout the 8 weeks of prospective antidepressant treatment (with escitalopram, fluoxetine, paroxetine controlled-release, sertraline, duloxetine delayed release, or venlafaxine extended release). Inadequate response during the prospective antidepressant treatment phase was defined as having persistent symptoms without substantial improvement throughout the course of treatment.
Patients in Study 1 (NCT01360645) were randomized to REXULTI 2 mg once a day or placebo. Patients in Study 2 (NCT01360632) were randomized to REXULTI 1 or 3 mg once a day or placebo. For patients randomized to REXULTI, all patients initiated treatment at 0.5 mg once daily during Week 1. At Week 2, the REXULTI dosage was increased to 1 mg in all treatment groups, and either maintained at 1 mg or increased to 2 mg or 3 mg once daily, based on treatment assignment, from Week 3 onwards. The dosages were then maintained for the 4 remaining weeks.
The primary endpoint was change from baseline to Week 6 in the Montgomery-Asberg Depression Rating Scale (MADRS), a 10-item clinician-related scale used to assess the degree of depressive symptomatology, with 0 representing no symptoms and 60 representing worst symptoms.
At randomization, the mean MADRS total score was 27. In Studies 1 and 2, REXULTI (plus ADT) 2 mg once daily and 3 mg once daily were superior to placebo plus ADT in reducing mean MADRS total scores. Results from the primary efficacy parameters for both fixed dose studies are shown below in Table 13. Figure 4 below shows the time course of response based on the primary efficacy measure (MADRS) in Study 1.
| Study | Treatment Group | N | Mean Baseline Score (SD) | LS Mean Change from Baseline (SE) | Placebo-subtracted Difference Difference (drug minus placebo) in least-squares mean change from baseline
(95% CI) |
|---|---|---|---|---|---|
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval | |||||
| 1 | REXULTI (2 mg/day) + ADT Dosages statistically significantly superior to placebo
|
175 | 26.9 (5.7) | -8.4 (0.6) | -3.2 (-4.9, -1.5) |
| Placebo + ADT | 178 | 27.3 (5.6) | -5.2 (0.6) | -- | |
| 2 | REXULTI (1 mg/day) + ADT | 211 | 26.5 (5.6) | -7.6 (0.5) | -1.3 (-2.7, 0.1) |
| REXULTI (3 mg/day) + ADT | 213 | 26.5 (5.3) | -8.3 (0.5) | -2.0 (-3.4, -0.5) | |
| Placebo + ADT | 203 | 26.5 (5.2) | -6.3 (0.5) | -- |
An examination of population subgroups did not suggest differential response based on age, gender, race, or choice of prospective antidepressant.
Figure 4 Change from Baseline in MADRS Total Score by Study Visit (Week) in Patients with MDD in Adults (Study 1)
Principal Display Panel 0.25 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Bottle Carton)
0.25 mg
30 Tablets
NDC 59148-035-13
REXULTI®
brexpiprazole
tablets
Rx only
Keep out of the
reach of children
DISPENSE THE
ACCOMPANYING
MEDICATION GUIDE
TO EACH PATIENT
2.6 Recommended Dosage in Patients With Renal Impairment (2.6 Recommended Dosage in Patients with Renal Impairment)
The maximum recommended dosage in patients with creatinine clearance CrCl<60 mL/minute is [see Use in Specific Populations (8.8), Clinical Pharmacology (12.3)].
2 mg orally once daily in patients with MDD or agitation associated with dementia due to Alzheimer's disease and
3 mg orally once daily in patients with schizophrenia
5.7 Pathological Gambling and Other Compulsive Behaviors
Post-marketing case reports suggest that patients can experience intense urges, particularly for gambling, and the inability to control these urges while taking REXULTI. Other compulsive urges, reported less frequently, include sexual urges, shopping, eating, or binge eating, and other impulsive or compulsive behaviors. Because patients may not recognize these behaviors as abnormal, it is important for prescribers to ask patients or their caregivers specifically about the development of new or intense gambling urges, compulsive sexual urges, compulsive shopping, binge or compulsive eating, or other urges while being treated with REXULTI. In some cases, although not all, urges were reported to have stopped when the dose was reduced, or the medication was discontinued. Compulsive behaviors may result in harm to the patient and others if not recognized. Consider dose reduction or stopping the medication if a patient develops such urges.
2.5 Recommended Dosage in Patients With Hepatic Impairment (2.5 Recommended Dosage in Patients with Hepatic Impairment)
The maximum recommended dosage in patients with moderate to severe hepatic impairment (Child-Pugh score ≥7) is [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2 mg orally once daily in patients with MDD or agitation associated with dementia due to Alzheimer's disease, and
3 mg orally once daily in patients with schizophrenia
7.1 Drugs Having Clinically Important Interactions With Rexulti (7.1 Drugs Having Clinically Important Interactions with REXULTI)
See Table 12 for clinically important drug interactions with REXULTI.
| Strong CYP3A4 Inhibitors | |
| Clinical Impact: | Concomitant use of REXULTI with strong CYP3A4 inhibitors increased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with a strong CYP3A4 inhibitor, reduce the REXULTI dosage [see Dosage and Administration (2.7)]. |
|
Strong CYP2D6 Inhibitors In the clinical studies examining the adjunctive use of REXULTI in the treatment of MDD, dosage was not adjusted for strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). Thus, CYP considerations are already factored into general dosing recommendations, and REXULTI may be administered without dosage adjustment in patients with MDD.
|
|
| Clinical Impact: | Concomitant use of REXULTI with strong CYP2D6 inhibitors increased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with a strong CYP2D6 inhibitor, reduce the REXULTI dosage [see Dosage and Administration (2.7)]. |
| Both CYP3A4 Inhibitors and CYP2D6 Inhibitors | |
| Clinical Impact: | Concomitant use of REXULTI with 1) a strong CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 2) a moderate CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 3) a strong CYP3A4 inhibitor and a moderate CYP2D6 inhibitor; or 4) a moderate CYP3A4 inhibitor and a moderate CYP2D6 inhibitor increased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with 1) a strong CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 2) a moderate CYP3A4 inhibitor and a strong CYP2D6 inhibitor; or 3) a strong CYP3A4 inhibitor and a moderate CYP2D6 inhibitor; or 4) a moderate CYP3A4 inhibitor and a moderate CYP2D6 inhibitor, decrease the REXULTI dosage [see Dosage and Administration (2.7)]. |
| Strong CYP3A4 Inducers | |
| Clinical Impact: | Concomitant use of REXULTI and a strong CYP3A4 inducer decreased the exposure of brexpiprazole compared to the use of REXULTI alone [see Clinical Pharmacology (12.3)]. |
| Intervention: | With concomitant use of REXULTI with a strong CYP3A4 inducer, increase the REXULTI dosage [see Dosage and Administration (2.7)]. |
14.3 Agitation Associated With Dementia Due to Alzheimer's Disease (14.3 Agitation Associated with Dementia Due to Alzheimer's Disease)
The efficacy of REXULTI in the treatment of agitation associated with dementia due to Alzheimer's disease was demonstrated in two 12-week, randomized, double-blind, placebo-controlled, fixed-dose studies (Study 6, NCT01862640 and Study 7, NCT03548584). In these studies, patients were required to:
- Have a diagnosis of probable Alzheimer's disease according to NINCDS-ADRDA criteria,
- Have a Mini-Mental State Examination (MMSE) score of ≥5 and ≤22 and have a total score of ≥4 by the agitation/aggression item of the NPI/NPI-NH, and
- Exhibit sufficient agitation behaviors at time of entry to warrant use of pharmacotherapy, after excluding other factors.
Patients in:
- Study 6 were randomized to an oral dosage of either REXULTI 1 mg once a day, REXULTI 2 mg once a day, or placebo. Patients in both REXULTI groups started on 0.25 mg once daily for approximately three days, then received 0.5 mg once daily for approximately 12 days. Subsequently, patients in the 1 mg group received 1 mg once daily for the remainder of the 12-week study, and patients in the 2 mg group received 1 mg once daily for approximately two weeks and then received 2 mg for the remainder of the study.
- Study 7 were randomized to an oral dose of either REXULTI 2 mg or 3 mg once a day (combined treatment arm) or placebo. Patients in both REXULTI groups started on 0.5 mg once daily for 7 days, then received 1 mg once daily for 7 days and then 2 mg once daily for 14 days. Subsequently, patients in the 2 mg group received 2 mg once daily for the remainder of the 12-week study, and patients in the 3 mg group received 3 mg once daily for the remainder of the study.
Study 6 included 433 patients with a mean age of 74 years old, and a range of 51 and 90 years old; 45% were male; 96%, 3%, and 1%, were White, Black or African American, and Asian, respectively; and 16% and 83% were Latino/Hispanic and not Latino/Hispanic, respectively. Study 7 included 345 patients with a mean age of 74 years old, and a range of 56 and 90 years old; 44% were male; 95%, 4%, and 1% were White, Black or African American, and Asian, respectively; and 31% and 69% were Latino/Hispanic and not Latino/Hispanic, respectively.
The primary efficacy endpoint in these two studies was the change from baseline in the Cohen-Mansfield Agitation Inventory total (CMAI) score at Week 12. The CMAI is a clinician rated questionnaire consisting of 29 items, which assess the frequency of manifestations of agitated behaviors in elderly patients, based on caregiver input. Three specific factors can be derived from the CMAI scale: 1) Aggressive Behavior (e.g., screaming, throwing things, cursing/verbal aggression, kicking, pushing scratching, hurting self or others); 2) Physically Non-Aggressive Behavior (e.g., repetitive mannerisms, general restlessness, pacing); and 3) Verbally Agitated Behavior (e.g., complaining, repetitive questions, constant requests for attention). Each CMAI behavior was rated on a scale of 1 (never) to 7 (very frequent agitated behaviors); the total CMAI scores range from 29 (best) to 203 (worst). A negative change indicates improvement.
In Trial 6, patients in the REXULTI 2 mg group showed improved total CMAI scores compared to patients in the placebo group at Week 12. In Trial 7, patients in the REXULTI 2 mg/3 mg group showed improved total CMAI scores compared to patients in the placebo group at Week 12.
As shown in Table 16 and Figure 8, the mean change from baseline in the total CMAI score after 12 weeks in the 2 mg/or 3 mg REXULTI group was statistically significantly superior to the placebo group. The 1 mg REXULTI group did not demonstrate significantly greater mean changes at baseline from the placebo group in the total CMAI score in this patient population. The 1 mg once day REXULTI dosage is not approved and is not recommended for the treatment of agitation associated with dementia due to Alzheimer's disease [see Dosage and Administration (2.4)].
| Study | Treatment Group | N | Mean Baseline Score (SD) |
LS Mean Change from Baseline (SE) |
Placebo-subtracted Difference Difference (drug minus placebo) in least-squares mean change from baseline
(95% CI) |
|---|---|---|---|---|---|
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval | |||||
| 6 | REXULTI 1 mg/day | 134 | 70.5 (16.0) | -17.6 (1.3) | 0.2 (-3.4, 3.9) |
| REXULTI 2 mg/day Dosages statistically significantly superior to placebo.
|
138 | 71.0 (16.6) | -21.6 (1.3) | -3.8 (-7.4, -0.2) | |
| Placebo | 131 | 72.2 (17.9) | -17.8 (1.3) | — | |
| 7 | REXULTI 2 mg/day or 3 mg/day | 225 | 80.6 (16.6) | -22.6 (1.1) | -5.3 (-8.8, -1.9) |
| Placebo | 116 | 79.2 (17.5) | -17.3 (1.4) | — |
Figure 8: Change from Baseline in Total CMAI Score by Study Week in Patients with Agitation Associated with Dementia Due to Alzheimer's Disease (Study 7)
7.2 Drugs Having No Clinically Important Interactions With Rexulti (7.2 Drugs Having No Clinically Important Interactions with REXULTI)
Based on pharmacokinetic studies, no dosage adjustment of REXULTI is required when administered concomitantly with CYP2B6 inhibitors (e.g., ticlopidine) or gastric pH modifiers (e.g., omeprazole). Additionally, no dosage adjustment for substrates of CYP2D6 (e.g., dextromethorphan), CYP3A4 (e.g., lovastatin), CYP2B6 (e.g., bupropion), BCRP (e.g., rosuvastatin), or P-gp (e.g., fexofenadine) is required when administered concomitantly with REXULTI.
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis (5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in the drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group.
Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Boxed Warning, Warnings and Precautions (5.3)].
5.2 Suicidal Thoughts and Behaviors in Children, Adolescents, and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and over 4,400 pediatric patients, the incidence of suicidal thoughts and behaviors in patients 24 years of age and younger was greater in antidepressant-treated patients than in placebo-treated patients. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1,000 patients treated are provided in Table 2.
No suicides occurred in any of the pediatric studies. There were suicides in the adult studies, but the number was not sufficient to reach any conclusion about antidepressant drug effect on suicide.
| Age Range (years) | Drug-Placebo Difference in Number of Patients with Suicidal Thoughts or Behaviors per 1,000 Patients Treated |
|---|---|
| Increases Compared to Placebo | |
| <18 | 14 additional patients |
| 18 to 24 | 5 additional patients |
| Decreases Compared to Placebo | |
| 25 to 64 | 1 fewer patient |
| ≥65 | 6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance studies in adults with MDD that antidepressants delay the recurrence of depression.
Monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing REXULTI, in patients whose depression is persistently worse or who are experiencing emergent suicidal thoughts or behaviors.
2.2 Recommended Dosage for Adjunctive Treatment of Major Depressive Disorder (adults) (2.2 Recommended Dosage for Adjunctive Treatment of Major Depressive Disorder (Adults))
The recommended starting REXULTI dosage for the adjunctive treatment of MDD in adults is 0.5 mg or 1 mg orally once daily. Titrate to 1 mg once daily, then titrate to the target dosage of 2 mg once daily (based on the patient's clinical response and tolerability, increase the dosage at weekly intervals). The maximum recommended daily dosage is 3 mg. Periodically reassess to determine the continued need and appropriate dosage for treatment.
2.4 Recommended Dosage for Agitation Associated With Dementia Due to Alzheimer's Disease (2.4 Recommended Dosage for Agitation Associated with Dementia Due to Alzheimer's Disease)
The recommended starting REXULTI dosage for the treatment of agitation associated with dementia due to Alzheimer's disease is 0.5 mg orally once daily on Days 1 to 7. Increase the dosage on Days 8 through 14 to 1 mg once daily, and on Day 15 to 2 mg once daily. The recommended target dose is 2 mg once daily. The dosage can be increased to the maximum recommended daily dosage of 3 mg once daily after at least 14 days, based on clinical response and tolerability.
5.3 Cerebrovascular Adverse Reactions Including Stroke in Elderly Patients With Dementia Related Psychosis (5.3 Cerebrovascular Adverse Reactions Including Stroke in Elderly Patients with Dementia-Related Psychosis)
In placebo-controlled trials in elderly patients with dementia, patients randomized to risperidone, aripiprazole, and olanzapine had a higher incidence of stroke and transient ischemic attack, including fatal stroke. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease [see Boxed Warning, Warnings and Precautions (5.1)].
2.7 Dosage Modifications for Cyp2d6 Poor Metabolizers and for Concomitant Use With Cyp Inhibitors Or Inducers (2.7 Dosage Modifications for CYP2D6 Poor Metabolizers and for Concomitant Use with CYP Inhibitors or Inducers)
Dosage modifications are recommended in patients who are known cytochrome P450 (CYP) 2D6 poor metabolizers and in patients taking concomitant CYP3A4 inhibitors, CYP2D6 inhibitors, or strong CYP3A4 inducers (see Table 1). If the concomitant drug is discontinued, adjust the REXULTI dosage to its original level. If the concomitant CYP3A4 inducer is discontinued, reduce the REXULTI dosage to the original level over 1 to 2 weeks [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
| Factors | Adjusted REXULTI Dosage |
|---|---|
| CYP2D6 Poor Metabolizers | |
| CYP2D6 poor metabolizers | Administer half of the recommended dosage. |
| Known CYP2D6 poor metabolizers taking strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Patients Taking CYP2D6 Inhibitors and/or CYP3A4 Inhibitors | |
| Strong CYP2D6 inhibitors In the clinical studies examining the use of REXULTI for the adjunctive treatment of MDD, dosage was not adjusted for strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine). Thus, CYP considerations are already factored into general dosing recommendations, and REXULTI may be administered without dosage adjustment in patients with MDD.
|
Administer half of the recommended dosage. |
| Strong CYP3A4 inhibitors | Administer half of the recommended dosage. |
| Strong/moderate CYP2D6 inhibitors with strong/moderate CYP3A4 inhibitors | Administer a quarter of the recommended dosage. |
| Patients Taking CYP3A4 Inducers | |
| Strong CYP3A4 inducers | Double the recommended dosage over 1 to 2 weeks. |
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis and Suicidal Thoughts and Behaviors (WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS)
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS
See full prescribing information for complete boxed warning.
- Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at increased risk of death. REXULTI is not approved for the treatment of patients with dementia-related psychosis without agitation associated with dementia due to Alzheimer's disease. (5.1)
- Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Safety and effectiveness of REXULTI have not been established in pediatric patients with MDD. (5.2, 8.4)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:48.630519 · Updated: 2026-03-14T22:28:06.461439