These Highlights Do Not Include All The Information Needed To Use Zevtera Safely And Effectively. See Full Prescribing Information For Zevtera.
2bd685df-7dc6-78a6-e063-6294a90a580b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ZEVTERA is a cephalosporin antibacterial indicated for the treatment of: Adult patients with Staphylococcus aureus bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis ( 1.1 ), Adult patients with acute bacterial skin and skin structure infections (ABSSSI) ( 1.2 ), and Adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) ( 1.3 ). To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria ( 1.4 ).
Indications and Usage
ZEVTERA is a cephalosporin antibacterial indicated for the treatment of: Adult patients with Staphylococcus aureus bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis ( 1.1 ), Adult patients with acute bacterial skin and skin structure infections (ABSSSI) ( 1.2 ), and Adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) ( 1.3 ). To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria ( 1.4 ).
Dosage and Administration
For treatment of pediatric patients with CABP, the recommended dosage of ZEVTERA is described in Table 2, based on patient age and weight [see Clinical Pharmacology (12.3) ] . The duration of treatment for CABP in pediatric patients (3 months to less than 18 years old) is 7 days to 14 days. Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5) ]. Table 2: Recommended Dosage Regimen in Pediatric Patients with CABP Pediatric Age Group Dose Duration of treatment for CABP in pediatric patients is 7 days to 14 days. Frequency 12 years to less than 18 years old 13.3 mg/kg (up to 667 mg/dose 13.3 mg/kg of ceftobiprole medocaril sodium is equivalent to 10 mg/kg of ceftobiprole; 20 mg/kg ceftobiprole medocaril sodium is equivalent to 15 mg/kg of ceftobiprole. 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole. ) Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old [see Dosage and Administration (2.5) ]. 3 months to less than 12 years old 20 mg/kg (up to 667 mg/dose ) Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5) ].
Warnings and Precautions
Increased Mortality with Unapproved use in Ventilator-Associated Bacterial Pneumonia (VABP) Patients: The safety and effectiveness of ZEVTERA for the treatment of VABP has not been established and the use of ZEVTERA for VABP is not approved ( 5.1 ). Hypersensitivity Reactions: Discontinue ZEVTERA if a hypersensitivity reaction occurs, and institute appropriate treatment ( 5.2 ). Seizures and other adverse central nervous system (CNS) reactions have been associated with the use of ZEVTERA. If seizures or other CNS adverse reactions occur, evaluate patients to determine whether ZEVTERA should be discontinued ( 5.3 ). Clostridioides difficile -associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including ZEVTERA. Evaluate if diarrhea occurs ( 5.4 ).
Contraindications
ZEVTERA is contraindicated in patients with a known history of severe hypersensitivity to ZEVTERA, or to other members of the cephalosporin class [see Warnings and Precautions (5.2) ].
Adverse Reactions
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section: Increased Mortality in Ventilator-Associated Bacterial Pneumonia Patients [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Contraindications (4) and Warning and Precautions (5.2) ] Seizures and Other Central Nervous System Reactions [see Warnings and Precautions (5.3) ] Clostridioides difficile -Associated Diarrhea [see Warnings and Precautions (5.4) ]
Drug Interactions
Organic Anion Transporting Polypeptide 1B1/1B3 (OATP1B1/OATP1B3) Substrates: ZEVTERA may increase the plasma concentrations of OATP1B1 and OATP1B3 substrates. Concomitant administration is not recommended ( 7.1 )
Storage and Handling
Store ZEVTERA vials refrigerated at 2 °C to 8 °C (36 °F to 46 °F) protected from light. Store in carton until time of use. ZEVTERA must be reconstituted and then further diluted prior to administration by intravenous infusion. Store reconstituted and diluted solution of ZEVTERA as described elsewhere in the labeling [see Dosage and Administration (2.7) ].
How Supplied
ZEVTERA (ceftobiprole medocaril sodium for injection), a white, yellowish to slightly brownish sterile powder for reconstitution, is supplied in a single-dose clear glass vial sealed with a rubber stopper (not made with natural rubber latex) and an aluminum seal with a flip-off cap. Each vial contains 667 mg ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole) and is supplied in a carton containing 10 single-dose vials (NDC# 68547-578-10).
Medication Information
Warnings and Precautions
Increased Mortality with Unapproved use in Ventilator-Associated Bacterial Pneumonia (VABP) Patients: The safety and effectiveness of ZEVTERA for the treatment of VABP has not been established and the use of ZEVTERA for VABP is not approved ( 5.1 ). Hypersensitivity Reactions: Discontinue ZEVTERA if a hypersensitivity reaction occurs, and institute appropriate treatment ( 5.2 ). Seizures and other adverse central nervous system (CNS) reactions have been associated with the use of ZEVTERA. If seizures or other CNS adverse reactions occur, evaluate patients to determine whether ZEVTERA should be discontinued ( 5.3 ). Clostridioides difficile -associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including ZEVTERA. Evaluate if diarrhea occurs ( 5.4 ).
Indications and Usage
ZEVTERA is a cephalosporin antibacterial indicated for the treatment of: Adult patients with Staphylococcus aureus bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis ( 1.1 ), Adult patients with acute bacterial skin and skin structure infections (ABSSSI) ( 1.2 ), and Adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) ( 1.3 ). To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria ( 1.4 ).
Dosage and Administration
For treatment of pediatric patients with CABP, the recommended dosage of ZEVTERA is described in Table 2, based on patient age and weight [see Clinical Pharmacology (12.3) ] . The duration of treatment for CABP in pediatric patients (3 months to less than 18 years old) is 7 days to 14 days. Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5) ]. Table 2: Recommended Dosage Regimen in Pediatric Patients with CABP Pediatric Age Group Dose Duration of treatment for CABP in pediatric patients is 7 days to 14 days. Frequency 12 years to less than 18 years old 13.3 mg/kg (up to 667 mg/dose 13.3 mg/kg of ceftobiprole medocaril sodium is equivalent to 10 mg/kg of ceftobiprole; 20 mg/kg ceftobiprole medocaril sodium is equivalent to 15 mg/kg of ceftobiprole. 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole. ) Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old [see Dosage and Administration (2.5) ]. 3 months to less than 12 years old 20 mg/kg (up to 667 mg/dose ) Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5) ].
Contraindications
ZEVTERA is contraindicated in patients with a known history of severe hypersensitivity to ZEVTERA, or to other members of the cephalosporin class [see Warnings and Precautions (5.2) ].
Adverse Reactions
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section: Increased Mortality in Ventilator-Associated Bacterial Pneumonia Patients [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Contraindications (4) and Warning and Precautions (5.2) ] Seizures and Other Central Nervous System Reactions [see Warnings and Precautions (5.3) ] Clostridioides difficile -Associated Diarrhea [see Warnings and Precautions (5.4) ]
Drug Interactions
Organic Anion Transporting Polypeptide 1B1/1B3 (OATP1B1/OATP1B3) Substrates: ZEVTERA may increase the plasma concentrations of OATP1B1 and OATP1B3 substrates. Concomitant administration is not recommended ( 7.1 )
Storage and Handling
Store ZEVTERA vials refrigerated at 2 °C to 8 °C (36 °F to 46 °F) protected from light. Store in carton until time of use. ZEVTERA must be reconstituted and then further diluted prior to administration by intravenous infusion. Store reconstituted and diluted solution of ZEVTERA as described elsewhere in the labeling [see Dosage and Administration (2.7) ].
How Supplied
ZEVTERA (ceftobiprole medocaril sodium for injection), a white, yellowish to slightly brownish sterile powder for reconstitution, is supplied in a single-dose clear glass vial sealed with a rubber stopper (not made with natural rubber latex) and an aluminum seal with a flip-off cap. Each vial contains 667 mg ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole) and is supplied in a carton containing 10 single-dose vials (NDC# 68547-578-10).
Description
ZEVTERA is a cephalosporin antibacterial indicated for the treatment of: Adult patients with Staphylococcus aureus bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis ( 1.1 ), Adult patients with acute bacterial skin and skin structure infections (ABSSSI) ( 1.2 ), and Adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) ( 1.3 ). To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria ( 1.4 ).
Section 42229-5
Reconstitution of ZEVTERA in the Vial
- For adult and pediatric patients 12 years of age and older, reconstitute ZEVTERA lyophilized powder in the vial with 10 mL of sterile water for injection or 10 mL of 5% dextrose injection.
- For pediatric patients aged 3 months to less than 12 years old, ZEVTERA lyophilized powder must be reconstituted with 10 mL of 5% dextrose injection.
- After reconstitution, shake the reconstituted ZEVTERA vial vigorously until dissolution is completed, which in some cases may take up to 10 minutes. The volume of the resulting reconstituted solution is approximately 10.6 mL.
- Allow any foam formed to dissipate and then inspect the reconstituted solution visually to ensure the product is in solution and particulate matter is absent.
- The reconstituted solution contains 66.7 mg/mL of ceftobiprole medocaril sodium and must be further diluted using aseptic technique with the appropriate diluent as described below, prior to administration. If it is not possible to dilute the reconstituted solution immediately, the reconstituted solution may be stored refrigerated for up to 24 hours, and at room temperature for up to 1 hour. Discard any unused reconstituted solution [see Dosage and Administration (2.6)].
1.4 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven, or strongly suspected, to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
There is no information on clinical signs and symptoms associated with an overdose of ZEVTERA. For patients who receive doses greater than the recommended dosage regimen and have unexpected adverse reactions associated with ZEVTERA, discontinue ZEVTERA, treat symptomatically, and institute general supportive treatment.
ZEVTERA can be removed by hemodialysis. However, no information is available on the use of hemodialysis to treat overdose [see Clinical Pharmacology (12.3)].
11 Description
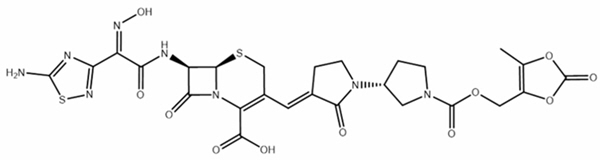
ZEVTERA (ceftobiprole medocaril sodium for injection) contains sodium salt of ceftobiprole medocaril, a semisynthetic, cephalosporin antibacterial, for intravenous use. Chemically, ceftobiprole medocaril is (6R,7R)-7-[[(2Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-hydroxyiminoacetyl]amino]-3-[(E)-[1-[(3R)-1-[(5-methyl-2-oxo-1,3-dioxol-4-yl)methoxycarbonyl]pyrrolidin-3-yl]-2-oxopyrrolidin-3-ylidene]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid. Its molecular weight is 690.6 g/mol. The empirical formula is C26H25N8NaO11S2.
Figure 1 Chemical Structure of ceftobiprole medocaril
ZEVTERA vials contain 667 mg of ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole). The powder for injection is a white, yellowish to slightly brownish sterile powder. Each vial includes inactive ingredient citric acid monohydrate (26.3 mg/vial) as a buffer component and sodium hydroxide (q.s.) as a pH adjustment agent. Each vial of ZEVTERA contains approximately 32 mg of sodium.
The pH of the reconstituted solution is 4.5–5.5.
16.1 How Supplied
ZEVTERA (ceftobiprole medocaril sodium for injection), a white, yellowish to slightly brownish sterile powder for reconstitution, is supplied in a single-dose clear glass vial sealed with a rubber stopper (not made with natural rubber latex) and an aluminum seal with a flip-off cap. Each vial contains 667 mg ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole) and is supplied in a carton containing 10 single-dose vials (NDC# 68547-578-10).
8.4 Pediatric Use
The safety and effectiveness of ZEVTERA have been established for the treatment of CABP in pediatric patients 3 months to less than 18 years. Use of ZEVTERA in this age group is supported by evidence from an adequate and well-controlled trial of ZEVTERA in adults, with additional pharmacokinetic, safety and efficacy data from pediatric trials [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.3)].
The safety and effectiveness of ZEVTERA have not been established for the treatment of CABP in pediatric patients less than 3 months of age.
The safety and effectiveness of ZEVTERA have not been established for the treatment of ABSSSI and SAB in pediatric patients.
8.5 Geriatric Use
Of the 835 adult CABP, ABSSSI, and SAB patients treated with ZEVTERA in the Trials 1, 2 and 3, 210 patients (25%) were 65 years of age and older, including 85 patients (10%) 75 years of age and older. No overall differences in safety or effectiveness of ZEVTERA were observed between patients 65 years of age and older and younger adult patients.
No clinically significant changes in the pharmacokinetics of ZEVTERA were observed in patients 65 years of age and older compared to younger adult patients [see Clinical Pharmacology (12.3)]. Dosage adjustment for geriatric patients should be based on renal function [see Dosage and Administration (2.3), Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
4 Contraindications
ZEVTERA is contraindicated in patients with a known history of severe hypersensitivity to ZEVTERA, or to other members of the cephalosporin class [see Warnings and Precautions (5.2)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section:
- Increased Mortality in Ventilator-Associated Bacterial Pneumonia Patients [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Contraindications (4) and Warning and Precautions (5.2)]
- Seizures and Other Central Nervous System Reactions [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.4)]
7 Drug Interactions
Organic Anion Transporting Polypeptide 1B1/1B3 (OATP1B1/OATP1B3) Substrates: ZEVTERA may increase the plasma concentrations of OATP1B1 and OATP1B3 substrates. Concomitant administration is not recommended (7.1)
12.2 Pharmacodynamics
Ceftobiprole exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
Ceftobiprole medocaril is the prodrug of the active moiety ceftobiprole. No clinically significant differences in the pharmacokinetics of ceftobiprole were observed with ZEVTERA following single or multiple dose administration. Ceftobiprole exhibits linear and time-independent pharmacokinetics. The Cmax and AUC increase proportionally with the dose over a range of 125 mg to 1000 mg (0.25 to 2 times the highest approved recommended adult dosage).
The mean pharmacokinetic parameters of ceftobiprole in healthy adults with normal renal function after single and multiple 2-hour IV infusions of ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) administered every 8 hours are summarized in Table 11. Pharmacokinetic parameters were similar for single and multiple dose administrations.
| Parameter | Multiple doses administered every 8 hours as a 2-hour infusion (n = 27) |
|---|---|
| Cmax = Maximum plasma concentration; AUC0-8h = Area under the plasma concentration-time curve from 0 to 8 h; Vss = Volume of distribution at steady state; CL = Clearance. | |
| Cmax (µg/mL) | 33.0 (4.83) |
| AUC0-8h (µg•h/mL) | 102 (11.9) |
| Distribution | |
| % Bound to human plasma protein | 16 |
| % ELF/unbound plasma | 15-19 |
| Vss (L) | 18 |
| Metabolism (Minimally Metabolized) | |
| t1/2 (h) | 3.3 (0.3) |
| CL (L/h) | 4.98 (0.582) |
| Excretion | |
| Major route of elimination | Renal |
| % excreted unchanged in urine | 83 |
1 Indications and Usage
ZEVTERA is a cephalosporin antibacterial indicated for the treatment of:
- Adult patients with Staphylococcus aureus bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis (1.1),
- Adult patients with acute bacterial skin and skin structure infections (ABSSSI) (1.2), and
- Adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) (1.3).
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria (1.4).
12.1 Mechanism of Action
ZEVTERA is an antibacterial drug [see Microbiology (12.4)]
1.1 Staphylococcus Aureus
ZEVTERA is indicated for the treatment of adult patients with Staphylococcus aureus bloodstream infection (bacteremia) (SAB), including those with right-sided infective endocarditis, caused by methicillin-susceptible and methicillin-resistant isolates.
16.2 Storage and Handling
Store ZEVTERA vials refrigerated at 2 °C to 8 °C (36 °F to 46 °F) protected from light. Store in carton until time of use.
ZEVTERA must be reconstituted and then further diluted prior to administration by intravenous infusion. Store reconstituted and diluted solution of ZEVTERA as described elsewhere in the labeling [see Dosage and Administration (2.7)].
14.1 Staphylococcus Aureus
The efficacy of ZEVTERA in the treatment of adult patients with SAB, including right-sided infective endocarditis, was demonstrated in a randomized, controlled, double-blind, multinational, multicenter trial (Trial 1, NCT03138733). In this trial, adult patients were randomized to either ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) IV every 6 hours from study Day 1 to Day 8, and 667 mg IV every 8 hours from study Day 9 onwards) or daptomycin (6 mg/kg up to 10 mg/kg IV every 24 hours) plus optional aztreonam for coverage of gram-negative co-infections (the comparator). Randomization was stratified by study site, dialysis status, and use of prior antibacterial drugs. The study was performed in two cohorts based on findings in animal studies [see Nonclinical Toxicology (13.2)]. The initial cohort was enrolled to receive a maximum treatment duration of 28 days. Based on a safety evaluation of cohort 1, patients could be enrolled in cohort 2 to receive a treatment duration of up to 42 days. The duration of study treatment was based on the investigator's clinical evaluation.
Study patients were required to have at least one positive blood culture for S. aureus obtained within 72 hours prior to randomization, signs and symptoms of bacteremia (fever, elevated white blood cell count or immature neutrophils, tachycardia, or hypotension), and at least one of the following conditions of complicated S. aureus bacteremia: chronic dialysis, persistent SAB, acute bacterial skin and skin structure infection, metastatic infections of native tissue, osteomyelitis, epidural or cerebral abscess, or definite native-valve right-sided infective endocarditis by Modified Duke's Criteria. Patients with uncomplicated S. aureus bacteremia, left-sided infective endocarditis, prosthetic heart valves, foreign body material that could not be removed, severe neutropenia, or pneumonia, were excluded from the study.
Upon entry, patients were classified for likelihood of endocarditis using the modified Duke's criteria (definite, possible, or rejected). Echocardiography, including a transesophageal echocardiogram, was to be performed during screening or within 7 days following study enrollment. Final diagnoses were to be made by the study investigators within 7 days of randomization. The final diagnosis for persistent S. aureus bacteremia was made by an independent treatment-blinded Data Review Committee (DRC).
A total of 390 patients (192 ZEVTERA, 198 daptomycin) with SAB were randomized from 60 centers in the USA, Europe, Latin America, and South Africa. The modified ITT (mITT) population was used for the primary efficacy analysis, comprising 387 patients (189 ZEVTERA, 198 daptomycin ± aztreonam) who received study drugs and had a baseline blood culture positive for S. aureus.
Patient demographic and baseline characteristics were balanced between the treatment groups. The median age among the 387 patients in the mITT population was 58 years, ranging from 19 to 91 years), with 31% aged ≥ 65 years, 69% of patients were male, and 96% were White.
Frequent conditions of complicated SAB included acute bacterial skin and skin structure infections (61%), intra-abdominal abscesses (14%), osteoarticular infections (13%), and patients on chronic dialysis (13%). A total of 6.5% of patients had definite right-sided infective endocarditis. A total of 24% had bacteremia caused by methicillin-resistant S. aureus (MRSA).
The primary efficacy outcome in the study was overall success at the post-treatment evaluation (PTE) visit at 70 days post-randomization in the mITT population, as assessed by the independent DRC.
Overall success required survival, symptom improvement, S. aureus bacteremia bloodstream clearance, no new S. aureus bacteremia complications, and no use of other potentially effective antibacterial drugs.
Overall success rates at the PTE visit in the mITT population were 69.8% (132/189) in patients treated with ZEVTERA and 68.7% (136/198) in patients treated with the comparator. The overall primary study outcome and in pre-defined subgroups in the mITT population is shown in Table 12.
| Group/Subgroup | ZEVTERA n/N (%) |
Daptomycin ± aztreonam n/N (%) |
Between-group Difference (%) (2-sided 95% CI) Between-group difference of ZEVTERA minus daptomycin ± aztreonam using Cochran-Mantel-Haenszel weights method adjusted for actual stratum (dialysis status and prior antibacterial treatment use).
|
|---|---|---|---|
| Overall | 132/189 (69.8) | 136/198 (68.7) | 2.0 (−7.1, 11.1) |
| MSSA (methicillin-susceptible) | 100/141 (70.9) | 97/146 (66.4) | 4.8 (−5.9, 15.5) |
| MRSA (methicillin-resistant) | 31/45 (68.9) | 38/49 (77.6) | −8.3 (−25.3, 8.6) |
| ABSSSI | 81/116 (69.8) | 80/121 (66.1) | 4.5 (−7.3, 16.3) |
| Definite right-sided infective endocarditis | 10/15 (66.7) | 7/10 (70.0) | −6.6 (−40.1, 27.0) |
For patients enrolled in Ukraine, overall success rates at the PTE visit in the mITT population were 77/84 (84.1%) in patients treated with ZEVTERA and 82/92 (89.1%) in patients treated with the comparator. For patients enrolled outside Ukraine, overall success rates at the PTE visit in the mITT population were 58/101 (57.4%) in patients treated with ZEVTERA and 54/106 (50.9%) in patients treated with the comparator.
All-cause mortality between randomization and the PTE visit was observed in 17/189 (9.0%) patients treated with ZEVTERA, and 18/198 (9.1%) patients treated with the comparator. Other key secondary efficacy outcomes included microbiological eradication at the PTE visit in the mITT population, which was achieved in 82% of patients treated with ZEVTERA and 77% of patients treated with the comparator, and the development of new S. aureus bacteremia complications, which occurred in 6% of patients treated with ZEVTERA and 6% of patients treated with the comparator. S. aureus bloodstream clearance, defined as 2 consecutive study days with negative blood cultures for S. aureus with no subsequent S. aureus relapse or reinfection, was achieved after a median of 4 days in each treatment group.
S. aureus bloodstream clearance in patients with MSSA was achieved after a median of 3 days in patients treated with ZEVTERA and after a median of 4 days in patients treated with the comparator.
S. aureus bloodstream clearance in patients with MRSA was achieved after a median of 5 days in each treatment group.
Failure of treatment due to relapse of S. aureus bacteremia was assessed by the DRC in two patients (1%) treated with ZEVTERA, and in four patients (2%) treated in the comparator arm. No increase in ceftobiprole MIC was observed for the two isolates from the ZEVTERA-treated patients.
5 Warnings and Precautions
- Increased Mortality with Unapproved use in Ventilator-Associated Bacterial Pneumonia (VABP) Patients: The safety and effectiveness of ZEVTERA for the treatment of VABP has not been established and the use of ZEVTERA for VABP is not approved (5.1).
- Hypersensitivity Reactions: Discontinue ZEVTERA if a hypersensitivity reaction occurs, and institute appropriate treatment (5.2).
- Seizures and other adverse central nervous system (CNS) reactions have been associated with the use of ZEVTERA. If seizures or other CNS adverse reactions occur, evaluate patients to determine whether ZEVTERA should be discontinued (5.3).
- Clostridioides difficile-associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including ZEVTERA. Evaluate if diarrhea occurs (5.4).
2 Dosage and Administration
- The recommended dosage of ZEVTERA for adult patients with SAB, ABSSSI and CABP is described in the table below (2.1):
Indication in Adults Dose Frequency SAB 667 mg Every 6 hours on Days 1 to 8 Every 8 hours from Day 9 ABSSSI 667 mg Every 8 hours CABP 667 mg Every 8 hours
- Duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP (2.1).
- Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL (2.1, 2.6).
- The recommended dosage of ZEVTERA for pediatric patients (3 months to less than 18 years old) with CABP is described below (2.2).
Pediatric Age Group for CABP Dose Frequency 12 years to less than 18 years old 13.3 mg/kg (up to 667 mg/dose) Every 8 Hours Greater than or equal to 3 months and less than 12 years old 20 mg/kg (up to 667 mg/dose) Every 8 Hours
- Duration of treatment in pediatric patients is 7 days to 14 days for CABP (2.1).
- Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for pediatric patients aged 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for pediatric patients aged 3 months to less than 12 years old (2.2, 2.6).
- Reduce the dosage in adult patients with CLCR less than 50 mL/min, including patients with CLCR less than 15 mL/min on hemodialysis (2.3 and 8.6).
- Increase the dosage in adult patients with CLCR greater than 150 mL/min (2.3).
- Reduce the dosage in pediatric patients aged 2 years old to less than 18 years old with eGFR less than 50 mL/min/1.73 m2 and greater than or equal to 15 mL/min/1.73 m2 (2.4 and 8.6).
- See Full Prescribing Information for instructions for preparation of ZEVTERA solution infusion solution (2.5).
3 Dosage Forms and Strengths
ZEVTERA (ceftobiprole medocaril sodium for injection) is available in a single-dose vial as a white, yellowish to slightly brownish, cake to broken cake or powder containing 667 mg of ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole) for reconstitution.
5.4 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial agents, including ZEVTERA, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile should be discontinued, if possible. Manage fluid and electrolyte levels as appropriate, supplement protein intake, monitor antibacterial treatment of C. difficile, and institute surgical evaluation as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions and altered laboratory tests have been identified during post-approval use of ZEVTERA and ceftobiprole outside of the United States, or other cephalosporin-class antibacterial drugs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Altered Laboratory Tests: Positive direct Coombs' test, false-positive test for urinary glucose.
Blood and lymphatic system disorders: Agranulocytosis, aplastic anemia.
Hepatobiliary disorders: Hepatic dysfunction including cholestasis.
Immune system disorders: Drug fever, serum sickness-like reaction.
Nervous system disorders: Myoclonus.
Renal and urinary disorders: Renal dysfunction, toxic nephropathy.
Vascular disorders: Hemorrhage, hypertension.
5.2 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, were observed in ZEVTERA-treated patients in clinical trials [see Adverse Reactions (6.1)]. Serious and occasionally fatal hypersensitivity reactions and serious skin reactions have been reported in patients receiving beta-lactam antibacterial drugs.
Before therapy with ZEVTERA is instituted, careful inquiry about previous hypersensitivity reactions to other cephalosporins, penicillins, or other beta-lactam antibacterial drugs should be made. Maintain clinical supervision if this product is to be given to a penicillin- or other beta-lactam-allergic patient, because cross sensitivity among beta-lactam antibacterial agents has been established. Discontinue ZEVTERA if a hypersensitivity reaction occurs, and institute appropriate treatment.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ZEVTERA was evaluated in adults in four controlled comparative phase 3 clinical trials (Trials 1 through 4) which included 1221 patients treated with ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) administered by IV infusion over 2 hours every 6 to 8 hours and 1248 patients treated with comparator for a treatment period up to 42 days. The median age of patients treated with ZEVTERA was 56 years, ranging between 18 and 95 years old. Patients treated with ZEVTERA were predominantly male (64%) and White (82%).
The safety of ZEVTERA was also evaluated in pediatric patients aged 3 months to less than 18 years in a controlled phase 3 clinical trial (Trial 5) which included 138 patients with CABP and hospital-acquired bacterial pneumonia (HABP) requiring hospitalization. Although HABP was included in the safety data, the safety and effectiveness of ZEVTERA for the treatment of HABP has not been established and ZEVTERA is not approved for the treatment of HABP.
Principal Display Panel 667 Mg Vial Box
NDC# 68547-578-10
Rx Only
10 single-dose vials
ZEVTERA® 667 mg/vial*
(ceftobiprole medocaril sodium for injection)
*equivalent to 500 mg ceftobiprole
For intravenous use only.
Reconstitute and further dilute before use.
Single-dose vial. Discard unused portion.
1.3 Community Acquired Bacterial Pneumonia
ZEVTERA is indicated for the treatment of adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) caused by susceptible isolates of the following gram-positive and gram-negative microorganisms: Staphylococcus aureus (methicillin-susceptible isolates), Streptococcus pneumoniae, Haemophilus influenzae, Haemophilus parainfluenzae, Escherichia coli, and Klebsiella pneumoniae.
13.2 Animal Toxicology And/or Pharmacology
Both rats and marmosets showed convulsions and thrombi and/or emboli with dosing durations greater than one month. In the 13-week intravenous rat study, clonic convulsions were noted at 500 mg/kg/day (2 times the MRHD based on BSA comparisons) with early deaths seen at 250 mg/kg in the males (1 time the MRHD based on BSA comparison). In the 13-week intravenous marmoset study, convulsions and thrombi were noted at multiple sites distal to the injection site (i.e., lung, and heart) at doses ≥ 50 mg/kg/day (0.1 times the MRHD, based on BSA comparisons). While dogs did not show convulsions in a 13-week toxicology study, catheter patency could only be maintained between 4 to 11 weeks in most animals at doses up to 32 mg/kg/day (0.4 times the MRHD based on BSA comparisons). Fibrin, erythrocytes and inflammatory cells within the lumens of the catheters were noted in the 13-week dog study, while thrombi were observed at the injection site in all species tested. Kidney findings were also noted in the 13-week studies in rats (tubular/duct eosinophilic bodies and presence of foreign material) at 1 times the MRHD.
5.5 Development of Drug Resistant Bacteria
Prescribing ZEVTERA in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.5 Preparation of Zevtera Infusion Solution
ZEVTERA must first be reconstituted in the vial and then further diluted prior to administration by intravenous infusion over a period of 2 hours. Aseptic technique must be followed in preparing the infusion solution. See additional instructions for reconstitution and dilution below.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. The solution for infusion should be clear to slightly opalescent and yellowish in color. Discard if discoloration or visible particles are observed.
2.7 Drug Compatibilities and Incompatibilities
ZEVTERA is compatible with 5% dextrose injection and 0.9 % sodium chloride injection [see Dosage and Administration (2.5)]. The compatibility of ZEVTERA with other drugs and infusion solutions other than 5% dextrose Injection or 0.9% sodium chloride Injection, has not been established.
ZEVTERA must not be mixed or administered simultaneously with calcium-containing solutions. Do not mix ZEVTERA with, or co-administer through, the same intravenous line or cannula with other drug products.
1.2 Acute Bacterial Skin and Skin Structure Infections
ZEVTERA is indicated for the treatment of adult patients with acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following gram-positive and gram-negative microorganisms: Staphylococcus aureus (methicillin-susceptible and methicillin-resistant isolates), Streptococcus pyogenes, and Klebsiella pneumoniae.
5.3 Seizures and Other Central Nervous System Reactions
Seizures and other adverse central nervous system (CNS) reactions have been reported during treatment with ZEVTERA and other cephalosporins [see Adverse Reactions (6.1)].
Nonconvulsive status epilepticus (NCSE), encephalopathy, coma, asterixis, neuromuscular excitability, and myoclonia have been reported with cephalosporins particularly in patients with a history of epilepsy or when recommended dosages of cephalosporins were exceeded due to renal impairment. Adjust ZEVTERA dosing based on creatinine clearance [see Dosage and Administration (2.3, 2.4)].
Anticonvulsant therapy should be continued in patients with known seizure disorders. If CNS adverse reactions, including seizures, occur, patients should undergo a neurological evaluation to determine whether ZEVTERA should be discontinued.
2.3 Recommended Dosage Regimen in Adult Patients With Renal Impairment
The recommended ZEVTERA dosage in adult patients with renal impairment (CLCR less than 50 mL/min), including patients receiving hemodialysis is shown in Table 3. The duration of treatment in adult patients with renal impairment is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL to adult patients with renal impairment [see Dosage and Administration (2.5)].
In adult patients with augmented renal clearance (CLCR greater than 150 mL/min), increase the ZEVTERA dosage to 667 mg every 6 hours.
| Indication Duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
|
Creatinine Clearance, CLCR (mL/min) Based on calculated creatinine clearance [see Use in Specific Populations (8.6)].
|
Dose 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole; 333 mg ceftobiprole medocaril sodium is equivalent to 250 mg of ceftobiprole.
|
Frequency Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL [see Dosage and Administration (2.5)].
|
|---|---|---|---|
| SAB | 30 to less than 50 mL/min | 667 mg | Every 8 hours on Days 1 to 8 |
| Every 12 hours from Day 9 | |||
| 15 to less than 30 mL/min | 333 mg | Every 8 hours on Days 1 to 8 | |
| Every 12 hours from Day 9 | |||
| Less than 15 mL/min, including hemodialysis Administer ZEVTERA after intermittent hemodialysis on hemodialysis days, because ceftobiprole is removed by hemodialysis.
|
333 mg | Every 24 hours | |
| ABSSSI or CABP | 30 to less than 50 | 667 mg | Every 12 hours |
| 15 to less than 30 | 333 mg | Every 12 hours | |
| Less than 15 mL/min, including hemodialysis | 333 mg | Every 24 hours |
14.2 Acute Bacterial Skin and Skin Structure Infections (absssi) in Adults
The efficacy of ZEVTERA in the treatment of adult patients with ABSSSI was demonstrated in a randomized, controlled, double-blind, multinational, multicenter trial (Trial 2, NCT03137173). Patients were randomized to either ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) IV every 8 hours or vancomycin plus aztreonam (vancomycin 1 gram or 15 mg/kg IV every 12 hours, aztreonam 1 gram IV every 12 hours). Randomization was stratified by study site and type of ABSSSI. Treatment duration was 5 to 14 days. Patients could receive concomitant metronidazole for suspected anaerobic infection. A switch to oral therapy was not allowed.
The study enrolled adult patients with ABSSSI (cellulitis/erysipelas, major cutaneous abscess, wound infection) with a lesion area of at least 75 cm2, systemic or regional signs of infection, and a requirement for IV antibiotic treatment. Patients with other forms of ABSSSI, uncomplicated skin and skin structure infections, or infections related to prosthetic devices, were excluded.
The primary endpoint, assessed in the ITT population, was early clinical response 48–72 hours after start of treatment. The ITT primary analysis population comprised all patients randomized. Early clinical response required a reduction of the primary skin lesion by at least 20%, survival for at least 72 hours, and the absence of concomitant antibacterial treatment or additional unplanned surgery.
The main secondary endpoint was investigator-assessed clinical success at the test-of-cure (TOC) visit 15 to 22 days after randomization and at least 5 days after the end-of-treatment.
A total of 679 patients (335 ZEVTERA, 344 vancomycin plus aztreonam) were randomized from 32 centers in the USA and Europe. The microbiological ITT (mITT) population included 506 patients with at least one valid baseline pathogen.
Patient demographic and baseline characteristics in the ITT population were balanced between the treatment groups. Approximately 95% of patients were classified as White, 59% were male, the median age was 50 years (range: 18 to 89), and the mean body mass index was 28 kg/m2, 62% of patients were enrolled in the USA, and 38% in Europe. A history of diabetes was present in 11% of patients and current injection drug use was reported in 33% of patients. The types of ABSSSI included cellulitis/erysipelas (33%), wound infection (39%), and major cutaneous abscesses (28%). The median area of the primary skin infection was 259 cm2.
The primary and main secondary outcomes are shown in Table 13.
| ZEVTERA n/N (%) |
Vancomycin plus aztreonam n/N (%) |
Between-group Difference (%) (2-sided 95% CI) Between-group difference of ZEVTERA minus vancomycin plus aztreonam, using Cochran-Mantel-Haenszel weights method adjusted for geographical region and actual type of ABSSSI.
|
|
|---|---|---|---|
| Primary endpoint analyses Early clinical response based on at least 20% reduction from baseline in the area of the primary lesion, survival for ≥ 72 hours from the time of administration of the first dose of study drug, no use of concomitant systemic antibacterial treatments or topical antibacterial administration on the primary lesion, and no additional unplanned surgical procedure for the ABSSSI after the start of study treatment.
|
|||
| Early clinical response at 48–72 hours after start of treatment (ITT) | 306/335 (91.3) | 303/344 (88.1) | 3.3 (−1.2, 7.8) |
| Main secondary endpoint analysis Clinical success was defined as complete (cured) or nearly complete (improved) resolution of baseline signs and symptoms of the primary infection, such that no further antibacterial treatment was needed.
|
|||
| Investigator-assessed clinical success at the TOC visit (ITT) | 302/335 (90.1) | 306/344 (89.0) | 1.0 (−3.5, 5.6) |
Clinical response rates at TOC by the most common baseline pathogens in the microbiological ITT (mITT) population, defined as all randomized patients with a baseline pathogen, are presented in Table 14.
| Early clinical response at 48–72 hours after start of treatment Early clinical response based on at least a 20% reduction from baseline in the area of the primary lesion, survival for ≥ 72 hours from the time of administration of the first dose of study drug, no use of concomitant systemic antibacterial treatments, or topical antibacterial administration on the primary lesion and no additional unplanned surgical procedure for the ABSSSI after the start of study treatment.
(mITT) |
Investigator-assessed clinical success at the TOC visit Clinical success was defined as complete (cured) or nearly complete (improved) resolution of baseline signs and symptoms of the primary infection such that no further antibacterial treatment was needed.
(mITT) |
|||
|---|---|---|---|---|
| Pathogen | ZEVTERA n/N (%) |
Vancomycin plus aztreonam n/N (%) |
ZEVTERA n/N (%) |
Vancomycin plus aztreonam n/N (%) |
| Any Gram-positive pathogen | 213/228 (93.4) | 220/244 (90.2) | 203/228 (89.0) | 217/244 (88.9) |
| Staphylococcus aureus | 186/197 (94.4) | 185/205 (90.2) | 174/197 (88.3) | 180/205 (87.8) |
| Methicillin-susceptible | 102/108 (94.4) | 112/124 (90.3) | 95/108 (88.0) | 107/124 (86.3) |
| Methicillin-resistant | 81/86 (94.2) | 67/73 (91.8) | 76/86 (88.4) | 66/73 (90.4) |
| Streptococcus pyogenes | 17/20 (85.0) | 23/24 (95.8) | 17/20 (85.0) | 22/24 (91.7) |
| Any Gram-negative pathogen | 26/27 (96.3) | 33/37 (89.2) | 24/27 (88.9) | 34/37 (91.9) |
| Enterobacterales | 16/16 (100.0) | 24/27 (88.9) | 15/16 (93.8) | 25/27 (92.6) |
| Klebsiella pneumoniae | 8/8 (100.0) | 5/5 (100.0) | 7/8 (87.5) | 5/5 (100.0) |
7.1 Organic Anion Transporting Polypeptide 1b1/1b3 (oatp1b1/oatp1b3) Substrates
ZEVTERA may increase the plasma concentrations of OATP1B1 and OATP1B3 substrates. Concomitant administration is not recommended [see Clinical Pharmacology (12.3)].
2.1 Recommended Dosage and Administration for Sab, Absssi and Cabp in Adult Patients
The recommended dosage of ZEVTERA for the treatment of adult patients with SAB, ABSSSI and CABP is described in Table 1. The duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL to adult patients [see Dosage and Administration (2.5)].
| Indication Duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
|
Dose | Frequency Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL [see Dosage and Administration (2.5)]
|
|---|---|---|
| SAB | 667 mg 667 mg of ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole.
|
Every 6 hours on Days 1 to 8 |
| Every 8 hours from Day 9 | ||
| ABSSSI | 667 mg | Every 8 hours |
| CABP | 667 mg | Every 8 hours |
5.1 Increased Mortality With Unapproved Use in Ventilator Associated Bacterial Pneumonia Patients
In Trial 4, an increase in mortality was seen in the subgroup of patients with ventilator-associated bacterial pneumonia (VABP) who were treated with ZEVTERA (35/103 [34%] versus 24/102 [24%] in comparator-treated patients) [see Adverse Reactions (6.1)]. The cause of this increased mortality has not been established. Generally, deaths were associated with complications of infection or underlying co-morbidities. The safety and effectiveness of ZEVTERA for the treatment of VABP has not been established and the use of ZEVTERA for VABP is not approved.
2.2 Recommended Dosage and Administration for Cabp in Pediatric Patients (3 Months to Less Than 18 Years Old)
For treatment of pediatric patients with CABP, the recommended dosage of ZEVTERA is described in Table 2, based on patient age and weight [see Clinical Pharmacology (12.3)]. The duration of treatment for CABP in pediatric patients (3 months to less than 18 years old) is 7 days to 14 days.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5)].
| Pediatric Age Group | Dose Duration of treatment for CABP in pediatric patients is 7 days to 14 days.
|
Frequency |
|---|---|---|
| 12 years to less than 18 years old | 13.3 mg/kg (up to 667 mg/dose 13.3 mg/kg of ceftobiprole medocaril sodium is equivalent to 10 mg/kg of ceftobiprole; 20 mg/kg ceftobiprole medocaril sodium is equivalent to 15 mg/kg of ceftobiprole. 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole. ) |
Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old [see Dosage and Administration (2.5)].
|
| 3 months to less than 12 years old | 20 mg/kg (up to 667 mg/dose) | Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5)].
|
2.4 Recommended Dosage Regimen for Cabp in Pediatric Patients (2 Years to Less Than 18 Years Old) With Renal Impairment
The recommended dosage of ZEVTERA in pediatric patients (2 years to less than 18 years old) with renal impairment (with eGFR less than 50 mL/min/1.73 m2 and greater than or equal to 15 mL/min/1.73 m2) is shown in Table 4. The duration of treatment for CABP in pediatric patients (2 years to less than 18 years old) with renal impairment is 7 days to 14 days.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for patients 2 years to less than 12 years old.
There is insufficient information to recommend dosage adjustments in pediatric patients 2 years of age and older with an eGFR less than 15 mL/min/1.73m2.
There is insufficient information to recommend dosage adjustments in pediatric patients less than 2 years of age with any degree of renal impairment.
| Pediatric Age Group Duration of treatment for CABP in pediatric patients (2 years to less than 18 years old) and with renal impairment is 7 days to 14 days.
|
eGFR (mL/min/1.73 m2) Calculate using a validated GFR estimating equation for the approved age of the pediatric population.
|
Dosage Regimen 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole; 333 mg ceftobiprole medocaril sodium is equivalent to 250 mg of ceftobiprole; 10 mg/kg of ceftobiprole medocaril sodium is equivalent to 7.5 mg/kg of ceftobiprole; 13.3 mg/kg ceftobiprole medocaril sodium is equivalent to 10 mg/kg of ceftobiprole.
|
|---|---|---|
| 12 years to less than 18 years old | 30 to less than 50 | 10 mg/kg (up to 667 mg) Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for pediatric patients 12 years to less than 18 years old [see Dosage and Administration (2.5)].
every 12 hours |
| 15 to less than 30 | 10 mg/kg (up to 333 mg) every 12 hours | |
| 6 years to less than 12 years old | 30 to less than 50 | 10 mg/kg (up to 667 mg) Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 5.33 mg/mL for pediatric patients 2 years to less than 12 years old [see Dosage and Administration (2.5)].
every 12 hours |
| 15 to less than 30 | 10 mg/kg (up to 333 mg) every 24 hours | |
| 2 years to less than 6 years old | 30 to less than 50 | 13.3 mg/kg every 12 hours (up to 667 mg) |
| 15 to less than 30 | 13.3 mg/kg every 24 hours (up to 333 mg) |
Structured Label Content
Section 42229-5 (42229-5)
Reconstitution of ZEVTERA in the Vial
- For adult and pediatric patients 12 years of age and older, reconstitute ZEVTERA lyophilized powder in the vial with 10 mL of sterile water for injection or 10 mL of 5% dextrose injection.
- For pediatric patients aged 3 months to less than 12 years old, ZEVTERA lyophilized powder must be reconstituted with 10 mL of 5% dextrose injection.
- After reconstitution, shake the reconstituted ZEVTERA vial vigorously until dissolution is completed, which in some cases may take up to 10 minutes. The volume of the resulting reconstituted solution is approximately 10.6 mL.
- Allow any foam formed to dissipate and then inspect the reconstituted solution visually to ensure the product is in solution and particulate matter is absent.
- The reconstituted solution contains 66.7 mg/mL of ceftobiprole medocaril sodium and must be further diluted using aseptic technique with the appropriate diluent as described below, prior to administration. If it is not possible to dilute the reconstituted solution immediately, the reconstituted solution may be stored refrigerated for up to 24 hours, and at room temperature for up to 1 hour. Discard any unused reconstituted solution [see Dosage and Administration (2.6)].
1.4 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven, or strongly suspected, to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
There is no information on clinical signs and symptoms associated with an overdose of ZEVTERA. For patients who receive doses greater than the recommended dosage regimen and have unexpected adverse reactions associated with ZEVTERA, discontinue ZEVTERA, treat symptomatically, and institute general supportive treatment.
ZEVTERA can be removed by hemodialysis. However, no information is available on the use of hemodialysis to treat overdose [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
ZEVTERA (ceftobiprole medocaril sodium for injection) contains sodium salt of ceftobiprole medocaril, a semisynthetic, cephalosporin antibacterial, for intravenous use. Chemically, ceftobiprole medocaril is (6R,7R)-7-[[(2Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-hydroxyiminoacetyl]amino]-3-[(E)-[1-[(3R)-1-[(5-methyl-2-oxo-1,3-dioxol-4-yl)methoxycarbonyl]pyrrolidin-3-yl]-2-oxopyrrolidin-3-ylidene]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid. Its molecular weight is 690.6 g/mol. The empirical formula is C26H25N8NaO11S2.
Figure 1 Chemical Structure of ceftobiprole medocaril
ZEVTERA vials contain 667 mg of ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole). The powder for injection is a white, yellowish to slightly brownish sterile powder. Each vial includes inactive ingredient citric acid monohydrate (26.3 mg/vial) as a buffer component and sodium hydroxide (q.s.) as a pH adjustment agent. Each vial of ZEVTERA contains approximately 32 mg of sodium.
The pH of the reconstituted solution is 4.5–5.5.
16.1 How Supplied
ZEVTERA (ceftobiprole medocaril sodium for injection), a white, yellowish to slightly brownish sterile powder for reconstitution, is supplied in a single-dose clear glass vial sealed with a rubber stopper (not made with natural rubber latex) and an aluminum seal with a flip-off cap. Each vial contains 667 mg ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole) and is supplied in a carton containing 10 single-dose vials (NDC# 68547-578-10).
8.4 Pediatric Use
The safety and effectiveness of ZEVTERA have been established for the treatment of CABP in pediatric patients 3 months to less than 18 years. Use of ZEVTERA in this age group is supported by evidence from an adequate and well-controlled trial of ZEVTERA in adults, with additional pharmacokinetic, safety and efficacy data from pediatric trials [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.3)].
The safety and effectiveness of ZEVTERA have not been established for the treatment of CABP in pediatric patients less than 3 months of age.
The safety and effectiveness of ZEVTERA have not been established for the treatment of ABSSSI and SAB in pediatric patients.
8.5 Geriatric Use
Of the 835 adult CABP, ABSSSI, and SAB patients treated with ZEVTERA in the Trials 1, 2 and 3, 210 patients (25%) were 65 years of age and older, including 85 patients (10%) 75 years of age and older. No overall differences in safety or effectiveness of ZEVTERA were observed between patients 65 years of age and older and younger adult patients.
No clinically significant changes in the pharmacokinetics of ZEVTERA were observed in patients 65 years of age and older compared to younger adult patients [see Clinical Pharmacology (12.3)]. Dosage adjustment for geriatric patients should be based on renal function [see Dosage and Administration (2.3), Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
ZEVTERA is contraindicated in patients with a known history of severe hypersensitivity to ZEVTERA, or to other members of the cephalosporin class [see Warnings and Precautions (5.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section:
- Increased Mortality in Ventilator-Associated Bacterial Pneumonia Patients [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Contraindications (4) and Warning and Precautions (5.2)]
- Seizures and Other Central Nervous System Reactions [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Organic Anion Transporting Polypeptide 1B1/1B3 (OATP1B1/OATP1B3) Substrates: ZEVTERA may increase the plasma concentrations of OATP1B1 and OATP1B3 substrates. Concomitant administration is not recommended (7.1)
12.2 Pharmacodynamics
Ceftobiprole exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
Ceftobiprole medocaril is the prodrug of the active moiety ceftobiprole. No clinically significant differences in the pharmacokinetics of ceftobiprole were observed with ZEVTERA following single or multiple dose administration. Ceftobiprole exhibits linear and time-independent pharmacokinetics. The Cmax and AUC increase proportionally with the dose over a range of 125 mg to 1000 mg (0.25 to 2 times the highest approved recommended adult dosage).
The mean pharmacokinetic parameters of ceftobiprole in healthy adults with normal renal function after single and multiple 2-hour IV infusions of ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) administered every 8 hours are summarized in Table 11. Pharmacokinetic parameters were similar for single and multiple dose administrations.
| Parameter | Multiple doses administered every 8 hours as a 2-hour infusion (n = 27) |
|---|---|
| Cmax = Maximum plasma concentration; AUC0-8h = Area under the plasma concentration-time curve from 0 to 8 h; Vss = Volume of distribution at steady state; CL = Clearance. | |
| Cmax (µg/mL) | 33.0 (4.83) |
| AUC0-8h (µg•h/mL) | 102 (11.9) |
| Distribution | |
| % Bound to human plasma protein | 16 |
| % ELF/unbound plasma | 15-19 |
| Vss (L) | 18 |
| Metabolism (Minimally Metabolized) | |
| t1/2 (h) | 3.3 (0.3) |
| CL (L/h) | 4.98 (0.582) |
| Excretion | |
| Major route of elimination | Renal |
| % excreted unchanged in urine | 83 |
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZEVTERA is a cephalosporin antibacterial indicated for the treatment of:
- Adult patients with Staphylococcus aureus bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis (1.1),
- Adult patients with acute bacterial skin and skin structure infections (ABSSSI) (1.2), and
- Adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) (1.3).
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEVTERA and other antibacterial drugs, ZEVTERA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria (1.4).
12.1 Mechanism of Action
ZEVTERA is an antibacterial drug [see Microbiology (12.4)]
1.1 Staphylococcus Aureus (1.1 Staphylococcus aureus)
ZEVTERA is indicated for the treatment of adult patients with Staphylococcus aureus bloodstream infection (bacteremia) (SAB), including those with right-sided infective endocarditis, caused by methicillin-susceptible and methicillin-resistant isolates.
16.2 Storage and Handling
Store ZEVTERA vials refrigerated at 2 °C to 8 °C (36 °F to 46 °F) protected from light. Store in carton until time of use.
ZEVTERA must be reconstituted and then further diluted prior to administration by intravenous infusion. Store reconstituted and diluted solution of ZEVTERA as described elsewhere in the labeling [see Dosage and Administration (2.7)].
14.1 Staphylococcus Aureus (14.1 Staphylococcus aureus)
The efficacy of ZEVTERA in the treatment of adult patients with SAB, including right-sided infective endocarditis, was demonstrated in a randomized, controlled, double-blind, multinational, multicenter trial (Trial 1, NCT03138733). In this trial, adult patients were randomized to either ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) IV every 6 hours from study Day 1 to Day 8, and 667 mg IV every 8 hours from study Day 9 onwards) or daptomycin (6 mg/kg up to 10 mg/kg IV every 24 hours) plus optional aztreonam for coverage of gram-negative co-infections (the comparator). Randomization was stratified by study site, dialysis status, and use of prior antibacterial drugs. The study was performed in two cohorts based on findings in animal studies [see Nonclinical Toxicology (13.2)]. The initial cohort was enrolled to receive a maximum treatment duration of 28 days. Based on a safety evaluation of cohort 1, patients could be enrolled in cohort 2 to receive a treatment duration of up to 42 days. The duration of study treatment was based on the investigator's clinical evaluation.
Study patients were required to have at least one positive blood culture for S. aureus obtained within 72 hours prior to randomization, signs and symptoms of bacteremia (fever, elevated white blood cell count or immature neutrophils, tachycardia, or hypotension), and at least one of the following conditions of complicated S. aureus bacteremia: chronic dialysis, persistent SAB, acute bacterial skin and skin structure infection, metastatic infections of native tissue, osteomyelitis, epidural or cerebral abscess, or definite native-valve right-sided infective endocarditis by Modified Duke's Criteria. Patients with uncomplicated S. aureus bacteremia, left-sided infective endocarditis, prosthetic heart valves, foreign body material that could not be removed, severe neutropenia, or pneumonia, were excluded from the study.
Upon entry, patients were classified for likelihood of endocarditis using the modified Duke's criteria (definite, possible, or rejected). Echocardiography, including a transesophageal echocardiogram, was to be performed during screening or within 7 days following study enrollment. Final diagnoses were to be made by the study investigators within 7 days of randomization. The final diagnosis for persistent S. aureus bacteremia was made by an independent treatment-blinded Data Review Committee (DRC).
A total of 390 patients (192 ZEVTERA, 198 daptomycin) with SAB were randomized from 60 centers in the USA, Europe, Latin America, and South Africa. The modified ITT (mITT) population was used for the primary efficacy analysis, comprising 387 patients (189 ZEVTERA, 198 daptomycin ± aztreonam) who received study drugs and had a baseline blood culture positive for S. aureus.
Patient demographic and baseline characteristics were balanced between the treatment groups. The median age among the 387 patients in the mITT population was 58 years, ranging from 19 to 91 years), with 31% aged ≥ 65 years, 69% of patients were male, and 96% were White.
Frequent conditions of complicated SAB included acute bacterial skin and skin structure infections (61%), intra-abdominal abscesses (14%), osteoarticular infections (13%), and patients on chronic dialysis (13%). A total of 6.5% of patients had definite right-sided infective endocarditis. A total of 24% had bacteremia caused by methicillin-resistant S. aureus (MRSA).
The primary efficacy outcome in the study was overall success at the post-treatment evaluation (PTE) visit at 70 days post-randomization in the mITT population, as assessed by the independent DRC.
Overall success required survival, symptom improvement, S. aureus bacteremia bloodstream clearance, no new S. aureus bacteremia complications, and no use of other potentially effective antibacterial drugs.
Overall success rates at the PTE visit in the mITT population were 69.8% (132/189) in patients treated with ZEVTERA and 68.7% (136/198) in patients treated with the comparator. The overall primary study outcome and in pre-defined subgroups in the mITT population is shown in Table 12.
| Group/Subgroup | ZEVTERA n/N (%) |
Daptomycin ± aztreonam n/N (%) |
Between-group Difference (%) (2-sided 95% CI) Between-group difference of ZEVTERA minus daptomycin ± aztreonam using Cochran-Mantel-Haenszel weights method adjusted for actual stratum (dialysis status and prior antibacterial treatment use).
|
|---|---|---|---|
| Overall | 132/189 (69.8) | 136/198 (68.7) | 2.0 (−7.1, 11.1) |
| MSSA (methicillin-susceptible) | 100/141 (70.9) | 97/146 (66.4) | 4.8 (−5.9, 15.5) |
| MRSA (methicillin-resistant) | 31/45 (68.9) | 38/49 (77.6) | −8.3 (−25.3, 8.6) |
| ABSSSI | 81/116 (69.8) | 80/121 (66.1) | 4.5 (−7.3, 16.3) |
| Definite right-sided infective endocarditis | 10/15 (66.7) | 7/10 (70.0) | −6.6 (−40.1, 27.0) |
For patients enrolled in Ukraine, overall success rates at the PTE visit in the mITT population were 77/84 (84.1%) in patients treated with ZEVTERA and 82/92 (89.1%) in patients treated with the comparator. For patients enrolled outside Ukraine, overall success rates at the PTE visit in the mITT population were 58/101 (57.4%) in patients treated with ZEVTERA and 54/106 (50.9%) in patients treated with the comparator.
All-cause mortality between randomization and the PTE visit was observed in 17/189 (9.0%) patients treated with ZEVTERA, and 18/198 (9.1%) patients treated with the comparator. Other key secondary efficacy outcomes included microbiological eradication at the PTE visit in the mITT population, which was achieved in 82% of patients treated with ZEVTERA and 77% of patients treated with the comparator, and the development of new S. aureus bacteremia complications, which occurred in 6% of patients treated with ZEVTERA and 6% of patients treated with the comparator. S. aureus bloodstream clearance, defined as 2 consecutive study days with negative blood cultures for S. aureus with no subsequent S. aureus relapse or reinfection, was achieved after a median of 4 days in each treatment group.
S. aureus bloodstream clearance in patients with MSSA was achieved after a median of 3 days in patients treated with ZEVTERA and after a median of 4 days in patients treated with the comparator.
S. aureus bloodstream clearance in patients with MRSA was achieved after a median of 5 days in each treatment group.
Failure of treatment due to relapse of S. aureus bacteremia was assessed by the DRC in two patients (1%) treated with ZEVTERA, and in four patients (2%) treated in the comparator arm. No increase in ceftobiprole MIC was observed for the two isolates from the ZEVTERA-treated patients.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Increased Mortality with Unapproved use in Ventilator-Associated Bacterial Pneumonia (VABP) Patients: The safety and effectiveness of ZEVTERA for the treatment of VABP has not been established and the use of ZEVTERA for VABP is not approved (5.1).
- Hypersensitivity Reactions: Discontinue ZEVTERA if a hypersensitivity reaction occurs, and institute appropriate treatment (5.2).
- Seizures and other adverse central nervous system (CNS) reactions have been associated with the use of ZEVTERA. If seizures or other CNS adverse reactions occur, evaluate patients to determine whether ZEVTERA should be discontinued (5.3).
- Clostridioides difficile-associated diarrhea (CDAD) has been reported with nearly all systemic antibacterial agents, including ZEVTERA. Evaluate if diarrhea occurs (5.4).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The recommended dosage of ZEVTERA for adult patients with SAB, ABSSSI and CABP is described in the table below (2.1):
Indication in Adults Dose Frequency SAB 667 mg Every 6 hours on Days 1 to 8 Every 8 hours from Day 9 ABSSSI 667 mg Every 8 hours CABP 667 mg Every 8 hours
- Duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP (2.1).
- Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL (2.1, 2.6).
- The recommended dosage of ZEVTERA for pediatric patients (3 months to less than 18 years old) with CABP is described below (2.2).
Pediatric Age Group for CABP Dose Frequency 12 years to less than 18 years old 13.3 mg/kg (up to 667 mg/dose) Every 8 Hours Greater than or equal to 3 months and less than 12 years old 20 mg/kg (up to 667 mg/dose) Every 8 Hours
- Duration of treatment in pediatric patients is 7 days to 14 days for CABP (2.1).
- Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for pediatric patients aged 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for pediatric patients aged 3 months to less than 12 years old (2.2, 2.6).
- Reduce the dosage in adult patients with CLCR less than 50 mL/min, including patients with CLCR less than 15 mL/min on hemodialysis (2.3 and 8.6).
- Increase the dosage in adult patients with CLCR greater than 150 mL/min (2.3).
- Reduce the dosage in pediatric patients aged 2 years old to less than 18 years old with eGFR less than 50 mL/min/1.73 m2 and greater than or equal to 15 mL/min/1.73 m2 (2.4 and 8.6).
- See Full Prescribing Information for instructions for preparation of ZEVTERA solution infusion solution (2.5).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
ZEVTERA (ceftobiprole medocaril sodium for injection) is available in a single-dose vial as a white, yellowish to slightly brownish, cake to broken cake or powder containing 667 mg of ceftobiprole medocaril sodium (equivalent to 500 mg of ceftobiprole) for reconstitution.
5.4 Clostridioides Difficile (5.4 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial agents, including ZEVTERA, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile should be discontinued, if possible. Manage fluid and electrolyte levels as appropriate, supplement protein intake, monitor antibacterial treatment of C. difficile, and institute surgical evaluation as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions and altered laboratory tests have been identified during post-approval use of ZEVTERA and ceftobiprole outside of the United States, or other cephalosporin-class antibacterial drugs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Altered Laboratory Tests: Positive direct Coombs' test, false-positive test for urinary glucose.
Blood and lymphatic system disorders: Agranulocytosis, aplastic anemia.
Hepatobiliary disorders: Hepatic dysfunction including cholestasis.
Immune system disorders: Drug fever, serum sickness-like reaction.
Nervous system disorders: Myoclonus.
Renal and urinary disorders: Renal dysfunction, toxic nephropathy.
Vascular disorders: Hemorrhage, hypertension.
5.2 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, were observed in ZEVTERA-treated patients in clinical trials [see Adverse Reactions (6.1)]. Serious and occasionally fatal hypersensitivity reactions and serious skin reactions have been reported in patients receiving beta-lactam antibacterial drugs.
Before therapy with ZEVTERA is instituted, careful inquiry about previous hypersensitivity reactions to other cephalosporins, penicillins, or other beta-lactam antibacterial drugs should be made. Maintain clinical supervision if this product is to be given to a penicillin- or other beta-lactam-allergic patient, because cross sensitivity among beta-lactam antibacterial agents has been established. Discontinue ZEVTERA if a hypersensitivity reaction occurs, and institute appropriate treatment.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ZEVTERA was evaluated in adults in four controlled comparative phase 3 clinical trials (Trials 1 through 4) which included 1221 patients treated with ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) administered by IV infusion over 2 hours every 6 to 8 hours and 1248 patients treated with comparator for a treatment period up to 42 days. The median age of patients treated with ZEVTERA was 56 years, ranging between 18 and 95 years old. Patients treated with ZEVTERA were predominantly male (64%) and White (82%).
The safety of ZEVTERA was also evaluated in pediatric patients aged 3 months to less than 18 years in a controlled phase 3 clinical trial (Trial 5) which included 138 patients with CABP and hospital-acquired bacterial pneumonia (HABP) requiring hospitalization. Although HABP was included in the safety data, the safety and effectiveness of ZEVTERA for the treatment of HABP has not been established and ZEVTERA is not approved for the treatment of HABP.
Principal Display Panel 667 Mg Vial Box (PRINCIPAL DISPLAY PANEL - 667 mg Vial Box)
NDC# 68547-578-10
Rx Only
10 single-dose vials
ZEVTERA® 667 mg/vial*
(ceftobiprole medocaril sodium for injection)
*equivalent to 500 mg ceftobiprole
For intravenous use only.
Reconstitute and further dilute before use.
Single-dose vial. Discard unused portion.
1.3 Community Acquired Bacterial Pneumonia (1.3 Community-Acquired Bacterial Pneumonia)
ZEVTERA is indicated for the treatment of adult and pediatric patients (3 months to less than 18 years old) with community-acquired bacterial pneumonia (CABP) caused by susceptible isolates of the following gram-positive and gram-negative microorganisms: Staphylococcus aureus (methicillin-susceptible isolates), Streptococcus pneumoniae, Haemophilus influenzae, Haemophilus parainfluenzae, Escherichia coli, and Klebsiella pneumoniae.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Both rats and marmosets showed convulsions and thrombi and/or emboli with dosing durations greater than one month. In the 13-week intravenous rat study, clonic convulsions were noted at 500 mg/kg/day (2 times the MRHD based on BSA comparisons) with early deaths seen at 250 mg/kg in the males (1 time the MRHD based on BSA comparison). In the 13-week intravenous marmoset study, convulsions and thrombi were noted at multiple sites distal to the injection site (i.e., lung, and heart) at doses ≥ 50 mg/kg/day (0.1 times the MRHD, based on BSA comparisons). While dogs did not show convulsions in a 13-week toxicology study, catheter patency could only be maintained between 4 to 11 weeks in most animals at doses up to 32 mg/kg/day (0.4 times the MRHD based on BSA comparisons). Fibrin, erythrocytes and inflammatory cells within the lumens of the catheters were noted in the 13-week dog study, while thrombi were observed at the injection site in all species tested. Kidney findings were also noted in the 13-week studies in rats (tubular/duct eosinophilic bodies and presence of foreign material) at 1 times the MRHD.
5.5 Development of Drug Resistant Bacteria (5.5 Development of Drug-Resistant Bacteria)
Prescribing ZEVTERA in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.5 Preparation of Zevtera Infusion Solution (2.5 Preparation of ZEVTERA Infusion Solution)
ZEVTERA must first be reconstituted in the vial and then further diluted prior to administration by intravenous infusion over a period of 2 hours. Aseptic technique must be followed in preparing the infusion solution. See additional instructions for reconstitution and dilution below.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. The solution for infusion should be clear to slightly opalescent and yellowish in color. Discard if discoloration or visible particles are observed.
2.7 Drug Compatibilities and Incompatibilities
ZEVTERA is compatible with 5% dextrose injection and 0.9 % sodium chloride injection [see Dosage and Administration (2.5)]. The compatibility of ZEVTERA with other drugs and infusion solutions other than 5% dextrose Injection or 0.9% sodium chloride Injection, has not been established.
ZEVTERA must not be mixed or administered simultaneously with calcium-containing solutions. Do not mix ZEVTERA with, or co-administer through, the same intravenous line or cannula with other drug products.
1.2 Acute Bacterial Skin and Skin Structure Infections
ZEVTERA is indicated for the treatment of adult patients with acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following gram-positive and gram-negative microorganisms: Staphylococcus aureus (methicillin-susceptible and methicillin-resistant isolates), Streptococcus pyogenes, and Klebsiella pneumoniae.
5.3 Seizures and Other Central Nervous System Reactions
Seizures and other adverse central nervous system (CNS) reactions have been reported during treatment with ZEVTERA and other cephalosporins [see Adverse Reactions (6.1)].
Nonconvulsive status epilepticus (NCSE), encephalopathy, coma, asterixis, neuromuscular excitability, and myoclonia have been reported with cephalosporins particularly in patients with a history of epilepsy or when recommended dosages of cephalosporins were exceeded due to renal impairment. Adjust ZEVTERA dosing based on creatinine clearance [see Dosage and Administration (2.3, 2.4)].
Anticonvulsant therapy should be continued in patients with known seizure disorders. If CNS adverse reactions, including seizures, occur, patients should undergo a neurological evaluation to determine whether ZEVTERA should be discontinued.
2.3 Recommended Dosage Regimen in Adult Patients With Renal Impairment (2.3 Recommended Dosage Regimen in Adult Patients with Renal Impairment)
The recommended ZEVTERA dosage in adult patients with renal impairment (CLCR less than 50 mL/min), including patients receiving hemodialysis is shown in Table 3. The duration of treatment in adult patients with renal impairment is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL to adult patients with renal impairment [see Dosage and Administration (2.5)].
In adult patients with augmented renal clearance (CLCR greater than 150 mL/min), increase the ZEVTERA dosage to 667 mg every 6 hours.
| Indication Duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
|
Creatinine Clearance, CLCR (mL/min) Based on calculated creatinine clearance [see Use in Specific Populations (8.6)].
|
Dose 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole; 333 mg ceftobiprole medocaril sodium is equivalent to 250 mg of ceftobiprole.
|
Frequency Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL [see Dosage and Administration (2.5)].
|
|---|---|---|---|
| SAB | 30 to less than 50 mL/min | 667 mg | Every 8 hours on Days 1 to 8 |
| Every 12 hours from Day 9 | |||
| 15 to less than 30 mL/min | 333 mg | Every 8 hours on Days 1 to 8 | |
| Every 12 hours from Day 9 | |||
| Less than 15 mL/min, including hemodialysis Administer ZEVTERA after intermittent hemodialysis on hemodialysis days, because ceftobiprole is removed by hemodialysis.
|
333 mg | Every 24 hours | |
| ABSSSI or CABP | 30 to less than 50 | 667 mg | Every 12 hours |
| 15 to less than 30 | 333 mg | Every 12 hours | |
| Less than 15 mL/min, including hemodialysis | 333 mg | Every 24 hours |
14.2 Acute Bacterial Skin and Skin Structure Infections (absssi) in Adults (14.2 Acute Bacterial Skin and Skin Structure Infections (ABSSSI) in Adults)
The efficacy of ZEVTERA in the treatment of adult patients with ABSSSI was demonstrated in a randomized, controlled, double-blind, multinational, multicenter trial (Trial 2, NCT03137173). Patients were randomized to either ZEVTERA 667 mg (equivalent to 500 mg of ceftobiprole) IV every 8 hours or vancomycin plus aztreonam (vancomycin 1 gram or 15 mg/kg IV every 12 hours, aztreonam 1 gram IV every 12 hours). Randomization was stratified by study site and type of ABSSSI. Treatment duration was 5 to 14 days. Patients could receive concomitant metronidazole for suspected anaerobic infection. A switch to oral therapy was not allowed.
The study enrolled adult patients with ABSSSI (cellulitis/erysipelas, major cutaneous abscess, wound infection) with a lesion area of at least 75 cm2, systemic or regional signs of infection, and a requirement for IV antibiotic treatment. Patients with other forms of ABSSSI, uncomplicated skin and skin structure infections, or infections related to prosthetic devices, were excluded.
The primary endpoint, assessed in the ITT population, was early clinical response 48–72 hours after start of treatment. The ITT primary analysis population comprised all patients randomized. Early clinical response required a reduction of the primary skin lesion by at least 20%, survival for at least 72 hours, and the absence of concomitant antibacterial treatment or additional unplanned surgery.
The main secondary endpoint was investigator-assessed clinical success at the test-of-cure (TOC) visit 15 to 22 days after randomization and at least 5 days after the end-of-treatment.
A total of 679 patients (335 ZEVTERA, 344 vancomycin plus aztreonam) were randomized from 32 centers in the USA and Europe. The microbiological ITT (mITT) population included 506 patients with at least one valid baseline pathogen.
Patient demographic and baseline characteristics in the ITT population were balanced between the treatment groups. Approximately 95% of patients were classified as White, 59% were male, the median age was 50 years (range: 18 to 89), and the mean body mass index was 28 kg/m2, 62% of patients were enrolled in the USA, and 38% in Europe. A history of diabetes was present in 11% of patients and current injection drug use was reported in 33% of patients. The types of ABSSSI included cellulitis/erysipelas (33%), wound infection (39%), and major cutaneous abscesses (28%). The median area of the primary skin infection was 259 cm2.
The primary and main secondary outcomes are shown in Table 13.
| ZEVTERA n/N (%) |
Vancomycin plus aztreonam n/N (%) |
Between-group Difference (%) (2-sided 95% CI) Between-group difference of ZEVTERA minus vancomycin plus aztreonam, using Cochran-Mantel-Haenszel weights method adjusted for geographical region and actual type of ABSSSI.
|
|
|---|---|---|---|
| Primary endpoint analyses Early clinical response based on at least 20% reduction from baseline in the area of the primary lesion, survival for ≥ 72 hours from the time of administration of the first dose of study drug, no use of concomitant systemic antibacterial treatments or topical antibacterial administration on the primary lesion, and no additional unplanned surgical procedure for the ABSSSI after the start of study treatment.
|
|||
| Early clinical response at 48–72 hours after start of treatment (ITT) | 306/335 (91.3) | 303/344 (88.1) | 3.3 (−1.2, 7.8) |
| Main secondary endpoint analysis Clinical success was defined as complete (cured) or nearly complete (improved) resolution of baseline signs and symptoms of the primary infection, such that no further antibacterial treatment was needed.
|
|||
| Investigator-assessed clinical success at the TOC visit (ITT) | 302/335 (90.1) | 306/344 (89.0) | 1.0 (−3.5, 5.6) |
Clinical response rates at TOC by the most common baseline pathogens in the microbiological ITT (mITT) population, defined as all randomized patients with a baseline pathogen, are presented in Table 14.
| Early clinical response at 48–72 hours after start of treatment Early clinical response based on at least a 20% reduction from baseline in the area of the primary lesion, survival for ≥ 72 hours from the time of administration of the first dose of study drug, no use of concomitant systemic antibacterial treatments, or topical antibacterial administration on the primary lesion and no additional unplanned surgical procedure for the ABSSSI after the start of study treatment.
(mITT) |
Investigator-assessed clinical success at the TOC visit Clinical success was defined as complete (cured) or nearly complete (improved) resolution of baseline signs and symptoms of the primary infection such that no further antibacterial treatment was needed.
(mITT) |
|||
|---|---|---|---|---|
| Pathogen | ZEVTERA n/N (%) |
Vancomycin plus aztreonam n/N (%) |
ZEVTERA n/N (%) |
Vancomycin plus aztreonam n/N (%) |
| Any Gram-positive pathogen | 213/228 (93.4) | 220/244 (90.2) | 203/228 (89.0) | 217/244 (88.9) |
| Staphylococcus aureus | 186/197 (94.4) | 185/205 (90.2) | 174/197 (88.3) | 180/205 (87.8) |
| Methicillin-susceptible | 102/108 (94.4) | 112/124 (90.3) | 95/108 (88.0) | 107/124 (86.3) |
| Methicillin-resistant | 81/86 (94.2) | 67/73 (91.8) | 76/86 (88.4) | 66/73 (90.4) |
| Streptococcus pyogenes | 17/20 (85.0) | 23/24 (95.8) | 17/20 (85.0) | 22/24 (91.7) |
| Any Gram-negative pathogen | 26/27 (96.3) | 33/37 (89.2) | 24/27 (88.9) | 34/37 (91.9) |
| Enterobacterales | 16/16 (100.0) | 24/27 (88.9) | 15/16 (93.8) | 25/27 (92.6) |
| Klebsiella pneumoniae | 8/8 (100.0) | 5/5 (100.0) | 7/8 (87.5) | 5/5 (100.0) |
7.1 Organic Anion Transporting Polypeptide 1b1/1b3 (oatp1b1/oatp1b3) Substrates (7.1 Organic Anion Transporting Polypeptide 1B1/1B3 (OATP1B1/OATP1B3) Substrates)
ZEVTERA may increase the plasma concentrations of OATP1B1 and OATP1B3 substrates. Concomitant administration is not recommended [see Clinical Pharmacology (12.3)].
2.1 Recommended Dosage and Administration for Sab, Absssi and Cabp in Adult Patients (2.1 Recommended Dosage and Administration for SAB, ABSSSI and CABP in Adult Patients)
The recommended dosage of ZEVTERA for the treatment of adult patients with SAB, ABSSSI and CABP is described in Table 1. The duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL to adult patients [see Dosage and Administration (2.5)].
| Indication Duration of treatment in adult patients is up to 42 days for SAB and 5 days to 14 days for ABSSSI and CABP.
|
Dose | Frequency Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL [see Dosage and Administration (2.5)]
|
|---|---|---|
| SAB | 667 mg 667 mg of ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole.
|
Every 6 hours on Days 1 to 8 |
| Every 8 hours from Day 9 | ||
| ABSSSI | 667 mg | Every 8 hours |
| CABP | 667 mg | Every 8 hours |
5.1 Increased Mortality With Unapproved Use in Ventilator Associated Bacterial Pneumonia Patients (5.1 Increased Mortality with Unapproved Use in Ventilator-Associated Bacterial Pneumonia Patients)
In Trial 4, an increase in mortality was seen in the subgroup of patients with ventilator-associated bacterial pneumonia (VABP) who were treated with ZEVTERA (35/103 [34%] versus 24/102 [24%] in comparator-treated patients) [see Adverse Reactions (6.1)]. The cause of this increased mortality has not been established. Generally, deaths were associated with complications of infection or underlying co-morbidities. The safety and effectiveness of ZEVTERA for the treatment of VABP has not been established and the use of ZEVTERA for VABP is not approved.
2.2 Recommended Dosage and Administration for Cabp in Pediatric Patients (3 Months to Less Than 18 Years Old) (2.2 Recommended Dosage and Administration for CABP in Pediatric Patients (3 months to less than 18 years old))
For treatment of pediatric patients with CABP, the recommended dosage of ZEVTERA is described in Table 2, based on patient age and weight [see Clinical Pharmacology (12.3)]. The duration of treatment for CABP in pediatric patients (3 months to less than 18 years old) is 7 days to 14 days.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5)].
| Pediatric Age Group | Dose Duration of treatment for CABP in pediatric patients is 7 days to 14 days.
|
Frequency |
|---|---|---|
| 12 years to less than 18 years old | 13.3 mg/kg (up to 667 mg/dose 13.3 mg/kg of ceftobiprole medocaril sodium is equivalent to 10 mg/kg of ceftobiprole; 20 mg/kg ceftobiprole medocaril sodium is equivalent to 15 mg/kg of ceftobiprole. 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole. ) |
Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old [see Dosage and Administration (2.5)].
|
| 3 months to less than 12 years old | 20 mg/kg (up to 667 mg/dose) | Every 8 Hours Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 5.33 mg/mL for patients greater than or equal to 3 months to less than 12 years old [see Dosage and Administration (2.5)].
|
2.4 Recommended Dosage Regimen for Cabp in Pediatric Patients (2 Years to Less Than 18 Years Old) With Renal Impairment (2.4 Recommended Dosage Regimen for CABP in Pediatric Patients (2 Years to Less than 18 Years Old) with Renal Impairment)
The recommended dosage of ZEVTERA in pediatric patients (2 years to less than 18 years old) with renal impairment (with eGFR less than 50 mL/min/1.73 m2 and greater than or equal to 15 mL/min/1.73 m2) is shown in Table 4. The duration of treatment for CABP in pediatric patients (2 years to less than 18 years old) with renal impairment is 7 days to 14 days.
Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for patients 12 years to less than 18 years old and at a concentration of 5.33 mg/mL for patients 2 years to less than 12 years old.
There is insufficient information to recommend dosage adjustments in pediatric patients 2 years of age and older with an eGFR less than 15 mL/min/1.73m2.
There is insufficient information to recommend dosage adjustments in pediatric patients less than 2 years of age with any degree of renal impairment.
| Pediatric Age Group Duration of treatment for CABP in pediatric patients (2 years to less than 18 years old) and with renal impairment is 7 days to 14 days.
|
eGFR (mL/min/1.73 m2) Calculate using a validated GFR estimating equation for the approved age of the pediatric population.
|
Dosage Regimen 667 mg ceftobiprole medocaril sodium is equivalent to 500 mg of ceftobiprole; 333 mg ceftobiprole medocaril sodium is equivalent to 250 mg of ceftobiprole; 10 mg/kg of ceftobiprole medocaril sodium is equivalent to 7.5 mg/kg of ceftobiprole; 13.3 mg/kg ceftobiprole medocaril sodium is equivalent to 10 mg/kg of ceftobiprole.
|
|---|---|---|
| 12 years to less than 18 years old | 30 to less than 50 | 10 mg/kg (up to 667 mg) Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 2.67 mg/mL for pediatric patients 12 years to less than 18 years old [see Dosage and Administration (2.5)].
every 12 hours |
| 15 to less than 30 | 10 mg/kg (up to 333 mg) every 12 hours | |
| 6 years to less than 12 years old | 30 to less than 50 | 10 mg/kg (up to 667 mg) Administer each prepared intravenous infusion solution of ZEVTERA over 2 hours at a concentration of 5.33 mg/mL for pediatric patients 2 years to less than 12 years old [see Dosage and Administration (2.5)].
every 12 hours |
| 15 to less than 30 | 10 mg/kg (up to 333 mg) every 24 hours | |
| 2 years to less than 6 years old | 30 to less than 50 | 13.3 mg/kg every 12 hours (up to 667 mg) |
| 15 to less than 30 | 13.3 mg/kg every 24 hours (up to 333 mg) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:30.681992 · Updated: 2026-03-14T22:40:41.023270