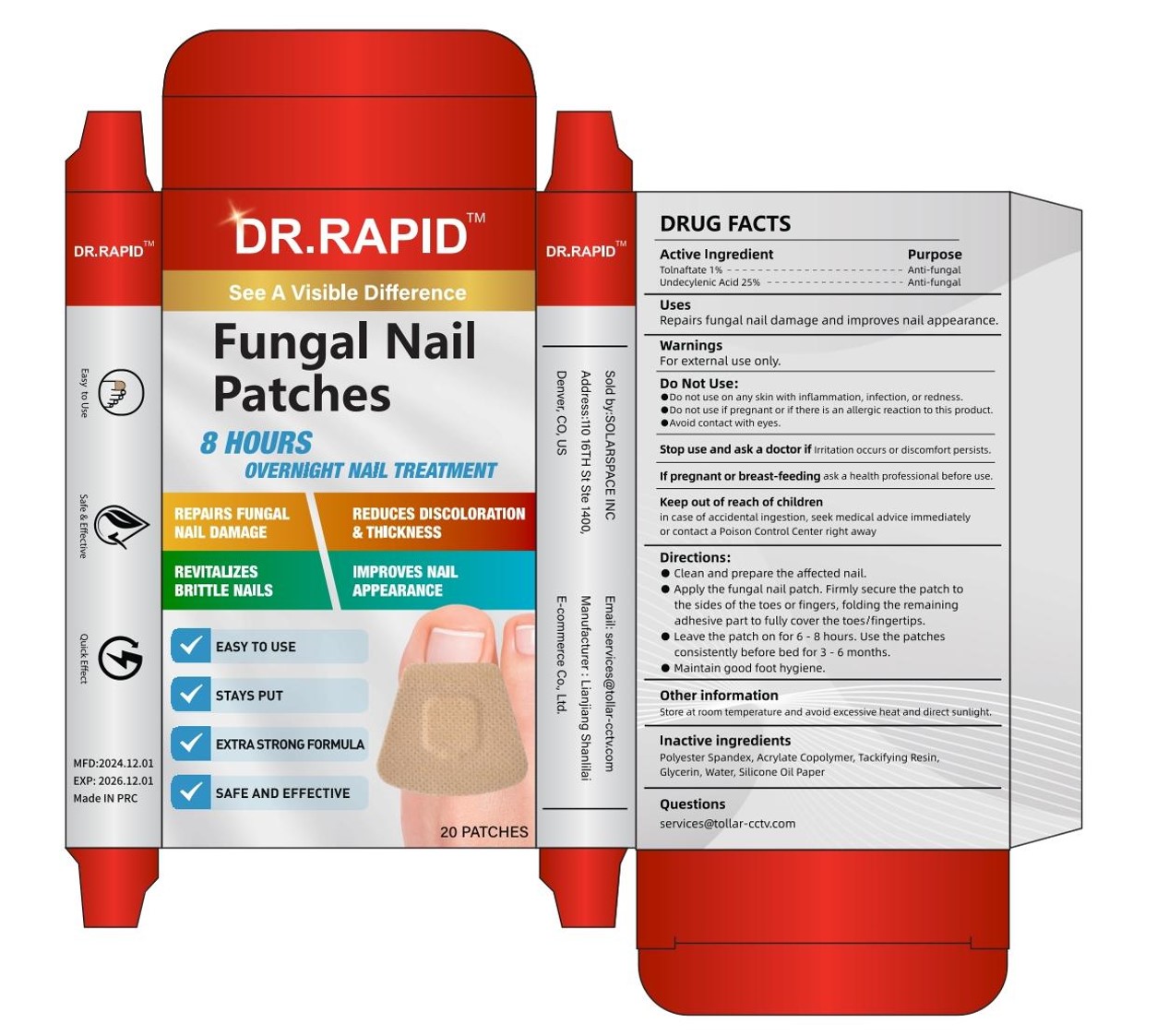
Fungal Nail Patches
299a63ad-f321-117c-e063-6394a90a3a22
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Fungal Nail Patches
Medication Information
Warnings and Precautions
For external use only.
Indications and Usage
Repairs fungal nail damage and improves nail appearance.
Dosage and Administration
Clean and prepare the affected nail.
Apply the fungal nail patch. Firmly secure the patch tothe sides of the toes or fingers, folding the remainingadhesive part to fully cover the toesffingertips.
Leave the patch on for 6 -8 hours. Use the patchesconsistently before bed for 3 -é months.
Maintain good foot hygiene.
Description
Fungal Nail Patches
Section 42229-5
Fungal Nail Patches
Section 44425-7
Store at room temperature and avoid excessive heat and direct sunlight.
Section 50565-1
in case of accidental ingestion, seek medical advice immediatelyor contact a Poison Control Center right away
Section 50566-9
Stop use and ask a doctor if lrritation occurs or discomfort persists
Section 50570-1
Do not use on any skin with inflammation,infectlon, or redness.
Do not use if preqnant or if there is an allergic reaction to this product
Avoid contact with eyes.
Section 51727-6
Polyester Spandex
Acrylate Copolymer
Tackifying Resin
Glycerin
Water
Silicone Oil Paper
Section 51945-4
Section 55105-1
Anti-fungal
Section 55106-9
TOLNAFTATE 1%
UNDECYLENIC ACID 25%
Structured Label Content
Indications and Usage (34067-9)
Repairs fungal nail damage and improves nail appearance.
Dosage and Administration (34068-7)
Clean and prepare the affected nail.
Apply the fungal nail patch. Firmly secure the patch tothe sides of the toes or fingers, folding the remainingadhesive part to fully cover the toesffingertips.
Leave the patch on for 6 -8 hours. Use the patchesconsistently before bed for 3 -é months.
Maintain good foot hygiene.
Warnings and Precautions (34071-1)
For external use only.
Section 42229-5 (42229-5)
Fungal Nail Patches
Section 44425-7 (44425-7)
Store at room temperature and avoid excessive heat and direct sunlight.
Section 50565-1 (50565-1)
in case of accidental ingestion, seek medical advice immediatelyor contact a Poison Control Center right away
Section 50566-9 (50566-9)
Stop use and ask a doctor if lrritation occurs or discomfort persists
Section 50570-1 (50570-1)
Do not use on any skin with inflammation,infectlon, or redness.
Do not use if preqnant or if there is an allergic reaction to this product
Avoid contact with eyes.
Section 51727-6 (51727-6)
Polyester Spandex
Acrylate Copolymer
Tackifying Resin
Glycerin
Water
Silicone Oil Paper
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Anti-fungal
Section 55106-9 (55106-9)
TOLNAFTATE 1%
UNDECYLENIC ACID 25%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:56.532246 · Updated: 2026-03-14T23:06:06.305155