These Highlights Do Not Include All The Information Needed To Use Dalbavancin For Injection Safely And Effectively. See Full Prescribing Information For Dalbavancin For Injection.
2c2ec45e-48a2-4887-8ab0-9244eb39a394
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dalbavancin for injection is a lipoglycopeptide antibacterial indicated for the treatment of adult and pediatric patients with acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible strains of Gram- positive microorganisms. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of dalbavancin for injection and other antibacterial drugs, dalbavancin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
Dosage and Administration
Dosage in Adult Patients ( 2.1 , 2.3 ) : Estimated Creatinine Clearance (CLcr) Single Dose Regimen 30 mL/min and above or on regular hemodialysis 1,500 mg Less than 30 mL/min and not on regular hemodialysis 1,125 mg Administer by intravenous infusion over 30 minutes ( 2.1 , 2.4 ) See Full Prescribing Information for instructions on reconstitution of lyophilized powder and preparation of injection ( 2.4 ) Dosage in Pediatric Patients with CLcr 30 mL/min/1.73m 2 and above ( 2.2 ) Age Range Dosage (Single Dose Regimen) Birth to less than 6 years 22.5 mg/kg (maximum of 1,500 mg) 6 to less than 18 years 18 mg/kg (maximum of 1,500 mg) Dosage adjustment in pediatric patients with CLcr less than 30 mL/min has not been studied.
Warnings and Precautions
Serious hypersensitivity (anaphylactic) and skin reactions have been reported in patients treated with dalbavancin for injection. If an allergic reaction occurs, discontinue treatment with dalbavancin for injection and institute appropriate therapy for the allergic reaction. Carefully monitor patients with known hypersensitivity to glycopeptides. ( 5.1 ) Rapid intravenous infusion of dalbavancin for injection can cause flushing of the upper body, urticaria, pruritus, rash, and/or back pain. Stopping or slowing the infusion may result in cessation of these reactions. ( 5.2 ) Alanine Aminotransferase (ALT) elevations with dalbavancin for injection treatment were reported in clinical trials. ( 5.3 , 6.1 ) Clostridioides difficile -associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including dalbavancin for injection. Evaluate if diarrhea occurs. ( 5.4 )
Contraindications
Dalbavancin for injection is contraindicated in patients with known hypersensitivity to dalbavancin.
Adverse Reactions
The following clinically significant adverse reactions are also discussed elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Infusion Related Reactions [see Warnings and Precautions ( 5.2 )] Hepatic Effects [see Warnings and Precautions ( 5.3 )] Clostridioides difficile -associated Diarrhea [see Warnings and Precautions ( 5.4 )]
Drug Interactions
No clinical drug-drug interaction studies have been conducted with dalbavancin for injection. There is minimal potential for drug-drug interactions between dalbavancin for injection and cytochrome P450 (CYP450) substrates, inhibitors, or inducers [see Clinical Pharmacology ( 12.3 )] .
Storage and Handling
Dalbavancin for injection is supplied as a white/off-white to pale yellow sterile lyophilized powder in a single-dose glass vial containing dalbavancin hydrochloride equivalent to 500 mg dalbavancin: Product Code Unit of Sale Strength 221012 NDC 65219-120-12 Individually packaged 500 mg per single-dose vial Dalbavancin for injection should be stored at 25ºC (77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Storage of the reconstituted and diluted solutions of dalbavancin for injection are described elsewhere in the prescribing information [see Dosage and Administration ( 2.4 )]. The container closure is not made with natural rubber latex.
How Supplied
Dalbavancin for injection is supplied as a white/off-white to pale yellow sterile lyophilized powder in a single-dose glass vial containing dalbavancin hydrochloride equivalent to 500 mg dalbavancin: Product Code Unit of Sale Strength 221012 NDC 65219-120-12 Individually packaged 500 mg per single-dose vial Dalbavancin for injection should be stored at 25ºC (77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Storage of the reconstituted and diluted solutions of dalbavancin for injection are described elsewhere in the prescribing information [see Dosage and Administration ( 2.4 )]. The container closure is not made with natural rubber latex.
Medication Information
Warnings and Precautions
Serious hypersensitivity (anaphylactic) and skin reactions have been reported in patients treated with dalbavancin for injection. If an allergic reaction occurs, discontinue treatment with dalbavancin for injection and institute appropriate therapy for the allergic reaction. Carefully monitor patients with known hypersensitivity to glycopeptides. ( 5.1 ) Rapid intravenous infusion of dalbavancin for injection can cause flushing of the upper body, urticaria, pruritus, rash, and/or back pain. Stopping or slowing the infusion may result in cessation of these reactions. ( 5.2 ) Alanine Aminotransferase (ALT) elevations with dalbavancin for injection treatment were reported in clinical trials. ( 5.3 , 6.1 ) Clostridioides difficile -associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including dalbavancin for injection. Evaluate if diarrhea occurs. ( 5.4 )
Dosage and Administration
Dosage in Adult Patients ( 2.1 , 2.3 ) : Estimated Creatinine Clearance (CLcr) Single Dose Regimen 30 mL/min and above or on regular hemodialysis 1,500 mg Less than 30 mL/min and not on regular hemodialysis 1,125 mg Administer by intravenous infusion over 30 minutes ( 2.1 , 2.4 ) See Full Prescribing Information for instructions on reconstitution of lyophilized powder and preparation of injection ( 2.4 ) Dosage in Pediatric Patients with CLcr 30 mL/min/1.73m 2 and above ( 2.2 ) Age Range Dosage (Single Dose Regimen) Birth to less than 6 years 22.5 mg/kg (maximum of 1,500 mg) 6 to less than 18 years 18 mg/kg (maximum of 1,500 mg) Dosage adjustment in pediatric patients with CLcr less than 30 mL/min has not been studied.
Contraindications
Dalbavancin for injection is contraindicated in patients with known hypersensitivity to dalbavancin.
Adverse Reactions
The following clinically significant adverse reactions are also discussed elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Infusion Related Reactions [see Warnings and Precautions ( 5.2 )] Hepatic Effects [see Warnings and Precautions ( 5.3 )] Clostridioides difficile -associated Diarrhea [see Warnings and Precautions ( 5.4 )]
Drug Interactions
No clinical drug-drug interaction studies have been conducted with dalbavancin for injection. There is minimal potential for drug-drug interactions between dalbavancin for injection and cytochrome P450 (CYP450) substrates, inhibitors, or inducers [see Clinical Pharmacology ( 12.3 )] .
Storage and Handling
Dalbavancin for injection is supplied as a white/off-white to pale yellow sterile lyophilized powder in a single-dose glass vial containing dalbavancin hydrochloride equivalent to 500 mg dalbavancin: Product Code Unit of Sale Strength 221012 NDC 65219-120-12 Individually packaged 500 mg per single-dose vial Dalbavancin for injection should be stored at 25ºC (77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Storage of the reconstituted and diluted solutions of dalbavancin for injection are described elsewhere in the prescribing information [see Dosage and Administration ( 2.4 )]. The container closure is not made with natural rubber latex.
How Supplied
Dalbavancin for injection is supplied as a white/off-white to pale yellow sterile lyophilized powder in a single-dose glass vial containing dalbavancin hydrochloride equivalent to 500 mg dalbavancin: Product Code Unit of Sale Strength 221012 NDC 65219-120-12 Individually packaged 500 mg per single-dose vial Dalbavancin for injection should be stored at 25ºC (77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Storage of the reconstituted and diluted solutions of dalbavancin for injection are described elsewhere in the prescribing information [see Dosage and Administration ( 2.4 )]. The container closure is not made with natural rubber latex.
Description
Dalbavancin for injection is a lipoglycopeptide antibacterial indicated for the treatment of adult and pediatric patients with acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible strains of Gram- positive microorganisms. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of dalbavancin for injection and other antibacterial drugs, dalbavancin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
Section 42229-5
Dilution:
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Dalbavancin for Injection 500 mg Vial Label
NDC 65219-120-12
Dalbavancin
for Injection
500 mg per vial
For Intravenous Infusion Only
Sterile Single-Dose Vial
Discard Unused Portion
Rx only
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of dalbavancin for injection and other antibacterial agents, dalbavancin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
Specific information is not available on the treatment of overdose with dalbavancin for injection, as dose- limiting toxicity has not been observed in clinical studies. In Phase 1 studies, healthy volunteers have been administered cumulative doses of up to 4,500 mg over a period of up to 8 weeks (not an approved dosing regimen), with no signs of toxicity or laboratory results of clinical concern.
Treatment of overdose with dalbavancin for injection should consist of observation and general supportive measures. Although no information is available specifically regarding the use of hemodialysis to treat overdose, in a Phase 1 study in patients with renal impairment less than 6% of the recommended dalbavancin dose was removed [see Clinical Pharmacology (12.3)].
11 Description
Dalbavancin for injection is a lipoglycopeptide antibacterial synthesized from a fermentation product of Nonomuraea species.
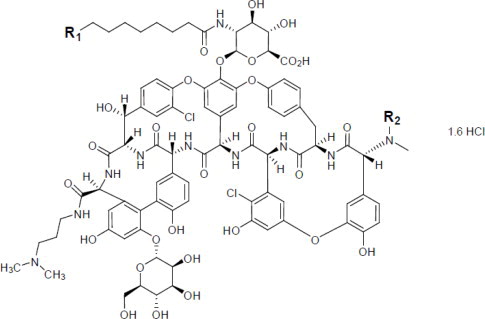
Dalbavancin is a mixture of five closely related active homologs (A0, A1, B0, B1, and B2); the component B0 is the major component of dalbavancin. The homologs share the same core structure and differ in the fatty acid side chain of the N-acylaminoglucuronic acid moiety (R1) structure and/or the presence of an additional methyl group (R2) on the terminal amino group (shown in the Figure 1 and Table 3 below).
|
*Anhydrous free base |
||||
| Dalbavancin | R1 | R2 | Molecular Formula | Molecular Weight* |
| A0 | CH(CH3)2 | H | C87H98N10O28Cl2 · 1.6 HCl | 1,802.7 |
| A1 | CH2CH2CH3 | H | C87H98N10O28Cl2 · 1.6 HCl | 1,802.7 |
| B0 | CH2CH(CH3)2 | H | C88H100N10O28Cl2 · 1.6 HCl | 1,816.7 |
| B1 | CH2CH2CH2CH3 | H | C88H100N10O28Cl2 · 1.6 HCl | 1,816.7 |
| B2 | CH2CH(CH3)2 | CH3 | C89H102N10O28Cl2 · 1.6 HCl | 1,830.7 |
The B0 INN chemical name is: 5,31-dichloro-38-de(methoxycarbonyl)-7-demethyl-19-deoxy-56-O- [2-deoxy-2-[(10-methylundecanoyl)amino]-β-D-glucopyranuronosyl]-38-[[3-(dimethylamino)propyl] carbamoyl]-42-O-α-D-mannopyranosyl-15-N-methyl(ristomycin A aglycone) hydrochloride.
Dalbavancin for injection is supplied in clear glass vials as a sterile, lyophilized, preservative-free, white to off-white to pale yellow solid. Each vial contains dalbavancin HCl equivalent to 500 mg of dalbavancin as the free base, plus lactose monohydrate (129 mg) and mannitol (129 mg) as excipients. Sodium hydroxide or hydrochloric acid may be added to adjust the pH at the time of manufacture. The powder is to be reconstituted and further diluted for IV infusion [see Dosage and Administration (2.4), How Supplied/Storage and Handling (16)].
8.4 Pediatric Use
The safety and effectiveness of dalbavancin for injection for the treatment of ABSSSI has been established in pediatric patients aged birth to less than 18 years. Use of dalbavancin for injection for this indication is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic and safety data in pediatric patients aged birth to less than 18 years [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
There is insufficient information to recommend dosage adjustment for pediatric patients with ABSSSI and CLcr less than 30 mL/min/1.73m2 [see Dosage and Administration (2.2)].
8.5 Geriatric Use
Of the 2,473 patients treated with dalbavancin for injection in Phase 2 and 3 clinical trials, 403 patients (16.3%) were 65 years of age or older. The efficacy and tolerability of dalbavancin for injection were similar to comparator regardless of age. The pharmacokinetics of dalbavancin for injection was not significantly altered with age; therefore, no dosage adjustment is necessary based on age alone.
Dalbavancin for injection is substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection in this age group.
4 Contraindications
Dalbavancin for injection is contraindicated in patients with known hypersensitivity to dalbavancin.
5.3 Hepatic Effects
In Phase 2 and 3 clinical trials, more dalbavancin for injection than comparator-treated subjects with normal baseline transaminase levels had post-baseline alanine aminotransferase (ALT) elevation greater than 3 times the upper limit of normal (ULN). Overall, abnormalities in liver tests (ALT, AST, bilirubin) were reported with similar frequency in the dalbavancin for injection and comparator arms [see Adverse Reactions (6.1)].
6 Adverse Reactions
The following clinically significant adverse reactions are also discussed elsewhere in the labeling:
8.6 Renal Impairment
In patients with renal impairment whose known CLcr is less than 30 mL/min and who are not receiving regularly scheduled hemodialysis, the recommended regimen for dalbavancin for injection is 1,125 mg, administered as a single dose. No dosage adjustment is recommended for patients receiving regularly scheduled hemodialysis, and dalbavancin for injection can be administered without regard to the timing of hemodialysis. There is insufficient information to recommend dosage adjustment for pediatric patients younger than 18 years with CLcr less than 30 mL/min/1.73m2 [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
The antibacterial activity of dalbavancin appears to best correlate with the ratio of area under the concentration-time curve to minimal inhibitory concentration (AUC/MIC) for Staphylococcus aureus based on animal models of infection [see Dosage and Administration (2.1), Clinical Pharmacology (12.3)].
1 Indication and Usage
Dalbavancin for injection is a lipoglycopeptide antibacterial indicated for the treatment of adult and pediatric patients with acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible strains of Gram- positive microorganisms. ( 1.1 )
To reduce the development of drug-resistant bacteria and maintain the effectiveness of dalbavancin for injection and other antibacterial drugs, dalbavancin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
8.7 Hepatic Impairment
No dosage adjustment of dalbavancin for injection is recommended for patients with mild hepatic impairment (Child-Pugh Class A). Caution should be exercised when prescribing dalbavancin for injection to patients with moderate or severe hepatic impairment (Child-Pugh Class B or C) as no data are available to determine the appropriate dosing in these patients [see Clinical Pharmacology (12.3)].
12.1 Mechanism of Action
Dalbavancin is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions
- Serious hypersensitivity (anaphylactic) and skin reactions have been reported in patients treated with dalbavancin for injection. If an allergic reaction occurs, discontinue treatment with dalbavancin for injection and institute appropriate therapy for the allergic reaction. Carefully monitor patients with known hypersensitivity to glycopeptides. ( 5.1 )
- Rapid intravenous infusion of dalbavancin for injection can cause flushing of the upper body, urticaria, pruritus, rash, and/or back pain. Stopping or slowing the infusion may result in cessation of these reactions. ( 5.2 )
- Alanine Aminotransferase (ALT) elevations with dalbavancin for injection treatment were reported in clinical trials. ( 5.3, 6.1 )
- Clostridioides difficile-associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including dalbavancin for injection. Evaluate if diarrhea occurs. ( 5.4 )
7.2 Drug Drug Interactions
No clinical drug-drug interaction studies have been conducted with dalbavancin for injection. There is minimal potential for drug-drug interactions between dalbavancin for injection and cytochrome P450 (CYP450) substrates, inhibitors, or inducers [see Clinical Pharmacology (12.3)].
2 Dosage and Administration
| Dosage in Adult Patients ( 2.1 , 2.3): | |
| Estimated Creatinine Clearance (CLcr) | Single Dose Regimen |
| 30 mL/min and above or on regular hemodialysis |
1,500 mg |
| Less than 30 mL/min and not on regular hemodialysis |
1,125 mg |
- Administer by intravenous infusion over 30 minutes ( 2.1 , 2.4 )
- See Full Prescribing Information for instructions on reconstitution of lyophilized powder and preparation of injection ( 2.4 )
| Dosage in Pediatric Patients with CLcr 30 mL/min/1.73m 2 and above ( 2.2 ) | |
| Age Range | Dosage (Single Dose Regimen) |
| Birth to less than 6 years | 22.5 mg/kg (maximum of 1,500 mg) |
| 6 to less than 18 years | 18 mg/kg (maximum of 1,500 mg) |
- Dosage adjustment in pediatric patients with CLcr less than 30 mL/min has not been studied.
3 Dosage Forms and Strengths
Dalbavancin for injection is supplied as a white/off-white to pale yellow lyophilized sterile powder for reconstitution in a single-dose clear glass vial containing dalbavancin hydrochloride equivalent to 500 mg of dalbavancin.
5.4 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported in users of nearly all systemic antibacterial drugs, including dalbavancin for injection, with severity ranging from mild diarrhea to fatal colitis. Treatment with antibacterial agents can alter the normal flora of the colon, and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile should be discontinued, if possible. Appropriate measures such as fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Post Marketing Experience
The following adverse reaction has been identified during post-approval use of dalbavancin. Because the reaction is reported voluntarily from a population of uncertain size, it is not possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
General disorders and administration site conditions: Back pain as an infusion-related reaction [See Warnings and Precautions ( 5.2 )].
5.1 Hypersensitivity Reactions
Serious hypersensitivity (anaphylactic) and skin reactions have been reported in patients treated with dalbavancin for injection. If an allergic reaction to dalbavancin for injection occurs, discontinue treatment with dalbavancin for injection and institute appropriate therapy for the allergic reaction. Before using dalbavancin for injection, inquire carefully about previous hypersensitivity reactions to other glycopeptides. Due to the possibility of cross-sensitivity, carefully monitor for signs of hypersensitivity during treatment with dalbavancin for injection in patients with a history of glycopeptide allergy [see Patient Counseling Information (17)].
5.2 Infusion Related Reactions
Dalbavancin for injection is administered via intravenous infusion, using a total infusion time of 30 minutes to minimize the risk of infusion-related reactions. Rapid intravenous infusions of dalbavancin for injection can cause flushing of the upper body, urticaria, pruritus, rash, and/or back pain. Stopping or slowing the infusion may result in cessation of these reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of dalbavancin for injection cannot be directly compared to rates in the clinical trials of another drug and may not reflect rates observed in practice.
2.4 Preparation and Administration
Dalbavancin for injection must be reconstituted with either Sterile Water for Injection, USP, or 5% Dextrose Injection, USP, and subsequently diluted only with 5% Dextrose Injection, USP, to a final concentration of 1 mg/mL to 5 mg/mL. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Reconstitution: dalbavancin for injection must be reconstituted under aseptic conditions, using 25 mL of either Sterile Water for Injection, USP, or 5% Dextrose Injection, USP, for each 500 mg vial. To avoid foaming, alternate between gentle swirling and inversion of the vial until its contents are completely dissolved. Do not shake. The reconstituted vial contains 20 mg/mL dalbavancin as a clear, colorless to yellow solution.
Reconstituted vials may be stored either refrigerated at 2°C to 8°C (36°F to 46°F), or at controlled room temperature 20°C to 25°C (68°F to 77°F). Do not freeze.
16 How Supplied/storage and Handling
Dalbavancin for injection is supplied as a white/off-white to pale yellow sterile lyophilized powder in a single-dose glass vial containing dalbavancin hydrochloride equivalent to 500 mg dalbavancin:
| Product Code | Unit of Sale | Strength |
| 221012 | NDC 65219-120-12 Individually packaged |
500 mg per single-dose vial |
Dalbavancin for injection should be stored at 25ºC (77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Storage of the reconstituted and diluted solutions of dalbavancin for injection are described elsewhere in the prescribing information [see Dosage and Administration (2.4)].
The container closure is not made with natural rubber latex.
7.1 Drug Laboratory Test Interactions
Drug-laboratory test interactions have not been reported. Dalbavancin for injection at therapeutic concentrations does not artificially prolong prothrombin time (PT) or activated partial thromboplastin time (aPTT).
13.2 Animal Toxicology And/or Pharmacology
Increases in serum levels of liver enzymes (ALT, AST), associated with microscopic findings in the liver were noted in toxicology studies in rats and dogs where dalbavancin was administered daily for 28 to 90 days. Hepatocellular necrosis was observed in dogs dosed at ≥10 mg/kg/day for longer than 2 months, i.e., at approximately 5 to 7 times the expected human dose on an exposure basis. Histiocytic vacuolation and hepatocyte necrosis were observed in rats dosed daily at 40 and 80 mg/kg/day, respectively, for 4 weeks, (approximately 3 and 6 times the expected human dose on an exposure basis, respectively). In addition, renal toxicity characterized by increases in serum BUN and creatinine and microscopic kidney findings was observed in rats and dogs at doses 5 to 7 times the expected human dose on an exposure basis. The relationship between these findings in the animal toxicology studies after 28 and 90 consecutive days of dosing to the indicated clinical dosing of 2 doses 7 days apart are unclear.
5.5 Development of Drug Resistant Bacteria
Prescribing dalbavancin for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
1.1 Acute Bacterial Skin and Skin Structure Infections
Dalbavancin for injection is indicated for the treatment of adult and pediatric patients with acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible strains of the following Gram-positive microorganisms: Staphylococcus aureus (including methicillin-susceptible and methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiae, Streptococcus anginosus group (including S. anginosus, S. intermedius, S. constellatus) and Enterococcus faecalis (vancomycin susceptible isolates).
2.3 Dosage Adjustments in Adult Patients With Clcr Less Than 30 Ml/min
In adult patients with renal impairment whose known CLcr is less than 30 mL/min and who are not receiving regularly scheduled hemodialysis, the recommended dosage regimen of dalbavancin for injection is 1,125 mg, administered as a single dose regimen.
No dosage adjustment is recommended for adult patients receiving regularly scheduled hemodialysis, and dalbavancin for injection can be administered without regard to the timing of hemodialysis [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
2.1 Recommended Dosage Regimen in Adult Patients With Clcr 30 Ml/min and Above
The recommended dosage regimen of dalbavancin for injection in adult patients with CLcr 30 mL/min and above is 1,500 mg, administered as a single dose regimen. Administer dalbavancin for injection over 30 minutes by intravenous infusion. For adult patients with CLcr less than 30 mL/min, dosage adjustment is required [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
2.2 Recommended Dosage Regimen in Pediatric Patients With Clcr 30 Ml/min/1.73m2
The recommended dosage regimen of dalbavancin for injection in pediatric patients with CLcr 30 mL/min/1.73m2 and above is a single dose regimen based on the age and weight of the pediatric patient (Table 1). Administer dalbavancin for injection over 30 minutes by intravenous infusion.
There is insufficient information to recommend dosage adjustment for pediatric patients younger than 18 years with CLcr less than 30 mL/min/1.73m2 [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.3)].
|
*Estimate CLcr or glomerular filtration rate (GFR) using an age-appropriate equation accepted for pediatric patients (birth to less than 18 years old) to define renal function impairment. |
|
| Age Range | Dosage (Single Dose Regimen) |
| Birth to less than 6 years | 22.5 mg/kg (maximum 1,500 mg) |
| 6 to less than 18 years | 18 mg/kg (maximum 1,500 mg) |
Structured Label Content
Section 42229-5 (42229-5)
Dilution:
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Dalbavancin for Injection 500 mg Vial Label
NDC 65219-120-12
Dalbavancin
for Injection
500 mg per vial
For Intravenous Infusion Only
Sterile Single-Dose Vial
Discard Unused Portion
Rx only
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of dalbavancin for injection and other antibacterial agents, dalbavancin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
Specific information is not available on the treatment of overdose with dalbavancin for injection, as dose- limiting toxicity has not been observed in clinical studies. In Phase 1 studies, healthy volunteers have been administered cumulative doses of up to 4,500 mg over a period of up to 8 weeks (not an approved dosing regimen), with no signs of toxicity or laboratory results of clinical concern.
Treatment of overdose with dalbavancin for injection should consist of observation and general supportive measures. Although no information is available specifically regarding the use of hemodialysis to treat overdose, in a Phase 1 study in patients with renal impairment less than 6% of the recommended dalbavancin dose was removed [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Dalbavancin for injection is a lipoglycopeptide antibacterial synthesized from a fermentation product of Nonomuraea species.
Dalbavancin is a mixture of five closely related active homologs (A0, A1, B0, B1, and B2); the component B0 is the major component of dalbavancin. The homologs share the same core structure and differ in the fatty acid side chain of the N-acylaminoglucuronic acid moiety (R1) structure and/or the presence of an additional methyl group (R2) on the terminal amino group (shown in the Figure 1 and Table 3 below).
|
*Anhydrous free base |
||||
| Dalbavancin | R1 | R2 | Molecular Formula | Molecular Weight* |
| A0 | CH(CH3)2 | H | C87H98N10O28Cl2 · 1.6 HCl | 1,802.7 |
| A1 | CH2CH2CH3 | H | C87H98N10O28Cl2 · 1.6 HCl | 1,802.7 |
| B0 | CH2CH(CH3)2 | H | C88H100N10O28Cl2 · 1.6 HCl | 1,816.7 |
| B1 | CH2CH2CH2CH3 | H | C88H100N10O28Cl2 · 1.6 HCl | 1,816.7 |
| B2 | CH2CH(CH3)2 | CH3 | C89H102N10O28Cl2 · 1.6 HCl | 1,830.7 |
The B0 INN chemical name is: 5,31-dichloro-38-de(methoxycarbonyl)-7-demethyl-19-deoxy-56-O- [2-deoxy-2-[(10-methylundecanoyl)amino]-β-D-glucopyranuronosyl]-38-[[3-(dimethylamino)propyl] carbamoyl]-42-O-α-D-mannopyranosyl-15-N-methyl(ristomycin A aglycone) hydrochloride.
Dalbavancin for injection is supplied in clear glass vials as a sterile, lyophilized, preservative-free, white to off-white to pale yellow solid. Each vial contains dalbavancin HCl equivalent to 500 mg of dalbavancin as the free base, plus lactose monohydrate (129 mg) and mannitol (129 mg) as excipients. Sodium hydroxide or hydrochloric acid may be added to adjust the pH at the time of manufacture. The powder is to be reconstituted and further diluted for IV infusion [see Dosage and Administration (2.4), How Supplied/Storage and Handling (16)].
8.4 Pediatric Use
The safety and effectiveness of dalbavancin for injection for the treatment of ABSSSI has been established in pediatric patients aged birth to less than 18 years. Use of dalbavancin for injection for this indication is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic and safety data in pediatric patients aged birth to less than 18 years [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
There is insufficient information to recommend dosage adjustment for pediatric patients with ABSSSI and CLcr less than 30 mL/min/1.73m2 [see Dosage and Administration (2.2)].
8.5 Geriatric Use
Of the 2,473 patients treated with dalbavancin for injection in Phase 2 and 3 clinical trials, 403 patients (16.3%) were 65 years of age or older. The efficacy and tolerability of dalbavancin for injection were similar to comparator regardless of age. The pharmacokinetics of dalbavancin for injection was not significantly altered with age; therefore, no dosage adjustment is necessary based on age alone.
Dalbavancin for injection is substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection in this age group.
4 Contraindications (4 CONTRAINDICATIONS)
Dalbavancin for injection is contraindicated in patients with known hypersensitivity to dalbavancin.
5.3 Hepatic Effects
In Phase 2 and 3 clinical trials, more dalbavancin for injection than comparator-treated subjects with normal baseline transaminase levels had post-baseline alanine aminotransferase (ALT) elevation greater than 3 times the upper limit of normal (ULN). Overall, abnormalities in liver tests (ALT, AST, bilirubin) were reported with similar frequency in the dalbavancin for injection and comparator arms [see Adverse Reactions (6.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are also discussed elsewhere in the labeling:
8.6 Renal Impairment
In patients with renal impairment whose known CLcr is less than 30 mL/min and who are not receiving regularly scheduled hemodialysis, the recommended regimen for dalbavancin for injection is 1,125 mg, administered as a single dose. No dosage adjustment is recommended for patients receiving regularly scheduled hemodialysis, and dalbavancin for injection can be administered without regard to the timing of hemodialysis. There is insufficient information to recommend dosage adjustment for pediatric patients younger than 18 years with CLcr less than 30 mL/min/1.73m2 [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
The antibacterial activity of dalbavancin appears to best correlate with the ratio of area under the concentration-time curve to minimal inhibitory concentration (AUC/MIC) for Staphylococcus aureus based on animal models of infection [see Dosage and Administration (2.1), Clinical Pharmacology (12.3)].
1 Indication and Usage (1 INDICATION AND USAGE)
Dalbavancin for injection is a lipoglycopeptide antibacterial indicated for the treatment of adult and pediatric patients with acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible strains of Gram- positive microorganisms. ( 1.1 )
To reduce the development of drug-resistant bacteria and maintain the effectiveness of dalbavancin for injection and other antibacterial drugs, dalbavancin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
8.7 Hepatic Impairment
No dosage adjustment of dalbavancin for injection is recommended for patients with mild hepatic impairment (Child-Pugh Class A). Caution should be exercised when prescribing dalbavancin for injection to patients with moderate or severe hepatic impairment (Child-Pugh Class B or C) as no data are available to determine the appropriate dosing in these patients [see Clinical Pharmacology (12.3)].
12.1 Mechanism of Action
Dalbavancin is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serious hypersensitivity (anaphylactic) and skin reactions have been reported in patients treated with dalbavancin for injection. If an allergic reaction occurs, discontinue treatment with dalbavancin for injection and institute appropriate therapy for the allergic reaction. Carefully monitor patients with known hypersensitivity to glycopeptides. ( 5.1 )
- Rapid intravenous infusion of dalbavancin for injection can cause flushing of the upper body, urticaria, pruritus, rash, and/or back pain. Stopping or slowing the infusion may result in cessation of these reactions. ( 5.2 )
- Alanine Aminotransferase (ALT) elevations with dalbavancin for injection treatment were reported in clinical trials. ( 5.3, 6.1 )
- Clostridioides difficile-associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including dalbavancin for injection. Evaluate if diarrhea occurs. ( 5.4 )
7.2 Drug Drug Interactions (7.2 Drug-Drug Interactions)
No clinical drug-drug interaction studies have been conducted with dalbavancin for injection. There is minimal potential for drug-drug interactions between dalbavancin for injection and cytochrome P450 (CYP450) substrates, inhibitors, or inducers [see Clinical Pharmacology (12.3)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
| Dosage in Adult Patients ( 2.1 , 2.3): | |
| Estimated Creatinine Clearance (CLcr) | Single Dose Regimen |
| 30 mL/min and above or on regular hemodialysis |
1,500 mg |
| Less than 30 mL/min and not on regular hemodialysis |
1,125 mg |
- Administer by intravenous infusion over 30 minutes ( 2.1 , 2.4 )
- See Full Prescribing Information for instructions on reconstitution of lyophilized powder and preparation of injection ( 2.4 )
| Dosage in Pediatric Patients with CLcr 30 mL/min/1.73m 2 and above ( 2.2 ) | |
| Age Range | Dosage (Single Dose Regimen) |
| Birth to less than 6 years | 22.5 mg/kg (maximum of 1,500 mg) |
| 6 to less than 18 years | 18 mg/kg (maximum of 1,500 mg) |
- Dosage adjustment in pediatric patients with CLcr less than 30 mL/min has not been studied.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Dalbavancin for injection is supplied as a white/off-white to pale yellow lyophilized sterile powder for reconstitution in a single-dose clear glass vial containing dalbavancin hydrochloride equivalent to 500 mg of dalbavancin.
5.4 Clostridioides Difficile (5.4 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported in users of nearly all systemic antibacterial drugs, including dalbavancin for injection, with severity ranging from mild diarrhea to fatal colitis. Treatment with antibacterial agents can alter the normal flora of the colon, and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile should be discontinued, if possible. Appropriate measures such as fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Post Marketing Experience
The following adverse reaction has been identified during post-approval use of dalbavancin. Because the reaction is reported voluntarily from a population of uncertain size, it is not possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
General disorders and administration site conditions: Back pain as an infusion-related reaction [See Warnings and Precautions ( 5.2 )].
5.1 Hypersensitivity Reactions
Serious hypersensitivity (anaphylactic) and skin reactions have been reported in patients treated with dalbavancin for injection. If an allergic reaction to dalbavancin for injection occurs, discontinue treatment with dalbavancin for injection and institute appropriate therapy for the allergic reaction. Before using dalbavancin for injection, inquire carefully about previous hypersensitivity reactions to other glycopeptides. Due to the possibility of cross-sensitivity, carefully monitor for signs of hypersensitivity during treatment with dalbavancin for injection in patients with a history of glycopeptide allergy [see Patient Counseling Information (17)].
5.2 Infusion Related Reactions (5.2 Infusion-Related Reactions)
Dalbavancin for injection is administered via intravenous infusion, using a total infusion time of 30 minutes to minimize the risk of infusion-related reactions. Rapid intravenous infusions of dalbavancin for injection can cause flushing of the upper body, urticaria, pruritus, rash, and/or back pain. Stopping or slowing the infusion may result in cessation of these reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of dalbavancin for injection cannot be directly compared to rates in the clinical trials of another drug and may not reflect rates observed in practice.
2.4 Preparation and Administration
Dalbavancin for injection must be reconstituted with either Sterile Water for Injection, USP, or 5% Dextrose Injection, USP, and subsequently diluted only with 5% Dextrose Injection, USP, to a final concentration of 1 mg/mL to 5 mg/mL. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Reconstitution: dalbavancin for injection must be reconstituted under aseptic conditions, using 25 mL of either Sterile Water for Injection, USP, or 5% Dextrose Injection, USP, for each 500 mg vial. To avoid foaming, alternate between gentle swirling and inversion of the vial until its contents are completely dissolved. Do not shake. The reconstituted vial contains 20 mg/mL dalbavancin as a clear, colorless to yellow solution.
Reconstituted vials may be stored either refrigerated at 2°C to 8°C (36°F to 46°F), or at controlled room temperature 20°C to 25°C (68°F to 77°F). Do not freeze.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Dalbavancin for injection is supplied as a white/off-white to pale yellow sterile lyophilized powder in a single-dose glass vial containing dalbavancin hydrochloride equivalent to 500 mg dalbavancin:
| Product Code | Unit of Sale | Strength |
| 221012 | NDC 65219-120-12 Individually packaged |
500 mg per single-dose vial |
Dalbavancin for injection should be stored at 25ºC (77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Storage of the reconstituted and diluted solutions of dalbavancin for injection are described elsewhere in the prescribing information [see Dosage and Administration (2.4)].
The container closure is not made with natural rubber latex.
7.1 Drug Laboratory Test Interactions (7.1 Drug-Laboratory Test Interactions)
Drug-laboratory test interactions have not been reported. Dalbavancin for injection at therapeutic concentrations does not artificially prolong prothrombin time (PT) or activated partial thromboplastin time (aPTT).
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Increases in serum levels of liver enzymes (ALT, AST), associated with microscopic findings in the liver were noted in toxicology studies in rats and dogs where dalbavancin was administered daily for 28 to 90 days. Hepatocellular necrosis was observed in dogs dosed at ≥10 mg/kg/day for longer than 2 months, i.e., at approximately 5 to 7 times the expected human dose on an exposure basis. Histiocytic vacuolation and hepatocyte necrosis were observed in rats dosed daily at 40 and 80 mg/kg/day, respectively, for 4 weeks, (approximately 3 and 6 times the expected human dose on an exposure basis, respectively). In addition, renal toxicity characterized by increases in serum BUN and creatinine and microscopic kidney findings was observed in rats and dogs at doses 5 to 7 times the expected human dose on an exposure basis. The relationship between these findings in the animal toxicology studies after 28 and 90 consecutive days of dosing to the indicated clinical dosing of 2 doses 7 days apart are unclear.
5.5 Development of Drug Resistant Bacteria (5.5 Development of Drug-Resistant Bacteria)
Prescribing dalbavancin for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
1.1 Acute Bacterial Skin and Skin Structure Infections
Dalbavancin for injection is indicated for the treatment of adult and pediatric patients with acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible strains of the following Gram-positive microorganisms: Staphylococcus aureus (including methicillin-susceptible and methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiae, Streptococcus anginosus group (including S. anginosus, S. intermedius, S. constellatus) and Enterococcus faecalis (vancomycin susceptible isolates).
2.3 Dosage Adjustments in Adult Patients With Clcr Less Than 30 Ml/min (2.3 Dosage Adjustments in Adult Patients with CLcr less than 30 mL/min)
In adult patients with renal impairment whose known CLcr is less than 30 mL/min and who are not receiving regularly scheduled hemodialysis, the recommended dosage regimen of dalbavancin for injection is 1,125 mg, administered as a single dose regimen.
No dosage adjustment is recommended for adult patients receiving regularly scheduled hemodialysis, and dalbavancin for injection can be administered without regard to the timing of hemodialysis [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
2.1 Recommended Dosage Regimen in Adult Patients With Clcr 30 Ml/min and Above (2.1 Recommended Dosage Regimen in Adult Patients with CLcr 30 mL/min and Above)
The recommended dosage regimen of dalbavancin for injection in adult patients with CLcr 30 mL/min and above is 1,500 mg, administered as a single dose regimen. Administer dalbavancin for injection over 30 minutes by intravenous infusion. For adult patients with CLcr less than 30 mL/min, dosage adjustment is required [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
2.2 Recommended Dosage Regimen in Pediatric Patients With Clcr 30 Ml/min/1.73m2 (2.2 Recommended Dosage Regimen in Pediatric Patients with CLcr 30 mL/min/1.73m2)
The recommended dosage regimen of dalbavancin for injection in pediatric patients with CLcr 30 mL/min/1.73m2 and above is a single dose regimen based on the age and weight of the pediatric patient (Table 1). Administer dalbavancin for injection over 30 minutes by intravenous infusion.
There is insufficient information to recommend dosage adjustment for pediatric patients younger than 18 years with CLcr less than 30 mL/min/1.73m2 [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.3)].
|
*Estimate CLcr or glomerular filtration rate (GFR) using an age-appropriate equation accepted for pediatric patients (birth to less than 18 years old) to define renal function impairment. |
|
| Age Range | Dosage (Single Dose Regimen) |
| Birth to less than 6 years | 22.5 mg/kg (maximum 1,500 mg) |
| 6 to less than 18 years | 18 mg/kg (maximum 1,500 mg) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:19.011345 · Updated: 2026-03-14T22:37:02.064259