2b9ac0cd-9dd1-4f78-a16d-9d40f5091c0a
34390-5
Human OTC Drug Label
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antihistamine
Medication Information
Purpose
Antihistamine
Description
Drug Facts
Uses
reduces hives and relieves itching due to hives (urticaria). This product will not prevent hives or an allergic skin reaction from occurring.
Section 42229-5
Drug Facts
Warnings
Severe Allergy Warning: Get emergency help immediately if you have hives along with any of the following symptoms:
- trouble swallowing
- dizziness or loss of consciousness
- swelling of tongue
- swelling in or around mouth
- trouble speaking
- drooling
- wheezing or problems breathing
These symptoms may be signs of anaphylactic shock. This condition can be life threatening if not treated by a health professional immediately. Symptoms of anaphylactic shock may occur when hives first appear or up to a few hours later.
Not a Substitute for Epinephrine. If your doctor has prescribed an epinephrine injector for "anaphylaxis" or severe allergy symptoms that could occur with your hives, never use this product as a substitute for the epinephrine injector. If you have been prescribed an epinephrine injector, you should carry it with you at all times.
Directions
For 5 mg:
| adults and children 6 years and over |
1 to 2 tablets once daily depending upon severity of symptoms; do not take more than 2 tablets in 24 hours |
| adults 65 years and over |
1 tablet once a day; do not take more than 1 tablet in 24 hours. |
| children under 6 years of age |
ask a doctor |
| consumers with liver or kidney disease |
ask a doctor |
For 10 mg:
| adults and children 6 years and over |
one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| adults 65 years and over |
ask a doctor |
| children under 6 years of age |
ask a doctor |
| consumers with liver or kidney disease |
ask a doctor |
Do Not Use
- to prevent hives from any known cause such as:
- foods
- insect stings
- medicines
- latex or rubber gloves
because this product will not stop hives from occurring. Avoiding the cause of your hives is the only way to prevent them. Hives can sometimes be serious. If you do not know the cause of your hives, see your doctor for a medical exam. Your doctor may be able to help you find a cause.
- if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Questions?
call 1-855-274-4122
Important: Read all product information before using. Keep this carton for important information.
Distributed by:
AUROHEALTH LLC
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Other Information
- store between 20° to 25°C (68° to 77°F)
- TAMPER EVIDENT: DO NOT USE IF SEAL OVER BOTTLE OPENING IS BROKEN OR MISSING.
Inactive Ingredients
colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, titanium dioxide
When Using This Product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Stop Use and Ask A Doctor If
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve after 3 days of treatment.
- the hives have lasted more than 6 weeks.
If Pregnant Or Breast Feeding:
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away. [1-800-222-1222]
Active Ingredient (in Each Tablet)
For 5 mg:
Cetirizine hydrochloride USP 5 mg
For 10 mg:
Cetirizine hydrochloride USP 10 mg
Ask A Doctor Before Use If You Have
- liver or kidney disease. Your doctor should determine if you need a different dose.
- hives that are an unusual color, look bruised or blistered
- hives that do not itch
Ask A Doctor Or Pharmacist Before Use If You Are
taking tranquilizers or sedatives.
Package Label Principal Display Panel 5 Mg (10 X 10 Blister Carton Label)
AUROHEALTH
NDC 58602-446-21
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP
5 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
100
(10x10's)
Unit dose Tablets Reduces Itching
5 mg each Due to Hives
Package Label Principal Display Panel 10 Mg (10 X 10 Blister Carton Label)
AUROHEALTH
NDC 58602-447-21
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP 10 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
100 Tablets
(10x10 Unit-dose) Reduces Itching
10 mg each Actual Size Due to Hives
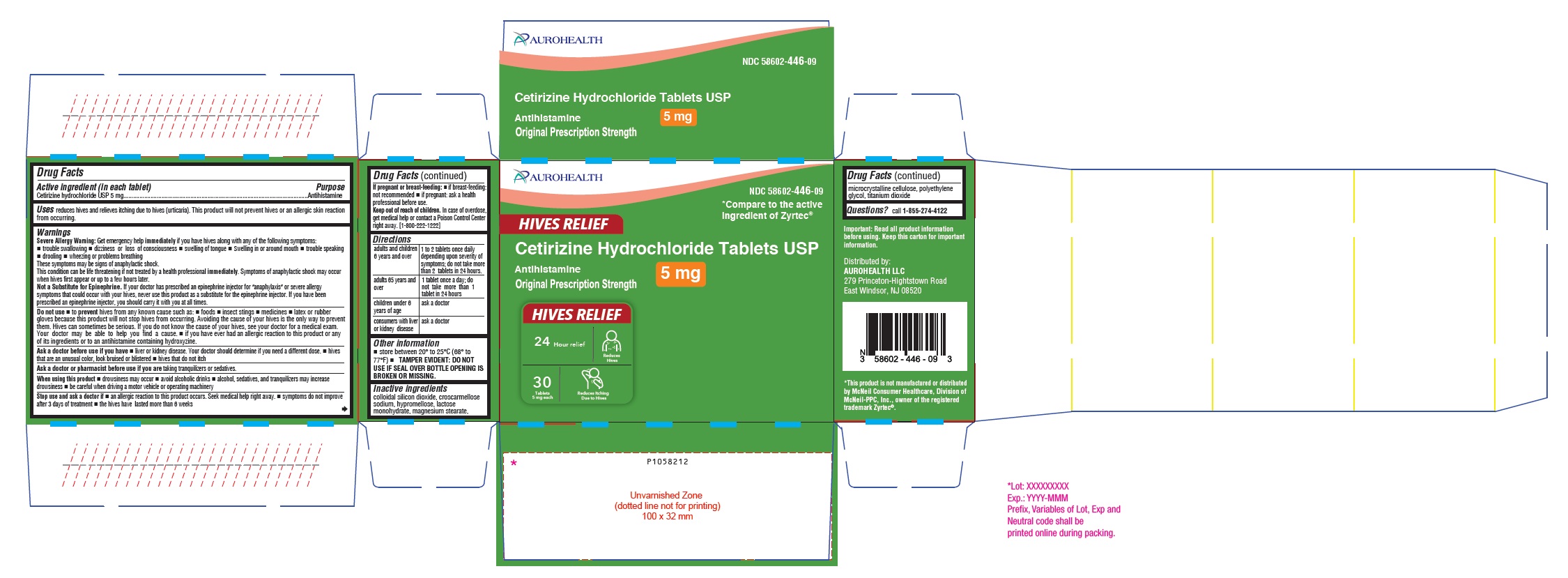
Package Label Principal Display Panel 5 Mg (30's Tablet Container Carton Label)
AUROHEALTH
NDC 58602-446-09
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP
5 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
30
Tablets Reduces Itching
5 mg each Due to Hives
Package Label Principal Display Panel 10 Mg (30's Tablet Container Carton Label)
AUROHEALTH
NDC 58602-447-09
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP 10 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
30
Tablets Reduces Itching
10 mg each Actual Size Due to Hives
Structured Label Content
Uses
reduces hives and relieves itching due to hives (urticaria). This product will not prevent hives or an allergic skin reaction from occurring.
Section 42229-5 (42229-5)
Drug Facts
Purpose
Antihistamine
Warnings
Severe Allergy Warning: Get emergency help immediately if you have hives along with any of the following symptoms:
- trouble swallowing
- dizziness or loss of consciousness
- swelling of tongue
- swelling in or around mouth
- trouble speaking
- drooling
- wheezing or problems breathing
These symptoms may be signs of anaphylactic shock. This condition can be life threatening if not treated by a health professional immediately. Symptoms of anaphylactic shock may occur when hives first appear or up to a few hours later.
Not a Substitute for Epinephrine. If your doctor has prescribed an epinephrine injector for "anaphylaxis" or severe allergy symptoms that could occur with your hives, never use this product as a substitute for the epinephrine injector. If you have been prescribed an epinephrine injector, you should carry it with you at all times.
Directions
For 5 mg:
| adults and children 6 years and over |
1 to 2 tablets once daily depending upon severity of symptoms; do not take more than 2 tablets in 24 hours |
| adults 65 years and over |
1 tablet once a day; do not take more than 1 tablet in 24 hours. |
| children under 6 years of age |
ask a doctor |
| consumers with liver or kidney disease |
ask a doctor |
For 10 mg:
| adults and children 6 years and over |
one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| adults 65 years and over |
ask a doctor |
| children under 6 years of age |
ask a doctor |
| consumers with liver or kidney disease |
ask a doctor |
Do Not Use (Do not use)
- to prevent hives from any known cause such as:
- foods
- insect stings
- medicines
- latex or rubber gloves
because this product will not stop hives from occurring. Avoiding the cause of your hives is the only way to prevent them. Hives can sometimes be serious. If you do not know the cause of your hives, see your doctor for a medical exam. Your doctor may be able to help you find a cause.
- if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Questions?
call 1-855-274-4122
Important: Read all product information before using. Keep this carton for important information.
Distributed by:
AUROHEALTH LLC
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Other Information (Other information)
- store between 20° to 25°C (68° to 77°F)
- TAMPER EVIDENT: DO NOT USE IF SEAL OVER BOTTLE OPENING IS BROKEN OR MISSING.
Inactive Ingredients (Inactive ingredients)
colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, titanium dioxide
When Using This Product (When using this product)
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve after 3 days of treatment.
- the hives have lasted more than 6 weeks.
If Pregnant Or Breast Feeding: (If pregnant or breast-feeding:)
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away. [1-800-222-1222]
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
For 5 mg:
Cetirizine hydrochloride USP 5 mg
For 10 mg:
Cetirizine hydrochloride USP 10 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- liver or kidney disease. Your doctor should determine if you need a different dose.
- hives that are an unusual color, look bruised or blistered
- hives that do not itch
Ask A Doctor Or Pharmacist Before Use If You Are (Ask a doctor or pharmacist before use if you are)
taking tranquilizers or sedatives.
Package Label Principal Display Panel 5 Mg (10 X 10 Blister Carton Label) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg (10 x 10 Blister Carton Label))
AUROHEALTH
NDC 58602-446-21
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP
5 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
100
(10x10's)
Unit dose Tablets Reduces Itching
5 mg each Due to Hives
Package Label Principal Display Panel 10 Mg (10 X 10 Blister Carton Label) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg (10 x 10 Blister Carton Label))
AUROHEALTH
NDC 58602-447-21
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP 10 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
100 Tablets
(10x10 Unit-dose) Reduces Itching
10 mg each Actual Size Due to Hives
Package Label Principal Display Panel 5 Mg (30's Tablet Container Carton Label) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg (30's Tablet Container Carton Label))
AUROHEALTH
NDC 58602-446-09
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP
5 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
30
Tablets Reduces Itching
5 mg each Due to Hives
Package Label Principal Display Panel 10 Mg (30's Tablet Container Carton Label) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg (30's Tablet Container Carton Label))
AUROHEALTH
NDC 58602-447-09
*Compare to the active
ingredient of Zyrtec®
HIVES RELIEF
Cetirizine Hydrochloride Tablets USP 10 mg
Antihistamine
Original Prescription Strength
HIVES RELIEF
24 Hour relief Reduces
Hives
30
Tablets Reduces Itching
10 mg each Actual Size Due to Hives
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:32.408287 · Updated: 2026-03-14T23:07:28.673990