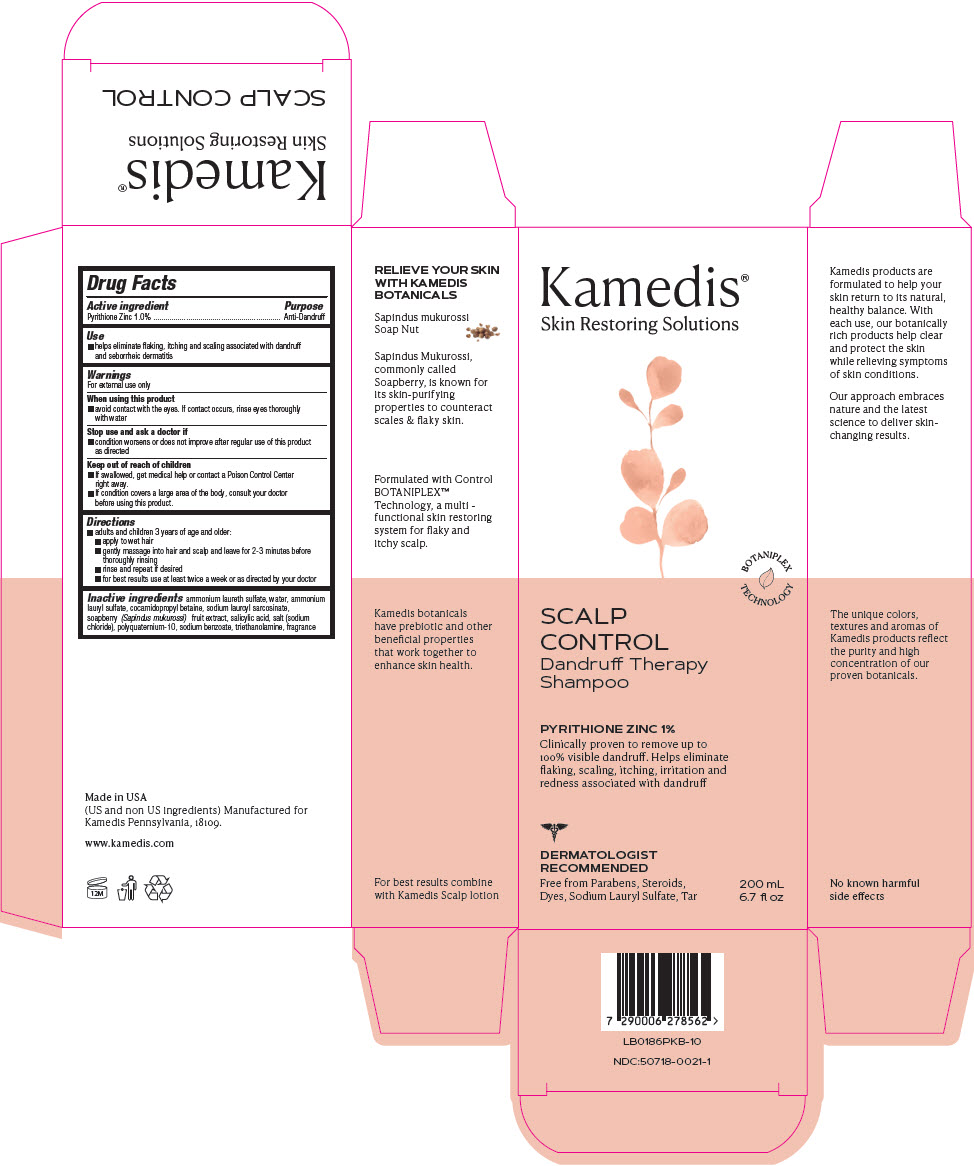
Control - Dandruff Therapy Shampoo
2a5fc8ae-1d98-496c-a6ca-922897e3ab47
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Pyrithione Zinc 1.0%
Purpose
Anti-Dandruff
Medication Information
Purpose
Anti-Dandruff
Description
Pyrithione Zinc 1.0%
Use
- helps eliminate flaking, itching and scaling associated with dandruff and seborrheic dermatitis
Section 42229-5
Made in USA
(US and non US ingredients) Manufactured for
Kamedis Pennsylvania, 18109.
www.kamedis.com
Warnings
For external use only
Directions
- adults and children 3 years of age and older:
- apply to wet hair
- gently massage into hair and scalp and leave for 2-3 minutes before thoroughly rinsing
- rinse and repeat if desired
- for best results use at least twice a week or as directed by your doctor
Drug Facts
Active Ingredient
Pyrithione Zinc 1.0%
Inactive Ingredients
ammonium laureth sulfate, water, ammonium lauryl sulfate, cocamidopropyl betaine, sodium lauroyl sarcosinate, soapberry (Sapindus mukurossi) fruit extract, salicylic acid, salt (sodium chloride), polyquaternium-10, sodium benzoate, triethanolamine, fragrance
When Using This Product
- avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with water
Stop Use and Ask A Doctor If
- condition worsens or does not improve after regular use of this product as directed
Keep Out of Reach of Children
- If swallowed, get medical help or contact a Poison Control Center right away.
- If condition covers a large area of the body, consult your doctor before using this product.
Principal Display Panel 200 Ml Bottle Carton
Kamedis®
Skin Restoring Solutions
BOTANIPLEX
TECHNOLOGY
SCALP
CONTROL
Dandruff Therapy
Shampoo
PYRITHIONE ZINC 1%
Clinically proven to remove up to
100% visible dandruff. Helps eliminate
flaking, scaling, itching, irritation and
redness associated with dandruff
DERMATOLOGIST
RECOMMENDED
Free from Parabens, Steroids,
Dyes, Sodium Lauryl Sulfate, Tar
200 mL
6.7 fl oz
Structured Label Content
Use
- helps eliminate flaking, itching and scaling associated with dandruff and seborrheic dermatitis
Section 42229-5 (42229-5)
Made in USA
(US and non US ingredients) Manufactured for
Kamedis Pennsylvania, 18109.
www.kamedis.com
Purpose
Anti-Dandruff
Warnings
For external use only
Directions
- adults and children 3 years of age and older:
- apply to wet hair
- gently massage into hair and scalp and leave for 2-3 minutes before thoroughly rinsing
- rinse and repeat if desired
- for best results use at least twice a week or as directed by your doctor
Drug Facts
Active Ingredient (Active ingredient)
Pyrithione Zinc 1.0%
Inactive Ingredients (Inactive ingredients)
ammonium laureth sulfate, water, ammonium lauryl sulfate, cocamidopropyl betaine, sodium lauroyl sarcosinate, soapberry (Sapindus mukurossi) fruit extract, salicylic acid, salt (sodium chloride), polyquaternium-10, sodium benzoate, triethanolamine, fragrance
When Using This Product (When using this product)
- avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with water
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens or does not improve after regular use of this product as directed
Keep Out of Reach of Children (Keep out of reach of children)
- If swallowed, get medical help or contact a Poison Control Center right away.
- If condition covers a large area of the body, consult your doctor before using this product.
Principal Display Panel 200 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 200 mL Bottle Carton)
Kamedis®
Skin Restoring Solutions
BOTANIPLEX
TECHNOLOGY
SCALP
CONTROL
Dandruff Therapy
Shampoo
PYRITHIONE ZINC 1%
Clinically proven to remove up to
100% visible dandruff. Helps eliminate
flaking, scaling, itching, irritation and
redness associated with dandruff
DERMATOLOGIST
RECOMMENDED
Free from Parabens, Steroids,
Dyes, Sodium Lauryl Sulfate, Tar
200 mL
6.7 fl oz
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:46.996489 · Updated: 2026-03-14T23:02:30.868416