Protex®
29a0a75c-a8a1-4d11-87ae-6d2de4fa9518
34390-5
HUMAN OTC DRUG LABEL
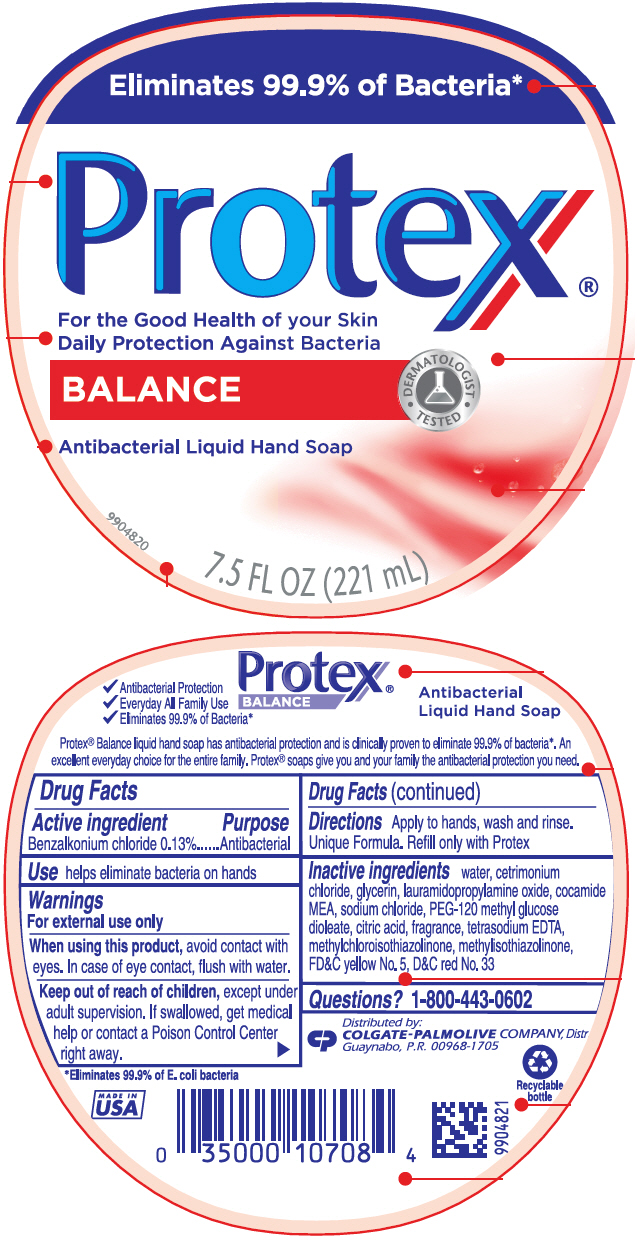
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antibacterial
Medication Information
Purpose
Antibacterial
Description
Drug Facts
Use
helps eliminate bacteria on hands
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children, except under adult supervision. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7
When using this product, avoid contact with eyes. In case of eye contact, flush with water.
Warnings
For external use only
Directions
Apply to hands, wash and rinse. Unique Formula. Refill only with Protex
Questions?
Call toll-free 1-800-443-0602
Active Ingredient
Benzalkonium chloride 0.13%
Inactive Ingredients
water, cetrimonium chloride, glycerin, lauramidopropylamine oxide, cocamide MEA, sodium chloride, PEG-120 methyl glucose dioleate, citric acid, fragrance, tetrasodium EDTA, methylchloroisothiazolinone, methylisothiazolinone, FD&C yellow No. 5, D&C red No. 33
Principal Display Panel 221 Ml Bottle Label
Eliminates 99.9% of Bacteria*
Protex®
For the Good Health of your Skin
Daily Protection Against Bacteria
BALANCE
• DERMATOLOGIST •
TESTED
Antibacterial Liquid Hand Soap
9904820
7.5 FL OZ (221 mL)
Structured Label Content
Use
helps eliminate bacteria on hands
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children, except under adult supervision. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7 (50567-7)
When using this product, avoid contact with eyes. In case of eye contact, flush with water.
Purpose
Antibacterial
Warnings
For external use only
Directions
Apply to hands, wash and rinse. Unique Formula. Refill only with Protex
Questions?
Call toll-free 1-800-443-0602
Active Ingredient (Active ingredient)
Benzalkonium chloride 0.13%
Inactive Ingredients (Inactive ingredients)
water, cetrimonium chloride, glycerin, lauramidopropylamine oxide, cocamide MEA, sodium chloride, PEG-120 methyl glucose dioleate, citric acid, fragrance, tetrasodium EDTA, methylchloroisothiazolinone, methylisothiazolinone, FD&C yellow No. 5, D&C red No. 33
Principal Display Panel 221 Ml Bottle Label (PRINCIPAL DISPLAY PANEL - 221 mL Bottle Label)
Eliminates 99.9% of Bacteria*
Protex®
For the Good Health of your Skin
Daily Protection Against Bacteria
BALANCE
• DERMATOLOGIST •
TESTED
Antibacterial Liquid Hand Soap
9904820
7.5 FL OZ (221 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:05.710858 · Updated: 2026-03-14T23:04:33.656231