These Highlights Do Not Include All The Information Needed To Use Xalkori®
2a51b0de-47d6-455e-a94c-d2c737b04ff7
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2 ) 9/2023
Indications and Usage
XALKORI is a kinase inhibitor indicated for the treatment of • adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are anaplastic lymphoma kinase (ALK) or ROS1-positive as detected by an FDA-approved test. ( 1.1 , 2.1 ) • pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic anaplastic large cell lymphoma (ALCL) that is ALK-positive. ( 1.2 , 2.3 ) o Limitations of Use: The safety and efficacy of XALKORI have not been established in older adults with relapsed or refractory, systemic ALK-positive ALCL. • adult and pediatric patients 1 year of age and older with unresectable, recurrent, or refractory inflammatory myofibroblastic tumor (IMT) that is ALK-positive. ( 1.3 , 2.3 )
Dosage and Administration
Warnings and Precautions
• Hepatotoxicity: Fatal hepatotoxicity has occurred. Monitor with periodic liver testing. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.2 , 2.6 , 5.1 ) • Interstitial Lung Disease (ILD)/Pneumonitis: Permanently discontinue in patients with ILD/pneumonitis. ( 2.6 , 5.2 ) • QT Interval Prolongation: Monitor electrocardiograms and electrolytes in patients who have a history of or predisposition for QTc prolongation, or who are taking medications that prolong QT. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.6 , 5.3 ) • Bradycardia: XALKORI can cause bradycardia. Monitor heart rate and blood pressure regularly. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.6 , 5.4 ) • Severe Visual Loss: XALKORI can cause visual changes including severe visual loss. Monitor and evaluate for ocular toxicity throughout treatment. Discontinue XALKORI in patients with severe visual loss. ( 2.2 , 2.6 , 5.5 ) • Gastrointestinal Toxicity in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT: XALKORI can cause severe nausea, vomiting, diarrhea, and stomatitis. Provide standard antiemetic and antidiarrheal agents. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.6 , 5.6 ) • Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. ( 5.7 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The recommended dosage modifications for adverse reactions for adult patients with NSCLC or IMT are provided in Table 4. Table 4. Recommended Dosage Reductions for Adverse Reactions for Adult Patients with NSCLC or IMT Using XALKORI Capsules or Pellets Dose Reduction Dose and Schedule First Dose Reduction 200 mg twice daily Second Dose Reduction 250 mg once daily Permanently discontinue XALKORI capsules or pellets if unable to tolerate 250 mg taken once daily. The recommended dosage modifications for adverse reactions for pediatric patients with ALCL or IMT and young adults with ALCL are based on body surface area and are provided in Table 5. Table 5. Recommended Dosage Reductions for Adverse Reactions for Pediatric Patients with ALCL or IMT and Young Adults with ALCL Using XALKORI Capsules or Pellets Body Surface Area (BSA) First Dose Reduction Second Dose Reduction Permanently discontinue in patients who are unable to tolerate XALKORI capsules or pellets after 2 dose reductions. Dosage Dosage Form and Strength to Achieve Recommended Dose Reduction Dosage Dosage Form and Strength to Achieve Recommended Dose Reduction 0.38 to 0.46 m 2 90 mg twice daily Pellets: 2 x 20 mg + 1 x 50 mg 70 mg twice daily Pellets: 1 x 20 mg + 1 x 50 mg 0.47 to 0.51 m 2 100 mg twice daily Pellets: 2 x 50 mg 80 mg twice daily Pellets: 4 x 20 mg 0.52 to 0.61 m 2 120 mg twice daily Pellets: 1 x 20 mg + 2 x 50 mg 90 mg twice daily Pellets: 2 x 20 mg + 1 x 50 mg 0.62 to 0.80 m 2 150 mg twice daily Pellets: 1 x 150 mg 120 mg twice daily Pellets: 1 x 20 mg + 2 x 50 mg 0.81 to 0.97 m 2 200 mg twice daily Pellets: 1 x 50 mg + 1 x 150 mg 150 mg twice daily Pellets: 1 x 150 mg 0.98 to 1.16 m 2 220 mg twice daily Pellets: 1 x 20 mg + 1 x 50 mg + 1 x 150 mg 170 mg twice daily Pellets: 1 x 20 mg + 1 x 150 mg 1.17 to 1.33 m 2 250 mg twice daily Pellets: 2 x 50 mg + 1 x 150 mg 200 mg twice daily Pellets: 1 x 50 mg + 1 x 150 mg 1.34 to 1.69 m 2 250 mg twice daily Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg 200 mg twice daily Pellets: 1 x 50 mg + 1 x 150 mg Or Capsule: 1 x 200 mg 1.7 m 2 or greater 400 mg twice daily Pellets: 2 x 50 mg + 2 x 150 mg Or Capsule: 2 x 200 mg 250 mg twice daily Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg Recommended Dosage Modifications for Hematologic Adverse Reactions for Adult Patients with NSCLC or IMT The recommended dosage modifications for hematologic adverse reactions for adult patients with NSCLC or IMT are provided in Table 6. Table 6. Adult Patients with NSCLC or IMT: XALKORI Dosage Modification – Hematologic Toxicities Except lymphopenia (unless associated with clinical events, e.g., opportunistic infections). Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. XALKORI Dosage Modification Grade 3 Withhold until recovery to Grade 2 or less, then resume at the same dosage. Grade 4 Withhold until recovery to Grade 2 or less, then resume at next lower dosage. Monitor complete blood counts including differential weekly for the first month of therapy and then at least monthly, with more frequent monitoring if Grade 3 or 4 abnormalities, fever, or infection occur. Recommended Dosage Modifications for Hematologic Adverse Reactions in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT The recommended dosage modifications for hematologic adverse reactions in pediatric and young adult patients with ALCL or pediatric patients with IMT are provided in Table 7. Table 7. Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT: XALKORI Dosage Modification for Hematologic Adverse Reactions Severity of Adverse Reaction XALKORI Dosage Modification Absolute Neutrophil Count (ANC) Less than 0.5 x 10 9 /L First occurrence: Withhold until recovery to ANC greater than 1.0 x 10 9 /L, then resume at the next lower dosage. Second occurrence: • Permanently discontinue for recurrence complicated by febrile neutropenia or infection. • For uncomplicated Grade 4 neutropenia, either permanently discontinue, or withhold until recovery to ANC greater than 1.0 x 10 9 /L, then resume at the next lower dosage. Permanently discontinue in patients who are unable to tolerate XALKORI after 2 dose reductions. Platelet Count 25 to 50 x 10 9 /L with concurrent bleeding Withhold until recovery to platelet count greater than 50 x 10 9 /L and bleeding resolves, then resume at the same dosage. Less than 25 x 10 9 /L Withhold until recovery to platelet count greater than 50 x 10 9 /L, then resume at the next lower dosage. Permanently discontinue for recurrence. Anemia Hemoglobin less than 8 g/dL Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the same dosage. Life-threatening anemia; urgent intervention indicated. Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the next lower dosage. Permanently discontinue for recurrence. Recommended Dosage Modifications for Non-Hematologic Adverse Reactions The recommended dosage modifications for non-hematologic adverse reactions are provided in Table 8. Table 8. All Patients: XALKORI Dosage Modification for Non-Hematologic Adverse Reactions Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. XALKORI Dosage Modification Hepatotoxicity [see Warnings and Precautions (5.1) ] Alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 1.5 times ULN Withhold until recovery to baseline or less than or equal to 3 times ULN, then resume at next lower dosage. ALT or AST greater than 3 times ULN with concurrent total bilirubin greater than 1.5 times ULN (in the absence of cholestasis or hemolysis) Permanently discontinue. Interstitial Lung Disease (Pneumonitis) [see Warnings and Precautions (5.2) ] Any grade drug-related interstitial lung disease/pneumonitis Permanently discontinue. QT Interval Prolongation [see Warnings and Precautions (5.3) ] QT corrected for heart rate (QTc) greater than 500 ms on at least 2 separate electrocardiograms (ECGs) Withhold until recovery to baseline or to a QTc less than 481 ms, then resume at next lower dosage. QTc greater than 500 ms or greater than or equal to 60 ms change from baseline with Torsade de pointes or polymorphic ventricular tachycardia or signs/symptoms of serious arrhythmia Permanently discontinue. Bradycardia [see Warnings and Precautions (5.4) ] Bradycardia Adult patients: Heart rate less than 60 beats per minute (bpm); Pediatric patients: Resting heart rate less than the 2.5 th percentile per age-specific norms. (symptomatic, may be severe and medically significant, medical intervention indicated) Withhold until recovery to a resting heart rate according to the patient’s age (based on the 2.5 th percentile per age-specific norms) as follows: • 1 to less than 2 years: 91 bpm or above • 2 to 3 years: 82 bpm or above • 4 to 5 years: 72 bpm or above • 6 to 8 years: 64 bpm or above • Older than 8 years: 60 bpm or above Evaluate concomitant medications known to cause bradycardia, as well as antihypertensive medications. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume at previous dose upon recovery to asymptomatic bradycardia or to the age-specific heart rate provided above. If no contributing concomitant medication is identified, or if contributing concomitant medications are not discontinued or dose adjusted, resume at reduced dose upon recovery to asymptomatic bradycardia or to the age-specific heart rate provided above. Bradycardia (life-threatening consequences, urgent intervention indicated) Permanently discontinue if no contributing concomitant medication is identified. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume at the second dose reduction level in Table 4 or 5 upon recovery to asymptomatic bradycardia or to the heart rate criteria listed for management of symptomatic or severe, medically significant bradycardia, with frequent monitoring. Permanently discontinue for recurrence. Ocular Toxicity, including Visual Loss [see Warnings and Precautions (5.5) ] Visual Symptoms, Grade 1 (mild symptoms) or Grade 2 (moderate symptoms affecting ability to perform age-appropriate activities of daily living) Monitor symptoms and report any symptoms to an eye specialist. Consider dose reduction for Grade 2 visual disorders. Visual Loss (Grade 3 or 4 Ocular Disorder, marked decrease in vision) Discontinue during evaluation of severe visual loss. Permanently discontinue XALKORI for Grade 3 or 4 ocular disorders or severe visual loss if no other cause found on evaluation. Gastrointestinal Toxicity Dosage modifications for gastrointestinal toxicity for pediatric patients with ALCL or IMT and young adults with ALCL only. [see Warnings and Precautions (5.6) ] Nausea (Grade 3: inadequate oral intake for more than 3 days, medical intervention required) Grade 3 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. Permanently discontinue in patients who are unable to tolerate XALKORI after 2 dose reductions. Vomiting (Grade 3: more than 6 episodes in 24 hours for more than 3 days, medical intervention required, i.e., tube feeding or hospitalization; Grade 4: life-threatening consequences, urgent intervention indicated) Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. Diarrhea (Grade 3: increase of 7 or more stools per day over baseline; incontinence; hospitalization indicated; Grade 4: life-threatening consequences, urgent intervention indicated) Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level.
Drug Interactions
• Strong CYP3A Inhibitors: Avoid concomitant use. ( 2.9 , 7.1 ) • Strong CYP3A Inducers: Avoid concomitant use. ( 7.1 ) • CYP3A Substrates: Avoid concomitant use with CYP3A substrates, where minimal concentration changes may lead to serious adverse reactions. ( 7.2 )
Storage and Handling
Capsules: • 200 mg capsules Hard gelatin capsule with pink opaque cap and white opaque body, printed with black ink "Pfizer" on the cap, "CRZ 200" on the body; available in: Bottles of 60 capsules: NDC 0069-8141-20 • 250 mg capsules Hard gelatin capsule with pink opaque cap and body, printed with black ink "Pfizer" on the cap, "CRZ 250" on the body; available in: Bottles of 60 capsules: NDC 0069-8140-20 Store at room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Oral Pellets: • 20 mg oral pellets Hard gelatin capsule, size 4, light blue opaque cap and white opaque body, printed with black ink “Pfizer” on the cap, “CRZ 20” on the body; available in: Bottles of 60 capsules: NDC 0069-0251-60 • 50 mg oral pellets Hard gelatin capsule, size 3, gray opaque cap and light gray opaque body, printed with black ink “Pfizer” on the cap, “CRZ 50” on the body; available in: Bottles of 60 capsules: NDC 0069-0507-60 • 150 mg oral pellets Hard gelatin capsule, size 0, light blue opaque cap and body, printed with black ink “Pfizer” on the cap, “CRZ 150” on the body; available in: Bottles of 60 capsules: NDC 0069-1500-60 Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
How Supplied
Capsules: • 200 mg capsules Hard gelatin capsule with pink opaque cap and white opaque body, printed with black ink "Pfizer" on the cap, "CRZ 200" on the body; available in: Bottles of 60 capsules: NDC 0069-8141-20 • 250 mg capsules Hard gelatin capsule with pink opaque cap and body, printed with black ink "Pfizer" on the cap, "CRZ 250" on the body; available in: Bottles of 60 capsules: NDC 0069-8140-20 Store at room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Oral Pellets: • 20 mg oral pellets Hard gelatin capsule, size 4, light blue opaque cap and white opaque body, printed with black ink “Pfizer” on the cap, “CRZ 20” on the body; available in: Bottles of 60 capsules: NDC 0069-0251-60 • 50 mg oral pellets Hard gelatin capsule, size 3, gray opaque cap and light gray opaque body, printed with black ink “Pfizer” on the cap, “CRZ 50” on the body; available in: Bottles of 60 capsules: NDC 0069-0507-60 • 150 mg oral pellets Hard gelatin capsule, size 0, light blue opaque cap and body, printed with black ink “Pfizer” on the cap, “CRZ 150” on the body; available in: Bottles of 60 capsules: NDC 0069-1500-60 Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Medication Information
Warnings and Precautions
• Hepatotoxicity: Fatal hepatotoxicity has occurred. Monitor with periodic liver testing. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.2 , 2.6 , 5.1 ) • Interstitial Lung Disease (ILD)/Pneumonitis: Permanently discontinue in patients with ILD/pneumonitis. ( 2.6 , 5.2 ) • QT Interval Prolongation: Monitor electrocardiograms and electrolytes in patients who have a history of or predisposition for QTc prolongation, or who are taking medications that prolong QT. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.6 , 5.3 ) • Bradycardia: XALKORI can cause bradycardia. Monitor heart rate and blood pressure regularly. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.6 , 5.4 ) • Severe Visual Loss: XALKORI can cause visual changes including severe visual loss. Monitor and evaluate for ocular toxicity throughout treatment. Discontinue XALKORI in patients with severe visual loss. ( 2.2 , 2.6 , 5.5 ) • Gastrointestinal Toxicity in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT: XALKORI can cause severe nausea, vomiting, diarrhea, and stomatitis. Provide standard antiemetic and antidiarrheal agents. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. ( 2.6 , 5.6 ) • Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. ( 5.7 , 8.1 , 8.3 )
Indications and Usage
XALKORI is a kinase inhibitor indicated for the treatment of • adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are anaplastic lymphoma kinase (ALK) or ROS1-positive as detected by an FDA-approved test. ( 1.1 , 2.1 ) • pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic anaplastic large cell lymphoma (ALCL) that is ALK-positive. ( 1.2 , 2.3 ) o Limitations of Use: The safety and efficacy of XALKORI have not been established in older adults with relapsed or refractory, systemic ALK-positive ALCL. • adult and pediatric patients 1 year of age and older with unresectable, recurrent, or refractory inflammatory myofibroblastic tumor (IMT) that is ALK-positive. ( 1.3 , 2.3 )
Dosage and Administration
Contraindications
None.
Adverse Reactions
The recommended dosage modifications for adverse reactions for adult patients with NSCLC or IMT are provided in Table 4. Table 4. Recommended Dosage Reductions for Adverse Reactions for Adult Patients with NSCLC or IMT Using XALKORI Capsules or Pellets Dose Reduction Dose and Schedule First Dose Reduction 200 mg twice daily Second Dose Reduction 250 mg once daily Permanently discontinue XALKORI capsules or pellets if unable to tolerate 250 mg taken once daily. The recommended dosage modifications for adverse reactions for pediatric patients with ALCL or IMT and young adults with ALCL are based on body surface area and are provided in Table 5. Table 5. Recommended Dosage Reductions for Adverse Reactions for Pediatric Patients with ALCL or IMT and Young Adults with ALCL Using XALKORI Capsules or Pellets Body Surface Area (BSA) First Dose Reduction Second Dose Reduction Permanently discontinue in patients who are unable to tolerate XALKORI capsules or pellets after 2 dose reductions. Dosage Dosage Form and Strength to Achieve Recommended Dose Reduction Dosage Dosage Form and Strength to Achieve Recommended Dose Reduction 0.38 to 0.46 m 2 90 mg twice daily Pellets: 2 x 20 mg + 1 x 50 mg 70 mg twice daily Pellets: 1 x 20 mg + 1 x 50 mg 0.47 to 0.51 m 2 100 mg twice daily Pellets: 2 x 50 mg 80 mg twice daily Pellets: 4 x 20 mg 0.52 to 0.61 m 2 120 mg twice daily Pellets: 1 x 20 mg + 2 x 50 mg 90 mg twice daily Pellets: 2 x 20 mg + 1 x 50 mg 0.62 to 0.80 m 2 150 mg twice daily Pellets: 1 x 150 mg 120 mg twice daily Pellets: 1 x 20 mg + 2 x 50 mg 0.81 to 0.97 m 2 200 mg twice daily Pellets: 1 x 50 mg + 1 x 150 mg 150 mg twice daily Pellets: 1 x 150 mg 0.98 to 1.16 m 2 220 mg twice daily Pellets: 1 x 20 mg + 1 x 50 mg + 1 x 150 mg 170 mg twice daily Pellets: 1 x 20 mg + 1 x 150 mg 1.17 to 1.33 m 2 250 mg twice daily Pellets: 2 x 50 mg + 1 x 150 mg 200 mg twice daily Pellets: 1 x 50 mg + 1 x 150 mg 1.34 to 1.69 m 2 250 mg twice daily Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg 200 mg twice daily Pellets: 1 x 50 mg + 1 x 150 mg Or Capsule: 1 x 200 mg 1.7 m 2 or greater 400 mg twice daily Pellets: 2 x 50 mg + 2 x 150 mg Or Capsule: 2 x 200 mg 250 mg twice daily Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg Recommended Dosage Modifications for Hematologic Adverse Reactions for Adult Patients with NSCLC or IMT The recommended dosage modifications for hematologic adverse reactions for adult patients with NSCLC or IMT are provided in Table 6. Table 6. Adult Patients with NSCLC or IMT: XALKORI Dosage Modification – Hematologic Toxicities Except lymphopenia (unless associated with clinical events, e.g., opportunistic infections). Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. XALKORI Dosage Modification Grade 3 Withhold until recovery to Grade 2 or less, then resume at the same dosage. Grade 4 Withhold until recovery to Grade 2 or less, then resume at next lower dosage. Monitor complete blood counts including differential weekly for the first month of therapy and then at least monthly, with more frequent monitoring if Grade 3 or 4 abnormalities, fever, or infection occur. Recommended Dosage Modifications for Hematologic Adverse Reactions in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT The recommended dosage modifications for hematologic adverse reactions in pediatric and young adult patients with ALCL or pediatric patients with IMT are provided in Table 7. Table 7. Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT: XALKORI Dosage Modification for Hematologic Adverse Reactions Severity of Adverse Reaction XALKORI Dosage Modification Absolute Neutrophil Count (ANC) Less than 0.5 x 10 9 /L First occurrence: Withhold until recovery to ANC greater than 1.0 x 10 9 /L, then resume at the next lower dosage. Second occurrence: • Permanently discontinue for recurrence complicated by febrile neutropenia or infection. • For uncomplicated Grade 4 neutropenia, either permanently discontinue, or withhold until recovery to ANC greater than 1.0 x 10 9 /L, then resume at the next lower dosage. Permanently discontinue in patients who are unable to tolerate XALKORI after 2 dose reductions. Platelet Count 25 to 50 x 10 9 /L with concurrent bleeding Withhold until recovery to platelet count greater than 50 x 10 9 /L and bleeding resolves, then resume at the same dosage. Less than 25 x 10 9 /L Withhold until recovery to platelet count greater than 50 x 10 9 /L, then resume at the next lower dosage. Permanently discontinue for recurrence. Anemia Hemoglobin less than 8 g/dL Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the same dosage. Life-threatening anemia; urgent intervention indicated. Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the next lower dosage. Permanently discontinue for recurrence. Recommended Dosage Modifications for Non-Hematologic Adverse Reactions The recommended dosage modifications for non-hematologic adverse reactions are provided in Table 8. Table 8. All Patients: XALKORI Dosage Modification for Non-Hematologic Adverse Reactions Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. XALKORI Dosage Modification Hepatotoxicity [see Warnings and Precautions (5.1) ] Alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 1.5 times ULN Withhold until recovery to baseline or less than or equal to 3 times ULN, then resume at next lower dosage. ALT or AST greater than 3 times ULN with concurrent total bilirubin greater than 1.5 times ULN (in the absence of cholestasis or hemolysis) Permanently discontinue. Interstitial Lung Disease (Pneumonitis) [see Warnings and Precautions (5.2) ] Any grade drug-related interstitial lung disease/pneumonitis Permanently discontinue. QT Interval Prolongation [see Warnings and Precautions (5.3) ] QT corrected for heart rate (QTc) greater than 500 ms on at least 2 separate electrocardiograms (ECGs) Withhold until recovery to baseline or to a QTc less than 481 ms, then resume at next lower dosage. QTc greater than 500 ms or greater than or equal to 60 ms change from baseline with Torsade de pointes or polymorphic ventricular tachycardia or signs/symptoms of serious arrhythmia Permanently discontinue. Bradycardia [see Warnings and Precautions (5.4) ] Bradycardia Adult patients: Heart rate less than 60 beats per minute (bpm); Pediatric patients: Resting heart rate less than the 2.5 th percentile per age-specific norms. (symptomatic, may be severe and medically significant, medical intervention indicated) Withhold until recovery to a resting heart rate according to the patient’s age (based on the 2.5 th percentile per age-specific norms) as follows: • 1 to less than 2 years: 91 bpm or above • 2 to 3 years: 82 bpm or above • 4 to 5 years: 72 bpm or above • 6 to 8 years: 64 bpm or above • Older than 8 years: 60 bpm or above Evaluate concomitant medications known to cause bradycardia, as well as antihypertensive medications. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume at previous dose upon recovery to asymptomatic bradycardia or to the age-specific heart rate provided above. If no contributing concomitant medication is identified, or if contributing concomitant medications are not discontinued or dose adjusted, resume at reduced dose upon recovery to asymptomatic bradycardia or to the age-specific heart rate provided above. Bradycardia (life-threatening consequences, urgent intervention indicated) Permanently discontinue if no contributing concomitant medication is identified. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume at the second dose reduction level in Table 4 or 5 upon recovery to asymptomatic bradycardia or to the heart rate criteria listed for management of symptomatic or severe, medically significant bradycardia, with frequent monitoring. Permanently discontinue for recurrence. Ocular Toxicity, including Visual Loss [see Warnings and Precautions (5.5) ] Visual Symptoms, Grade 1 (mild symptoms) or Grade 2 (moderate symptoms affecting ability to perform age-appropriate activities of daily living) Monitor symptoms and report any symptoms to an eye specialist. Consider dose reduction for Grade 2 visual disorders. Visual Loss (Grade 3 or 4 Ocular Disorder, marked decrease in vision) Discontinue during evaluation of severe visual loss. Permanently discontinue XALKORI for Grade 3 or 4 ocular disorders or severe visual loss if no other cause found on evaluation. Gastrointestinal Toxicity Dosage modifications for gastrointestinal toxicity for pediatric patients with ALCL or IMT and young adults with ALCL only. [see Warnings and Precautions (5.6) ] Nausea (Grade 3: inadequate oral intake for more than 3 days, medical intervention required) Grade 3 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. Permanently discontinue in patients who are unable to tolerate XALKORI after 2 dose reductions. Vomiting (Grade 3: more than 6 episodes in 24 hours for more than 3 days, medical intervention required, i.e., tube feeding or hospitalization; Grade 4: life-threatening consequences, urgent intervention indicated) Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. Diarrhea (Grade 3: increase of 7 or more stools per day over baseline; incontinence; hospitalization indicated; Grade 4: life-threatening consequences, urgent intervention indicated) Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level.
Drug Interactions
• Strong CYP3A Inhibitors: Avoid concomitant use. ( 2.9 , 7.1 ) • Strong CYP3A Inducers: Avoid concomitant use. ( 7.1 ) • CYP3A Substrates: Avoid concomitant use with CYP3A substrates, where minimal concentration changes may lead to serious adverse reactions. ( 7.2 )
Storage and Handling
Capsules: • 200 mg capsules Hard gelatin capsule with pink opaque cap and white opaque body, printed with black ink "Pfizer" on the cap, "CRZ 200" on the body; available in: Bottles of 60 capsules: NDC 0069-8141-20 • 250 mg capsules Hard gelatin capsule with pink opaque cap and body, printed with black ink "Pfizer" on the cap, "CRZ 250" on the body; available in: Bottles of 60 capsules: NDC 0069-8140-20 Store at room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Oral Pellets: • 20 mg oral pellets Hard gelatin capsule, size 4, light blue opaque cap and white opaque body, printed with black ink “Pfizer” on the cap, “CRZ 20” on the body; available in: Bottles of 60 capsules: NDC 0069-0251-60 • 50 mg oral pellets Hard gelatin capsule, size 3, gray opaque cap and light gray opaque body, printed with black ink “Pfizer” on the cap, “CRZ 50” on the body; available in: Bottles of 60 capsules: NDC 0069-0507-60 • 150 mg oral pellets Hard gelatin capsule, size 0, light blue opaque cap and body, printed with black ink “Pfizer” on the cap, “CRZ 150” on the body; available in: Bottles of 60 capsules: NDC 0069-1500-60 Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
How Supplied
Capsules: • 200 mg capsules Hard gelatin capsule with pink opaque cap and white opaque body, printed with black ink "Pfizer" on the cap, "CRZ 200" on the body; available in: Bottles of 60 capsules: NDC 0069-8141-20 • 250 mg capsules Hard gelatin capsule with pink opaque cap and body, printed with black ink "Pfizer" on the cap, "CRZ 250" on the body; available in: Bottles of 60 capsules: NDC 0069-8140-20 Store at room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Oral Pellets: • 20 mg oral pellets Hard gelatin capsule, size 4, light blue opaque cap and white opaque body, printed with black ink “Pfizer” on the cap, “CRZ 20” on the body; available in: Bottles of 60 capsules: NDC 0069-0251-60 • 50 mg oral pellets Hard gelatin capsule, size 3, gray opaque cap and light gray opaque body, printed with black ink “Pfizer” on the cap, “CRZ 50” on the body; available in: Bottles of 60 capsules: NDC 0069-0507-60 • 150 mg oral pellets Hard gelatin capsule, size 0, light blue opaque cap and body, printed with black ink “Pfizer” on the cap, “CRZ 150” on the body; available in: Bottles of 60 capsules: NDC 0069-1500-60 Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Description
Dosage and Administration ( 2 ) 9/2023
Section 42229-5
Limitations of Use: The safety and efficacy of XALKORI have not been established in older adults with relapsed or refractory, systemic ALK-positive ALCL.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: Sep 2023 | |||
|
MEDICATION GUIDE |
|||
|
XALKORI® (zal-KOR-ee)
|
XALKORI® (zal-KOR-ee)
|
||
|
What is the most important information I should know about XALKORI?
|
|||
|
|
||
|
|||
|
|
||
|
In addition, for children or young adults taking XALKORI to treat anaplastic large cell lymphoma (ALCL) or children taking XALKORI to treat inflammatory myofibroblastic tumor (IMT): |
|||
See "What are possible side effects of XALKORI?" for more information about side effects. |
|||
|
What is XALKORI?
It is not known if XALKORI is safe and effective in children younger than 1 year of age with ALCL or IMT, or in any children with NSCLC. |
|||
|
Before taking XALKORI, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. XALKORI can affect the way other medicines work, and other medicines can affect how XALKORI works. |
|||
|
How should I take XALKORI?
XALKORI comes as capsules and oral pellets.
|
|||
|
What should I avoid while taking XALKORI?
|
|||
|
What are the possible side effects of XALKORI?
The most common side effects of XALKORI in adults with NSCLC include: |
|||
|
|
||
|
The most common side effects of XALKORI in people with ALCL include: |
|||
|
|
||
|
The most common side effects of XALKORI in adults with IMT include: |
|||
|
|||
|
The most common side effects of XALKORI in children with IMT include: |
|||
|
|
||
|
XALKORI may cause fertility problems in females and males, which may affect the ability to have children. Talk to your healthcare provider if you have concerns about fertility. |
|||
|
How should I store XALKORI?
Keep XALKORI and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of XALKORI.
|
|||
|
What are the ingredients in XALKORI?
LAB-0441-12.0 |
Section 43683-2
|
Dosage and Administration (2) |
9/2023 |
11 Description
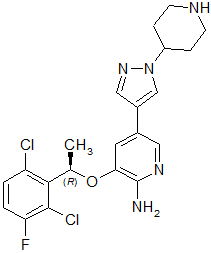
Crizotinib is a kinase inhibitor. The molecular formula for crizotinib is C21H22Cl2FN5O and the molecular weight is 450.34 daltons. Crizotinib is described chemically as (R)-3-[1-(2,6-Dichloro-3-fluorophenyl)ethoxy]-5-[1-(piperidin-4-yl)-1H-pyrazol-4-yl]pyridin-2-amine.
The chemical structure of crizotinib is shown below:
Crizotinib is a white to pale-yellow powder with a pKa of 9.4 (piperidinium cation) and 5.6 (pyridinium cation). The solubility of crizotinib in aqueous media decreases over the range pH 1.6 to pH 8.2 from greater than 10 mg/mL to less than 0.1 mg/mL. The log of the distribution coefficient (octanol/water) at pH 7.4 is 1.65.
Capsules:
XALKORI (crizotinib) capsules for oral administration are supplied as printed hard-shell capsules containing 250 mg or 200 mg of crizotinib together with colloidal silicon dioxide, microcrystalline cellulose, anhydrous dibasic calcium phosphate, sodium starch glycolate, magnesium stearate, and hard gelatin capsule shells as inactive ingredients.
The pink opaque capsule shell components contain gelatin, titanium dioxide, and red iron oxide. The white opaque capsule shell components contain gelatin and titanium dioxide. The printing ink contains shellac, propylene glycol, strong ammonia solution, potassium hydroxide, and black iron oxide.
Oral Pellets:
XALKORI (crizotinib) oral pellets for oral administration are supplied as 20 mg, 50 mg, 150 mg of crizotinib contained in hard gelatin capsules. The inactive ingredients in the uncoated pellets are poloxamer and stearyl alcohol. The film-coating consists of hypromellose, glyceryl monostearate, medium chain triglycerides, polyethylene glycol/macrogol, sucrose, and talc.
5.4 Bradycardia
Symptomatic bradycardia can occur in patients receiving XALKORI. Across clinical trials in patients with NSCLC, bradycardia occurred in 13% of 1719 patients treated with XALKORI. Grade 3 syncope occurred in 2.4% of XALKORI-treated patients and in 0.6% of the chemotherapy-treated patients [see Adverse Reactions (6.1)].
In Study ADVL0912, among 121 patients ages 1 to ≤21 years treated with XALKORI, bradycardia was reported in 14%, including Grade 3 bradycardia in 0.8% of patients. Of the 26 patients with ALCL treated with XALKORI, bradycardia (all Grade 1) was reported in 19%. Of the 14 pediatric patients with IMT treated with XALKORI, bradycardia was reported in 14% of patients, including Grade 3 bradycardia in 7% of patients.
Avoid using XALKORI in combination with other medications known to cause bradycardia (e.g., beta-blockers, nondihydropyridine-calcium channel blockers, clonidine, and digoxin) to the extent possible. Monitor heart rate and blood pressure regularly. If bradycardia occurs, re-evaluate for the use of concomitant medications known to cause bradycardia. Withhold, reduce dose, or permanently discontinue XALKORI for bradycardia as recommended [see Dosage and Administration (2.6)].
8.4 Pediatric Use
The safety and effectiveness of XALKORI have been established in pediatric patients 1 year of age and older with relapsed or refractory, systemic ALK-positive ALCL or with unresectable, recurrent, or refractory ALK-positive IMT [see Adverse Reactions (6.1), Clinical Studies (14.2, 14.3)]. The safety and effectiveness have not been established in pediatric patients younger than 1 year of age with ALCL or with IMT, or in any pediatric patients with NSCLC.
In a study that evaluated XALKORI in combination with chemotherapy in pediatric patients with newly diagnosed ALCL (Study ANHL12P1; NCT01979536), 13 of 66 (20%) patients had a Grade 2 or higher thromboembolic event, including pulmonary embolism in 6%. The safety and effectiveness of XALKORI in combination with chemotherapy have not been established in patients with newly diagnosed ALCL.
8.5 Geriatric Use
Of the total number of patients with ALK-positive metastatic NSCLC in clinical studies of XALKORI (n=1669), 16% were 65 years or older and 3.8% were 75 years or older. No overall differences in safety or effectiveness were observed between these patients and younger patients.
Clinical studies of XALKORI in patients with ROS1-positive metastatic NSCLC did not include sufficient numbers of patients age 65 years and older to determine whether they respond differently from younger patients.
2.4 Administration
-
•Administer XALKORI capsules or pellets orally, twice daily, with or without food.
-
•If a dose of XALKORI capsules or pellets is missed, make up that dose unless the next dose is due within 6 hours.
-
•If vomiting occurs after taking a dose of XALKORI capsules or pellets, do not take an additional dose. Take the next dose at the regular scheduled time.
XALKORI Capsules
-
•Swallow XALKORI capsules whole, with or without food twice daily.
-
•Do not chew, crush or split XALKORI capsules.
XALKORI Pellets
-
•XALKORI pellets are supplied encapsulated in shells.
-
•Do not chew or crush XALKORI pellets.
-
•Do not swallow XALKORI pellets encapsulated in the shell.
-
•XALKORI pellets can be administered by 2 options:
-
1.Open shell(s) containing XALKORI pellets and empty the contents directly into the patient’s mouth.
-
2.Open shell(s) containing XALKORI pellets and empty the contents into a consumer-supplied oral dosing aid (e.g., spoon, medicine cup). Administer XALKORI pellets via the dosing aid directly into the patient’s mouth.
-
•Immediately after administration, give a sufficient amount of water to ensure that all medication is swallowed.
5.1 Hepatotoxicity
Drug-induced hepatotoxicity with fatal outcome occurred in 0.1% of the 1719 patients treated with XALKORI for NSCLC across clinical trials [see Adverse Reactions (6.1)]. Concurrent elevations in ALT or AST ≥3 times the ULN and total bilirubin ≥2 times the ULN, with normal alkaline phosphatase, occurred in <1% of patients treated with XALKORI. Increased ALT or AST >5 times the ULN occurred in 11% and 6% of patients, respectively. One percent (1.0%) of patients required permanent discontinuation due to elevated transaminases. Increased transaminases generally occurred within the first 2 months of treatment.
In Study ADVL0912, of 121 patients ages 1 to ≤21 years treated with XALKORI for relapsed or refractory tumors including ALCL and IMT, 71% and 79% had increases of AST and ALT, respectively, with increased ALT or AST >5 times the ULN in 6% each. Of the 26 patients with ALCL treated with XALKORI, 65% and 81% had increases of AST and ALT, respectively, with increases >5 times the ULN in 4% each. Of the 14 pediatric patients with IMT treated with XALKORI, 71% had increases of AST and 71% had increases of ALT.
In Study A8081013, of the 7 adult patients with IMT treated with XALKORI, 57% and 43% had increases of AST and ALT, respectively.
Monitor liver function tests, including ALT, AST, and total bilirubin, every 2 weeks during the first 2 months of treatment, then once a month, and as clinically indicated, with more frequent repeat testing for increased liver transaminases, alkaline phosphatase, or total bilirubin in patients who develop increased transaminases. Withhold, reduce dose, or permanently discontinue XALKORI for hepatotoxicity as recommended [see Dosage and Administration (2.6)].
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Hepatotoxicity [see Warnings and Precautions (5.1)]
-
•Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.2)]
-
•QT Interval Prolongation [see Warnings and Precautions (5.3)]
-
•Bradycardia [see Warnings and Precautions (5.4)]
-
•Severe Visual Loss [see Warnings and Precautions (5.5)]
-
•Gastrointestinal Toxicity in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT [see Warnings and Precautions (5.6)]
7 Drug Interactions
8.7 Renal Impairment
Increased exposure to crizotinib occurred in patients with pre-existing severe renal impairment (CLcr less than 30 mL/min calculated using the modified Cockcroft-Gault equation for adult patients and the Schwartz equation for pediatric patients) not requiring dialysis, therefore reduce dosage of XALKORI in these patients [see Dosage and Administration (2.8), Clinical Pharmacology (12.3)]. No dose adjustment is recommended in patients with mild to moderate renal impairment (CLcr 30 to 89 mL/min).
Instructions for Use
INSTRUCTIONS FOR USE
XALKORI® [zal-KOR-ee]
(crizotinib)
oral pellets
This Instructions for Use contains information on how to give or take XALKORI oral pellets. Read this Instructions for Use each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider or pharmacist about your or your child’s medical condition or treatment.
Important information you need to know before giving or taking XALKORI oral pellets:
-
•XALKORI oral pellets come in a capsule “shell” that must be opened before giving or taking a dose. Do not swallow the shell containing the oral pellets. Do not chew or crush the oral pellets.
-
•XALKORI oral pellets come in 3 dosage strengths: 20 mg, 50 mg, and 150 mg. Your healthcare provider may combine different strengths for your prescribed dose. No more than 4 XALKORI oral pellet shells are to be used for a single dose.
-
•Your healthcare provider will decide the right dose of XALKORI oral pellets for you or your child. Follow your healthcare provider’s instructions for the dose of XALKORI oral pellets to give your child or for you to take.
-
•Empty XALKORI oral pellets from the shells as described in Steps 1 to 4 below.
-
•Check the expiration date on the bottle containing XALKORI oral pellets. Do not use XALKORI oral pellets after the expiration date on the bottle has passed.
-
•Ask your healthcare provider or pharmacist if you are not sure how to prepare and give or take the prescribed dose of XALKORI oral pellets.
Supplies needed to give or take XALKORI oral pellets:
-
•XALKORI oral pellet(s), as prescribed by your healthcare provider.
-
•Spoon or medicine cup (optional). See Step 4 “Giving or taking XALKORI oral pellets”.
Preparing XALKORI oral pellets (Steps 1 to 3):
|
Step 1 |
Remove the number of XALKORI oral pellet shell(s) needed for the prescribed dose from each bottle(s). |
|
Step 2 |
|
|
Figure A |
|
|
Step 3 |
Carefully hold and twist the top and bottom parts of the shell in opposite directions and pull apart to open the shell (see Figure B). Figure B |
Giving or taking XALKORI oral pellets (Step 4): There are 2 options for giving or taking the oral pellets.
|
Step 4 |
Option 1 (Pour directly into the mouth) |
|
|
Figure C |
||
|
Option 2 (Pour from a spoon or medicine cup) |
|
|
|
Figure D |
||
|
After Step 4 is completed, other drinks or foods can be given or taken, except grapefruit juice, grapefruit, or supplements containing grapefruit extract. |
Storing XALKORI oral pellets:
-
•Store XALKORI oral pellets at room temperature between 68°F to 77°F (20°C to 25°C).
-
•Keep XALKORI oral pellets and all medicines out of the reach of children.
Disposing of empty XALKORI oral pellet shells:
-
•Dispose of (throw away) the empty XALKORI oral pellet shell(s) in the household trash.
-
•Ask your pharmacist how to throw away medicines you no longer use or are expired.
For more information, go to www.Pfizer.com or call 1-800-438-1985.
LAB-1526-1.0
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Issued: 09/2023
12.3 Pharmacokinetics
Following XALKORI 250 mg capsules twice daily, steady-state was reached within 15 days with a median accumulation ratio of 4.8. Steady-state minimum concentration (Cmin.ss) and AUC increased in a greater than dose-proportional manner over the dose range of 200 mg to 300 mg twice daily (0.8 to 1.2 times the approved recommended dosage).
2.1 Patient Selection
Select patients for the treatment of metastatic NSCLC with XALKORI based on the presence of ALK or ROS1 positivity in tumor specimens [see Clinical Studies (14.1, 14.2, 14.3)].
Information on FDA-approved tests for the detection of ALK and ROS1 rearrangements in NSCLC is available at http://www.fda.gov/companiondiagnostics.
2.3 Recommended Dosage
The recommended dosage of XALKORI is provided in Table 1.
|
Indication |
Recommended Dosage of XALKORI |
|
ALK- or ROS1-Positive Metastatic NSCLC |
Adults: |
|
Relapsed or Refractory, Systemic ALK-Positive ALCL |
Pediatric Patients and Young Adults: See Table 2 for Recommended Dosage based on body surface area for pediatric patients and young adults with ALCL for the capsules and oral pellets.
|
|
Unresectable, Recurrent, or Refractory ALK-Positive IMT |
Adults: |
|
Pediatric Patients: See Table 3 for Recommended Dosage based on body surface area for pediatric patients with IMT for the capsules and oral pellets.
|
Recommended Dosage for Adult Patients with ALK- or ROS1-Positive Metastatic NSCLC
-
•The recommended dosage for adult patients with ALK- or ROS1-positive metastatic NSCLC is XALKORI capsules 250 mg orally, twice daily, with or without food until disease progression or unacceptable toxicity occurs.
-
•For adults who cannot swallow capsules, the recommended dosage of XALKORI pellets is 250 mg (2 x 50 mg + 1 x 150 mg) orally, twice daily, with or without food until disease progression or unacceptable toxicity occurs.
Recommended Dosage for Pediatric and Young Adult Patients with ALK-Positive ALCL
-
•The recommended dosage for pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic ALK-positive ALCL is based on body surface area (BSA) and is provided in Table 2.
-
•Administer XALKORI capsules or pellets orally, twice daily, with or without food until disease progression or unacceptable toxicity occurs.
Table 2 provides the dosage based on body surface area (BSA) for XALKORI capsules or pellets.
|
Body Surface Area (BSA) |
Recommended XALKORI Dosage to Achieve 280 mg/m2 Twice Daily |
Dose Strength Combinations of XALKORI Pellets to Administer |
Dose Strength Combinations of XALKORI Capsules to Administer |
|
0.38 to 0.46 m2 |
120 mg twice daily |
1 x 20 mg + 2 x 50 mg |
--- |
|
0.47 to 0.51 m2 |
140 mg twice daily |
2 x 20 mg + 2 x 50 mg |
--- |
|
0.52 to 0.61 m2 |
150 mg twice daily |
1 x 150 mg |
--- |
|
0.62 to 0.80 m2 |
200 mg twice daily |
1 x 50 mg + 1 x 150 mg |
--- |
|
0.81 to 0.97 m2 |
250 mg twice daily |
2 x 50 mg + 1 x 150 mg |
--- |
|
0.98 to 1.16 m2 |
300 mg twice daily |
2 x 150 mg |
--- |
|
1.17 to 1.33 m2 |
350 mg twice daily |
1 x 50 mg + 2 x 150 mg |
--- |
|
1.34 to 1.51 m2 |
400 mg twice daily |
2 x 50 mg + 2 x 150 mg |
2 x 200 mg |
|
1.52 to 1.69 m2 |
450 mg twice daily |
3 x 150 mg |
1 x 200 mg + 1 x 250 mg |
|
1.7 m2 or greater |
500 mg twice daily |
1 x 50 mg + 3 x 150 mg |
2 x 250 mg |
Recommended Dosage for Pediatric and Adult Patients with ALK-Positive IMT
-
•The recommended dosage for adult patients with unresectable, recurrent, or refractory ALK-positive IMT is provided in Table 1.
-
•The recommended dosage for pediatric patients 1 year of age and older with unresectable, recurrent, or refractory ALK-positive IMT is based on BSA and is provided in Table 3.
-
•Administer XALKORI capsules or pellets orally twice daily, with or without food, until disease progression or unacceptable toxicity occurs.
Table 3 provides the dosage based on BSA for XALKORI capsules or pellets.
|
Body Surface Area (BSA) |
Recommended XALKORI Dosage to Achieve 280 mg/m2 Twice Daily |
Dose Strength Combinations of XALKORI Pellets to Administer
No more than 4 oral pellet shells are to be used for a single dose.
|
Dose Strength Combinations of XALKORI Capsules to Administer |
|
0.38 to 0.46 m2 |
120 mg twice daily |
1 x 20 mg + 2 x 50 mg |
--- |
|
0.47 to 0.51 m2 |
140 mg twice daily |
2 x 20 mg + 2 x 50 mg |
--- |
|
0.52 to 0.61 m2 |
150 mg twice daily |
1 x 150 mg |
--- |
|
0.62 to 0.80 m2 |
200 mg twice daily |
1 x 50 mg + 1 x 150 mg |
--- |
|
0.81 to 0.97 m2 |
250 mg twice daily |
2 x 50 mg + 1 x 150 mg |
--- |
|
0.98 to 1.16 m2 |
300 mg twice daily |
2 x 150 mg |
--- |
|
1.17 to 1.33 m2 |
350 mg twice daily |
1 x 50 mg + 2 x 150 mg |
--- |
|
1.34 to 1.51 m2 |
400 mg twice daily |
2 x 50 mg + 2 x 150 mg |
2 x 200 mg |
|
1.52 to 1.69 m2 |
450 mg twice daily |
3 x 150 mg |
1 x 200 mg + 1 x 250 mg |
|
1.7 m2 or greater |
500 mg twice daily |
1 x 50 mg + 3 x 150 mg |
2 x 250 mg |
5.5 Severe Visual Loss
Across all clinical trials in patients with NSCLC, the incidence of Grade 4 visual field defect with visual loss was 0.2% of 1719 patients [see Adverse Reactions (6.1)]. Optic atrophy and optic nerve disorder have been reported as potential causes of visual loss.
In Study ADVL0912, visual disorders occurred in 46% of 121 patients with XALKORI, including 65% of 26 patients with ALCL and 50% of 14 patients with IMT. Of the 56 patients who experienced visual disorders, one pediatric patient with IMT experienced Grade 3 myopic optic nerve disorder. The most common visual symptoms were blurred vision and visual impairment.
Assessment of visual symptoms for all patients is recommended monthly during treatment. Report new visual symptoms to an eye specialist.
For pediatric and young adult patients with ALCL or pediatric patients with IMT, obtain baseline and follow-up ophthalmologic examinations including retinal examination within 1 month of starting XALKORI and every 3 months thereafter. The ophthalmological evaluation should consist of best corrected visual acuity, retinal photographs, visual fields, optical coherence tomography (OCT), and other evaluations as appropriate [see Dosage and Administration (2.6), Adverse Reactions (6.1)].
Permanently discontinue XALKORI for Grade 3 or 4 ocular disorders or severe visual loss (best corrected vision less than 20/200 in one or both eyes) unless another cause is identified [see Dosage and Administration (2.6)]. There is insufficient information to characterize the risks of resumption of XALKORI in patients who develop visual symptoms or visual loss. A decision to resume XALKORI should consider the potential benefits versus risks to the patient.
8.6 Hepatic Impairment
Crizotinib concentrations increased in patients with pre-existing moderate (any AST and total bilirubin greater than 1.5 times ULN and less than or equal to 3 times ULN) or severe (any AST and total bilirubin greater than 3 times ULN) hepatic impairment [see Clinical Pharmacology (12.3)]. Reduce XALKORI dosage in patients with moderate or severe hepatic impairment [see Dosage and Administration (2.7)]. No dose adjustment is recommended in patients with pre-existing mild hepatic impairment (AST > ULN and total bilirubin less than or equal to 1 times ULN or any AST and total bilirubin greater than 1 times ULN but less than or equal to1.5 times ULN).
1 Indications and Usage
XALKORI is a kinase inhibitor indicated for the treatment of
-
•adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are anaplastic lymphoma kinase (ALK) or ROS1-positive as detected by an FDA-approved test. (1.1, 2.1)
-
•pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic anaplastic large cell lymphoma (ALCL) that is ALK-positive. (1.2, 2.3)
-
oLimitations of Use: The safety and efficacy of XALKORI have not been established in older adults with relapsed or refractory, systemic ALK-positive ALCL.
-
-
•adult and pediatric patients 1 year of age and older with unresectable, recurrent, or refractory inflammatory myofibroblastic tumor (IMT) that is ALK-positive. (1.3, 2.3)
12.1 Mechanism of Action
Crizotinib is an inhibitor of receptor tyrosine kinases including ALK, Hepatocyte Growth Factor Receptor (HGFR, c-Met), ROS1 (c-ros), and Recepteur d'Origine Nantais (RON). Translocations can affect the ALK gene resulting in the expression of oncogenic fusion proteins. The formation of ALK fusion proteins results in activation and dysregulation of the gene's expression and signaling which can contribute to increased cell proliferation and survival in tumors expressing these proteins. Crizotinib demonstrated concentration-dependent inhibition of ALK, ROS1, and c-Met phosphorylation in cell-based assays using tumor cell lines and demonstrated antitumor activity in mice bearing tumor xenografts that expressed echinoderm microtubule-associated protein-like 4 (EML4)- or nucleophosmin (NPM)-ALK fusion proteins or c-Met.
In vitro, crizotinib induced apoptosis and inhibited proliferation and ALK-mediated signaling in ALCL-derived cell lines (containing NPM-ALK) at clinically achievable exposures. In vivo data obtained in an ALCL-derived mouse model showed complete regression of the tumor at a dose of 100 mg/kg once daily.
5.7 Embryo Fetal Toxicity
Based on its mechanism of action, XALKORI can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, oral administration of crizotinib in pregnant rats during organogenesis at exposures similar to those observed with the maximum recommended human dose resulted in embryotoxicity and fetotoxicity. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with XALKORI and for 45 days following the last dose. Advise male patients with female partners of reproductive potential to use condoms during treatment with XALKORI and for 90 days after the last dose [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
5 Warnings and Precautions
-
•Hepatotoxicity: Fatal hepatotoxicity has occurred. Monitor with periodic liver testing. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.2, 2.6, 5.1)
-
•Interstitial Lung Disease (ILD)/Pneumonitis: Permanently discontinue in patients with ILD/pneumonitis. (2.6, 5.2)
-
•QT Interval Prolongation: Monitor electrocardiograms and electrolytes in patients who have a history of or predisposition for QTc prolongation, or who are taking medications that prolong QT. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.6, 5.3)
-
•Bradycardia: XALKORI can cause bradycardia. Monitor heart rate and blood pressure regularly. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.6, 5.4)
-
•Severe Visual Loss: XALKORI can cause visual changes including severe visual loss. Monitor and evaluate for ocular toxicity throughout treatment. Discontinue XALKORI in patients with severe visual loss. (2.2, 2.6, 5.5)
-
•Gastrointestinal Toxicity in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT: XALKORI can cause severe nausea, vomiting, diarrhea, and stomatitis. Provide standard antiemetic and antidiarrheal agents. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.6, 5.6)
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.7, 8.1, 8.3)
2 Dosage and Administration
3 Dosage Forms and Strengths
Capsules:
-
•200 mg: hard gelatin capsule, size 1, white opaque body and pink opaque cap, with "Pfizer" on the cap and "CRZ 200" on the body.
-
•250 mg: hard gelatin capsule, size 0, pink opaque cap and body, with "Pfizer" on the cap and "CRZ 250" on the body.
Oral Pellets:
-
•20 mg: hard gelatin capsule, size 4, white opaque body and light blue opaque cap, printed with black ink “Pfizer” on the cap and “CRZ 20” on the body.
-
•50 mg: hard gelatin capsule, size 3, light gray opaque body and gray opaque cap, printed with black ink “Pfizer” on the cap and “CRZ 50” on the body.
-
•150 mg: hard gelatin capsule, size 0, light blue opaque body and cap, printed with black ink “Pfizer” on the cap and “CRZ 150” on the body.
5.3 Qt Interval Prolongation
QTc prolongation can occur in patients treated with XALKORI. Across clinical trials in patients with NSCLC, 2.1% of 1616 patients had QTcF (corrected QT for heart rate by the Fridericia method) greater than or equal to 500 ms and 5% of 1582 patients had an increase from baseline QTcF greater than or equal to 60 ms by automated machine-read evaluation of ECGs.
In Study ADVL0912, QTc prolongation was reported as an adverse reaction in 4.1% of patients, including 8% of patients with ALCL and 7% of pediatric patients with IMT.
Avoid use of XALKORI in patients with congenital long QT syndrome. Monitor ECGs and electrolytes in patients with congestive heart failure, bradyarrhythmias, electrolyte abnormalities, or who are taking medications that are known to prolong the QT interval. Withhold, reduce dose, or permanently discontinue XALKORI for QT/QTc interval prolongation as recommended [see Dosage and Administration (2.6), Clinical Pharmacology (12.2)].
6.2 Postmarketing Experience
The following additional adverse reaction has been identified during post-approval use of XALKORI. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
Investigations: Increased blood creatine phosphokinase
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data in the Warnings and Precautions reflect exposure to XALKORI in 1719 patients with NSCLC who received XALKORI 250 mg twice daily enrolled on Studies 1 (including an additional 109 patients who crossed over from the control arm), 2, 3, a single-arm trial (n=1063) of ALK-positive NSCLC, and an additional ALK-positive NSCLC expansion cohort of a dose finding study (n=154). The data also reflect exposure to XALKORI in 121 patients ages 1 to ≤21 years with relapsed or refractory tumors, including 26 patients with systemic ALCL and 14 pediatric patients with IMT, in a single-arm trial (Study ADVL0912). The data are also described for 7 adult patients with IMT treated with XALKORI in a single-arm trial (Study A8081013).
7.4 Drugs That Cause Bradycardia
XALKORI can cause bradycardia. Avoid concomitant use of XALKORI with drugs that cause bradycardia (e.g., beta-blockers, non-dihydropyridine calcium channel blockers, clonidine, and digoxin) [see Warnings and Precautions (5.4)].
17 Patient Counseling Information
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide; Instructions for Use).
16 How Supplied/storage and Handling
Capsules:
-
•200 mg capsules
Hard gelatin capsule with pink opaque cap and white opaque body, printed with black ink "Pfizer" on the cap, "CRZ 200" on the body; available in:
Bottles of 60 capsules: NDC 0069-8141-20 -
•250 mg capsules
Hard gelatin capsule with pink opaque cap and body, printed with black ink "Pfizer" on the cap, "CRZ 250" on the body; available in:
Bottles of 60 capsules: NDC 0069-8140-20
Store at room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Oral Pellets:
-
•20 mg oral pellets
Hard gelatin capsule, size 4, light blue opaque cap and white opaque body, printed with black ink “Pfizer” on the cap, “CRZ 20” on the body; available in:
Bottles of 60 capsules: NDC 0069-0251-60 -
•50 mg oral pellets
Hard gelatin capsule, size 3, gray opaque cap and light gray opaque body, printed with black ink “Pfizer” on the cap, “CRZ 50” on the body; available in:
Bottles of 60 capsules: NDC 0069-0507-60 -
•150 mg oral pellets
Hard gelatin capsule, size 0, light blue opaque cap and body, printed with black ink “Pfizer” on the cap, “CRZ 150” on the body; available in:
Bottles of 60 capsules: NDC 0069-1500-60
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
7.3 Drugs That Prolong the Qt Interval
XALKORI can prolong the QT/QTc interval. Avoid concomitant use of XALKORI with drugs that prolong the QT interval [see Warnings and Precautions (5.3), Clinical Pharmacology (12.2)].
5.2 Interstitial Lung Disease/pneumonitis
Severe, life-threatening, or fatal interstitial lung disease (ILD)/pneumonitis can occur in patients treated with XALKORI. Across clinical trials in patients with NSCLC (n=1719), 2.9% of XALKORI-treated patients had ILD of any grade, 1% had Grade 3 or 4 ILD, and 0.5% had fatal ILD [see Adverse Reactions (6.1)]. Interstitial lung disease generally occurred within 3 months after the initiation of XALKORI.
In Study ADVL0912, among 121 patients ages 1 to ≤21 years with relapsed or refractory tumors, including ALCL and IMT, ILD occurred in 0.8% of patients.
Monitor patients for pulmonary symptoms indicative of ILD/pneumonitis. Exclude other potential causes of ILD/pneumonitis, and permanently discontinue XALKORI in patients diagnosed with drug-related ILD/pneumonitis [see Dosage and Administration (2.6)].
2.6 Dosage Modifications for Adverse Reactions
The recommended dosage modifications for adverse reactions for adult patients with NSCLC or IMT are provided in Table 4.
|
Dose Reduction |
Dose and Schedule |
|
First Dose Reduction |
200 mg twice daily |
|
Second Dose Reduction |
250 mg once daily |
|
Permanently discontinue XALKORI capsules or pellets if unable to tolerate 250 mg taken once daily. |
The recommended dosage modifications for adverse reactions for pediatric patients with ALCL or IMT and young adults with ALCL are based on body surface area and are provided in Table 5.
|
Body Surface Area (BSA) |
First Dose Reduction |
Second Dose Reduction
Permanently discontinue in patients who are unable to tolerate XALKORI capsules or pellets after 2 dose reductions.
|
||
|
Dosage |
Dosage Form and Strength to Achieve Recommended Dose Reduction |
Dosage |
Dosage Form and Strength to Achieve Recommended Dose Reduction |
|
|
0.38 to 0.46 m2 |
90 mg twice daily |
Pellets: 2 x 20 mg + 1 x 50 mg |
70 mg twice daily |
Pellets: 1 x 20 mg + 1 x 50 mg |
|
0.47 to 0.51 m2 |
100 mg twice daily |
Pellets: 2 x 50 mg |
80 mg twice daily |
Pellets: 4 x 20 mg |
|
0.52 to 0.61 m2 |
120 mg twice daily |
Pellets: 1 x 20 mg + 2 x 50 mg |
90 mg twice daily |
Pellets: 2 x 20 mg + 1 x 50 mg |
|
0.62 to 0.80 m2 |
150 mg twice daily |
Pellets: 1 x 150 mg |
120 mg twice daily |
Pellets: 1 x 20 mg + 2 x 50 mg |
|
0.81 to 0.97 m2 |
200 mg twice daily |
Pellets: 1 x 50 mg + 1 x 150 mg |
150 mg twice daily |
Pellets: 1 x 150 mg |
|
0.98 to 1.16 m2 |
220 mg twice daily |
Pellets: 1 x 20 mg + 1 x 50 mg + 1 x 150 mg |
170 mg twice daily |
Pellets: 1 x 20 mg + 1 x 150 mg |
|
1.17 to 1.33 m2 |
250 mg twice daily |
Pellets: 2 x 50 mg + 1 x 150 mg |
200 mg twice daily |
Pellets: 1 x 50 mg + 1 x 150 mg |
|
1.34 to 1.69 m2 |
250 mg twice daily |
Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg |
200 mg twice daily |
Pellets: 1 x 50 mg + 1 x 150 mg Or Capsule: 1 x 200 mg |
|
1.7 m2 or greater |
400 mg twice daily |
Pellets: 2 x 50 mg + 2 x 150 mg Or Capsule: 2 x 200 mg |
250 mg twice daily |
Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg |
Recommended Dosage Modifications for Hematologic Adverse Reactions for Adult Patients with NSCLC or IMT
The recommended dosage modifications for hematologic adverse reactions for adult patients with NSCLC or IMT are provided in Table 6.
|
Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0.
|
XALKORI Dosage Modification |
|
Grade 3 |
Withhold until recovery to Grade 2 or less, then resume at the same dosage. |
|
Grade 4 |
Withhold until recovery to Grade 2 or less, then resume at next lower dosage. |
Monitor complete blood counts including differential weekly for the first month of therapy and then at least monthly, with more frequent monitoring if Grade 3 or 4 abnormalities, fever, or infection occur.
Recommended Dosage Modifications for Hematologic Adverse Reactions in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT
The recommended dosage modifications for hematologic adverse reactions in pediatric and young adult patients with ALCL or pediatric patients with IMT are provided in Table 7.
|
Severity of Adverse Reaction |
XALKORI Dosage Modification |
|
Absolute Neutrophil Count (ANC) |
|
|
Less than 0.5 x 109/L |
First occurrence: Withhold until recovery to ANC greater than 1.0 x 109/L, then resume at the next lower dosage.
|
|
Platelet Count |
|
|
25 to 50 x 109/L with concurrent bleeding |
Withhold until recovery to platelet count greater than 50 x 109/L and bleeding resolves, then resume at the same dosage. |
|
Less than 25 x 109/L |
Withhold until recovery to platelet count greater than 50 x 109/L, then resume at the next lower dosage. Permanently discontinue for recurrence. |
|
Anemia |
|
|
Hemoglobin less than 8 g/dL |
Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the same dosage. |
|
Life-threatening anemia; urgent intervention indicated. |
Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the next lower dosage. Permanently discontinue for recurrence. |
Recommended Dosage Modifications for Non-Hematologic Adverse Reactions
The recommended dosage modifications for non-hematologic adverse reactions are provided in Table 8.
|
Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0.
|
XALKORI Dosage Modification |
|
Hepatotoxicity [see Warnings and Precautions (5.1)] |
|
|
Alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 1.5 times ULN |
Withhold until recovery to baseline or less than or equal to 3 times ULN, then resume at next lower dosage. |
|
ALT or AST greater than 3 times ULN with concurrent total bilirubin greater than 1.5 times ULN (in the absence of cholestasis or hemolysis) |
Permanently discontinue. |
|
Interstitial Lung Disease (Pneumonitis) [see Warnings and Precautions (5.2)] |
|
|
Any grade drug-related interstitial lung disease/pneumonitis |
Permanently discontinue. |
|
QT Interval Prolongation [see Warnings and Precautions (5.3)] |
|
|
QT corrected for heart rate (QTc) greater than 500 ms on at least 2 separate electrocardiograms (ECGs) |
Withhold until recovery to baseline or to a QTc less than 481 ms, then resume at next lower dosage. |
|
QTc greater than 500 ms or greater than or equal to 60 ms change from baseline with Torsade de pointes or polymorphic ventricular tachycardia or signs/symptoms of serious arrhythmia |
Permanently discontinue. |
|
Bradycardia [see Warnings and Precautions (5.4)] |
|
|
Bradycardia Adult patients: Heart rate less than 60 beats per minute (bpm); Pediatric patients: Resting heart rate less than the 2.5th percentile per age-specific norms. (symptomatic, may be severe and medically significant, medical intervention indicated)
|
Withhold until recovery to a resting heart rate according to the patient’s age (based on the 2.5th percentile per age-specific norms) as follows:
Evaluate concomitant medications known to cause bradycardia, as well as antihypertensive medications. |
|
Bradycardia (life-threatening consequences, urgent intervention indicated) |
Permanently discontinue if no contributing concomitant medication is identified. |
|
Ocular Toxicity, including Visual Loss [see Warnings and Precautions (5.5)] |
|
|
Visual Symptoms, Grade 1 (mild symptoms) or Grade 2 (moderate symptoms affecting ability to perform age-appropriate activities of daily living) |
Monitor symptoms and report any symptoms to an eye specialist. Consider dose reduction for Grade 2 visual disorders. |
|
Visual Loss (Grade 3 or 4 Ocular Disorder, marked decrease in vision) |
Discontinue during evaluation of severe visual loss. |
|
Gastrointestinal Toxicity Dosage modifications for gastrointestinal toxicity for pediatric patients with ALCL or IMT and young adults with ALCL only.
[see Warnings and Precautions (5.6)]
|
|
|
Nausea (Grade 3: inadequate oral intake for more than 3 days, medical intervention required) |
Grade 3 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. Permanently discontinue in patients who are unable to tolerate XALKORI after 2 dose reductions.
|
|
Vomiting (Grade 3: more than 6 episodes in 24 hours for more than 3 days, medical intervention required, i.e., tube feeding or hospitalization; Grade 4: life-threatening consequences, urgent intervention indicated) |
Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. |
|
Diarrhea (Grade 3: increase of 7 or more stools per day over baseline; incontinence; hospitalization indicated; Grade 4: life-threatening consequences, urgent intervention indicated) |
Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. |
Principal Display Panel 20 Mg Oral Pellets Label
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
NDC 0069-0251-60
Pfizer
XALKORI®
(crizotinib) oral pellets
20 mg
Open capsule shell to administer pellets.
Do not swallow capsule whole.
Do not crush or chew pellets.
60 Capsules
Rx only
Principal Display Panel 50 Mg Oral Pellets Label
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
NDC 0069-0507-60
Pfizer
XALKORI®
(crizotinib) oral pellets
50 mg
Open capsule shell to administer pellets.
Do not swallow capsule whole.
Do not crush or chew pellets.
60 Capsules
Rx only
2.8 Dosage Modification for Severe Renal Impairment
The recommended dosage of XALKORI in patients with severe renal impairment [creatinine clearance (CLcr) less than 30 mL/min, calculated using the modified Cockcroft-Gault equation for adult patients and the Schwartz equation for pediatric patients] not requiring dialysis is the second dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
Principal Display Panel 150 Mg Oral Pellets Label
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
NDC 0069-1500-60
Pfizer
XALKORI®
(crizotinib) oral pellets
150 mg
Open capsule shell to administer pellets.
Do not swallow capsule whole.
Do not crush or chew pellets.
60 Capsules
Rx only
2.2 Recommended Testing During Treatment With Xalkori
-
•Monitor liver function tests, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), and total bilirubin, every 2 weeks during the first 2 months of treatment, then once a month, and as clinically indicated, with more frequent repeat testing for increased liver transaminases, alkaline phosphatase, or total bilirubin in patients who develop increased transaminases [see Warnings and Precautions (5.1)].
-
•Monitor complete blood counts including differential weekly for the first month of therapy and then at least monthly, with more frequent monitoring if Grade 3 or 4 abnormalities, fever, or infection occur [see Adverse Reactions (6.1)].
-
•For pediatric and young adult patients with ALCL or pediatric patients with IMT, obtain baseline and follow-up ophthalmologic examinations including retinal examination within 1 month of starting XALKORI and every 3 months thereafter [see Warnings and Precautions (5.5)].
Principal Display Panel 200 Mg Capsule Bottle Label
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
Pfizer
NDC 0069-8141-20
XALKORI®
(crizotinib) capsules
200 mg
Swallow capsule whole
60 Capsules
Rx only
Principal Display Panel 250 Mg Capsule Bottle Label
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
Pfizer
NDC 0069-8140-20
XALKORI®
(crizotinib) capsules
250 mg
Swallow capsule whole
60 Capsules
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with crizotinib have not been conducted.
Crizotinib was genotoxic in an in vitro micronucleus assay in Chinese Hamster Ovary cultures, in an in vitro human lymphocyte chromosome aberration assay, and in in vivo rat bone marrow micronucleus assays. Crizotinib was not mutagenic in vitro in the bacterial reverse mutation (Ames) assay.
No specific studies with crizotinib have been conducted in animals to evaluate the effect on fertility; however, crizotinib is considered to have the potential to impair reproductive function and fertility in humans based on findings in repeat-dose toxicity studies in the rat. Findings observed in the male reproductive tract included testicular pachytene spermatocyte degeneration in rats given greater than or equal to 50 mg/kg/day for 28 days (greater than 1.7 times the recommended human dose based on AUC). Findings observed in the female reproductive tract included single-cell necrosis of ovarian follicles of a rat given 500 mg/kg/day (approximately 10 times the recommended human dose based on body surface area) for 3 days.
1.1 Alk Or Ros1 Positive Metastatic Non Small Cell Lung Cancer
XALKORI is indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are anaplastic lymphoma kinase (ALK) or ROS1-positive as detected by an FDA-approved test [see Dosage and Administration (2.1)].
2.7 Dosage Modifications for Moderate and Severe Hepatic Impairment
The recommended dose of XALKORI in patients with moderate hepatic impairment [any aspartate aminotransferase (AST) and total bilirubin greater than 1.5 times the upper limit of normal (ULN) and less than or equal to 3 times ULN] is the first dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
The recommended dose of XALKORI in patients with severe hepatic impairment (any AST and total bilirubin greater than 3 times ULN) is the second dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL [see Dosage and Administration (2.6), Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2.9 Dosage Modification for Concomitant Use of Strong Cyp3a Inhibitors
Avoid concomitant use of strong CYP3A inhibitors [see Drug Interactions (7.1)]. If concomitant use of strong CYP3A inhibitors is unavoidable, reduce the dose of XALKORI to the second dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL. After discontinuation of a strong CYP3A inhibitor, resume the XALKORI dose used prior to initiating the strong CYP3A inhibitor.
1.2 Relapsed Or Refractory, Systemic Alk Positive Anaplastic Large Cell Lymphoma
XALKORI is indicated for the treatment of pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic anaplastic large cell lymphoma (ALCL) that is ALK-positive.
14.2 Relapsed Or Refractory, Systemic Alk Positive Anaplastic Large Cell Lymphoma
The efficacy of XALKORI was evaluated in Study ADVL0912 (NCT00939770), a multicenter, single arm, open-label study in patients 1 to ≤21 years of age that included 26 patients with relapsed or refractory, systemic ALK-positive ALCL after at least one systemic treatment. ALK-positive status (confirmation of an ALK fusion) was determined locally by immunohistochemistry or fluorescence in situ hybridization. The study excluded patients with primary cutaneous ALCL or central nervous system involvement by lymphoma.
Patients received XALKORI 280 mg/m2 (20 patients) or 165 mg/m2 (6 patients) orally twice daily until disease progression or unacceptable toxicity. Patients were permitted to discontinue XALKORI to undergo hematopoietic stem cell transplantation (HSCT).
Of the 26 patients evaluated, the median age was 11 years (range: 3 to 20); 69% were male, 54% were White, 19% Black, 8% Asian. Patient enrollment by age category was 4 patients from 3 to <6 years, 11 patients from 6 to <12 years, 7 patients from 12 to <18 years, and 4 patients from 18 to ≤21 years.
All patients had received multi-agent systemic therapy, 2 (8%) had received a prior HSCT and 4 (15%) had received at least 3 prior therapies.
Efficacy was based on objective response rate and duration of response, as assessed by an independent review committee (Table 19). The median time to first response was 3.9 weeks (range: 3.5 to 9.1 weeks).
| Efficacy Parameter | N=26 |
|---|---|
| CI=confidence interval; N/n=number of patients | |
|
Objective response rate (95% CI, %) Based on Lugano Classification.
,
95% CI based on Wilson score method.
|
88% (71, 96) |
|
Complete response, n |
21 (81%) |
|
Partial response, n |
2 (8%) |
|
Duration of response Of 23 patients with objective response, 2 had disease progression and the remainder (91% of responding patients) were censored.
|
|
|
Patients maintaining response at 3 months, n/N |
13/23 (57%) |
|
Patients maintaining response at 6 months, n/N |
9/23 (39%) |
|
Patients maintaining response at 12 months, n/N |
5/23 (22%) |
1.3 Unresectable, Recurrent, Or Refractory Alk Positive Inflammatory Myofibroblastic Tumor
XALKORI is indicated for the treatment of adult and pediatric patients 1 year of age and older with unresectable, recurrent, or refractory inflammatory myofibroblastic tumor (IMT) that is ALK-positive.
2.5 Concomitant Treatments for Pediatric and Young Adult Patients With Alcl Or Pediatric Patients With Imt
Antiemetics are recommended prior to and during treatment with XALKORI to prevent nausea and vomiting. Provide standard antiemetic and antidiarrheal agents for gastrointestinal toxicities.
Consider intravenous or oral hydration for patients at risk of dehydration, and replace electrolytes as clinically indicated [see Warnings and Precautions (5.6)].
5.6 Gastrointestinal Toxicity in Pediatric and Young Adult Patients With Alcl Or Pediatric Patients With Imt
XALKORI can cause severe gastrointestinal toxicities in pediatric and young adult patients with ALCL or pediatric patients with IMT [see Adverse Reactions (6.1)]. In patients with ALCL (n=26), gastrointestinal toxicity occurred in 100% of patients; Grade 3 gastrointestinal toxicity occurred in 27% of patients and included diarrhea, nausea, vomiting, and stomatitis. In pediatric patients with IMT (n=14), vomiting occurred in 93%, nausea occurred in 86%, and diarrhea occurred in 64% of patients.
Provide standard antiemetic and antidiarrheal agents for gastrointestinal toxicities in pediatric and young adult patients with ALCL or pediatric patients with IMT. Antiemetics are recommended prior to and during treatment with XALKORI to prevent nausea and vomiting. If patients develop Grade 3 nausea lasting 3 days or Grade 3 or 4 diarrhea or vomiting despite maximum medical therapy, withhold XALKORI until resolved, and then resume at the next lower dose level. Consider supportive care such as hydration, electrolyte supplementation, and nutritional support as clinically indicated [see Dosage and Administration (2.6)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use: The safety and efficacy of XALKORI have not been established in older adults with relapsed or refractory, systemic ALK-positive ALCL.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: Sep 2023 | |||
|
MEDICATION GUIDE |
|||
|
XALKORI® (zal-KOR-ee)
|
XALKORI® (zal-KOR-ee)
|
||
|
What is the most important information I should know about XALKORI?
|
|||
|
|
||
|
|||
|
|
||
|
In addition, for children or young adults taking XALKORI to treat anaplastic large cell lymphoma (ALCL) or children taking XALKORI to treat inflammatory myofibroblastic tumor (IMT): |
|||
See "What are possible side effects of XALKORI?" for more information about side effects. |
|||
|
What is XALKORI?
It is not known if XALKORI is safe and effective in children younger than 1 year of age with ALCL or IMT, or in any children with NSCLC. |
|||
|
Before taking XALKORI, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. XALKORI can affect the way other medicines work, and other medicines can affect how XALKORI works. |
|||
|
How should I take XALKORI?
XALKORI comes as capsules and oral pellets.
|
|||
|
What should I avoid while taking XALKORI?
|
|||
|
What are the possible side effects of XALKORI?
The most common side effects of XALKORI in adults with NSCLC include: |
|||
|
|
||
|
The most common side effects of XALKORI in people with ALCL include: |
|||
|
|
||
|
The most common side effects of XALKORI in adults with IMT include: |
|||
|
|||
|
The most common side effects of XALKORI in children with IMT include: |
|||
|
|
||
|
XALKORI may cause fertility problems in females and males, which may affect the ability to have children. Talk to your healthcare provider if you have concerns about fertility. |
|||
|
How should I store XALKORI?
Keep XALKORI and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of XALKORI.
|
|||
|
What are the ingredients in XALKORI?
LAB-0441-12.0 |
Section 43683-2 (43683-2)
|
Dosage and Administration (2) |
9/2023 |
11 Description (11 DESCRIPTION)
Crizotinib is a kinase inhibitor. The molecular formula for crizotinib is C21H22Cl2FN5O and the molecular weight is 450.34 daltons. Crizotinib is described chemically as (R)-3-[1-(2,6-Dichloro-3-fluorophenyl)ethoxy]-5-[1-(piperidin-4-yl)-1H-pyrazol-4-yl]pyridin-2-amine.
The chemical structure of crizotinib is shown below:
Crizotinib is a white to pale-yellow powder with a pKa of 9.4 (piperidinium cation) and 5.6 (pyridinium cation). The solubility of crizotinib in aqueous media decreases over the range pH 1.6 to pH 8.2 from greater than 10 mg/mL to less than 0.1 mg/mL. The log of the distribution coefficient (octanol/water) at pH 7.4 is 1.65.
Capsules:
XALKORI (crizotinib) capsules for oral administration are supplied as printed hard-shell capsules containing 250 mg or 200 mg of crizotinib together with colloidal silicon dioxide, microcrystalline cellulose, anhydrous dibasic calcium phosphate, sodium starch glycolate, magnesium stearate, and hard gelatin capsule shells as inactive ingredients.
The pink opaque capsule shell components contain gelatin, titanium dioxide, and red iron oxide. The white opaque capsule shell components contain gelatin and titanium dioxide. The printing ink contains shellac, propylene glycol, strong ammonia solution, potassium hydroxide, and black iron oxide.
Oral Pellets:
XALKORI (crizotinib) oral pellets for oral administration are supplied as 20 mg, 50 mg, 150 mg of crizotinib contained in hard gelatin capsules. The inactive ingredients in the uncoated pellets are poloxamer and stearyl alcohol. The film-coating consists of hypromellose, glyceryl monostearate, medium chain triglycerides, polyethylene glycol/macrogol, sucrose, and talc.
5.4 Bradycardia
Symptomatic bradycardia can occur in patients receiving XALKORI. Across clinical trials in patients with NSCLC, bradycardia occurred in 13% of 1719 patients treated with XALKORI. Grade 3 syncope occurred in 2.4% of XALKORI-treated patients and in 0.6% of the chemotherapy-treated patients [see Adverse Reactions (6.1)].
In Study ADVL0912, among 121 patients ages 1 to ≤21 years treated with XALKORI, bradycardia was reported in 14%, including Grade 3 bradycardia in 0.8% of patients. Of the 26 patients with ALCL treated with XALKORI, bradycardia (all Grade 1) was reported in 19%. Of the 14 pediatric patients with IMT treated with XALKORI, bradycardia was reported in 14% of patients, including Grade 3 bradycardia in 7% of patients.
Avoid using XALKORI in combination with other medications known to cause bradycardia (e.g., beta-blockers, nondihydropyridine-calcium channel blockers, clonidine, and digoxin) to the extent possible. Monitor heart rate and blood pressure regularly. If bradycardia occurs, re-evaluate for the use of concomitant medications known to cause bradycardia. Withhold, reduce dose, or permanently discontinue XALKORI for bradycardia as recommended [see Dosage and Administration (2.6)].
8.4 Pediatric Use
The safety and effectiveness of XALKORI have been established in pediatric patients 1 year of age and older with relapsed or refractory, systemic ALK-positive ALCL or with unresectable, recurrent, or refractory ALK-positive IMT [see Adverse Reactions (6.1), Clinical Studies (14.2, 14.3)]. The safety and effectiveness have not been established in pediatric patients younger than 1 year of age with ALCL or with IMT, or in any pediatric patients with NSCLC.
In a study that evaluated XALKORI in combination with chemotherapy in pediatric patients with newly diagnosed ALCL (Study ANHL12P1; NCT01979536), 13 of 66 (20%) patients had a Grade 2 or higher thromboembolic event, including pulmonary embolism in 6%. The safety and effectiveness of XALKORI in combination with chemotherapy have not been established in patients with newly diagnosed ALCL.
8.5 Geriatric Use
Of the total number of patients with ALK-positive metastatic NSCLC in clinical studies of XALKORI (n=1669), 16% were 65 years or older and 3.8% were 75 years or older. No overall differences in safety or effectiveness were observed between these patients and younger patients.
Clinical studies of XALKORI in patients with ROS1-positive metastatic NSCLC did not include sufficient numbers of patients age 65 years and older to determine whether they respond differently from younger patients.
2.4 Administration
-
•Administer XALKORI capsules or pellets orally, twice daily, with or without food.
-
•If a dose of XALKORI capsules or pellets is missed, make up that dose unless the next dose is due within 6 hours.
-
•If vomiting occurs after taking a dose of XALKORI capsules or pellets, do not take an additional dose. Take the next dose at the regular scheduled time.
XALKORI Capsules
-
•Swallow XALKORI capsules whole, with or without food twice daily.
-
•Do not chew, crush or split XALKORI capsules.
XALKORI Pellets
-
•XALKORI pellets are supplied encapsulated in shells.
-
•Do not chew or crush XALKORI pellets.
-
•Do not swallow XALKORI pellets encapsulated in the shell.
-
•XALKORI pellets can be administered by 2 options:
-
1.Open shell(s) containing XALKORI pellets and empty the contents directly into the patient’s mouth.
-
2.Open shell(s) containing XALKORI pellets and empty the contents into a consumer-supplied oral dosing aid (e.g., spoon, medicine cup). Administer XALKORI pellets via the dosing aid directly into the patient’s mouth.
-
•Immediately after administration, give a sufficient amount of water to ensure that all medication is swallowed.
5.1 Hepatotoxicity
Drug-induced hepatotoxicity with fatal outcome occurred in 0.1% of the 1719 patients treated with XALKORI for NSCLC across clinical trials [see Adverse Reactions (6.1)]. Concurrent elevations in ALT or AST ≥3 times the ULN and total bilirubin ≥2 times the ULN, with normal alkaline phosphatase, occurred in <1% of patients treated with XALKORI. Increased ALT or AST >5 times the ULN occurred in 11% and 6% of patients, respectively. One percent (1.0%) of patients required permanent discontinuation due to elevated transaminases. Increased transaminases generally occurred within the first 2 months of treatment.
In Study ADVL0912, of 121 patients ages 1 to ≤21 years treated with XALKORI for relapsed or refractory tumors including ALCL and IMT, 71% and 79% had increases of AST and ALT, respectively, with increased ALT or AST >5 times the ULN in 6% each. Of the 26 patients with ALCL treated with XALKORI, 65% and 81% had increases of AST and ALT, respectively, with increases >5 times the ULN in 4% each. Of the 14 pediatric patients with IMT treated with XALKORI, 71% had increases of AST and 71% had increases of ALT.
In Study A8081013, of the 7 adult patients with IMT treated with XALKORI, 57% and 43% had increases of AST and ALT, respectively.
Monitor liver function tests, including ALT, AST, and total bilirubin, every 2 weeks during the first 2 months of treatment, then once a month, and as clinically indicated, with more frequent repeat testing for increased liver transaminases, alkaline phosphatase, or total bilirubin in patients who develop increased transaminases. Withhold, reduce dose, or permanently discontinue XALKORI for hepatotoxicity as recommended [see Dosage and Administration (2.6)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Hepatotoxicity [see Warnings and Precautions (5.1)]
-
•Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.2)]
-
•QT Interval Prolongation [see Warnings and Precautions (5.3)]
-
•Bradycardia [see Warnings and Precautions (5.4)]
-
•Severe Visual Loss [see Warnings and Precautions (5.5)]
-
•Gastrointestinal Toxicity in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
Increased exposure to crizotinib occurred in patients with pre-existing severe renal impairment (CLcr less than 30 mL/min calculated using the modified Cockcroft-Gault equation for adult patients and the Schwartz equation for pediatric patients) not requiring dialysis, therefore reduce dosage of XALKORI in these patients [see Dosage and Administration (2.8), Clinical Pharmacology (12.3)]. No dose adjustment is recommended in patients with mild to moderate renal impairment (CLcr 30 to 89 mL/min).
Instructions for Use
INSTRUCTIONS FOR USE
XALKORI® [zal-KOR-ee]
(crizotinib)
oral pellets
This Instructions for Use contains information on how to give or take XALKORI oral pellets. Read this Instructions for Use each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider or pharmacist about your or your child’s medical condition or treatment.
Important information you need to know before giving or taking XALKORI oral pellets:
-
•XALKORI oral pellets come in a capsule “shell” that must be opened before giving or taking a dose. Do not swallow the shell containing the oral pellets. Do not chew or crush the oral pellets.
-
•XALKORI oral pellets come in 3 dosage strengths: 20 mg, 50 mg, and 150 mg. Your healthcare provider may combine different strengths for your prescribed dose. No more than 4 XALKORI oral pellet shells are to be used for a single dose.
-
•Your healthcare provider will decide the right dose of XALKORI oral pellets for you or your child. Follow your healthcare provider’s instructions for the dose of XALKORI oral pellets to give your child or for you to take.
-
•Empty XALKORI oral pellets from the shells as described in Steps 1 to 4 below.
-
•Check the expiration date on the bottle containing XALKORI oral pellets. Do not use XALKORI oral pellets after the expiration date on the bottle has passed.
-
•Ask your healthcare provider or pharmacist if you are not sure how to prepare and give or take the prescribed dose of XALKORI oral pellets.
Supplies needed to give or take XALKORI oral pellets:
-
•XALKORI oral pellet(s), as prescribed by your healthcare provider.
-
•Spoon or medicine cup (optional). See Step 4 “Giving or taking XALKORI oral pellets”.
Preparing XALKORI oral pellets (Steps 1 to 3):
|
Step 1 |
Remove the number of XALKORI oral pellet shell(s) needed for the prescribed dose from each bottle(s). |
|
Step 2 |
|
|
Figure A |
|
|
Step 3 |
Carefully hold and twist the top and bottom parts of the shell in opposite directions and pull apart to open the shell (see Figure B). Figure B |
Giving or taking XALKORI oral pellets (Step 4): There are 2 options for giving or taking the oral pellets.
|
Step 4 |
Option 1 (Pour directly into the mouth) |
|
|
Figure C |
||
|
Option 2 (Pour from a spoon or medicine cup) |
|
|
|
Figure D |
||
|
After Step 4 is completed, other drinks or foods can be given or taken, except grapefruit juice, grapefruit, or supplements containing grapefruit extract. |
Storing XALKORI oral pellets:
-
•Store XALKORI oral pellets at room temperature between 68°F to 77°F (20°C to 25°C).
-
•Keep XALKORI oral pellets and all medicines out of the reach of children.
Disposing of empty XALKORI oral pellet shells:
-
•Dispose of (throw away) the empty XALKORI oral pellet shell(s) in the household trash.
-
•Ask your pharmacist how to throw away medicines you no longer use or are expired.
For more information, go to www.Pfizer.com or call 1-800-438-1985.
LAB-1526-1.0
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Issued: 09/2023
12.3 Pharmacokinetics
Following XALKORI 250 mg capsules twice daily, steady-state was reached within 15 days with a median accumulation ratio of 4.8. Steady-state minimum concentration (Cmin.ss) and AUC increased in a greater than dose-proportional manner over the dose range of 200 mg to 300 mg twice daily (0.8 to 1.2 times the approved recommended dosage).
2.1 Patient Selection
Select patients for the treatment of metastatic NSCLC with XALKORI based on the presence of ALK or ROS1 positivity in tumor specimens [see Clinical Studies (14.1, 14.2, 14.3)].
Information on FDA-approved tests for the detection of ALK and ROS1 rearrangements in NSCLC is available at http://www.fda.gov/companiondiagnostics.
2.3 Recommended Dosage
The recommended dosage of XALKORI is provided in Table 1.
|
Indication |
Recommended Dosage of XALKORI |
|
ALK- or ROS1-Positive Metastatic NSCLC |
Adults: |
|
Relapsed or Refractory, Systemic ALK-Positive ALCL |
Pediatric Patients and Young Adults: See Table 2 for Recommended Dosage based on body surface area for pediatric patients and young adults with ALCL for the capsules and oral pellets.
|
|
Unresectable, Recurrent, or Refractory ALK-Positive IMT |
Adults: |
|
Pediatric Patients: See Table 3 for Recommended Dosage based on body surface area for pediatric patients with IMT for the capsules and oral pellets.
|
Recommended Dosage for Adult Patients with ALK- or ROS1-Positive Metastatic NSCLC
-
•The recommended dosage for adult patients with ALK- or ROS1-positive metastatic NSCLC is XALKORI capsules 250 mg orally, twice daily, with or without food until disease progression or unacceptable toxicity occurs.
-
•For adults who cannot swallow capsules, the recommended dosage of XALKORI pellets is 250 mg (2 x 50 mg + 1 x 150 mg) orally, twice daily, with or without food until disease progression or unacceptable toxicity occurs.
Recommended Dosage for Pediatric and Young Adult Patients with ALK-Positive ALCL
-
•The recommended dosage for pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic ALK-positive ALCL is based on body surface area (BSA) and is provided in Table 2.
-
•Administer XALKORI capsules or pellets orally, twice daily, with or without food until disease progression or unacceptable toxicity occurs.
Table 2 provides the dosage based on body surface area (BSA) for XALKORI capsules or pellets.
|
Body Surface Area (BSA) |
Recommended XALKORI Dosage to Achieve 280 mg/m2 Twice Daily |
Dose Strength Combinations of XALKORI Pellets to Administer |
Dose Strength Combinations of XALKORI Capsules to Administer |
|
0.38 to 0.46 m2 |
120 mg twice daily |
1 x 20 mg + 2 x 50 mg |
--- |
|
0.47 to 0.51 m2 |
140 mg twice daily |
2 x 20 mg + 2 x 50 mg |
--- |
|
0.52 to 0.61 m2 |
150 mg twice daily |
1 x 150 mg |
--- |
|
0.62 to 0.80 m2 |
200 mg twice daily |
1 x 50 mg + 1 x 150 mg |
--- |
|
0.81 to 0.97 m2 |
250 mg twice daily |
2 x 50 mg + 1 x 150 mg |
--- |
|
0.98 to 1.16 m2 |
300 mg twice daily |
2 x 150 mg |
--- |
|
1.17 to 1.33 m2 |
350 mg twice daily |
1 x 50 mg + 2 x 150 mg |
--- |
|
1.34 to 1.51 m2 |
400 mg twice daily |
2 x 50 mg + 2 x 150 mg |
2 x 200 mg |
|
1.52 to 1.69 m2 |
450 mg twice daily |
3 x 150 mg |
1 x 200 mg + 1 x 250 mg |
|
1.7 m2 or greater |
500 mg twice daily |
1 x 50 mg + 3 x 150 mg |
2 x 250 mg |
Recommended Dosage for Pediatric and Adult Patients with ALK-Positive IMT
-
•The recommended dosage for adult patients with unresectable, recurrent, or refractory ALK-positive IMT is provided in Table 1.
-
•The recommended dosage for pediatric patients 1 year of age and older with unresectable, recurrent, or refractory ALK-positive IMT is based on BSA and is provided in Table 3.
-
•Administer XALKORI capsules or pellets orally twice daily, with or without food, until disease progression or unacceptable toxicity occurs.
Table 3 provides the dosage based on BSA for XALKORI capsules or pellets.
|
Body Surface Area (BSA) |
Recommended XALKORI Dosage to Achieve 280 mg/m2 Twice Daily |
Dose Strength Combinations of XALKORI Pellets to Administer
No more than 4 oral pellet shells are to be used for a single dose.
|
Dose Strength Combinations of XALKORI Capsules to Administer |
|
0.38 to 0.46 m2 |
120 mg twice daily |
1 x 20 mg + 2 x 50 mg |
--- |
|
0.47 to 0.51 m2 |
140 mg twice daily |
2 x 20 mg + 2 x 50 mg |
--- |
|
0.52 to 0.61 m2 |
150 mg twice daily |
1 x 150 mg |
--- |
|
0.62 to 0.80 m2 |
200 mg twice daily |
1 x 50 mg + 1 x 150 mg |
--- |
|
0.81 to 0.97 m2 |
250 mg twice daily |
2 x 50 mg + 1 x 150 mg |
--- |
|
0.98 to 1.16 m2 |
300 mg twice daily |
2 x 150 mg |
--- |
|
1.17 to 1.33 m2 |
350 mg twice daily |
1 x 50 mg + 2 x 150 mg |
--- |
|
1.34 to 1.51 m2 |
400 mg twice daily |
2 x 50 mg + 2 x 150 mg |
2 x 200 mg |
|
1.52 to 1.69 m2 |
450 mg twice daily |
3 x 150 mg |
1 x 200 mg + 1 x 250 mg |
|
1.7 m2 or greater |
500 mg twice daily |
1 x 50 mg + 3 x 150 mg |
2 x 250 mg |
5.5 Severe Visual Loss
Across all clinical trials in patients with NSCLC, the incidence of Grade 4 visual field defect with visual loss was 0.2% of 1719 patients [see Adverse Reactions (6.1)]. Optic atrophy and optic nerve disorder have been reported as potential causes of visual loss.
In Study ADVL0912, visual disorders occurred in 46% of 121 patients with XALKORI, including 65% of 26 patients with ALCL and 50% of 14 patients with IMT. Of the 56 patients who experienced visual disorders, one pediatric patient with IMT experienced Grade 3 myopic optic nerve disorder. The most common visual symptoms were blurred vision and visual impairment.
Assessment of visual symptoms for all patients is recommended monthly during treatment. Report new visual symptoms to an eye specialist.
For pediatric and young adult patients with ALCL or pediatric patients with IMT, obtain baseline and follow-up ophthalmologic examinations including retinal examination within 1 month of starting XALKORI and every 3 months thereafter. The ophthalmological evaluation should consist of best corrected visual acuity, retinal photographs, visual fields, optical coherence tomography (OCT), and other evaluations as appropriate [see Dosage and Administration (2.6), Adverse Reactions (6.1)].
Permanently discontinue XALKORI for Grade 3 or 4 ocular disorders or severe visual loss (best corrected vision less than 20/200 in one or both eyes) unless another cause is identified [see Dosage and Administration (2.6)]. There is insufficient information to characterize the risks of resumption of XALKORI in patients who develop visual symptoms or visual loss. A decision to resume XALKORI should consider the potential benefits versus risks to the patient.
8.6 Hepatic Impairment
Crizotinib concentrations increased in patients with pre-existing moderate (any AST and total bilirubin greater than 1.5 times ULN and less than or equal to 3 times ULN) or severe (any AST and total bilirubin greater than 3 times ULN) hepatic impairment [see Clinical Pharmacology (12.3)]. Reduce XALKORI dosage in patients with moderate or severe hepatic impairment [see Dosage and Administration (2.7)]. No dose adjustment is recommended in patients with pre-existing mild hepatic impairment (AST > ULN and total bilirubin less than or equal to 1 times ULN or any AST and total bilirubin greater than 1 times ULN but less than or equal to1.5 times ULN).
1 Indications and Usage (1 INDICATIONS AND USAGE)
XALKORI is a kinase inhibitor indicated for the treatment of
-
•adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are anaplastic lymphoma kinase (ALK) or ROS1-positive as detected by an FDA-approved test. (1.1, 2.1)
-
•pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic anaplastic large cell lymphoma (ALCL) that is ALK-positive. (1.2, 2.3)
-
oLimitations of Use: The safety and efficacy of XALKORI have not been established in older adults with relapsed or refractory, systemic ALK-positive ALCL.
-
-
•adult and pediatric patients 1 year of age and older with unresectable, recurrent, or refractory inflammatory myofibroblastic tumor (IMT) that is ALK-positive. (1.3, 2.3)
12.1 Mechanism of Action
Crizotinib is an inhibitor of receptor tyrosine kinases including ALK, Hepatocyte Growth Factor Receptor (HGFR, c-Met), ROS1 (c-ros), and Recepteur d'Origine Nantais (RON). Translocations can affect the ALK gene resulting in the expression of oncogenic fusion proteins. The formation of ALK fusion proteins results in activation and dysregulation of the gene's expression and signaling which can contribute to increased cell proliferation and survival in tumors expressing these proteins. Crizotinib demonstrated concentration-dependent inhibition of ALK, ROS1, and c-Met phosphorylation in cell-based assays using tumor cell lines and demonstrated antitumor activity in mice bearing tumor xenografts that expressed echinoderm microtubule-associated protein-like 4 (EML4)- or nucleophosmin (NPM)-ALK fusion proteins or c-Met.
In vitro, crizotinib induced apoptosis and inhibited proliferation and ALK-mediated signaling in ALCL-derived cell lines (containing NPM-ALK) at clinically achievable exposures. In vivo data obtained in an ALCL-derived mouse model showed complete regression of the tumor at a dose of 100 mg/kg once daily.
5.7 Embryo Fetal Toxicity (5.7 Embryo-Fetal Toxicity)
Based on its mechanism of action, XALKORI can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, oral administration of crizotinib in pregnant rats during organogenesis at exposures similar to those observed with the maximum recommended human dose resulted in embryotoxicity and fetotoxicity. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with XALKORI and for 45 days following the last dose. Advise male patients with female partners of reproductive potential to use condoms during treatment with XALKORI and for 90 days after the last dose [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Hepatotoxicity: Fatal hepatotoxicity has occurred. Monitor with periodic liver testing. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.2, 2.6, 5.1)
-
•Interstitial Lung Disease (ILD)/Pneumonitis: Permanently discontinue in patients with ILD/pneumonitis. (2.6, 5.2)
-
•QT Interval Prolongation: Monitor electrocardiograms and electrolytes in patients who have a history of or predisposition for QTc prolongation, or who are taking medications that prolong QT. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.6, 5.3)
-
•Bradycardia: XALKORI can cause bradycardia. Monitor heart rate and blood pressure regularly. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.6, 5.4)
-
•Severe Visual Loss: XALKORI can cause visual changes including severe visual loss. Monitor and evaluate for ocular toxicity throughout treatment. Discontinue XALKORI in patients with severe visual loss. (2.2, 2.6, 5.5)
-
•Gastrointestinal Toxicity in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT: XALKORI can cause severe nausea, vomiting, diarrhea, and stomatitis. Provide standard antiemetic and antidiarrheal agents. Temporarily suspend, dose reduce, or permanently discontinue XALKORI. (2.6, 5.6)
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.7, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules:
-
•200 mg: hard gelatin capsule, size 1, white opaque body and pink opaque cap, with "Pfizer" on the cap and "CRZ 200" on the body.
-
•250 mg: hard gelatin capsule, size 0, pink opaque cap and body, with "Pfizer" on the cap and "CRZ 250" on the body.
Oral Pellets:
-
•20 mg: hard gelatin capsule, size 4, white opaque body and light blue opaque cap, printed with black ink “Pfizer” on the cap and “CRZ 20” on the body.
-
•50 mg: hard gelatin capsule, size 3, light gray opaque body and gray opaque cap, printed with black ink “Pfizer” on the cap and “CRZ 50” on the body.
-
•150 mg: hard gelatin capsule, size 0, light blue opaque body and cap, printed with black ink “Pfizer” on the cap and “CRZ 150” on the body.
5.3 Qt Interval Prolongation (5.3 QT Interval Prolongation)
QTc prolongation can occur in patients treated with XALKORI. Across clinical trials in patients with NSCLC, 2.1% of 1616 patients had QTcF (corrected QT for heart rate by the Fridericia method) greater than or equal to 500 ms and 5% of 1582 patients had an increase from baseline QTcF greater than or equal to 60 ms by automated machine-read evaluation of ECGs.
In Study ADVL0912, QTc prolongation was reported as an adverse reaction in 4.1% of patients, including 8% of patients with ALCL and 7% of pediatric patients with IMT.
Avoid use of XALKORI in patients with congenital long QT syndrome. Monitor ECGs and electrolytes in patients with congestive heart failure, bradyarrhythmias, electrolyte abnormalities, or who are taking medications that are known to prolong the QT interval. Withhold, reduce dose, or permanently discontinue XALKORI for QT/QTc interval prolongation as recommended [see Dosage and Administration (2.6), Clinical Pharmacology (12.2)].
6.2 Postmarketing Experience
The following additional adverse reaction has been identified during post-approval use of XALKORI. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
Investigations: Increased blood creatine phosphokinase
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data in the Warnings and Precautions reflect exposure to XALKORI in 1719 patients with NSCLC who received XALKORI 250 mg twice daily enrolled on Studies 1 (including an additional 109 patients who crossed over from the control arm), 2, 3, a single-arm trial (n=1063) of ALK-positive NSCLC, and an additional ALK-positive NSCLC expansion cohort of a dose finding study (n=154). The data also reflect exposure to XALKORI in 121 patients ages 1 to ≤21 years with relapsed or refractory tumors, including 26 patients with systemic ALCL and 14 pediatric patients with IMT, in a single-arm trial (Study ADVL0912). The data are also described for 7 adult patients with IMT treated with XALKORI in a single-arm trial (Study A8081013).
7.4 Drugs That Cause Bradycardia
XALKORI can cause bradycardia. Avoid concomitant use of XALKORI with drugs that cause bradycardia (e.g., beta-blockers, non-dihydropyridine calcium channel blockers, clonidine, and digoxin) [see Warnings and Precautions (5.4)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide; Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Capsules:
-
•200 mg capsules
Hard gelatin capsule with pink opaque cap and white opaque body, printed with black ink "Pfizer" on the cap, "CRZ 200" on the body; available in:
Bottles of 60 capsules: NDC 0069-8141-20 -
•250 mg capsules
Hard gelatin capsule with pink opaque cap and body, printed with black ink "Pfizer" on the cap, "CRZ 250" on the body; available in:
Bottles of 60 capsules: NDC 0069-8140-20
Store at room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Oral Pellets:
-
•20 mg oral pellets
Hard gelatin capsule, size 4, light blue opaque cap and white opaque body, printed with black ink “Pfizer” on the cap, “CRZ 20” on the body; available in:
Bottles of 60 capsules: NDC 0069-0251-60 -
•50 mg oral pellets
Hard gelatin capsule, size 3, gray opaque cap and light gray opaque body, printed with black ink “Pfizer” on the cap, “CRZ 50” on the body; available in:
Bottles of 60 capsules: NDC 0069-0507-60 -
•150 mg oral pellets
Hard gelatin capsule, size 0, light blue opaque cap and body, printed with black ink “Pfizer” on the cap, “CRZ 150” on the body; available in:
Bottles of 60 capsules: NDC 0069-1500-60
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
7.3 Drugs That Prolong the Qt Interval (7.3 Drugs That Prolong the QT Interval)
XALKORI can prolong the QT/QTc interval. Avoid concomitant use of XALKORI with drugs that prolong the QT interval [see Warnings and Precautions (5.3), Clinical Pharmacology (12.2)].
5.2 Interstitial Lung Disease/pneumonitis (5.2 Interstitial Lung Disease/Pneumonitis)
Severe, life-threatening, or fatal interstitial lung disease (ILD)/pneumonitis can occur in patients treated with XALKORI. Across clinical trials in patients with NSCLC (n=1719), 2.9% of XALKORI-treated patients had ILD of any grade, 1% had Grade 3 or 4 ILD, and 0.5% had fatal ILD [see Adverse Reactions (6.1)]. Interstitial lung disease generally occurred within 3 months after the initiation of XALKORI.
In Study ADVL0912, among 121 patients ages 1 to ≤21 years with relapsed or refractory tumors, including ALCL and IMT, ILD occurred in 0.8% of patients.
Monitor patients for pulmonary symptoms indicative of ILD/pneumonitis. Exclude other potential causes of ILD/pneumonitis, and permanently discontinue XALKORI in patients diagnosed with drug-related ILD/pneumonitis [see Dosage and Administration (2.6)].
2.6 Dosage Modifications for Adverse Reactions
The recommended dosage modifications for adverse reactions for adult patients with NSCLC or IMT are provided in Table 4.
|
Dose Reduction |
Dose and Schedule |
|
First Dose Reduction |
200 mg twice daily |
|
Second Dose Reduction |
250 mg once daily |
|
Permanently discontinue XALKORI capsules or pellets if unable to tolerate 250 mg taken once daily. |
The recommended dosage modifications for adverse reactions for pediatric patients with ALCL or IMT and young adults with ALCL are based on body surface area and are provided in Table 5.
|
Body Surface Area (BSA) |
First Dose Reduction |
Second Dose Reduction
Permanently discontinue in patients who are unable to tolerate XALKORI capsules or pellets after 2 dose reductions.
|
||
|
Dosage |
Dosage Form and Strength to Achieve Recommended Dose Reduction |
Dosage |
Dosage Form and Strength to Achieve Recommended Dose Reduction |
|
|
0.38 to 0.46 m2 |
90 mg twice daily |
Pellets: 2 x 20 mg + 1 x 50 mg |
70 mg twice daily |
Pellets: 1 x 20 mg + 1 x 50 mg |
|
0.47 to 0.51 m2 |
100 mg twice daily |
Pellets: 2 x 50 mg |
80 mg twice daily |
Pellets: 4 x 20 mg |
|
0.52 to 0.61 m2 |
120 mg twice daily |
Pellets: 1 x 20 mg + 2 x 50 mg |
90 mg twice daily |
Pellets: 2 x 20 mg + 1 x 50 mg |
|
0.62 to 0.80 m2 |
150 mg twice daily |
Pellets: 1 x 150 mg |
120 mg twice daily |
Pellets: 1 x 20 mg + 2 x 50 mg |
|
0.81 to 0.97 m2 |
200 mg twice daily |
Pellets: 1 x 50 mg + 1 x 150 mg |
150 mg twice daily |
Pellets: 1 x 150 mg |
|
0.98 to 1.16 m2 |
220 mg twice daily |
Pellets: 1 x 20 mg + 1 x 50 mg + 1 x 150 mg |
170 mg twice daily |
Pellets: 1 x 20 mg + 1 x 150 mg |
|
1.17 to 1.33 m2 |
250 mg twice daily |
Pellets: 2 x 50 mg + 1 x 150 mg |
200 mg twice daily |
Pellets: 1 x 50 mg + 1 x 150 mg |
|
1.34 to 1.69 m2 |
250 mg twice daily |
Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg |
200 mg twice daily |
Pellets: 1 x 50 mg + 1 x 150 mg Or Capsule: 1 x 200 mg |
|
1.7 m2 or greater |
400 mg twice daily |
Pellets: 2 x 50 mg + 2 x 150 mg Or Capsule: 2 x 200 mg |
250 mg twice daily |
Pellets: 2 x 50 mg + 1 x 150 mg Or Capsule: 1 x 250 mg |
Recommended Dosage Modifications for Hematologic Adverse Reactions for Adult Patients with NSCLC or IMT
The recommended dosage modifications for hematologic adverse reactions for adult patients with NSCLC or IMT are provided in Table 6.
|
Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0.
|
XALKORI Dosage Modification |
|
Grade 3 |
Withhold until recovery to Grade 2 or less, then resume at the same dosage. |
|
Grade 4 |
Withhold until recovery to Grade 2 or less, then resume at next lower dosage. |
Monitor complete blood counts including differential weekly for the first month of therapy and then at least monthly, with more frequent monitoring if Grade 3 or 4 abnormalities, fever, or infection occur.
Recommended Dosage Modifications for Hematologic Adverse Reactions in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT
The recommended dosage modifications for hematologic adverse reactions in pediatric and young adult patients with ALCL or pediatric patients with IMT are provided in Table 7.
|
Severity of Adverse Reaction |
XALKORI Dosage Modification |
|
Absolute Neutrophil Count (ANC) |
|
|
Less than 0.5 x 109/L |
First occurrence: Withhold until recovery to ANC greater than 1.0 x 109/L, then resume at the next lower dosage.
|
|
Platelet Count |
|
|
25 to 50 x 109/L with concurrent bleeding |
Withhold until recovery to platelet count greater than 50 x 109/L and bleeding resolves, then resume at the same dosage. |
|
Less than 25 x 109/L |
Withhold until recovery to platelet count greater than 50 x 109/L, then resume at the next lower dosage. Permanently discontinue for recurrence. |
|
Anemia |
|
|
Hemoglobin less than 8 g/dL |
Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the same dosage. |
|
Life-threatening anemia; urgent intervention indicated. |
Withhold until recovery to hemoglobin 8 g/dL or more, then resume at the next lower dosage. Permanently discontinue for recurrence. |
Recommended Dosage Modifications for Non-Hematologic Adverse Reactions
The recommended dosage modifications for non-hematologic adverse reactions are provided in Table 8.
|
Severity of Adverse Reaction Grade based on National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE), version 4.0.
|
XALKORI Dosage Modification |
|
Hepatotoxicity [see Warnings and Precautions (5.1)] |
|
|
Alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 1.5 times ULN |
Withhold until recovery to baseline or less than or equal to 3 times ULN, then resume at next lower dosage. |
|
ALT or AST greater than 3 times ULN with concurrent total bilirubin greater than 1.5 times ULN (in the absence of cholestasis or hemolysis) |
Permanently discontinue. |
|
Interstitial Lung Disease (Pneumonitis) [see Warnings and Precautions (5.2)] |
|
|
Any grade drug-related interstitial lung disease/pneumonitis |
Permanently discontinue. |
|
QT Interval Prolongation [see Warnings and Precautions (5.3)] |
|
|
QT corrected for heart rate (QTc) greater than 500 ms on at least 2 separate electrocardiograms (ECGs) |
Withhold until recovery to baseline or to a QTc less than 481 ms, then resume at next lower dosage. |
|
QTc greater than 500 ms or greater than or equal to 60 ms change from baseline with Torsade de pointes or polymorphic ventricular tachycardia or signs/symptoms of serious arrhythmia |
Permanently discontinue. |
|
Bradycardia [see Warnings and Precautions (5.4)] |
|
|
Bradycardia Adult patients: Heart rate less than 60 beats per minute (bpm); Pediatric patients: Resting heart rate less than the 2.5th percentile per age-specific norms. (symptomatic, may be severe and medically significant, medical intervention indicated)
|
Withhold until recovery to a resting heart rate according to the patient’s age (based on the 2.5th percentile per age-specific norms) as follows:
Evaluate concomitant medications known to cause bradycardia, as well as antihypertensive medications. |
|
Bradycardia (life-threatening consequences, urgent intervention indicated) |
Permanently discontinue if no contributing concomitant medication is identified. |
|
Ocular Toxicity, including Visual Loss [see Warnings and Precautions (5.5)] |
|
|
Visual Symptoms, Grade 1 (mild symptoms) or Grade 2 (moderate symptoms affecting ability to perform age-appropriate activities of daily living) |
Monitor symptoms and report any symptoms to an eye specialist. Consider dose reduction for Grade 2 visual disorders. |
|
Visual Loss (Grade 3 or 4 Ocular Disorder, marked decrease in vision) |
Discontinue during evaluation of severe visual loss. |
|
Gastrointestinal Toxicity Dosage modifications for gastrointestinal toxicity for pediatric patients with ALCL or IMT and young adults with ALCL only.
[see Warnings and Precautions (5.6)]
|
|
|
Nausea (Grade 3: inadequate oral intake for more than 3 days, medical intervention required) |
Grade 3 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. Permanently discontinue in patients who are unable to tolerate XALKORI after 2 dose reductions.
|
|
Vomiting (Grade 3: more than 6 episodes in 24 hours for more than 3 days, medical intervention required, i.e., tube feeding or hospitalization; Grade 4: life-threatening consequences, urgent intervention indicated) |
Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. |
|
Diarrhea (Grade 3: increase of 7 or more stools per day over baseline; incontinence; hospitalization indicated; Grade 4: life-threatening consequences, urgent intervention indicated) |
Grade 3 or 4 (despite maximum medical therapy): Withhold until resolved, and then resume at the next lower dose level. |
Principal Display Panel 20 Mg Oral Pellets Label (PRINCIPAL DISPLAY PANEL - 20 mg Oral Pellets Label)
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
NDC 0069-0251-60
Pfizer
XALKORI®
(crizotinib) oral pellets
20 mg
Open capsule shell to administer pellets.
Do not swallow capsule whole.
Do not crush or chew pellets.
60 Capsules
Rx only
Principal Display Panel 50 Mg Oral Pellets Label (PRINCIPAL DISPLAY PANEL - 50 mg Oral Pellets Label)
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
NDC 0069-0507-60
Pfizer
XALKORI®
(crizotinib) oral pellets
50 mg
Open capsule shell to administer pellets.
Do not swallow capsule whole.
Do not crush or chew pellets.
60 Capsules
Rx only
2.8 Dosage Modification for Severe Renal Impairment
The recommended dosage of XALKORI in patients with severe renal impairment [creatinine clearance (CLcr) less than 30 mL/min, calculated using the modified Cockcroft-Gault equation for adult patients and the Schwartz equation for pediatric patients] not requiring dialysis is the second dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
Principal Display Panel 150 Mg Oral Pellets Label (PRINCIPAL DISPLAY PANEL - 150 mg Oral Pellets Label)
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
NDC 0069-1500-60
Pfizer
XALKORI®
(crizotinib) oral pellets
150 mg
Open capsule shell to administer pellets.
Do not swallow capsule whole.
Do not crush or chew pellets.
60 Capsules
Rx only
2.2 Recommended Testing During Treatment With Xalkori (2.2 Recommended Testing During Treatment with XALKORI)
-
•Monitor liver function tests, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), and total bilirubin, every 2 weeks during the first 2 months of treatment, then once a month, and as clinically indicated, with more frequent repeat testing for increased liver transaminases, alkaline phosphatase, or total bilirubin in patients who develop increased transaminases [see Warnings and Precautions (5.1)].
-
•Monitor complete blood counts including differential weekly for the first month of therapy and then at least monthly, with more frequent monitoring if Grade 3 or 4 abnormalities, fever, or infection occur [see Adverse Reactions (6.1)].
-
•For pediatric and young adult patients with ALCL or pediatric patients with IMT, obtain baseline and follow-up ophthalmologic examinations including retinal examination within 1 month of starting XALKORI and every 3 months thereafter [see Warnings and Precautions (5.5)].
Principal Display Panel 200 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 200 mg Capsule Bottle Label)
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
Pfizer
NDC 0069-8141-20
XALKORI®
(crizotinib) capsules
200 mg
Swallow capsule whole
60 Capsules
Rx only
Principal Display Panel 250 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 250 mg Capsule Bottle Label)
ALWAYS DISPENSE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
Pfizer
NDC 0069-8140-20
XALKORI®
(crizotinib) capsules
250 mg
Swallow capsule whole
60 Capsules
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with crizotinib have not been conducted.
Crizotinib was genotoxic in an in vitro micronucleus assay in Chinese Hamster Ovary cultures, in an in vitro human lymphocyte chromosome aberration assay, and in in vivo rat bone marrow micronucleus assays. Crizotinib was not mutagenic in vitro in the bacterial reverse mutation (Ames) assay.
No specific studies with crizotinib have been conducted in animals to evaluate the effect on fertility; however, crizotinib is considered to have the potential to impair reproductive function and fertility in humans based on findings in repeat-dose toxicity studies in the rat. Findings observed in the male reproductive tract included testicular pachytene spermatocyte degeneration in rats given greater than or equal to 50 mg/kg/day for 28 days (greater than 1.7 times the recommended human dose based on AUC). Findings observed in the female reproductive tract included single-cell necrosis of ovarian follicles of a rat given 500 mg/kg/day (approximately 10 times the recommended human dose based on body surface area) for 3 days.
1.1 Alk Or Ros1 Positive Metastatic Non Small Cell Lung Cancer (1.1 ALK- or ROS1-Positive Metastatic Non-Small Cell Lung Cancer)
XALKORI is indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are anaplastic lymphoma kinase (ALK) or ROS1-positive as detected by an FDA-approved test [see Dosage and Administration (2.1)].
2.7 Dosage Modifications for Moderate and Severe Hepatic Impairment
The recommended dose of XALKORI in patients with moderate hepatic impairment [any aspartate aminotransferase (AST) and total bilirubin greater than 1.5 times the upper limit of normal (ULN) and less than or equal to 3 times ULN] is the first dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
The recommended dose of XALKORI in patients with severe hepatic impairment (any AST and total bilirubin greater than 3 times ULN) is the second dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL [see Dosage and Administration (2.6), Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2.9 Dosage Modification for Concomitant Use of Strong Cyp3a Inhibitors (2.9 Dosage Modification for Concomitant Use of Strong CYP3A Inhibitors)
Avoid concomitant use of strong CYP3A inhibitors [see Drug Interactions (7.1)]. If concomitant use of strong CYP3A inhibitors is unavoidable, reduce the dose of XALKORI to the second dose reduction shown in Table 4 for adult patients with NSCLC or IMT and Table 5 for pediatric patients with ALCL or IMT and young adults with ALCL. After discontinuation of a strong CYP3A inhibitor, resume the XALKORI dose used prior to initiating the strong CYP3A inhibitor.
1.2 Relapsed Or Refractory, Systemic Alk Positive Anaplastic Large Cell Lymphoma (1.2 Relapsed or Refractory, Systemic ALK-Positive Anaplastic Large Cell Lymphoma)
XALKORI is indicated for the treatment of pediatric patients 1 year of age and older and young adults with relapsed or refractory, systemic anaplastic large cell lymphoma (ALCL) that is ALK-positive.
14.2 Relapsed Or Refractory, Systemic Alk Positive Anaplastic Large Cell Lymphoma (14.2 Relapsed or Refractory, Systemic ALK-Positive Anaplastic Large Cell Lymphoma)
The efficacy of XALKORI was evaluated in Study ADVL0912 (NCT00939770), a multicenter, single arm, open-label study in patients 1 to ≤21 years of age that included 26 patients with relapsed or refractory, systemic ALK-positive ALCL after at least one systemic treatment. ALK-positive status (confirmation of an ALK fusion) was determined locally by immunohistochemistry or fluorescence in situ hybridization. The study excluded patients with primary cutaneous ALCL or central nervous system involvement by lymphoma.
Patients received XALKORI 280 mg/m2 (20 patients) or 165 mg/m2 (6 patients) orally twice daily until disease progression or unacceptable toxicity. Patients were permitted to discontinue XALKORI to undergo hematopoietic stem cell transplantation (HSCT).
Of the 26 patients evaluated, the median age was 11 years (range: 3 to 20); 69% were male, 54% were White, 19% Black, 8% Asian. Patient enrollment by age category was 4 patients from 3 to <6 years, 11 patients from 6 to <12 years, 7 patients from 12 to <18 years, and 4 patients from 18 to ≤21 years.
All patients had received multi-agent systemic therapy, 2 (8%) had received a prior HSCT and 4 (15%) had received at least 3 prior therapies.
Efficacy was based on objective response rate and duration of response, as assessed by an independent review committee (Table 19). The median time to first response was 3.9 weeks (range: 3.5 to 9.1 weeks).
| Efficacy Parameter | N=26 |
|---|---|
| CI=confidence interval; N/n=number of patients | |
|
Objective response rate (95% CI, %) Based on Lugano Classification.
,
95% CI based on Wilson score method.
|
88% (71, 96) |
|
Complete response, n |
21 (81%) |
|
Partial response, n |
2 (8%) |
|
Duration of response Of 23 patients with objective response, 2 had disease progression and the remainder (91% of responding patients) were censored.
|
|
|
Patients maintaining response at 3 months, n/N |
13/23 (57%) |
|
Patients maintaining response at 6 months, n/N |
9/23 (39%) |
|
Patients maintaining response at 12 months, n/N |
5/23 (22%) |
1.3 Unresectable, Recurrent, Or Refractory Alk Positive Inflammatory Myofibroblastic Tumor (1.3 Unresectable, Recurrent, or Refractory ALK-Positive Inflammatory Myofibroblastic Tumor)
XALKORI is indicated for the treatment of adult and pediatric patients 1 year of age and older with unresectable, recurrent, or refractory inflammatory myofibroblastic tumor (IMT) that is ALK-positive.
2.5 Concomitant Treatments for Pediatric and Young Adult Patients With Alcl Or Pediatric Patients With Imt (2.5 Concomitant Treatments for Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT)
Antiemetics are recommended prior to and during treatment with XALKORI to prevent nausea and vomiting. Provide standard antiemetic and antidiarrheal agents for gastrointestinal toxicities.
Consider intravenous or oral hydration for patients at risk of dehydration, and replace electrolytes as clinically indicated [see Warnings and Precautions (5.6)].
5.6 Gastrointestinal Toxicity in Pediatric and Young Adult Patients With Alcl Or Pediatric Patients With Imt (5.6 Gastrointestinal Toxicity in Pediatric and Young Adult Patients with ALCL or Pediatric Patients with IMT)
XALKORI can cause severe gastrointestinal toxicities in pediatric and young adult patients with ALCL or pediatric patients with IMT [see Adverse Reactions (6.1)]. In patients with ALCL (n=26), gastrointestinal toxicity occurred in 100% of patients; Grade 3 gastrointestinal toxicity occurred in 27% of patients and included diarrhea, nausea, vomiting, and stomatitis. In pediatric patients with IMT (n=14), vomiting occurred in 93%, nausea occurred in 86%, and diarrhea occurred in 64% of patients.
Provide standard antiemetic and antidiarrheal agents for gastrointestinal toxicities in pediatric and young adult patients with ALCL or pediatric patients with IMT. Antiemetics are recommended prior to and during treatment with XALKORI to prevent nausea and vomiting. If patients develop Grade 3 nausea lasting 3 days or Grade 3 or 4 diarrhea or vomiting despite maximum medical therapy, withhold XALKORI until resolved, and then resume at the next lower dose level. Consider supportive care such as hydration, electrolyte supplementation, and nutritional support as clinically indicated [see Dosage and Administration (2.6)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:22.162142 · Updated: 2026-03-14T22:29:41.730396